Abstract
Brucella spp. were isolated from an abortion case submitted for laboratory examination 8 months after the first clinical symptoms appeared in a kennel consisting of 31 dogs. Pathological investigations revealed the parallel presence of necrotic placentitis and the strong immunostaining of trophoblast cells by immunohistochemistry (IHC) using hyperimmune rabbit anti-Brucella canis primary antibodies. The rapid slide agglutination test was positive in 7 of 31 (23%) cases. The organism B. canis was successfully cultured from the blood, tissues, or vaginal swabs of only 3 of 31 (10%) cases. The isolated strains were identified as B. canis based on their colony morphology and agglutination with R sera. The strains were initially misidentified as B. suis with the “Bruce-ladder” method, and were subsequently correctly identified as B. canis with a single nucleotide polymorphism (SNP) typing test. Three culture-positive cases and 3 culture-negative cases with histories of reproductive disorders were selected and examined for the presence of B. canis infection using histopathology, IHC, and polymerase chain reaction (PCR) assays. Characteristic histologic lesions were found in all of the 6 animals, whereas IHC and PCR yielded positive results only in single cases from both groups. The results imply that all cases of canine abortion should be examined for brucellosis by bacterial culture of aborted fetuses and placentas. Immunohistochemical examination of placentas is also recommended because it is a quick and sensitive technique compared with bacterial culture. Multiple methods (i.e., serology, blood, and genital bacterial cultures) should be applied simultaneously and repeatedly for the reliable screening of B. canis infection in live individuals.
Brucella canis is the etiologic agent of canine brucellosis and has been recognized as the cause of great economic loss in kennels. Although occasional Brucella abortus, Brucella melitensis, and Brucella suis infections have been reported in dogs, B. canis is the main cause of canine brucellosis. 16 Canine brucellosis is a contagious disease with venereal, oronasal, or conjunctival modes of transmission that produces late-term abortion in females, and epididymitis and prostatitis in males. 10,16 Nonreproductive lesions are less common and include ophthalmitis, diskospondylitis, lymphadenopathy, and splenomegaly. Asymptomatic infection frequently occurs as well. 10,16 Although B. canis affects human beings, there are few cases reported, and the illness is often mild. 10,14
A definitive diagnosis of B. canis infection requires simultaneous application of different laboratory techniques due to the lack of a single, highly reliable diagnostic method. The current report describes the first known outbreak of B. canis infection in Hungary, and the associated challenging diagnostic aspects of B. canis infection despite the use of state-of-the-art, modern diagnostic techniques.
The breeding kennel population consisted of 31 dogs of various ages: 22 Havanese (20 females, 2 males), 8 Beagles (7 females, 1 male), and 1 male German Shepherd. The clinical history and time of laboratory examinations are indicated on Table 1. After the diagnosis of B. canis abortion (index case) in a female (case 1), samples were obtained from every resident animal at a 1-month interval and submitted for laboratory examination. Important abortifacient agents other than Brucella species were ruled out by examination of the aborted fetuses and placentas (index case) for the presence of Chlamydiaceae, Neospora caninum, Toxoplasma gondii, Leptospira spp., and Coxiella burnetii by immunohistochemistry (IHC) and Canid herpesvirus 1 by polymerase chain reaction (PCR; data not shown).
Selective Brucella agar plates a containing 5% heat-inactivated horse serum b were inoculated directly with 2× 100 μl of anticoagulated (sodium citrate), nonlysed whole blood, as well as swabs, and 100 μl of homogenized organs (separate samples of uterus, testes, and epididymis, and individually pooled samples of liver-spleen and mesenteric-bronchial-sublingual lymph nodes). The plates were incubated at 37°C in 5% CO2 for 7 days. In addition, 1-ml aliquots of blood were cultured indirectly at 37°C for 21 days in trypticase soy broth c before 100 μl of inoculum being transferred to Brucella agar plates on days 7, 14, and 21 and cultured as described previously. Growth of Brucella spp. was checked daily. Routine biochemical and growth-based typing tests were performed on the isolates. 1,2 The utilization of 95 carbon sources was examined with a commercial test. d Slide agglutination tests were carried out using anti-Brucella polyclonal sera (against B. abortus) and antirough (anti-R) monospecific sera (against B. ovis) with single bacterial colonies. Sera were produced, and the examinations were conducted as described previously. 4
Clinical history and laboratory examinations in the affected dog breeding kennel. *

IHC = immunohistochemistry; PCR = polymerase chain reaction.
DNA was extracted with a commercial DNA isolation kit e from 1 distinct colony of each isolated strain according to the manufacturer's recommendations. Two novel PCR systems, the “Bruce-ladder” method 13 and single nucleotide polymorphism (SNP) typing, 6 were used for the molecular typing of the strains. All PCR reactions were performed in 25-μl total volume, containing 1 μl of target DNA, 5 μl of GoTaq buffer, f 3 μl of MgCl2 (25 mmol), g 1 μl of deoxyribonucleotide triphosphate (10 mmol), g 1 μl of each primer (6.25 pmol/μl: Bruce ladder; 10 pmol/μl: SNP typing), and 0.4 μl of GoTaq polymerase (5 U/μl). f The PCR assays consisted of steps as described previously. 6,7,13 Spleen and liver samples of 6 selected dogs (cases 1–6) were screened by PCR using 2 primer pairs (BMEI0535, BMEII0428) from the “Bruce-ladder” method 13 and a primer pair dedicated to the 16S–23S ribosomal DNA interspacer, 11 as described previously.
Serum samples were tested with a commercial rapid slide agglutination test (RSAT) before and after 2-mercapto-ethanol pretreatment (2ME-RSAT) h for the presence of B. canis–specific (R-type) antibodies, whereas the Rose Bengal test (RBT), i serum agglutination test (SAT), j and complement fixation test (CFT) k were used for the detection of smooth (S)-type antibodies.
After gross pathological examination, tissue samples were collected for histologic and IHC examination from the index case (placenta, fetal lung, liver, kidney, and spleen) and from cases 1–6 (heart, lung, liver, spleen, kidney, small and large intestine, testes, epididymis, ovaries, uterus, and mesenteric and sublingual lymph nodes). Four-micron thick sections of formalin-fixed and paraffin-embedded tissue samples were stained with hematoxylin and eosin, and examined by light microscopy. Hyperimmune rabbit serum was produced for IHC by the intravenous infection of 2 animals with 4 × 109 colony-forming units of live bacteria obtained from the isolated B. canis strains in accordance with all applicable institutional and national guidelines. This serum reacted with B. canis and B. ovis in a SAT, but it was negative with B. abortus and B. suis (data not shown). Deparaffinized sections were incubated overnight at 4°C with the hyperimmune serum diluted to 1:20,000, and antibody binding was detected by a horseradish peroxidase–labeled polymer. l Serial tissue sections from each case were rinsed with phosphate buffered solution. Tissue sections from an uninfected dog were used as negative controls.
Results are summarized on Table 2. Clinical symptoms of reproductive disorders were recognized in 10 of 31 (32%) animals during the course of the outbreak. Failure of conception occurred in 4 animals, whereas late-term abortions or premature litters were presented by 6 animals. Brucella canis was recovered from the blood culture and organs or vaginal samples only in a total of 3 of 31 (10%) animals (cases 1–3). Throat swabs remained negative in all cases. After 48 hr of incubation, the isolated colonies were small, translucent, yellow-colored, convex with an entire edge, and had a creamy consistency. The Gram-negative and modified acid-fast, small coccobacilli demonstrated oxidase, catalase, and urease activity, but failed to produce H2S. All isolates grew in the presence of 20 mg/ml thionin and basic fuchsin. The isolates could not be identified as either B. suis or B. canis by the traditional biochemical tests or the carbon-source utilization test. The strains showed rough colony morphology with crystal violet staining, weak agglutination with anti-Brucella polyclonal sera, and strong agglutination with anti-R monospecific sera. Isolates were misidentified as B. suis with the “Bruce-ladder” method in the first round of molecular examinations. However, subsequent SNP typing correctly identified all strains as B. canis. Brucella canis–specific antibodies were found in 7 of 31 (23%) cases. Four animals became seronegative 1 month after the first test, whereas only 1 animal became seropositive in the second test. The RBT, SAT, and CFT tests for anti-S antibodies yielded negative results.
Gross pathological changes were absent in the aborted fetuses, placenta (index case), and euthanized dogs. A moderate diffuse lymphohistiocytic periportal hepatitis was found microscopically in one of the fetuses. Severe multifocal coagulative necrosis accompanied with minimal-to-moderate infiltration by macrophages and a few neutrophil granulocytes were detected in the placenta (index case). Strong immunolabeling with the hyperimmune rabbit anti-B. canis serum was observed within almost every trophoblast cell in the placentas (Fig. 1), but immunolabeling was minimal or absent in areas of coagulative necrosis. Histologic examination of the selected dog cases revealed mild-to-severe multifocal lymphohistiocytic inflammation of multiple organs, including hepatitis accompanied by the necrosis of a few hepatocytes, meningitis (cases 1–4), interstitial pneumonia (cases 3–6), endometritis (cases 1, 5, 6), and interstitial mastitis (case 6). Bilateral diffuse lymphohistiocytic epididymitis (Fig. 2) and severe subacute-to-chronic prostatitis affecting several lobules (Fig. 2 inset) were detected in a stud dog (case 3). Lymphoid hyperplasia was found in tonsils and lymph nodes of each case, whereas scant multinucleated giant cells were detected in lymph nodes of case 1. Immunolabeling was only found in a few macrophages and giant cells within the tonsils and lymph nodes of case 1 (Fig. 1 inset). Immunostaining was absent in canine fetal organs, of cases 2–6, and in the negative control sections.
Summary of the results of serologic, bacteriological, immunohistochemical, histologic, and polymerase chain reaction examinations of dogs with positive laboratory findings. *
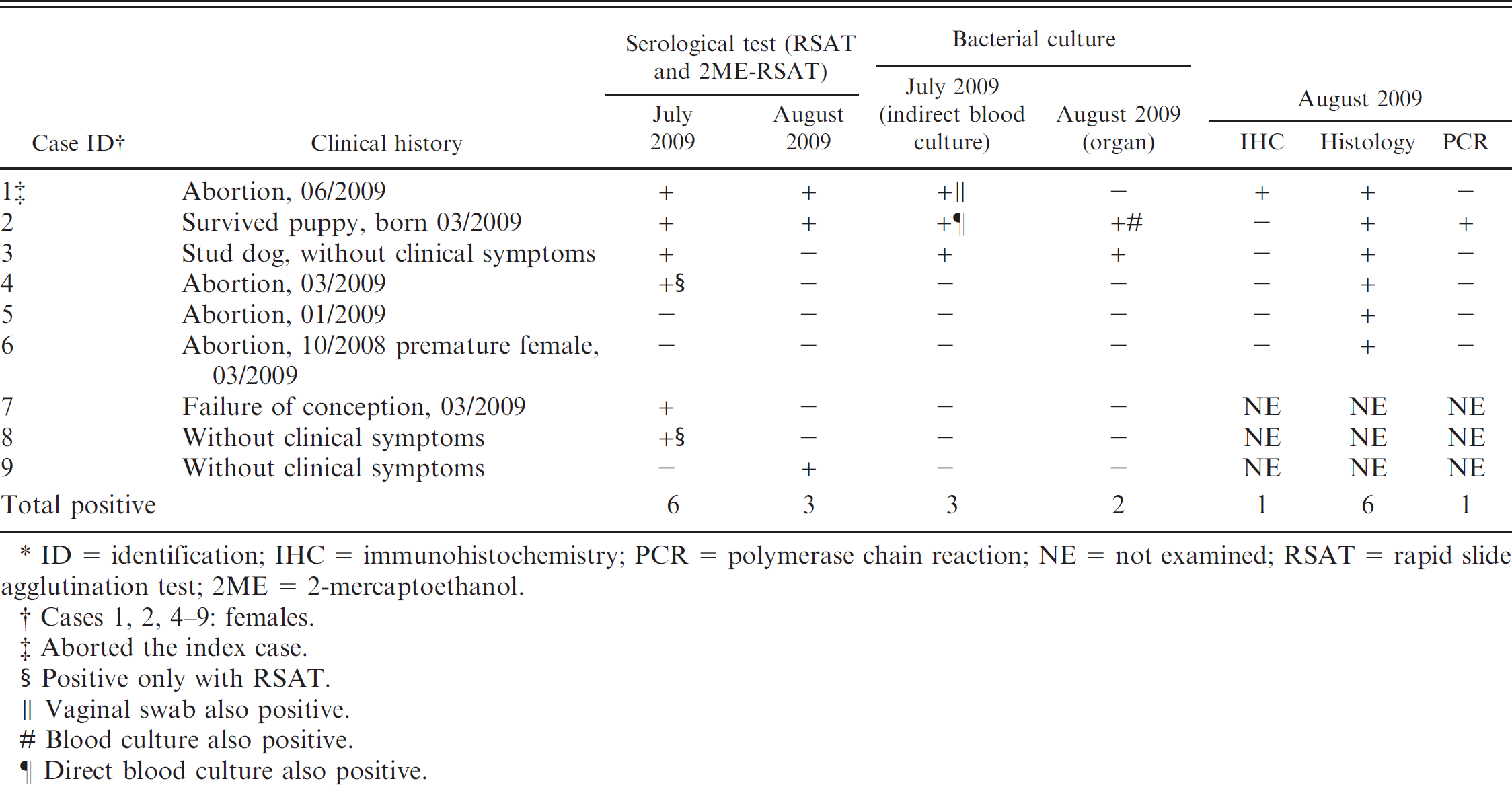
ID = identification; IHC = immunohistochemistry; PCR = polymerase chain reaction; NE = not examined; RSAT = rapid slide agglutination test; 2ME = 2-mercaptoethanol.
Cases 1, 2, 4–9: females.
Aborted the index case.
Positive only with RSAT.
Vaginal swab also positive.
Blood culture also positive.
Direct blood culture also positive.
It seems most likely that the infection was introduced to the affected kennel by an imported pregnant female (case 6). The abortion of this imported animal was the first obvious reproductive disorder. Sporadic reproductive clinical symptoms were detected by the breeder during the 8 months preceding the first laboratory examination. Similar, long-lasting (1 year) reproductive clinical symptoms were reported from a Canadian kennel in the clinical history of a recently published canine brucellosis case. 3
In the present study, the highest number of B. canis–infected animals was detected with RSAT. Subclinical infection was found in 4 of 7 (57%) cases, which is compatible with the results of other studies. 10,16 Because detectable antibodies appear only 4–8 weeks after infection and can be absent in chronically infected dogs, a significant number of cases may remain negative with RSAT. 11 Even a culture-positive stud dog (case 3) was found to be seronegative in the current study. These results also support previous findings, indicating that increasing specificity due to 2ME pretreatment could decrease the sensitivity of serologic testing. 11 The ratio of seropositive animals was rather low (23%) in the current study, which is in contrast with the result of a recent publication about canine brucellosis, where 61% of the animals were seropositive in the affected kennel. 3 The indirect fluorescence antibody test used in that study may be more sensitive compared with the commercial RSAT used in the current study.
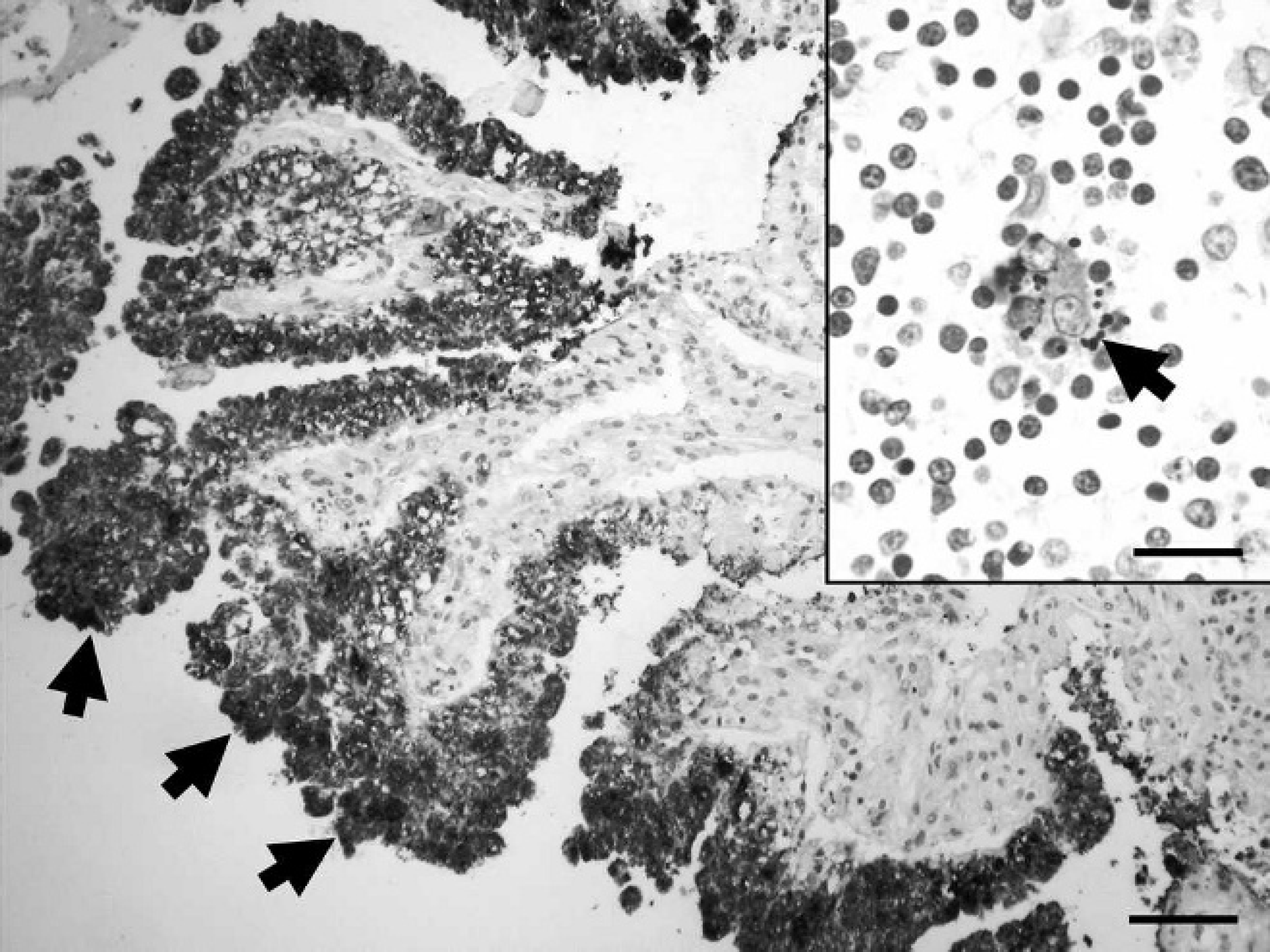
Aborted canine placenta. Strong immunolabeling in trophoblast cells (arrows) indicates the presence of Brucella canis. Immunohistochemistry using hyperimmune rabbit anti–B. canis primary antibodies, hematoxylin counterstain. Bar = 150 μm. Inset: Lymph node, aborted female, case 1. Dot-shaped immunolabeling in the cytoplasm of a giant cell (arrow). Immunohistochemistry using hyperimmune rabbit anti–B. canis primary antibodies, hematoxylin counterstain. Bar = 40 μm.
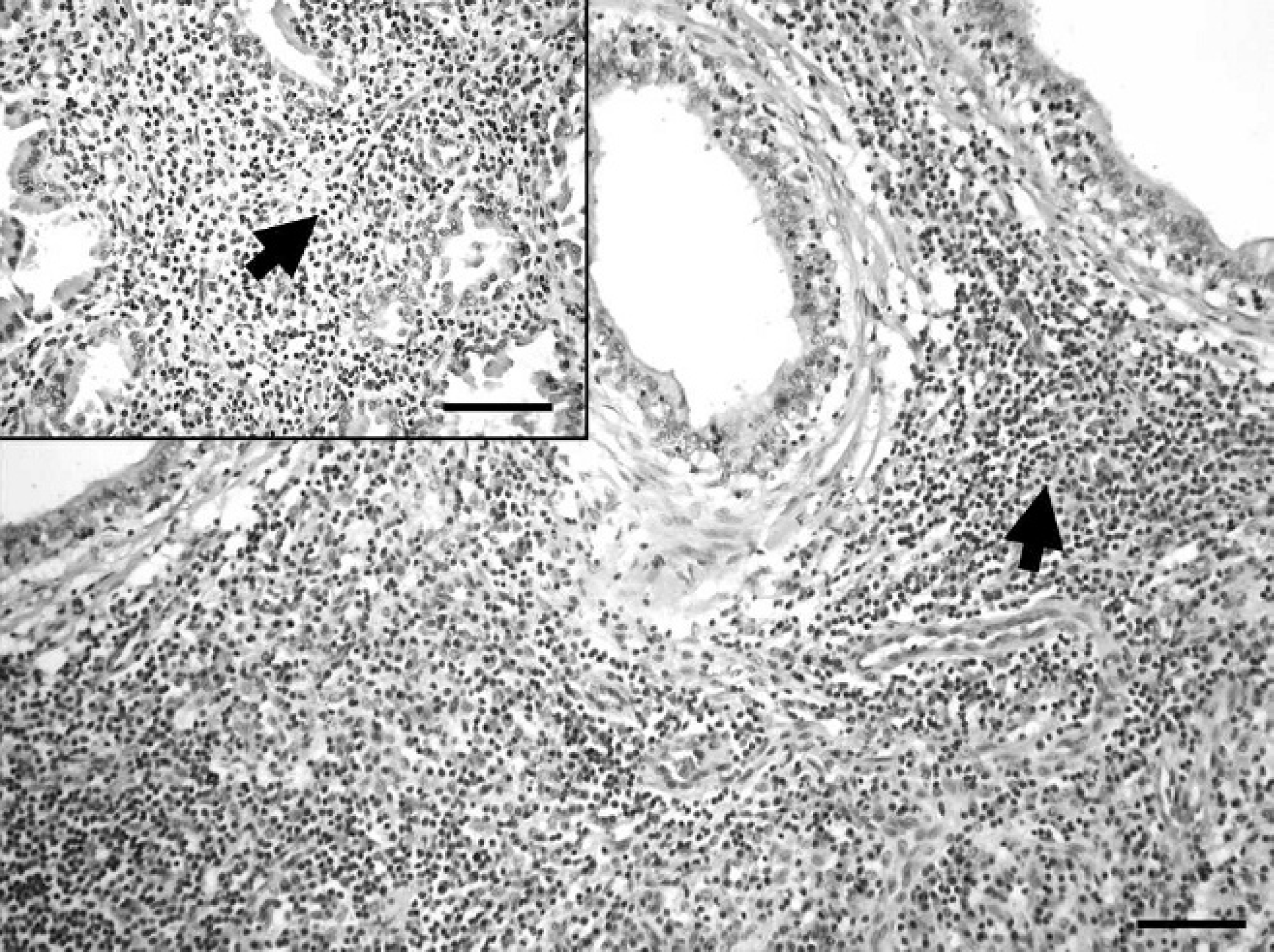
Canine epididymis, case 3. Severe lymphohistiocytic inflammation (arrow). Hematoxylin and eosin. Bar = 200 μm. Inset: Canine prostata, case 3. Severe lymphohistiocytic inflammation (arrow). Hematoxylin and eosin. Bar = 200 μm.
The serologic status of every examined case changed over time, with 2 exceptions. One of these was a recently aborted female (case 1) and the other was a 5-month-old puppy (case 2). The puppy was the sole survivor from a stillborn litter, and it was the only animal presenting persistent bacteriemia. In this case, the infection may have been contracted during or after delivery, when large numbers of B. canis are excreted in uterine fluids. 10 This case highlights the risk of keeping and breeding puppies born in an infected kennel, because these animals may serve as a permanent source of infection for both human beings and dogs. Less than half of the seropositive animals were found to be positive by bacteriology. Intermittent bacteriemia can be frequently found in dogs infected with B. canis, 10,16 and it is often responsible for negative culture results also observed in the present study.
Similarly to a previous study, 3 examination of colony morphology was the only test found that was capable of differentiating B. suis from B. canis from the available biochemical and growth-based tests. In the past, B. canis was considered a biotype of B. suis, 15 and there is a strong genetic relationship between them, 5 which can explain the failure of the “Bruce-ladder” method to distinguish between them. The recently published SNP typing test overcame this failure. 12 The direct detection of Brucella spp. from organs by PCR was unsuccessful in the majority of cases. According to these results, a more sensitive PCR protocol should be taken into consideration.
The histologic lesions and IHC results observed in the current study in aborted fetal membranes and in adult dogs were similar to previous reports. 3,8–10 Characteristic histologic lesions were found in all 6 examined animals. Even 2 females (cases 5, 6), who aborted or presented premature birth several months earlier, were positive by histology, whereas all other laboratory examinations remained negative. In accordance with the observation of a previous report, 3 the histologic lesions were not associated with clinical signs of disease in most of the cases of the present study. Because there is no commercially available B. canis–specific antibody applicable in an IHC test, a hyperimmune rabbit serum against the isolated B. canis strain was produced. The presence of B. canis under the detection limit of IHC could explain that all fetal and the majority of adult dog organs presented negative results.
In conclusion, the clinical signs of canine brucellosis in the present case were not serious, and they were confined to sporadic reproductive disorders. This highlights the importance of reminding breeders of the clinical signs of canine brucellosis and their responsibility to prevent intrapopulation and interpopulation spread of the disease and possible human infections. Surviving puppies can be an important source of B. canis infection, as they can become permanent carriers and shedders of the pathogen. All cases of canine abortion should be examined for brucellosis by bacterial culture of the fetuses and placentas. Examination of aborted placentas with IHC is also recommended because it is a quick and sensitive technique compared with bacteriology. Multiple methods (i.e., serology, blood, and genital bacteriology) should be applied simultaneously and repeatedly for the reliable screening of B. canis infection in live individuals.
Footnotes
a.
Oxoid Ltd., Cambridge, United Kingdom.
b.
Invitrogen Corp., Carlsbad, CA.
c.
Difco, BD Diagnostic Systems, Sparks, MD.
d.
MicroLog™ MicroStation™ System, GN2 Microplates; Biolog Inc., Hayward, CA.
e.
QIAamp DNA Mini Kit, QIAquick Gel Extraction Kit; Qiagen Inc., Valencia, CA.
f.
Promega Corp., Madison, WI.
g.
Fermentas Inc., Burlington, Canada.
h.
D-Tec CB, Synbiotics Corp., San Diego, CA.
i.
Vircell S.L., Granada, Spain.
j.
Veterinary Laboratories Agency, Weybridge, United Kingdom.
k.
Institute Virion/Serion GmbH, Würzburg, Germany.
l.
EnVision™+ Kit, Dako Denmark A/S, Glostrup, Denmark.
