Abstract
Expression of vascular endothelial growth factor (VEGF), its receptors (flt-1 and flk-1), and basic fibroblast growth factor (bFGF) in canine hemangiosarcoma (HSA) and hemangiomas was investigated by immunohistochemical analysis. In addition, expression of the mRNAs of VEGF, flt-1, flk-1, and flg-1 (a receptor for bFGF), was analyzed by reverse transcriptase polymerase chain reaction (RT-PCR) and in situ hybridization (ISH) with cRNA probes. VEGF, bFGF, flt-1, and flk-1 were immunohistochemically detected in the neoplastic cells in HSAs; the staining intensity was stronger in HSAs than in hemangiomas. On the other hand, the neoplastic cells in hemangiomas exhibited very weak or no expression of VEGF, although they showed moderate expression of flt-1 and flk-1. The mRNAs of VEGF, flt-1, flk-1, and flg-1 were detected in the neoplastic cells in HSAs by ISH and RT-PCR. However, VEGF mRNA was not detectable in the neoplastic cells in hemangiomas by ISH, although it was detected in the inflammatory cells in the tumors by RT-PCR. Moreover, the HSAs that showed intense staining for flk-1 had a high proliferative activity, which was reflected as a high Ki-67 positive index. These results suggest that the expression of the growth factors and their receptors, especially flk-1, might be associated with the malignant proliferation of HSAs.
Growth factors are an important group of extracellular stimuli because they play essential roles in the regulation of cell proliferation and growth. Most known growth factors are either polypeptides or glycoproteins; they are mitogenic and induce cell division. The actions of growth factors are mediated by receptors present on their target cells. 3, 28 Generally in normal tissues the concentrations of growth factors are minimal; 9 however, their expression is reinduced during a pathologic change. 4 For example, angiogenesis plays a critical role in important pathologic processes such as wound healing and inflammation and is controlled by angiogenesis-stimulating growth factors. These factors are involved in endothelial cell proliferation, differentiation, and migration, as well as vessel formation. In addition, vascular endothelial growth factor (VEGF) has been found to be associated with the proliferative phase in human infantile hemangioma. 2
Hemangiosarcoma (HSA) is a malignant neoplasm of vascular endothelial cells. Canine HSA most commonly arises in the spleen, right atrium, or subcutis. Local infiltration and systemic metastases are the common growth patterns. Various therapeutic regimens have been recommended for dogs with HSA; however, the median survival time is slightly more than 6 months. 5, 11, 19, 25 The mechanism of abnormal growth of HSA is unclear. A recent study suggests the role of VEGF and its receptors in the paracrine or autocrine mechanism of proliferation in human angiosarcoma. 12 Further, recent findings have shown that basic fibroblast growth factor (bFGF) and its receptor play an important role in the growth and progression of human angiosarcoma. 29
In this study, we investigated the expression of VEGF, bFGF, and their receptors to determine their involvement in canine vascular tumors by immunohistochemical analysis and by detection of their mRNAs with in situ hybridization (ISH).
Materials and Methods
Samples and classification of canine vascular neoplasms
A total of 73 HSAs (66 samples were primary lesions, 3 samples were primary lesions and metastatic lesions, and 1 sample was only metastatic lesion) and 15 hemangiomas were examined. In addition, 3 granulation tissues were investigated as proangiogenic endothelial in an active phase of angiogenesis. All samples were collected between 1998 and 2005 from the Veterinary Teaching Hospital of Gifu University and private animal hospitals. The samples were removed surgically, and snap frozen samples were collected from 8 of the 73 HSAs and 3 of the 15 hemangiomas for reverse transcriptase polymerase chain reaction (RT-PCR) and stored at -80°C until use. The samples were immediately fixed in 10% neutral buffered formalin and embedded in paraffin. Tissue sections were stained with hematoxylin and eosin (HE). The diagnosis of HSA or hemangioma was confirmed by review of slides stained with HE. Subsequently, serial sections of all putative vascular neoplasms were confirmed based on the expression of von Willebrand factor by immunostaining using a specific antibody (anti-von Willebrand factor rabbit antibody; Dako Cytomation, Glostrup, Denmark). The HSAs were classified into 2 types (well differentiated or poorly differentiated) based on the degree of differentiation of the tumor cells, as described in a previous study. 22
Immunohistochemical analysis of VEGF, bFGF, flt-1, and flk-1
Immunohistochemical staining was performed for VEGF, flt-1, and flk-1 in all samples and for bFGF in 41 samples. Anti-VEGF antibody (Santa Cruz Biotechnology, Santa Cruz, CA) is a mouse affinity-purified monoclonal IgG2a antibody corresponding to amino acids 1–140 of human VEGF. Anti-flt-1 antibody (Santa Cruz Biotechnology) is a rabbit affinity-purified polyclonal antibody raised against a peptide mapped at the carboxy terminal of human flt-1. Anti-flt-1 antibody (Santa Cruz Biotechnology) is a mouse monoclonal IgG1 antibody against a carboxy-terminal peptide (amino acids 1158–1345) of mouse flk-1. Anti-bFGF antibody (Santa Cruz Biotechnology) is a rabbit polyclonal IgG antibody raised against an epitope mapped within an internal region of human FGF-2.
Sections with a thickness of 3 μm were deparaffinized in xylene and rehydrated in graded ethanol. For antigen retrieval of flt-1 and flk-1, the sections were immersed in Target Retrieval Solution (Dako Cytomation) and heated for 15 minutes at 120°C in an autoclave. For demasking of VEGF and bFGF, the sections were digested with 0.1% trypsin in Tris-HCl buffer (pH 7.0) for 15 minutes at 37°C. Endogenous peroxidase was quenched with 0.3% hydrogen peroxidase in methanol for 30 minutes at room temperature. To prevent the binding of nonspecific proteins to the primary antibody, the sections were treated with Protein Block Serum-Free (Dako Cytomation); they were then incubated with the primary antibody overnight at 4°C. For the negative control, the primary antibody was omitted and replaced with phosphate-buffered saline (PBS). The sections were then washed with PBS containing 0.1% Tween 20 (Sigma-Aldrich, St. Louis, MO) and incubated for 30 minutes at room temperature with the appropriate secondary antibodies (EnVision™+ System HRP; Dako Cytomation). The binding was detected with diaminobenzidine and 0.02% hydrogen peroxidase in 0.1 M Tris-HCl (pH 7.0). Finally the sections were washed in distilled water and counterstained with Mayer's hematoxylin.
For evaluation of the cell growth fraction, immunolabeling of Ki-67 was performed as described by Sakai et al (2001). 20
Scoring and grouping of the immunohistochemical results
The immunohistochemical staining results were divided into the following 4 categories based on the total percentage of neoplastic cells staining positively and the staining intensity: - = nonstained neoplastic cells; ± = neoplastic cells with weak to intermediate staining; + = <50% neoplastic cells with intense staining; and ++ = >50% neoplastic cells with intense staining. Further, the immunohistochemical staining results for VEGF, flt-1, and flk-1 were grouped into 2 categories (strong [++] and weak [-, ±, and +]). The Ki-67-positive cells were counted in a total of 1,000 cells (from 10 high-power fields) to obtain the Ki-67 positive index (Ki-67 PI).
Detection of mRNA expression by RT-PCR
Using the RNeasy kit as per the manufacturer's protocol (Qiagen, Valencia, CA), total RNA was extracted from 8 frozen HSA samples, 3 frozen hemangioma samples, and 1 frozen sample of a normal spleen, which was located adjacent to the neoplasm and was confirmed as nonneoplastic by histopathology. Then cDNA was synthesized using ThermoScript reverse transcriptase (Invitrogen, Carlsbad, CA). The PCR was carried out in a Takara PCR thermal cycler MP (Takara, Ohtsu, Japan). For all the amplifications, the PCR mixture was composed of 1X PCR reaction buffer (15 mM Tris-HCl [pH 8.0] and 50 mM KCl), 0.5 μM of each primer pair, 250 μM of each dNTP, and 1.25 U AmpliTaq Gold DNA polymerase (Applied Biosystems, Foster City, CA). The MgCl2 concentration in the reaction mixtures was 3 mM for VEGF, 2.5 mM for flt-1, and 1.5 mM for flk-1, flg-1, and glyceraldehyde-3-phosphate dehydrogenase (GAPDH). After activating the AmpliTaq Gold, the amplifications were performed under the following conditions: denaturing (15 seconds at 95°C), annealing (30 seconds), and extension (1 minute at 72°C) for 40 cycles, except in the case of flg-1 (35 cycles). The annealing temperatures used were 55°C for VEGF, 62°C for flt-1 and flk-1, and 58°C for flg-1. 4, 21 The expression of GAPDH was used as an internal control for RNA integrity. The cDNA encoding GAPDH was amplified for 30 cycles (15 seconds at 94°C, 30 seconds at 55°C, and 1 minute at 72°C) using the primers described previously by Grone et al. 10 The PCR primer sets and products sizes are shown in Table 1. All primers were canine-specific sequences, except in the case of flg-1, for which a human-specific sequence was used. The products of the PCR amplifications were analyzed on a 1.5% agarose gel stained with ethidium bromide and visualized under ultraviolet light.
Primer sets for RT-PCR amplification and generation of template for cRNA probes.

Italics indicate SP6, T7, or T3 core promoter sequences.
GAPDH = glyceraldehyde-3-phosphate dehydrogenase; RT-PCR = reverse transcriptase polymerase chain reaction; VEGF = vascular endothelial growth factor.
Preparation of cRNA probes for in situ hybridization
For hybridization probes, we generated amplicons from the cDNA of frozen normal canine spleens with SP6, T3, or T7 RNA polymerase that recognizes sequence-adapted primers. 13 The sequences of the adapted primers are shown in Table 1. PCR amplifications were carried out in a Takara PCR thermal cycler MP with AmpliTaq Gold DNA polymerase. The amplification conditions are shown in Table 2. Approximately 0.5 μg of amplified DNA (Fig. 1) was used as the template. Antisense and sense digoxigenin-labeled riboprobes were generated by in vitro transcription using a DIG RNA labeling kit (Roche Diagnostics, Mannheim, Germany) as per the manufacturer's protocol.
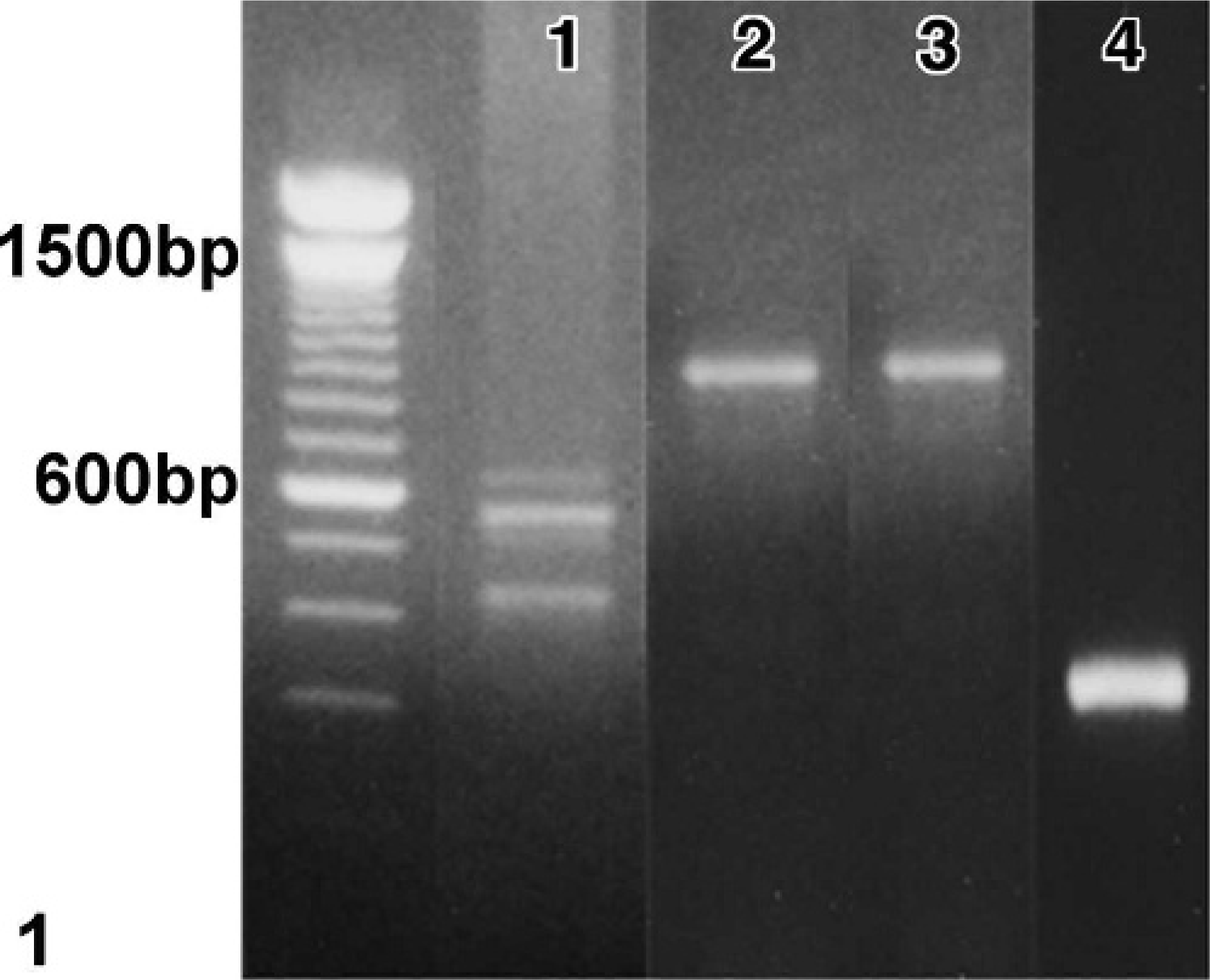
Normal spleen, canine. Amplified DNA used as a template for digoxigenin-labeled riboprobes. Lane 1, vascular endothelial growth factor (VEGF); 2, flt-1; 3, flk-1; 4, flg-1. Lane on the extreme left, molecular size markers. The predicted sizes of the amplification products were 481, 553, 613, and 685 bp for VEGF and 1,036 bp for flt-1, 1,015 bp for flk-1, and 327 bp for flg-1.
Amplification conditions for generation of template for cRNA probes.

VEGF = vascular endothelial growth factor.
In situ hybridization
We selected 4 HSA samples, 1 hemangioma sample, and 1 granulation tissue sample for further evaluation of mRNA expression by performing ISH on the formalin-fixed paraffin-embedded sections. After the tissue sections were deparaffinized and rehydrated, the basic proteins were removed by incubation in 0.2 N HCl for 20 minutes. For the detection of VEGF, flk-1, and flg-1 mRNAs, the sections were then digested with 10 μg/ml proteinase K (Takara) for 10 minutes at room temperature; for the detection of flt-1 mRNA, the sections were digested with 500 μg/ml pepsin (Wako Pure Chemical Industries, Osaka, Japan) for 30 minutes at 37°C. The slides were dehydrated by sequential immersion in 95% and 100% ethanol and were air dried. They were then hybridized with 2 μg/ml antisense or sense probe in DIG Easy Hyb Granules (Roche Diagnostics) for 2 hours at 42°C. After hybridization, the slides were washed in 2X SSC for 20 minutes at 50°C. After washing, the sections were subjected to immunohistochemical analysis for the detection of the hybridized probes by using an alkaline phosphatase–conjugated anti-digoxigenin antibody (Roche Diagnostics). The alkaline phosphatase reaction was visualized with 5-bromo-4-chloro-3-indolyl phosphate, nitroblue tetrazolium, and iodonitrotetrazolium (Dako Cytomation). Subsequently, the sections were counterstained with Mayer's hematoxylin. The brown color indicated positive staining.
Statistical analysis
Significant differences between the histopathologic classification and the expression of VEGF, flt-1, or flk-1 in the neoplastic cells were analyzed by Fisher's exact test; those between the Ki-67 PI and the histopathologic classification or the expression of VEGF, flt-1, or flk-1 in the neoplastic cells were analyzed by the Mann-Whitney U test. A P < 0.05 level of confidence was considered significant.
Results
Breed, sex, and age of animals and sites of hemangiosarcoma and hemangioma
The canine HSA study group comprised 27 females, 40 males, and 3 animals of unrecorded gender with an average age of 9.7 years (range, 2–15 years). The most common breeds were Golden Retrievers (n = 17), Siberian Huskies (n = 8), Labrador Retrievers (n = 5), Shibas (n = 5), Shetland Sheepdogs (n = 4), and Beagles (n = 4). The primary location of the HSA in the 73 dogs was the spleen (n = 44), skin (n = 17), liver (n = 2), kidney (n = 2), and nictitans, auricle of the right atrium, oral mucosa, or left femora (n = 1 each). The hemangioma study group comprised 5 females, 8 males, and 2 animals of unrecorded gender with an average age of 8.1 years (range, 3–16 years). Eight different breeds were represented: Shetland Sheepdogs (n = 4) and Golden Retrievers (n = 2) were the most common. The locations of the hemangioma in the 15 dogs included the skin (n = 13), and nictitans or external ear (n = 1 each).
Histopathology
Based on the histopathology, the morphology of the neoplastic endothelial cells was variable and ranged from a spindle shape to polygonal to ovoid; the growth pattern was recognizable as vascular clefts or channels in the tumor. In some areas, an acellular and brightly eosinophilic stroma was observed between the clefts or channels. On the other hand, there were solid areas that were indistinguishable from a fibrosarcoma or other poorly differentiated sarcomas. In this study, we considered the sarcomas to be of a well-differentiated type when the cells had a fairly uniform morphology and were arranged in cords and vascular channels (Fig. 2). Neoplasms were considered to be of the poorly differentiated type when the cells were pleomorphic and formed clefts or solid sheets rather than channels (Fig. 3a). 22 In the morphologic classification of the HSAs, 43 samples were classified as well differentiated and 30 samples as poorly differentiated. The hemangiomas were well circumscribed and composed of variably sized vascular spaces filled with blood and lined by a single layer of uniform endothelial cells (Fig. 4). The von Willebrand factor was detected by immunohistochemical analysis in all the samples (Fig. 3b).
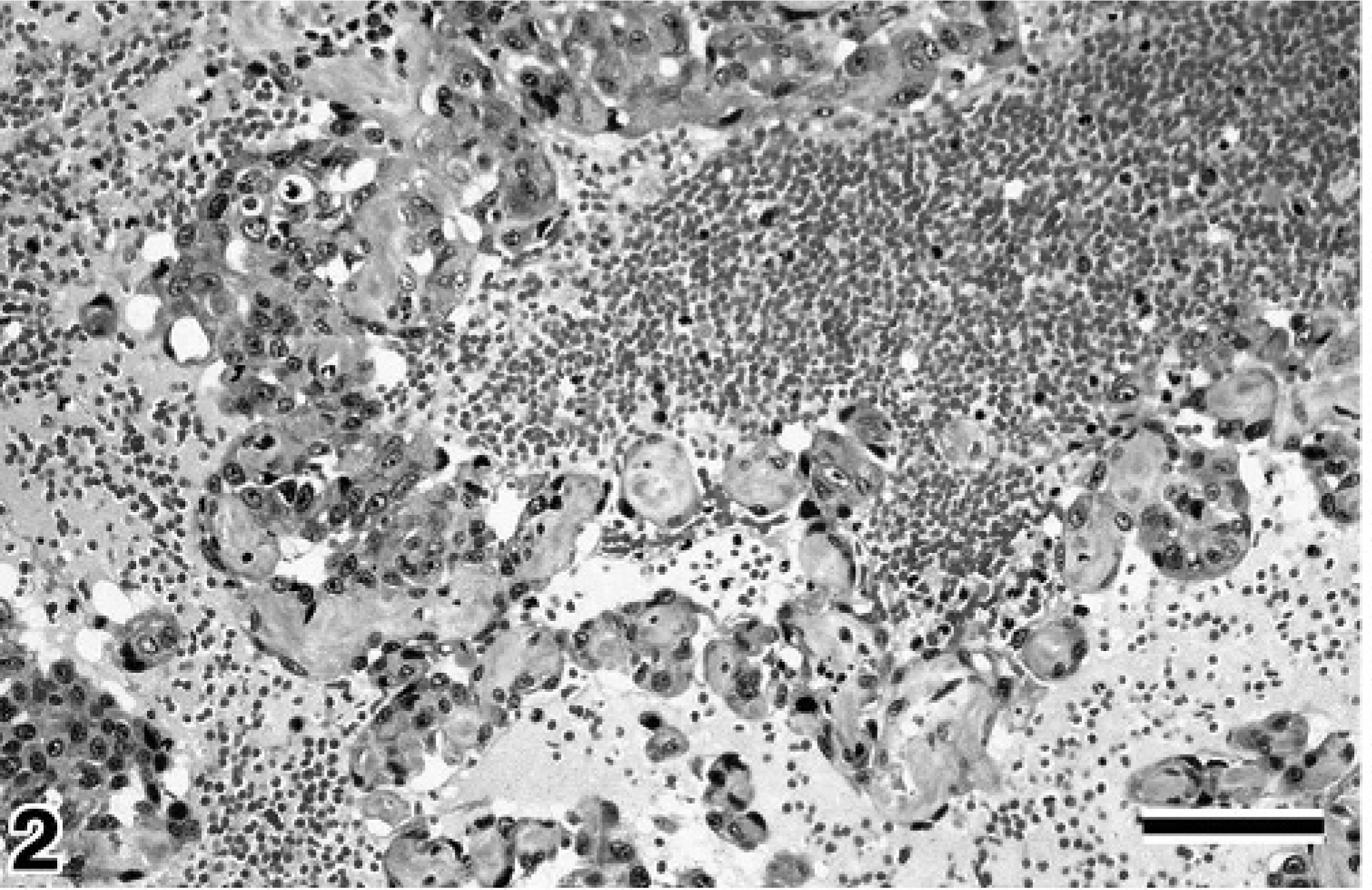
Splenic mass, canine. Representative of hemangiosarcoma, well-differentiated type. Irregularly shaped and sized vessels with plump endothelial cells lining and filling trabeculae between lumens. HE. Bar = 50 μm.
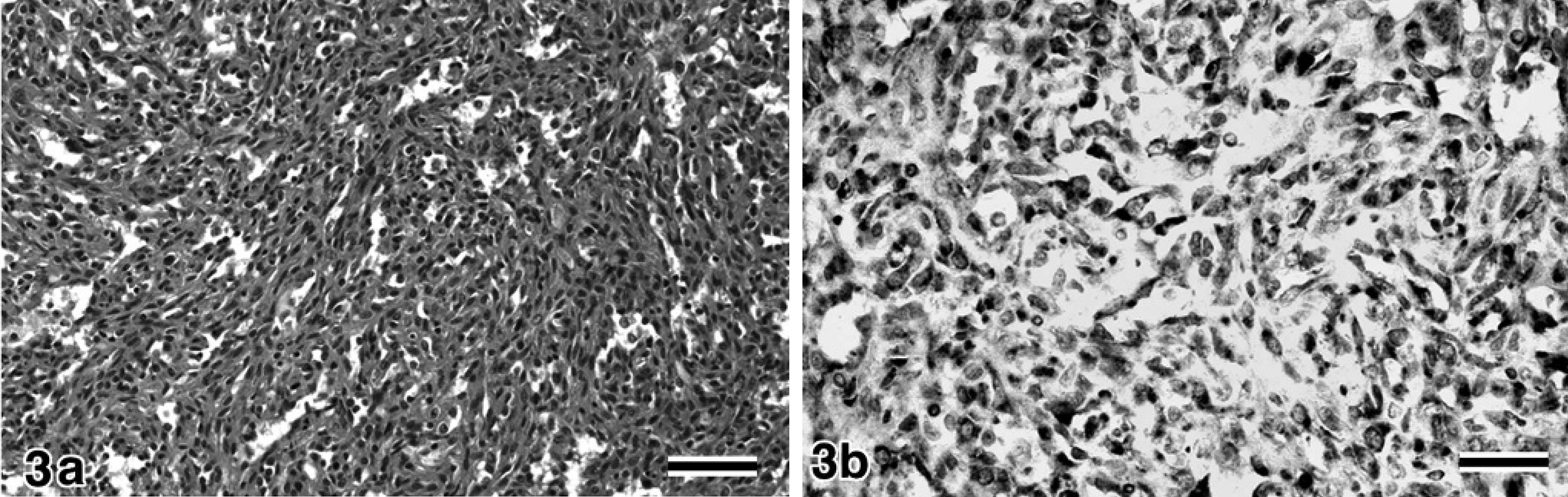
Spleen, canine. Representative of hemangiosarcoma, poorly differentiated type.
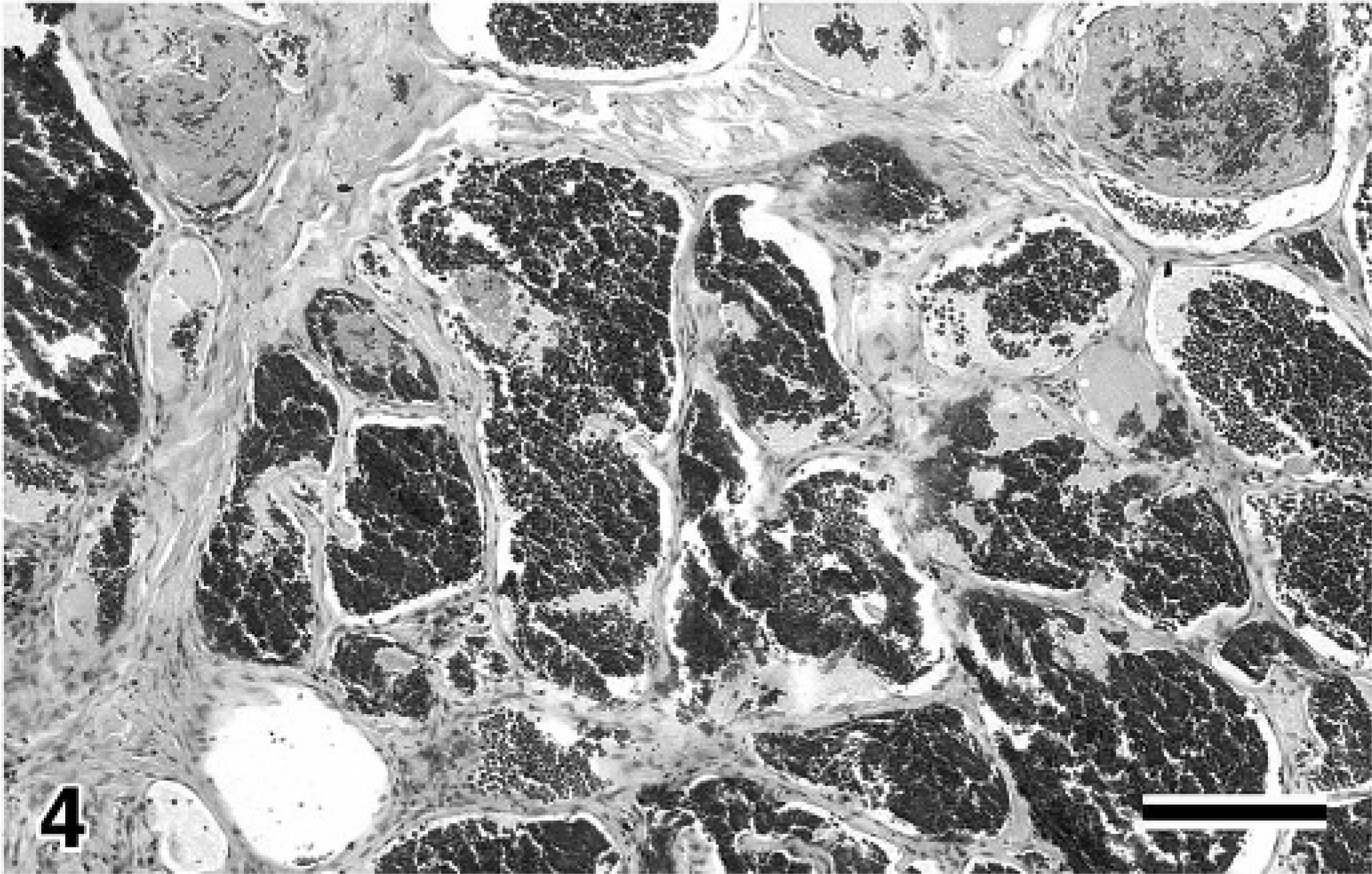
Skin, canine. Representative of hemangioma. Uniformly sized vessels containing red blood cells and lined by inconspicuous endothelial cells. HE. Bar = 150 μm.
Immunohistochemical analysis and in situ hybridization
In the granulation tissue, intense staining of VEGF, flt-1, flk-1, and bFGF was detected in the cytoplasm of newly formed vascular endothelial cells, fibroblasts, and inflammatory cells, including macrophages and lymphocytes but not neutrophils (Fig. 5).
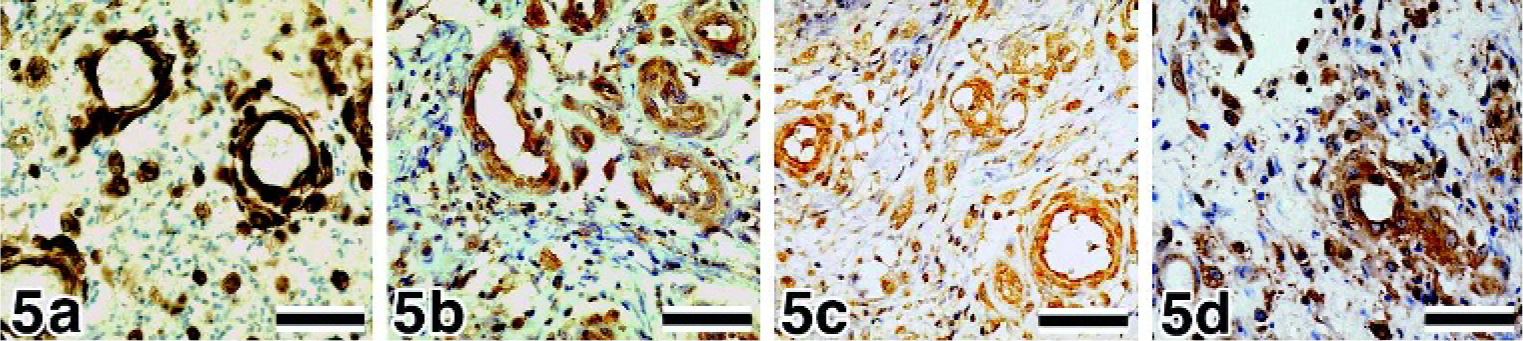
Skin, canine, granulation tissue.
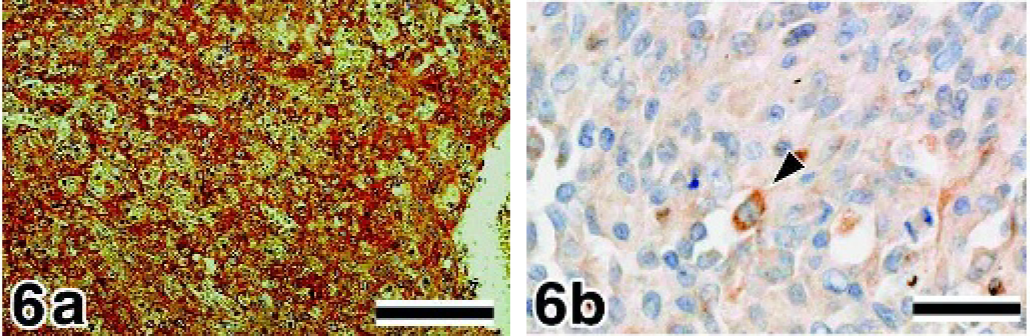
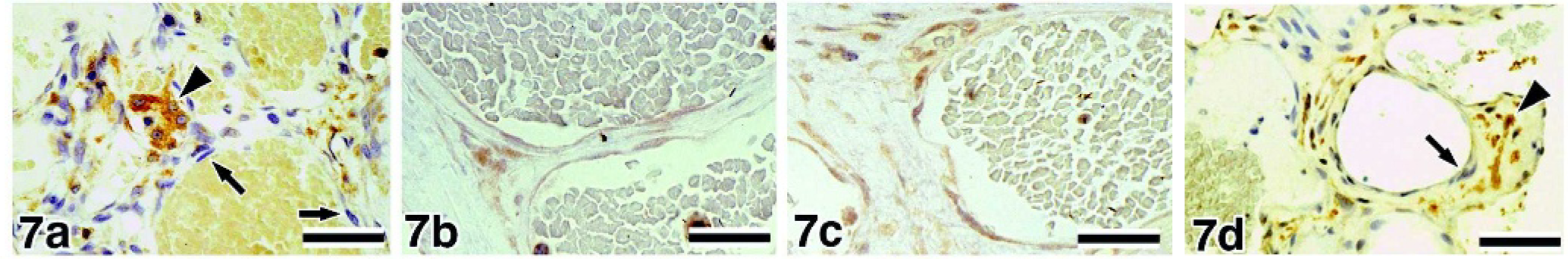
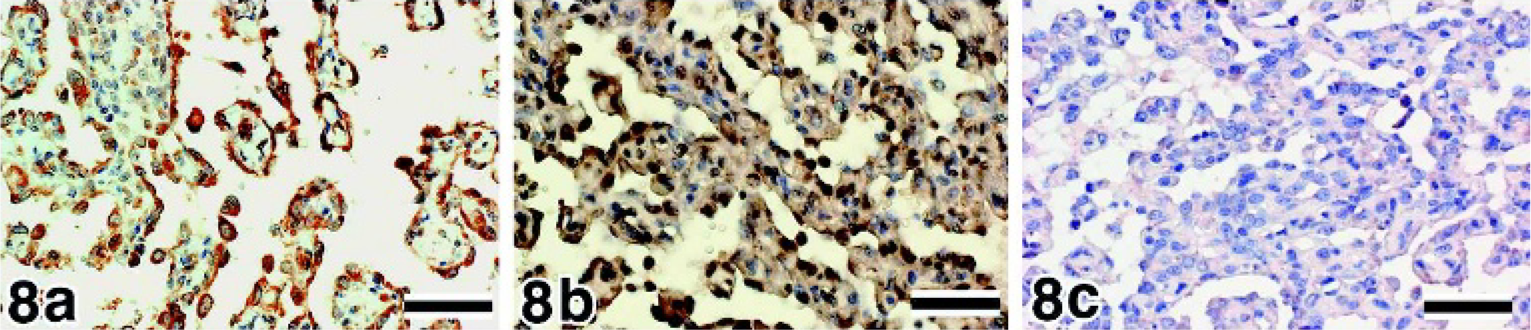
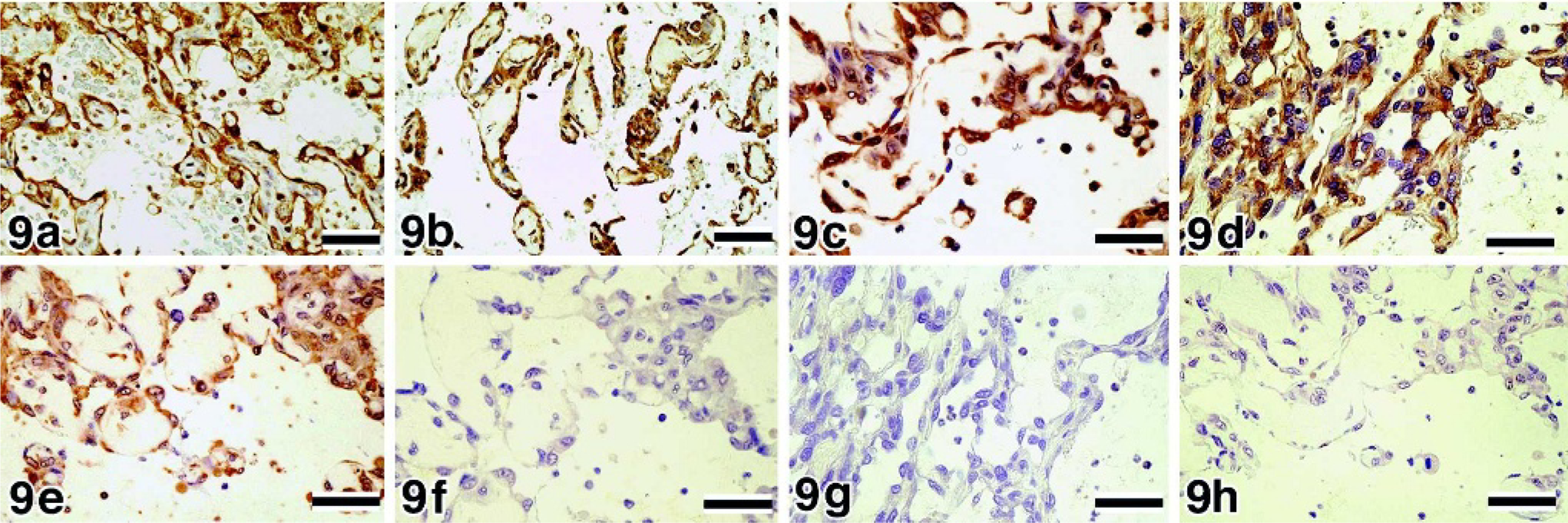
The neoplastic cells of the HSAs and hemangiomas revealed cytoplasmic immunoreactivity of VEGF, flt-1, flk-1, and bFGF (Figs. 6, 7, 8a, 9a, and 9b). As observed in the granulation tissue, intense staining of VEGF, flt-1, flk-1, and bFGF was detected in the fibroblasts and inflammatory cells that had infiltrated into the tumor. The immunoreactivities of VEGF, flt-1, flk-1, and bFGF in the HSAs and hemangiomas are summarized in Table 3.
Spleen, canine, hemangiosarcoma, poorly differentiated type.
Skin, canine, hemangioma.
Spleen, canine, hemangiosarcoma, well-differentiated type.
Spleen, canine, hemangiosarcoma, well-differentiated type.
Expression of VEGF, bFGF, flt-1, and flk-1 in HSA and hemangioma.
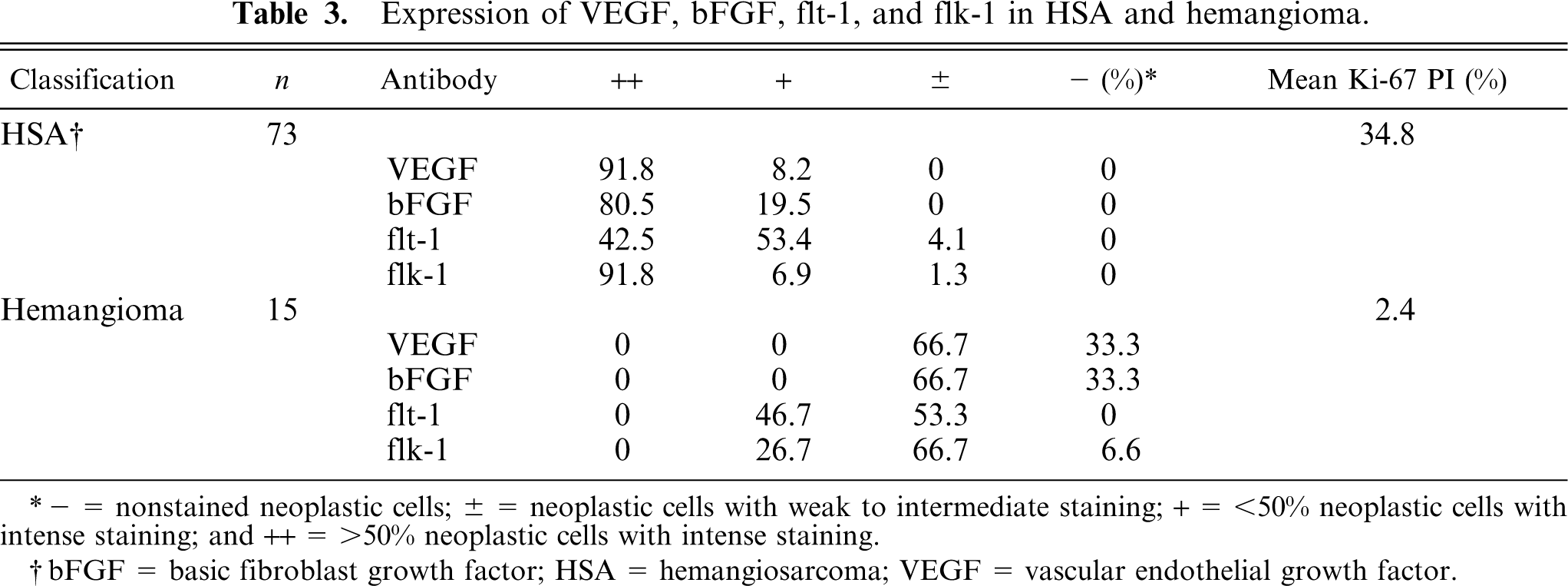
− = nonstained neoplastic cells; ± = neoplastic cells with weak to intermediate staining; + = <50% neoplastic cells with intense staining; and ++ = >50% neoplastic cells with intense staining.
bFGF = basic fibroblast growth factor; HSA = hemangiosarcoma; VEGF = vascular endothelial growth factor.
In the immunostaining of VEGF, 67 of the 73 samples showed ++ immunoreactivity (Fig. 6a), and 6 HSA samples showed + immunoreactivity; there was no weakly (±) or negatively (-) stained sample. On the other hand, all hemangioma samples clearly exhibited a lower number of positive tumor cells and less intense staining (± or -) of VEGF (Fig. 7a). In the immunostaining of flt-1, 31 of the 73 HSA samples showed ++ immunoreactivity (Fig. 8a), 39 samples showed + immunoreactivity, and 3 samples showed ± immunoreactivity (Fig. 6b). The neoplastic cells in the hemangioma samples showed + or ± immunoreactivity (Fig. 7b); none of the samples showed intense staining. In the immunostaining of flk-1, all HSA samples showed positive staining; 67 of the 73 samples showed ++ immunoreactivity (Fig. 9a), 5 samples showed + immunoreactivity, and 1 sample showed ± immunoreactivity. None of the HSA samples showed negative staining. In the hemangioma samples, 26.7% samples showed + immunoreactivity of flk-1, and 66.7% and 6.6% samples showed ± and - immunoreactivity, respectively (Fig. 7c). In the immunostaining of bFGF, 80.5% and 19.5% HSA samples showed ++ and + immunoreactivity, respectively (Fig. 9b). In hemangiomas, the immunoreactivity of bFGF was ± in 66.7% of samples and - in 33.3% of samples (Fig. 7d).
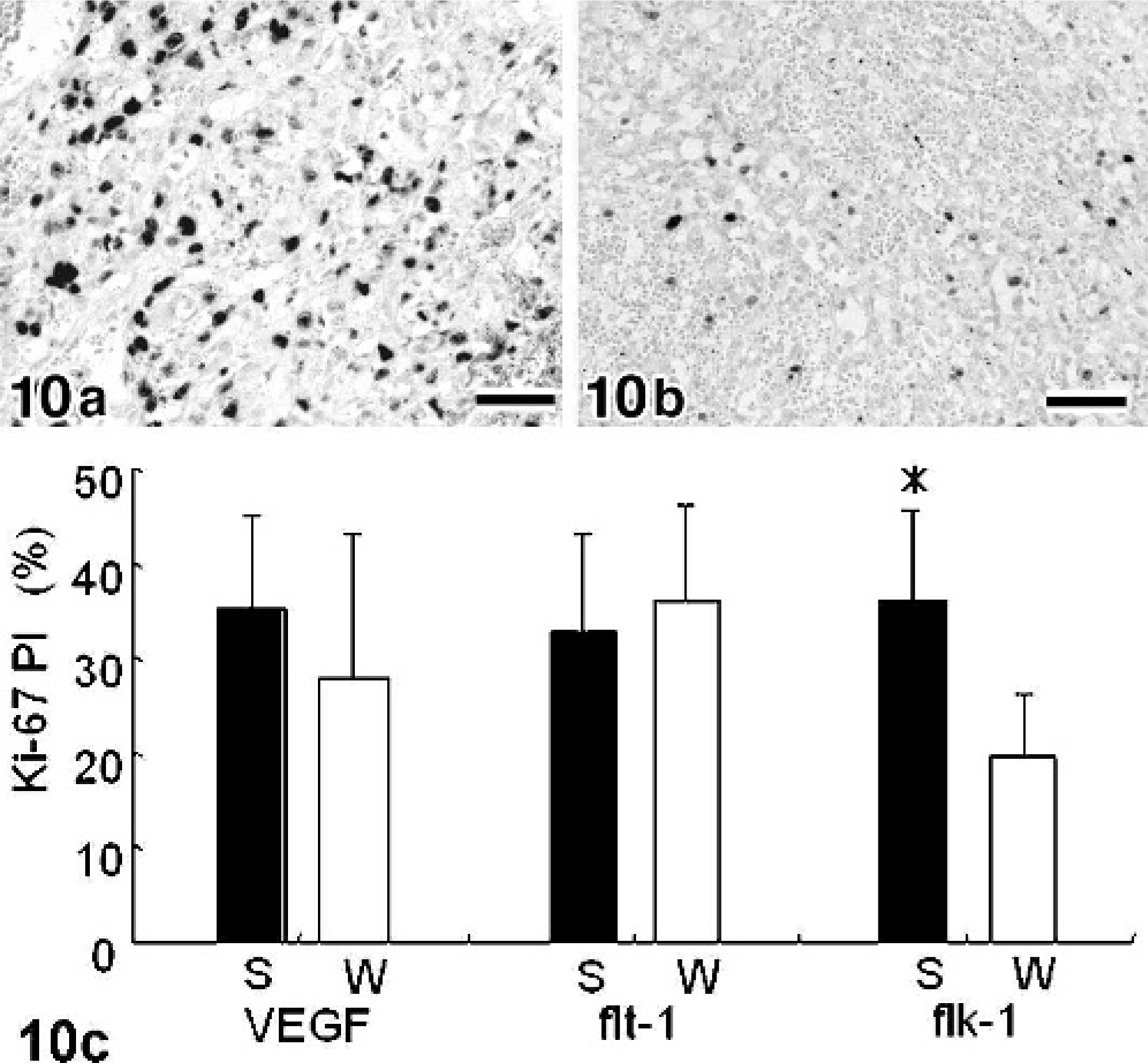
The Ki-67 PI of the HSA samples ranged from 8.4% to 57.4% (mean ± SD = 34.8% ± 10.4%). In contrast with HSA, the Ki-67 PI of the hemangioma samples ranged from 0.4% to 6.8%, (mean ± SD = 2.4% ± 2.2%) (Table 3). The Ki-67 PI of the HSA samples was significantly higher than that of the hemangioma samples. The mean ± SD of Ki-67 PI in 43 well-differentiated HSAs and 30 poorly differentiated HSAs was 33.3% ± 10.7% and 36.8% ± 9.7%, respectively. There was no significant difference in the Ki-67 PI between the histologic subclasses. In the expression of flk-1, the mean ± SD of Ki-67 PI of the strong expression group was 36.1% ± 9.6% (n = 67) (Fig. 10a). In contrast, the mean ± SD of Ki-67 PI of the weak expression group was 19.7% ± 6.6% (n = 6) (Fig. 10b). The mean Ki-67 PI of the strong expression group was significantly higher than that of the weak expression group (P < 0.001). There were no significant differences between the mean Ki-67 PI values of the groups that showed strong and weak expression of VEGF and flt-1 (Fig. 10c).
Spleen, canine, hemangiosarcoma (HSA).
We succeeded in detecting the mRNA expression of VEGF, flt-1, flk-1, and flg-1 in the HSAs by ISH (Figs. 8b and 9c–e). The mRNA was mostly localized in the cytoplasm and rarely in the nucleus (Fig. 8b). VEGF was not detected in the neoplastic cells in hemangiomas, although the inflammatory cells were positive for VEGF. The sense probes did not produce any positive signals (Figs. 8c and 9f–h).
Expression of VEGF, flt-1, flk-1, and flg-1 mRNA
The results of RT-PCR are shown in Fig. 11. The RNA integrity was confirmed by the sufficient amplification of GAPDH. Following amplification of the VEGF genes by RT-PCR, 2 major bands (573 and 441 bp) and 2 minor or very faint bands (513 and 645 bp) were detected in all but 1 HSA sample and in the normal spleen sample. In all HSA samples tested by RT-PCR, intense staining of VEGF was detected in the cytoplasm of neoplastic cells by immunohistochemistry. Two major bands were detected in only 1 hemangioma sample. Although inflammatory cells and stromal fibroblasts were positive, very faint staining of VEGF was detected in the cytoplasm of this sample by immunohistochemistry. Amplification with flt-1-specific primers yielded a single 996-bp product. flt-1 was detected in all the HSA and normal spleen samples and in 2 hemangioma samples. Amplification with flk-1-specific primers yielded a single 975-bp product. Similar to flt-1 expression, flk-1 was detected in all the HSA and normal spleen samples and in 2 hemangioma samples. In all HSA and hemangioma samples tested by RT-PCR, positive staining of flt-1 and flk-1 was detected in the cytoplasm of neoplastic cells, although the mRNAs of flt-1 and flk-1 were not detected by RT-PCR in 1 sample. Amplification with flg-1-specific primers yielded a single 287-bp product in all the samples.
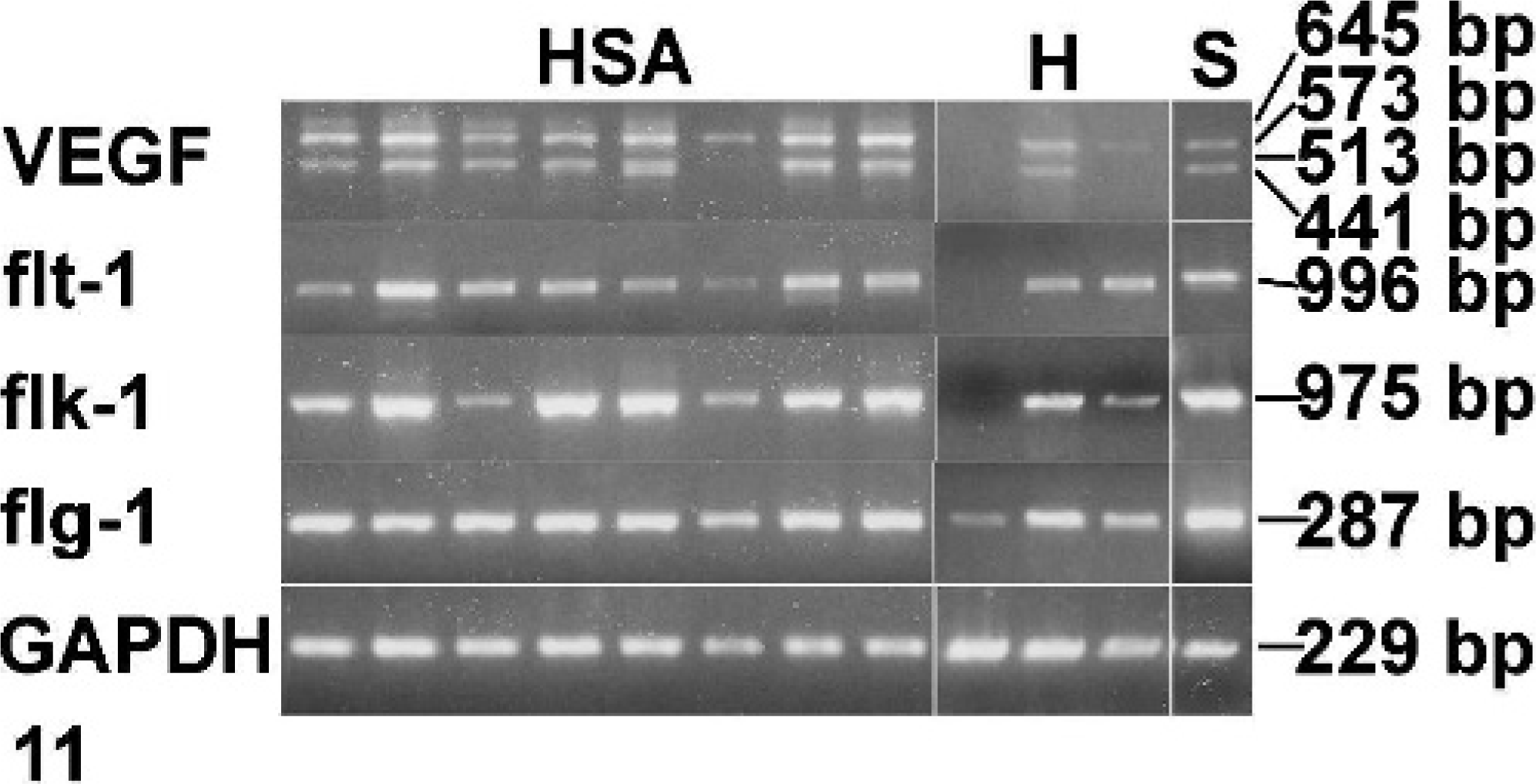
Reverse transcriptase polymerase chain reaction of vascular endothelial growth factor, flt-1, flk-1, and flg-1. cDNA was made using RNA from frozen samples of hemangiosarcomas (HSA, n = 8), hemangioma (H, n = 3) and normal canine spleen (S, n = 1). Amplification of a 229-bp fragment from glyceraldehyde-3-phosphate dehydrogenase was performed to verify the RNA integrity and as a loading control for the reaction.
Discussion
The proliferation mechanism of HSA has not been well elucidated, although HSA is one of the most aggressive neoplasms encountered in veterinary or human medicine. The purpose of this study was to obtain basic knowledge of the involvement of key growth factors such as VEGF and bFGF and their receptors in the angiogenesis of canine HSA. Angiogenesis, which is the pathophysiologic formation of new blood vessels from ordinary blood vessels, is indispensable for inflammation, organ repair, and tumor growth. 7, 8, 14 In angiogenesis, the expression of the VEGF gene is induced in endothelial cells, pericytes, and inflammatory cells to produce secretory forms of VEGF. VEGF may act on its receptors on endothelial cells in autocrine and paracrine manners, thereby causing the proliferation of endothelial cells, which may lead to angiogenesis. 17 VEGF also acts on inflammatory cells via its receptors in an autocrine manner, presumably to promote their migration. 15 In our results of immunohistochemical analysis and ISH in granulation tissue, the newly formed vascular endothelial cells were intensely positive for VEGF, flt-1, flk-1, and bFGF, and they were positive for flg-1 in ISH; in addition, both growth factors and their receptors were expressed in fibroblasts and inflammatory cells, except in neutrophils, and this may reflect the angiogenic mechanism.
In this study, both the growth factors and their receptors were expressed in the neoplastic cells of most canine HSAs, and the staining intensities of flt-1 and flk-1 were stronger in the HSAs than in the hemangiomas. On the other hand, the neoplastic cells of the hemangiomas showed moderate expression of flt-1 and flk-1, whereas the neoplastic cells exhibited very weak or no VEGF expression in most hemangiomas. In 1 sample, the mRNAs of flt-1 and flk-1 were not detectable even by RT-PCR. These results suggest that the expression of both the growth factor and its receptor might be associated with the malignant proliferation of canine HSA. The immunolocalization of VEGF in the paraffin-embedded specimens of canine HSA showed that the neoplastic cells were positive for VEGF. This suggests 2 possibilities: neoplastic endothelial cells themselves produce VEGF, or a receptor-binding form of VEGF is secreted by nonneoplastic cells, such as macrophages, in tumors. The results of ISH for VEGF mRNA show that the mRNA was detected in neoplastic endothelial cells; therefore, we concluded that neoplastic endothelial cells themselves produce VEGF. Inflammatory cells, except neutrophils, also expressed VEGF mRNA. A recent study suggests that the molecular biology and the function of the VEGF signaling system are virtually identical in healthy and disease conditions, 21 and the role of VEGF and flt-1 in the pathogenesis of human angiosarcoma is in a paracrine and/or autocrine manner. 12 Others have also shown that canine HSA-derived cell lines express both the growth factors and their receptors and have the ability to promote their own growth in an autocrine and/or paracrine manner. 1, 27 Our results indicate that the proliferation of canine HSA cells is maintained by the VEGF secreted from the malignant endothelial cells and inflammatory cells that had infiltrated into the tumor, and an autocrine or paracrine mechanism might be associated with the proliferation of the malignant endothelial cells. In contrast, small amounts of the VEGF and bFGF proteins were detected in the hemangioma cells, although VEGF mRNA could be detected in 2 hemangiomas by RT-PCR. In the immunostaining of the VEGF and bFGF proteins, inflammatory cells and stromal fibroblasts were positive; therefore, the mRNA detected in the hemangiomas by RT-PCR might originate from inflammatory cells or stromal fibroblasts. The bFGF protein was also found in the neoplastic cells of canine HSA. It has been suggested that bFGF synthesized in the neoplastic endothelial cells plays an important role in the growth and progression of human angiosarcoma. 29 In this study, the flg-1 protein could not be detected because we did not have any primary antibody that reacted with canine flg-1. However, we were able to detect flg-1 mRNA in the tumor cells by ISH. This result suggests that bFGF and flg-1, along with VEGF and its receptors, might also be associated with the malignant proliferation of canine HSA.
Biochemically flt-1 shows a VEGF affinity that is at least 10-fold higher than that shown by flk-1; however, flk-1 has a tyrosine kinase activity that is 10-fold higher than that of flt-1. 16, 18, 23, 24, 26 flt-1 shows weak tyrosine autophosphorylation in response to VEGF; however, it is able to regulate the activity of VEGF in a negative fashion as a “decoy” receptor on the vascular endothelium. 6 In contrast, flk-1 is the major mediator of the mitogenic, angiogenic, and permeability-enhancing effects of VEGF. 6 In the HSAs studied here, groups showing a strong immunoreactivity of flk-1 have a higher Ki-67 PI, which reflects the growth fraction, than groups showing a weak immunoreactivity of flk-1. Our immunohistochemical findings suggest that the local concentration of VEGF in HSAs is higher than that in hemangiomas; stromal cells in hemangiomas produce VEGF, but neoplastic cells do not. We hypothesized that the high VEGF concentration in the microenvironment of HSA causes its saturated binding to flt-1, which has high affinity for VEGF. The residual VEGF then binds to flk-1, followed by the induction of proliferation. On the contrary, VEGF secretion from the stromal cells in a hemangioma may be inadequate for the activation of flt-1. Therefore, the flk-1 pathway might be associated with the high proliferative activity of canine HSA.
In conclusion, the present results indicate the presence of an autocrine or paracrine mechanism of proliferation in canine HSA. VEGF secreted from neoplastic cells binds to its receptor on the neoplastic endothelial cells and promotes their proliferation. These results suggest that the expression of both the growth factor and its receptor, especially flk-1, might be associated with the malignant proliferation of HSAs. Moreover, bFGF synthesized in the neoplastic cells also plays an important role in the proliferation of canine HSA. These results may also provide a theoretic explanation for the success of the inhibitory activities of VEGF and bFGF in tumor therapy.
