Abstract
A primary cerebral hemangiosarcoma was identified in a 6-week-old, female, cross-breed dog. Grossly, the tumor mass was poorly demarcated from the adjacent neuropil, hemorrhagic, and caused effacement of the right dorsolateral cerebral hemisphere. Microscopically, the tumor was composed of an infiltrative mass of small vascular channels lined by neoplastic endothelial cells that stained variably with factor VIII-related antigen and negatively with glial fibrillary acidic protein. This is the first description of a primary intracranial hemangiosarcoma in an immature dog.
Hemangiosarcoma is a common neoplasm in the dog, accounting for up to 7% of all tumors in published reports. 8 Primary sites for hemangiosarcoma are most commonly noted as the spleen, right atrium, and liver. 1, 10 The median age for clinical presentation is 11 years, 12 and at the time of presentation 80% of subjects have metastasis to distant organs such as liver and lung. 8 In 14% of cases there is metastasis to the brain, making hemangiosarcoma the most common intracranial metastatic sarcoma in dogs. Primary intracranial vascular tumors, however, are rare in dogs. A single incidence has been included in one case series. 2 In a retrospective review examining tumors in juvenile dogs (less than 6 months old), hematopoetic and intracranial neoplasms represented the 2 most common categories of neoplasia. Hemangiosarcoma occurred at extra cranial locations in 8% of cases, but intracranial ocurrence was not reported. 5
A 6 week-old, female, mixed breed dog (Rottweiler/Pittbull cross) was presented to the Atlantic Veterinary College with a history of intractable, recurrent seizuring of 4-hour duration. No other litter mates had similar signs. On presentation the dog was alert, mildly dehydrated, and continuously barked or cried. Its hematocrit, blood glucose, and blood urea nitrogen were within normal limits. Further diagnostic evaluation was not pursued, and the case was submitted for full necropsy and histologic examination. Aside from changes in the central nervous system there were no gross or histologic abnormalities found following full necropsy.
The brain was removed and fixed for 48 hours in formalin before detailed examination. The right cerebral hemisphere was largely replaced by a friable, hemorrhagic mass (Fig. 1). The left lateral ventricle and mesencephalic aqueduct were dilated, and the right lateral ventricle was partially obliterated by the mass. There was caudoventral compression and coning of the cerebellum. Histologically, there was prominent distension of the overlying meninges (Fig. 2) by a neoplastic infiltrate. Multifocal cellular aggregates extended into the adjacent neuropil, often in an angiocentric pattern. The cortex of the right dorsal cerebral hemisphere was effaced by a nonencapsulated and poorly demarcated cellular mass focally contiguous with the meningeal infiltrate. The mass was composed of multifocal cellular sheets, with ill-defined, erythrocyte-filled vascular channels lined by attenuated neoplastic cells (Fig. 3). Typically they were polygonal- to spindle-shaped, with a single nucleus that ranged from ovoid to reniform. There was moderate anisocytosis and anisokaryosis. Nuclei had finely stippled chromatin with 1–2 inconspicuous nucleoli. Mitotic figures were uncommon and averaged 1 per 10 random high-power fields (40×). Large coalescent foci of hemorrhage, necrosis, and thrombosis (Fig. 2) in the neuropil corresponded to the grossly hemorrhagic appearance of the tumor. Immunohistochemical staining of the tissue with antibodies against factor VIII–related antigen and glial fibrillary acidic protein (GFAP) was performed. A variable granular cytoplasmic staining with factor VIII–related antigen was present in the neoplastic cells (Fig. 4), supporting an endothelial origin. The adjacent neuropil stained intensely and diffusely with GFAP, whereas neoplastic infiltrates were GFAP negative. The findings were most consistent with hemangiosarcoma.
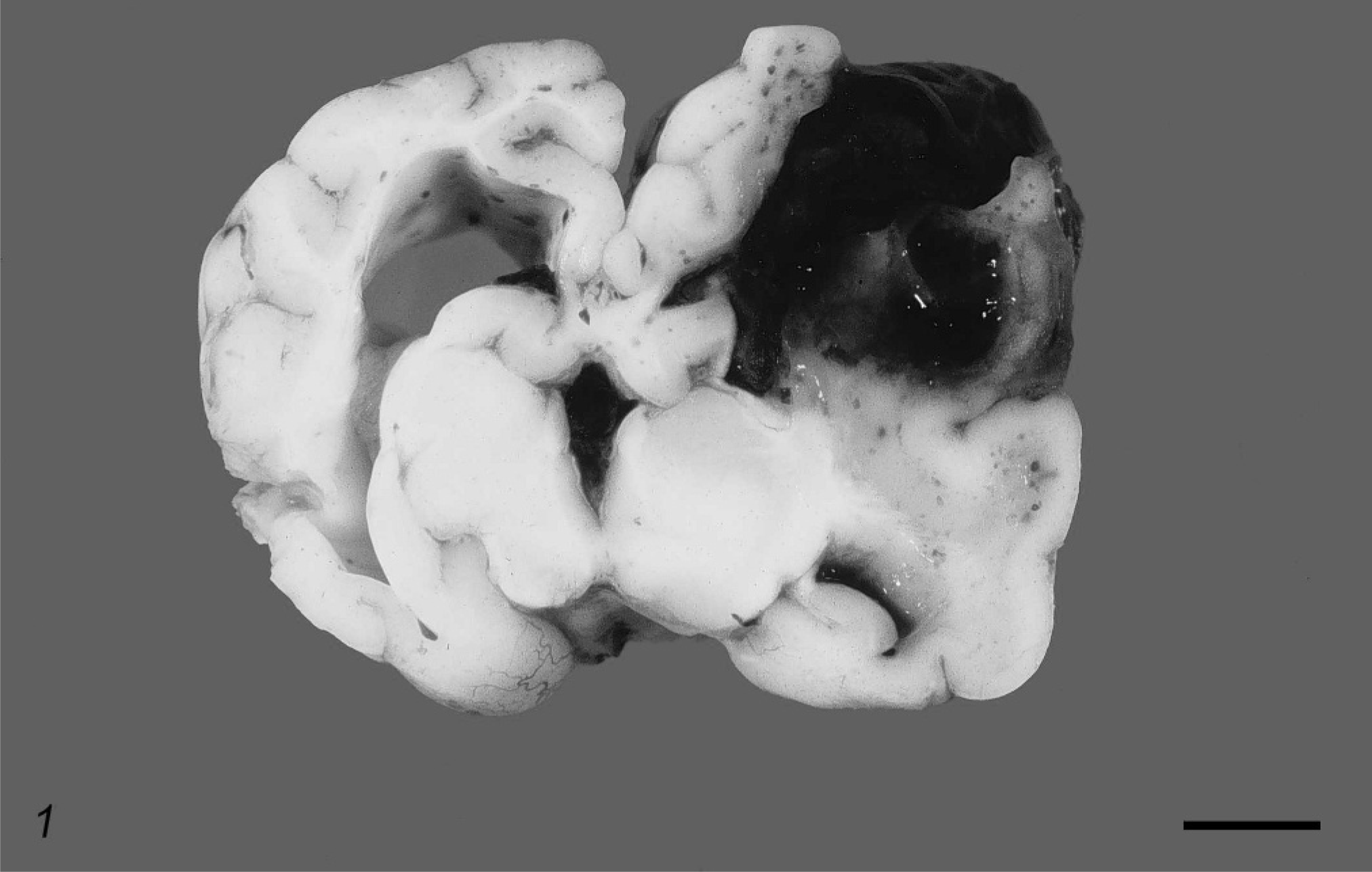
Cerebrum. Canine. There is a dark red mass that obliterates the dorsolateral region of the right hemisphere. The ventricles are dilated. Bar = 1 cm.
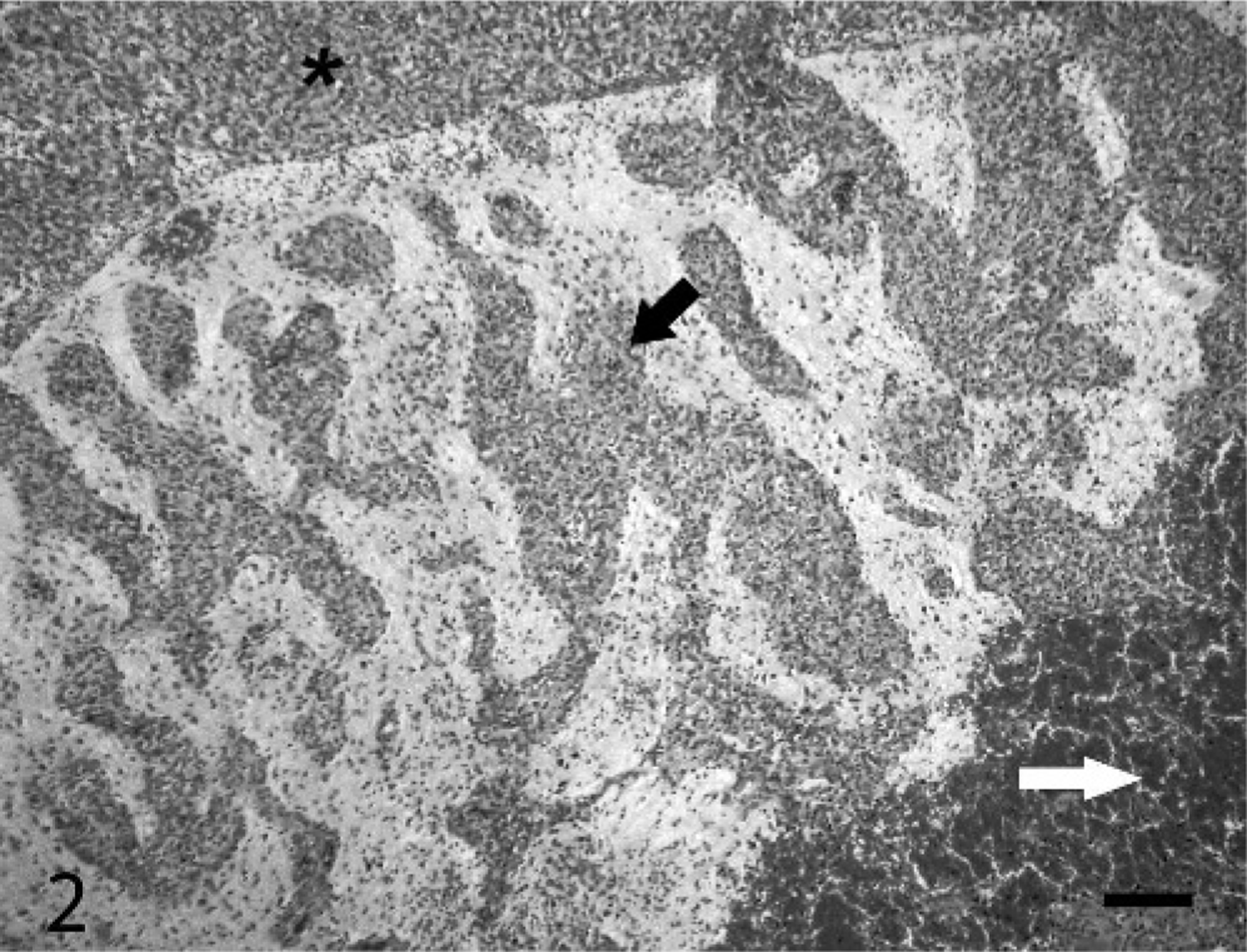
Cerebrum. Canine. Extensive infiltrative, highly cellular infiltrates (black arrow). Overlying meninges are sharply demarcated (asterisk). There is a large central core of hemorrhage and necrosis (white arrow). HE. Bar = 175 μm.
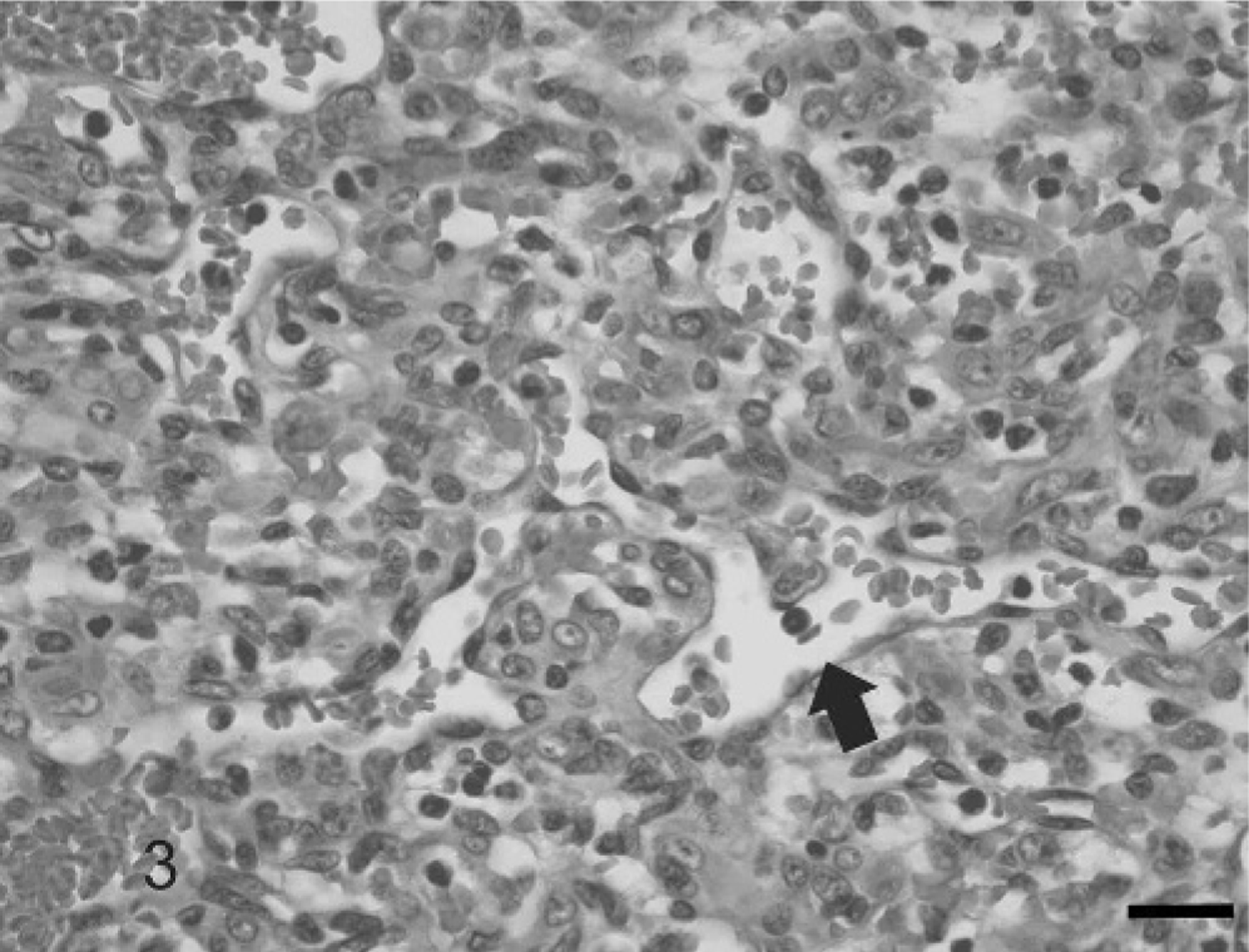
Cerebrum. Canine. Meningeal infiltrate is composed of sheets of cells often forming vascular channels variably filled with erythrocytes (arrow). HE. Bar = 40 μm.
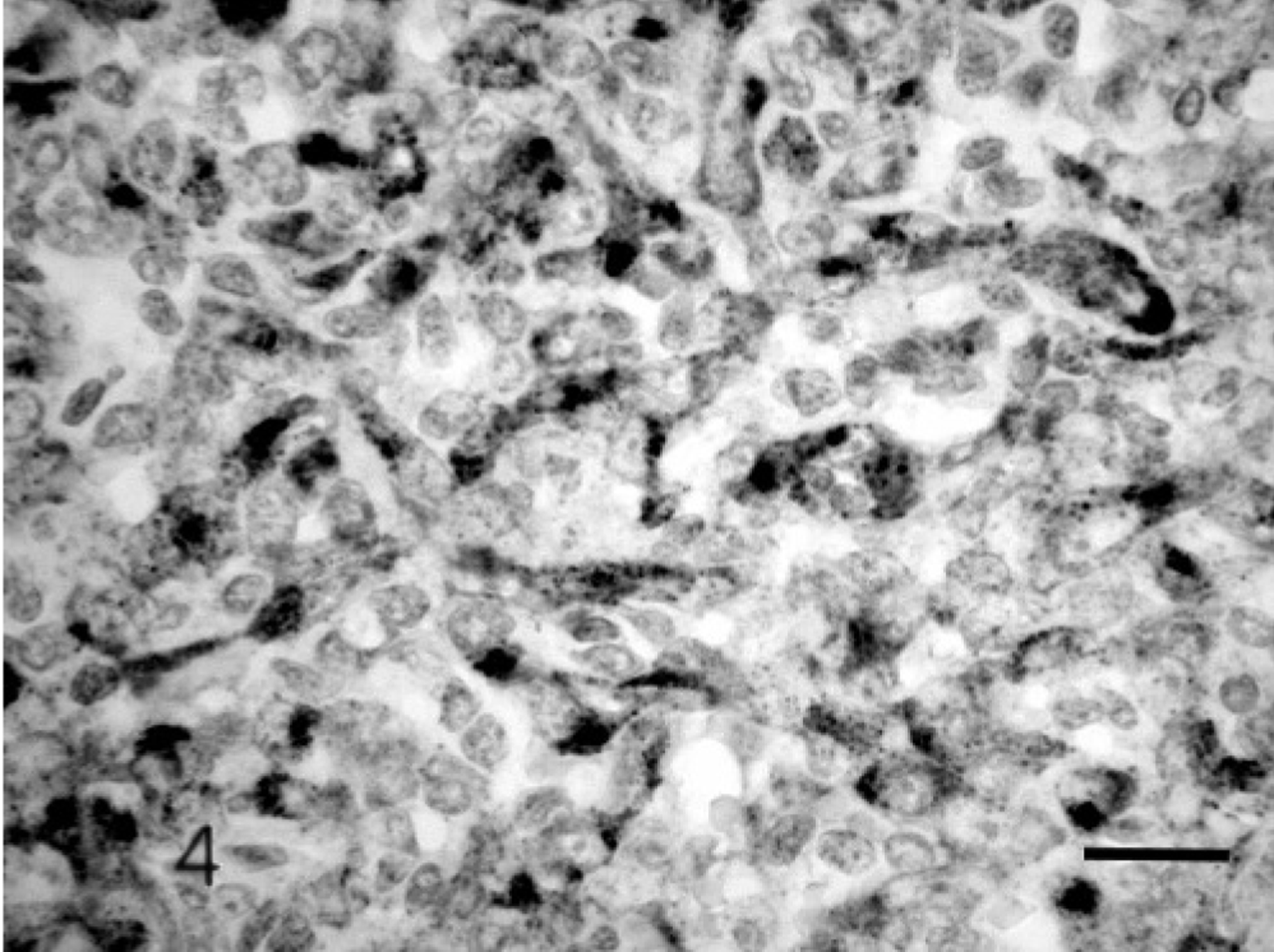
Cerebrum. Canine. Meningeal infiltrate. There is a variable granular cytoplasmic staining of neoplastic cells. Factor VIII–related antigen immunostain. Bar = 20 μm.
The differential diagnosis included other forms of neoplasia, such as angioendotheliosis, or vascular malformations. Angioendotheliosis of the dog is lymphoid in origin, and cells are consistently factor VIII–related antigen negative. 3 Briefly, vascular malformations in humans are divided into congenital malformations of the brain parenchyma (which include arteriovenous malformation, cavernous hemangioma, and capillary telangectasis) and congenital malformations of the meninges (which include arteriovenous malformation, venous angioma, or cavernous hemangioma). Of these, arteriovenous malformations appear histologically as tortous masses of blood vessels. These may reach significant sizes involving large portions of an individual cerebral hemisphere and often have a propensity to hemorrhage. A rare familial occurrence has been reported. Although they can be associated with adjacent neuropil compression, invasiveness is not a feature. Cavernous hemangiomas consist of closely clustered enlarged capillary channels (caverns) with a single layer of endothelium without normal intervening brain parenchyma or mature vessel wall elements. Capillary telangiectasis is usually an incidental finding and is composed of loosely aggregated, variably ectatic capillaries.
Vascular malformations are uncommonly noted in domestic animals. 9, 11 A recent description of 5 cerebral vascular hamartomas in the dog 9 identified masses as discrete aggregates of blood vessels with minimal to no compression of the surrounding parenchyma and a lack of invasiveness. For humans, brain tumors are the most common form of solid malignancy in children 4 with an increasing incidence in western countries. 6 Intracranial vascular tumors are uncommon. The exception is hemangioblastoma, which accounts for 7.3% of neoplasms in the posterior fossa. This is a benign mass closely associated with the meninges, which are composed of stromal cells containing characteristic lipid-laden intracytoplasmic vacuoles. No such cell population was noted in the current case.
Neoplasia of the immature dog is not an uncommon finding, however, intracranial neoplasm in immature dogs is rarely reported. In a retrospective study of 69 dogs less than 6 months old, 11 intracranial tumors were noted, with meningiomas accounting for the largest subset (4 cases). Other tumors included glioma, hamartoma, lipoma, astrocytoma, ependyoma, and choroid plexus papilloma. 5 In the current case a final diagnosis of hemangiosarcoma was reached. There was a distinct, highly abnormal cellular infiltration of the meninges that extended into and multifocally effaced the neuropil. The tumor was composed of vascular spaces lined by atypical cells that stained variably with factor VIII–related antigen, a finding most consistent with cells of an endothelial origin.
Primary intracranial hemangiosarcoma is rare in dogs. A review of the literature identified sporadic cases included in retrospective analyses. 2, 11, 13 These included a single case involving the brain stem of an 11-year-old dog 13 and a solitary intracranial mass in a dog more than 5 years old. 3 The two most common canine brain tumors are astrocytomas and meningiomas, which occur largely in dogs more than 5 years old. 7, 13 Hematopoetic tumors are the most common category of intracranial neoplasms in dogs less than 6 months old, 5 however, to the authors' knowledge, this is the first published case of intracranial hemangiosarcoma in a juvenile dog.
Footnotes
Acknowledgements
We acknowledge Drs. P. Hanna, A. Bourqe and L. Miller for review of the initial document and R. Taylor, D. O'Connell, E. Daley, and S. Ebbett for technical assistance.
