Abstract
A total of 210 frog samples originating either from a mass mortality (1991/1992) or from routine postmortem investigations of the years 1990 to 2004 were examined retrospectively for a possible involvement of Chlamydiae. For a prevalence study of Chlamydia in a selected Swiss amphibian population, 403 samples from free-ranging Rana temporaria were examined. Histopathology, immunohistochemistry using a monoclonal antibody against chlamydial lipopolysaccharide, and a 16S rRNA polymerase chain reaction (PCR) followed by DNA sequencing were performed on the formalin-fixed and paraffin-embedded tissues. Using PCR, 8 of 54 (14.8%) frog samples from the mass mortality (1991/1992) were positive for Chlamydia suis S45. A control group of healthy Xenopus laevis had 3 of 38 positive samples, sequenced as C suis S45 (2/3) and an endosymbiont of Acanthamoeba species UWE1 (1/ 3). Chlamydophila pneumoniae TW-183 was detected from exotic frogs kept in a zoo. Of the frogs collected for the prevalence study, 6 of 238 (2.5%) tested positive, 1 each for C suis S45, Cp pneumoniae TW-183, and uncultured Chlamydiales CRG22, and the remaining 3 revealed Chlamydophila abortus S26/3. In immunohistochemistry, there were 2 positive labeling reactions, 1 in intestine and the other in the epithelium coating the body cavity, both testing positive for Cp pneumoniae TW-183 in PCR. Histologically there were no lesions recorded being characteristic for Chlamydia. Although there is a prevalence of Chlamydia in Swiss frogs, no connection to a mass mortality (1991/1992) could be established. For the first time, C suis S45 and Cp abortus S26/3 were detected in frog material.
Introduction
In the past few decades, there have been sporadic reports of frog and amphibian mortalities from all over the world. The population declines exceeded normal fluctuations, and a discussion about possible reasons was started.10,15 In some of the events, the chytrid fungus Batrachochytrium dendrobatidis could be identified as the causative agent.2,3,21 Other possible factors contributing to the mass declines were infection with Ranavirus, increased UV-B radiation, or habitat and climate changes.5,6,26
In northern Switzerland there were mass mortalities of amphibians in the winter of 1990/1991 and to a lesser extent in 1992. The species affected more than any other was the common frog (Rana temporaria). In springtime, an unusually high number of dead animals were found in ponds after the ice had melted. A governmental study investigated many factors, such as several parameters of water quality and environmental effects, but no search for specific pathogens was undertaken.13 The conclusion of the study was that the mortality occurred due to an oxygen deficit and poor water quality.
The first reports of chlamydiosis in amphibians described sudden death with inflammation of liver, spleen, kidney, and heart in Xenopus laevis due to naturally and experimental infection.16,20,28 The detection of Chlamydiae in these early studies before polymerase chain reaction (PCR) was based on inclusion morphology and histologic lesions. Therefore, the agent was assumed to be Chlamydia psittaci. Only since 1998 has the use of such new molecular methods as PCR provided an opportunity to differentiate the different species. Berger and colleagues were the first to identify Chlamydophila pneumoniae by PCR in the lung of a Giant Barred Frog with chronic mononuclear pneumonia.4 Reed et al. reported an epizootic in a breeding colony of Xenopus tropicalis due to Cp pneumoniae in combination with the chytrid fungus B dendrobatidis.23 In 2001, Hotzel et al. showed by comparison of all currently known isolates of Cp pneumoniae that the isolate they found in African frogs (Cryptohylax gresshoffi) was closest to the koala biovar.14
Because Chlamydia in frogs have been reported mostly after the event in 1991/1992, animals from the mass decline in Switzerland were investigated for the involvement of Chlamydia. Because all of the described incidents were single cases or local events, this is the first study to include more animals from different sources and years. One aim of the study was to investigate whether there could have been an involvement of Chlamydia in the mass mortality of R temporaria on the basis of retrospective material; another aim was to find out if there is a prevalence of Chlamydia in selected Swiss frog populations at present.
Materials and Methods
Cases
The study was performed on formalin-fixed and paraffin-embedded tissue blocks. The retrospective material was archived in the institute from routine diagnostic investigations, showing great differences in the number and amount of organs being sampled and state of tissue preservation. Many different organs were put on 1 paraffin block for economic reasons. Information about the archived data was very restricted, and the names of the amphibian species and the number of animals sampled per case were mostly unknown. The number of blocks was taken as number of samples, and the samples were divided into Groups A to F, as listed in Table 1. The number of individuals in each group could only be estimated for Groups A to D; exact data were only present for Groups E and F.
Sample groups.
Group A consisted of 54 samples of R temporaria from a mass mortality in 1991/1992. The remainder of the retrospective material was divided into another 3 groups: free-ranging amphibians of various species (Group B, 40 samples), Xenopus frogs originating from laboratories (Group C, 33 samples), and animals kept in zoos or by private owners (Group D, 83 samples). Because all these animals had a history of sudden death, they were compared with a clinically healthy control group of 19 laboratory Xenopus frogs being sacrificed after oocyte harvesting (Group E, 38 samples).
For the prevalence study, 126 common frogs (R temporaria) were collected in fall 2004 and spring 2005 (Group F, 238 samples) during the times of migration. Because all amphibians are protected by law in Switzerland, a permit (No. 4071) from the department for nature protection and landscape conservation was issued, which allowed sampling only in designated areas with dense populations.
Histology and immunohistochemistry
The reported macroscopic findings of the retrospective cases (Groups A−D) were summarized from the records and compared with the histopathologic findings in organs with macroscopic lesions using HE-stained sections. Of the R temporaria collected for the prevalence study (Group F) during the migrations, a much wider range of organs was sampled at necropsy; histopathology was performed on heart, spleen, stomach, intestine, liver, lung, kidney, muscle, and skin.
On sections from each paraffin block available, immunohistochemistry was performed to label Chlamydia using a monoclonal antibody directed against chlamydial LPS (AC-I, Progen, Heidelberg), counterstained by hematoxylin. The tissue processing and labeling protocol was followed as described by Soldati et al., with the exception that microwave pretreatment in citrate buffer was extended to 20 minutes.25 To reduce the large amounts of nonspecific pigment, slides were bleached in 10% hydrogen peroxide for 48 hours before immunohistochemistry was done.
DNA extraction for PCR
A tissue section of 25μm was cut from each paraffin block and placed into a sterile microcentrifuge tube. To avoid cross-contamination, an empty paraffin block was cut in between the sample blocks, the microtome cleaned with xylene, and gloves changed after each block. To remove paraffin, 200 μl of extraction buffer (0.5% Tween-20, 1mM EDTA, 50 mM Tris-HCl, pH 8.5) was added and the tube incubated at 95°C for 10 minutes, vortexing regularly. After centrifugation (13,200 rpm, 30 min, 4°C), the paraffin cap was carefully removed from the tube. After adding proteinase K (2 μl of a 19 mg/ml solution, Roche Diagnostics, Rotkreuz, Switzerland), the samples were incubated overnight at 55°C. They were then boiled at 95°C for 10 minutes to inactivate the enzyme activity and be ready for use as a template in the PCR.1
16S PCR for Chlamydia
An order-specific rRNA PCR was used for the frog material, amplifying a 278-bp fragment within the conserved region of the 16S gene. The primer pair consisted of 16S IGF (5′-GATGAGGCATGCAAGTCGAACG-3′) and 16S IGR (5′-CCAGTGTTGGCGGTCAATCTCTC-3′), modified from Everett.12 The reactions with a total volume of 50 μl consisted of 1x Taq buffer (Applied Biosystems, Rotkreuz, Switzerland), 1 μl of template, 1 μM of each primer (Microsynth, Balgach, Switzerland), 200 μM of each dNTP (Roche, Basel, Switzerland), 3.5 mM MgCl2,and 2 U of AmpliTaq Gold DNA Polymerase (Applied Biosystems, Rotkreuz, Switzerland). All reactions were performed with a TGradient thermocycler (Biometra GmbH, Göttingen, Germany), starting with an initial denaturation at 95°C for 15 minutes, followed by 40 cycles of denaturation at 94°C for 30 seconds, primer annealing at 70°C for 30 seconds, extension at 72°C for 45 seconds, and a final extension step at 72°C for 5 minutes.
For negative control, template DNA was substituted by water in the reaction, and a stock solution of genomic Chlamydophila abortus DNA served as positive control. The PCR products were electrophoresed and visualized on a 1.5% agarose gel (Invitrogen Life Technology, Carlsbad, Germany) stained with ethidium bromide. In case of positive reaction, purification of DNA was performed using Qiagen's MinElute Extraction Kit (Qiagen, Hombrechtikon, Switzerland).
OmpA PCR for Chlamydia
A nested PCR amplifying the ompA region was done to confirm the 16S results and to enable a comparison of sequences with other known serovars, predominantly in case of Cp pneumoniae. A primary genus-specific PCR was followed by a secondary, with primer pairs 191CHOMP/CHOMP336 and 201CHOMP/CHOMP271, respectively, as described in Schiller et al.24 In case of positive reaction, species-specific primers were used, 201CHOMP/TRACH269 for C suis and 201CHOMP/KOALA312 for Cp pneumoniae.17
Sequencing of the results was carried out on an Abi377 sequencing device (Applied Biosystems, Foster City, California, USA). The received results were compared with the sequences available in GenBank, using the BLAST search from the National Center for Biotechnology Information.
Results
A range of different chlamydial species was detected in the amphibian tissues using the 16S PCR analysis, as listed in Table 2. In Group A from the mass mortality, 8 of 54 samples were positive for C suis S45 (14,8%). Of Group D consisting of animals privately owned or kept in zoos, 3 of 83 were positive (3.6%), all for Cp pneumoniae TW-183. Due to the arrangement and number of organs present on the paraffin block, it was assumed that all the positive samples came from individual frogs. In the clinically healthy control Xenopus of Group E, 3 samples of 38 also tested positive (7.9%), 2 for C suis S45 and the third sample for endosymbiont of Acanthamoeba species UWE1. In Group F of the R temporaria collected for the prevalence study, 6 samples of a total of 238 (2.5%) were positive, 1 sample each for C suis S45, Cp pneumoniae TW-183, and uncultured Chlamydiales CRG22; the other 3 revealed C abortus S26/3. Except in the case of an endosymbiont of Acanthamoeba species UWE1 with only 90% similarity, the sequences obtained had an identity of 98−100% with the data published in GenBank. More than 70% of the samples were tested repeatedly in the 16S PCR, resulting in the same sequence each time.
Results of16S polymerase chain reaction.
In the nested PCR amplifying the OmpA region, only 4 samples gave a positive reaction, and the data from sequencing confirmed the results from 16S: 2 samples of Group D (Cp pneumoniae TW-183, frog species unknown) and 2 samples of Group F (Cp pneumoniae and C abortus S26/3, R temporaria). Here again, the samples came from 4 individual frogs. The sensitivity of the OmpA reaction proved to be much lower, needing 1,000 templates/μl compared to 1 template/μl in 16S.
Histologically, the retrospective material showed a high degree of autolysis and therefore was sometimes difficult to interpret. Frogs with a history of sudden death generally showed a poor body condition at necropsy, their absent fat reserves and empty intestinal tracts indicating anorexia. In nearly half of the cases, these findings were accompanied with either edematous tissues or ascites in body cavities. The presence of parasites, such as intestinal and pulmonary helminths and protozoa in the kidneys, was very common, but interestingly, the surrounding tissues showed no reaction to the infestations. Inflammatory processes of different ages and qualities were found in several organs, mainly kidney, spleen, and liver, with the kidneys often being involved in acute interstitial nephritis with masses of heterophils present. The lesions were not characteristic for Chlamydia, and there was no correlation between PCR positivity and histological changes.
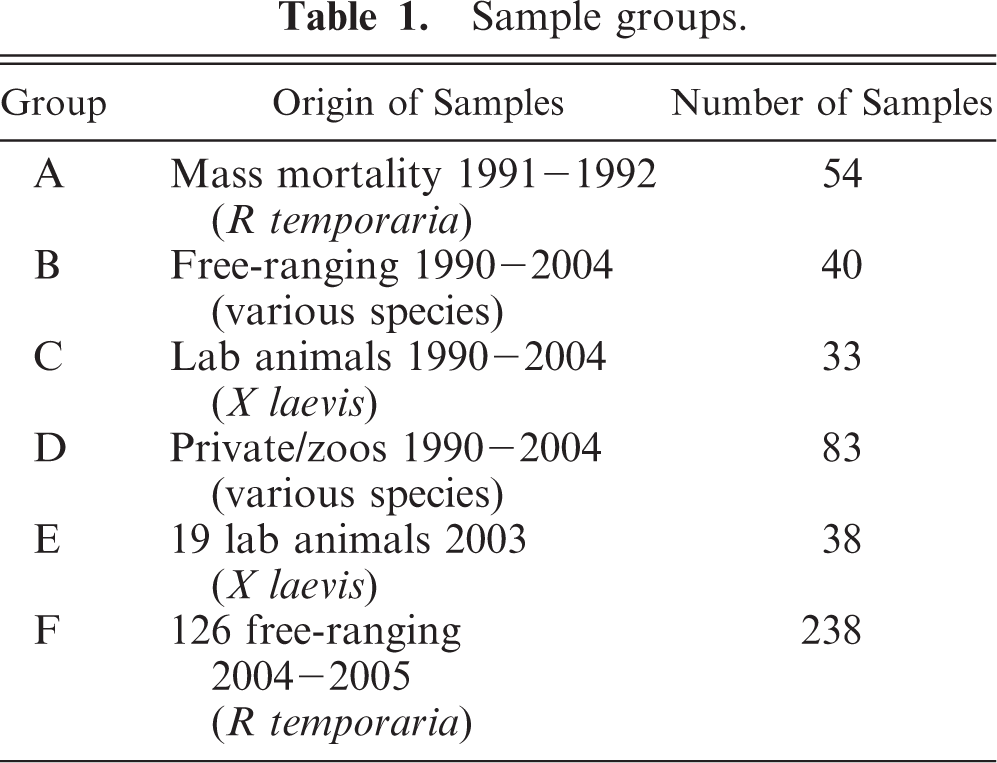
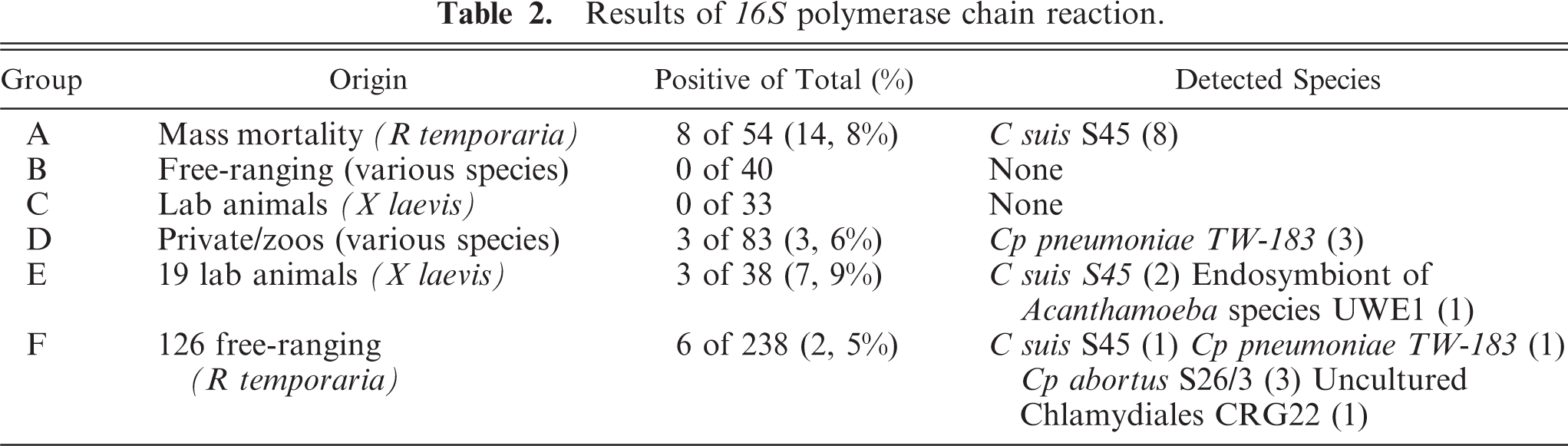
In immunohistochemistry, there were only 2 positive reactions in 2 individual frogs from Group D. One sample showed positive reaction with clearly visible chlamydial inclusions in a macrophage of the small intestine, accompanied with a mild chronic inflammation (Figs. 1–3). The other positive reaction was found in a mesothelial cell coating the body cavity without inflammatory process (Fig. 4). Both samples came from individual frogs in Group D.
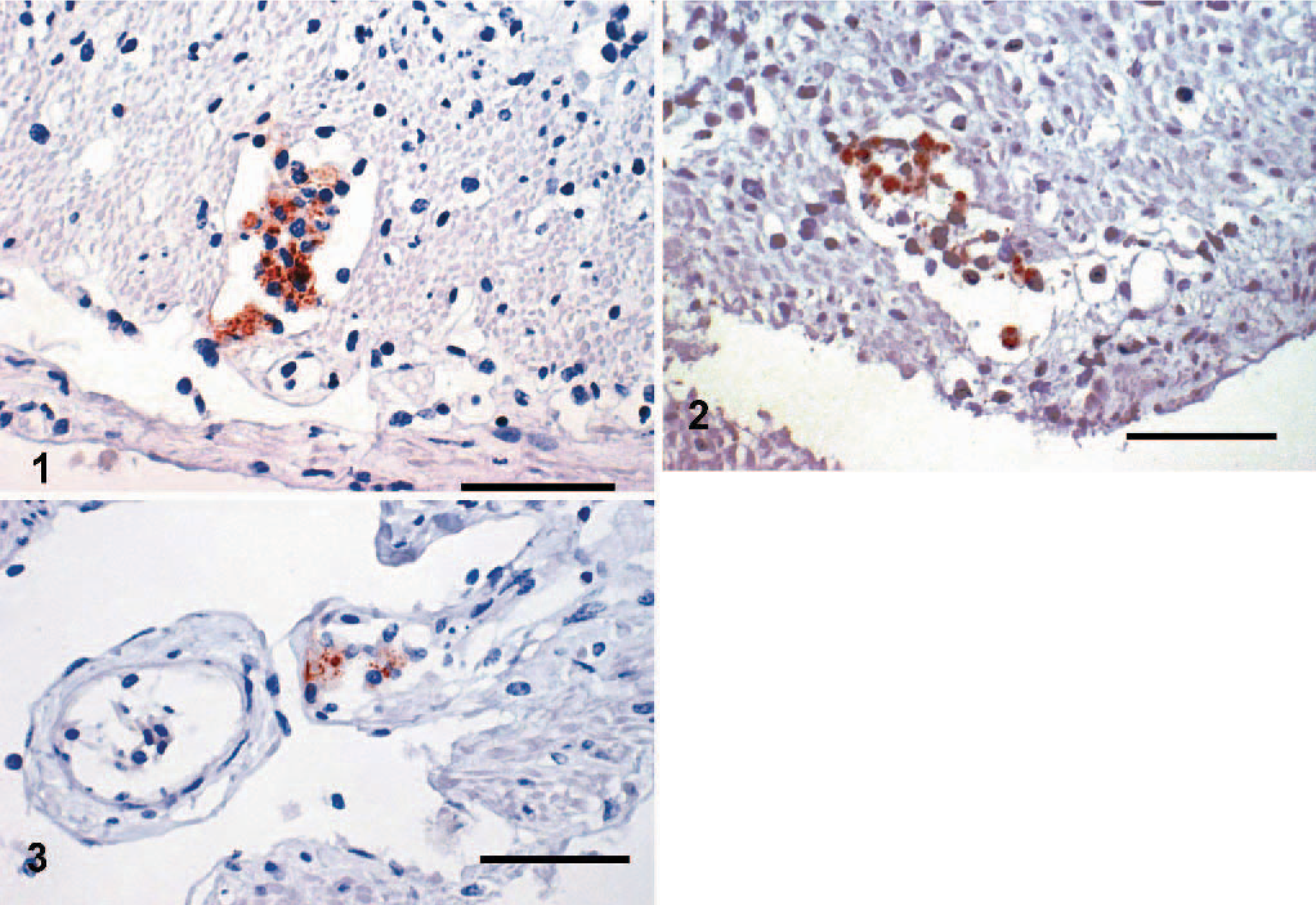
Subserosa of small intestine, pouched frog (Gastrotheca spp.), case No. 11. Mild chronic inflammation, macrophages with numerous chlamydial inclusions. Immunohistochemistry for chlamydial LPS, counterstain hematoxylin.
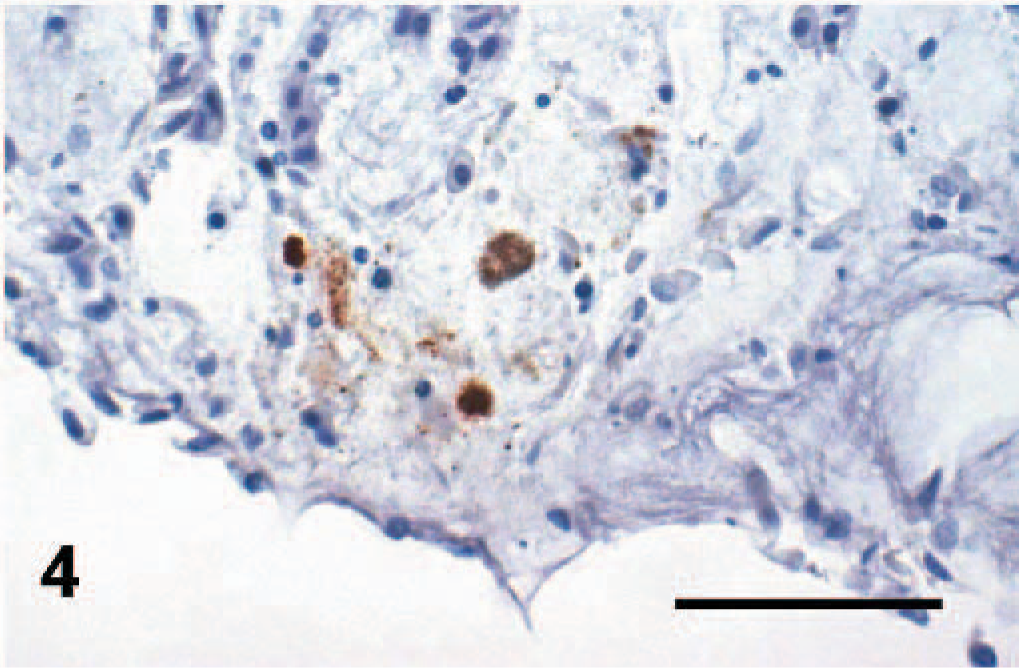
Mesothelium in body cavity, frog species unknown, case No. 9. Mild positive reaction of chlamydial inclusions in a macrophage within mesothelium. Immunohistochemistry for chlamydial lipopolysaccharide, counterstain hematoxylin.
Discussion
Earlier reports about chlamydiosis in amphibians were all based on single cases or events. This was the first study to include more material and a larger variety of tissue samples. The studies before 1989 were based on analysis by immunohistochemistry and morphology of chlamydial inclusion bodies, so the chlamydial agent was assumed to be C psittaci.16,20,28 In 1999, Berger et al. detected Cp pneumoniae in the lung of a single Giant Barred Frog by PCR, and this finding was later confirmed by other authors.4,7,14,23 With the 16S PCR used in our study, more Chlamydia species were detectable than expected. Although Cp pneumoniae had been known to affect amphibians, Chlamydia suis and Cp abortus were found in amphibian material for the first time.
The most surprising fact was to consistently find C suis S45 in the positive samples from the mass mortality in 1991/1992. The sequence we found showed a 98−99% identity with isolates assigned to samples by O. Kolbl from feces of a healthy pig in the early 1960s (porcine serovar of Chlamydia trachomatis).18,19 After Everett et al. reclassified the taxonomy of all chlamydiae and chlamydiaceae in 1999, this strain was listed as C suis S45 (GenBank Number U73110).11,12 The reason why all positive samples from the time of the mass mortality revealed C suis S45 can not be answered sufficiently at this stage. An alignment and comparison of all these sequences showed that the 1 to 2 differences in nucleotides always occurred in the same positions. The chance of repeating amplification mistakes on the same spot is very small. We believe there could be another unknown strain concealed behind the sequence that is currently known as C suis S45.
Cp abortus S26/3 (GenBank Number CR848038) was found in 3 samples from the years 2004 and 2005 (Group F) but in none from the mass mortality (Group A). Because strain S26/3 has also been used as a positive control in the 16S PCR, the samples were tested repeatedly, always giving the same result. Cp abortus is known to cause abortion and reproductive problems in ruminants and pigs; a single case of detection in amphibians was described by Bodetti et al.7,8 Switzerland is a small country with an intense agriculture where farm animals are always proximate to amphibian habitats, as well as settlement areas. Therefore, pathogens only have to travel short distances between species. Feasible routes for amphibian infection could be C suis spread in fecal material in the form of manure and C abortus shed in large amounts in placenta and amniotic fluid during abortions. In addition, it is well documented that Chlamydia can easily cross species barriers, such as Cp abortus causing abortion in humans.22 In Cp pneumoniae, the way of infection for amphibians is not so easy to explain. Earlier reports documented serious disease and mass deaths due to Cp pneumoniae.16,20 However, the positive frogs of this study showed no signs of disease, and in the areas where collecting was done, there were no dead animals found.
Two of the samples positive for Cp pneumoniae in 16S PCR were also confirmed in immunohistochemistry, 1 of them showing positive reaction in the intestine for the first time. In the 16S PCR, the result showed closest (99%) homology with the published sequence for Cp pneumoniae TW-183 (GenBank Number AE017160). The fragment length of 278 bp was too short to permit the differentiation between species spectre strains of Cp pneumoniae.9,27 Thus, we can not say whether we are dealing with an amphibian-specific strain or frog infection by a human strain. The positive immunohistochemistry of intestine is shown in Figs. 1 through 3. The other positive reaction was found in mesothelium originating from the body cavity of a pouched frog imported to the Zurich Zoo from an unknown source. (Fig. 4)
In histopathology, the types of inflammation attributed to Chlamydia in amphibians vary greatly, ranging from acute,16 lymphohistiocytic,22 mononuclear, and lymphocytic20 to granulomatous.23 A common finding of all authors was the involvement of liver, spleen, and kidney into inflammatory processes. Another study of our institute confirmed Chlamydia spp. to be a causative agent of granulomatous inflammation in reptiles.25
We could not always detect signs of inflammation associated with infection. In the sample of intestine staining positive in immunohistochemistry, there was mild chronic inflammation of the subserosa with a macrophage containing clearly visible inclusions. On the contrary, the other sample with positive reaction in mesothelial cells showed no inflammation of any type but also a macrophage with inclusions (Fig. 3). Partly, this could be due to the different levels of sensitivity between immunohistochemistry for lipopolysaccharide and PCR for the 16S fragment. PCR is inherently more sensitive, and in our experience, with properly fixed samples, fragments of approximately 300 bp can be readily detected after extraction from formalin-fixed and paraffin-embedded material. The number of lipopolysaccharide molecules, even in a small inclusion, is very large, so that immunodetection of Chlamydiae does not pose a problem, even if antibody access to the antigen is restricted in paraffin-embedded samples.
Of the 126 R temporaria collected for the prevalence study (Group F), only 6 samples were positive in PCR, indicating that there is a low prevalence for Chlamydia spp. in the Swiss amphibian population. It is documented that the common frog (R temporaria) has disappeared from many habitats, while in other areas its populations grew so intensively that they became a problem for other frog species. The exact reason for the fluctuations could not be determined, so they were, in the absence of specific pathogens, normally explained with environmental changes. The governmental study into the mass mortality of R temporaria (1991/1992) considered different parameters of water quality and environmental factors but included no search for specific pathogens.13 Since then, no other die-offs were noticed in the Swiss amphibian population, and therefore the mass mortality must be considered a single event. The finding of Chlamydia spp. in some of the samples from the die-off is in itself interesting, providing additional proof that amphibians can carry and spread Chlamydia. The absence of histopathologic lesions and the low number of positive samples rather indicate that the mass mortality was caused by something else.
We were also able to detect Chlamydia in a control group of X laevis (Group E) housed in a laboratory for oocyte collection. The frogs could not be examined before death but were reported to be clinically healthy and had no history of increased death rate. In all 3 cases it was a combined sample of spleen and skin testing positive in the 16S PCR, with none of the organs showing any histologic changes. The clinical importance of Chlamydia spp. in amphibians remains unclear, and additional studies of wider range will have to be done. If inapparent infection is possible, as indicated by these healthy frogs, amphibians could represent a vector and/or reservoir for Chlamydia spp. Especially in cases of Cp pneumoniae infection, which is of potential zoonotic risk and where no animal reservoir as such has yet been identified, additional investigations would be worthwhile.
Footnotes
Acknowledgements
We thank the laboratory staff of the Institute of Veterinary Pathology and the Institute of Clinical Pathology for technical assistance.
