Abstract
Immunohistochemistry (IHC) and in situ hybridization (ISH) can be used either to detect or to differentiate between Eastern equine encephalitis virus (EEEV) and West Nile virus (WNV) within formalin-fixed, paraffin-embedded (FFPE) brain tissue of horses. To compare the diagnostic sensitivity and specificity of ISH and IHC, FFPE brain tissue from 20 EEEV-positive horses and 16 WNV-positive horses were tested with both EEEV and WNV oligoprobes and EEEV- and WNV-specific antibodies. Reverse transcription polymerase chain reaction (RT-PCR) for detection of EEEV and WNV was used as the gold standard to confirm infection. All horses that tested positive for EEEV by RT-PCR also tested positive by IHC and ISH, except for 1 case that was false-negative by ISH. In contrast, all horses that tested positive for WNV by RT-PCR tested negative by IHC and only 2 horses tested positive by ISH. No false-positives were detected with either method for both viruses. Both IHC and ISH are highly specific and sensitive diagnostic methods to detect EEEV in equine FFPE brain tissues, although neither appear effective for the diagnosis of WNV in equine neurologic cases.
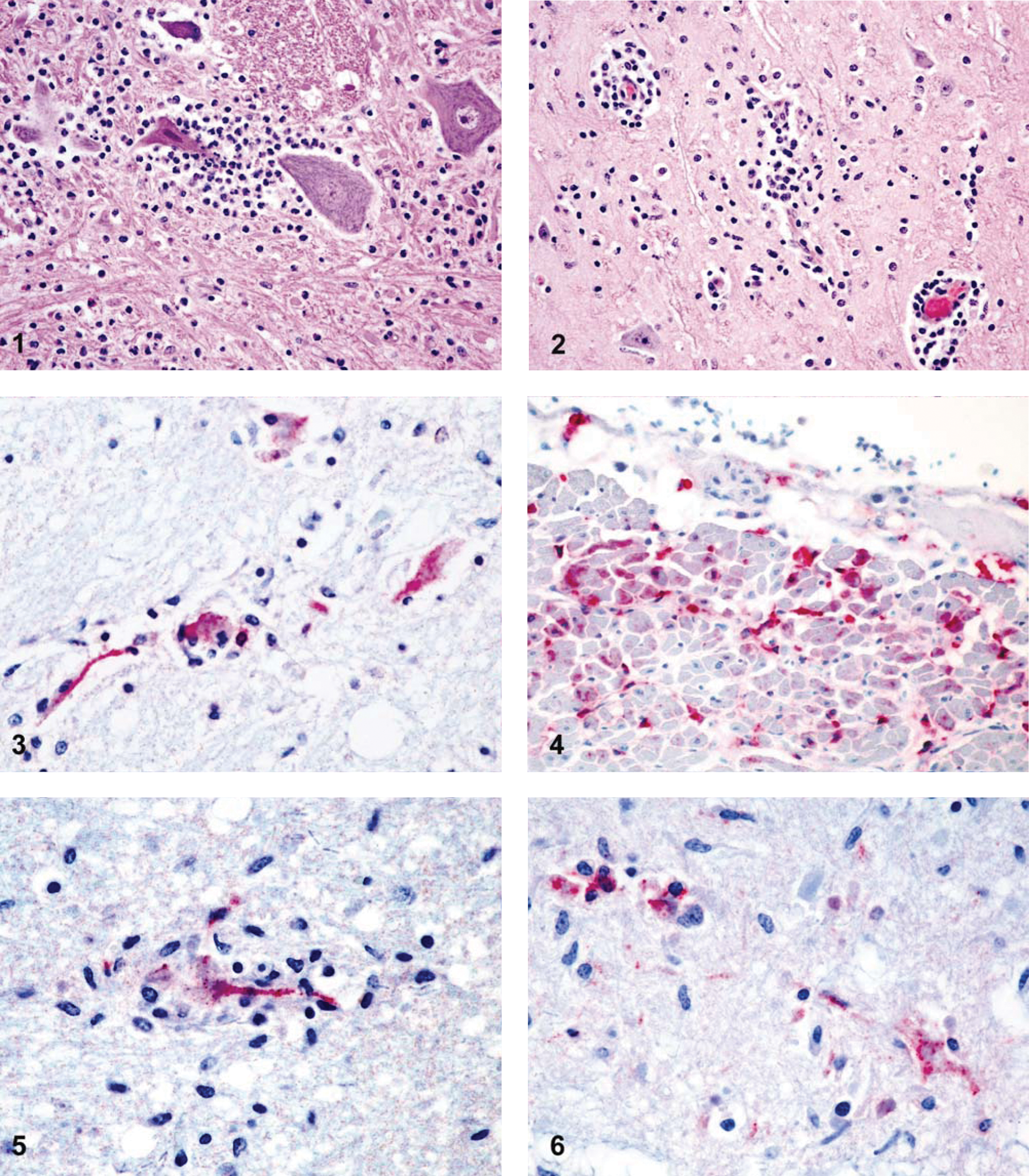
Eastern equine encephalitis virus (EEEV) and West Nile virus (WNV) are arthropod-borne viruses (arboviruses) that cause neurologic disease in horses, human beings, and other vertebrates. Horses are considered dead-end hosts for both viruses, but may present with clinical signs of disease. 15 Neurologic signs due to EEEV are variable and may include hyperexcitability or other sensory changes, anorexia, ataxia, paresis, paralysis, and obtunded mentation.5,7,15 Brain lesions in horses affected by EEEV are variably characterized by suppurative or nonsuppurative encephalitis with neuronal necrosis and neutrophilic neuronophagia (Fig. 1).9,12,13 Clinical signs of WNV in horses may range from subclinical infection to muscle fasciculations and motor abnormalities that progress to complete paralysis. 12 Lesion morphology and distribution include a mild to moderate, nonsuppurative encephalomyelitis with moderate to severe hemorrhage that tends to involve the lower brain stem and ventral horns of the thoracolumbar spinal cord (Fig. 2).2,3
Results of immunohistochemistry (IHC) and in situ hybridization (ISH).
Both viruses are endemic among horses in the United States, with a most recent yearly mortality of 247 EEEV cases and 125 WNV cases being reported by the U.S. Division of Agriculture, Animal and Plant Health Inspection Service.19,20 Transmission of EEEV and WNV is dependent on mosquito vector viability (particularly Culex sp.) and the presence of avian hosts.1,6 Vaccination has reduced the frequency and spread of outbreaks for both diseases among the horse population, but the potential for high morbidity and mortality in susceptible horses and human beings generates continued interest in these viruses.4,10
The objective of the current study was to evaluate the diagnostic sensitivity and specificity of automated in situ hybridization (ISH) and immunohistochemistry (IHC) for detecting EEEV and WNV in formalin-fixed, paraffin-embedded (FFPE) equine brain tissues using reverse transcription polymerase chain reaction (RT-PCR) as a confirmatory test. Because there are no pathognomonic clinical signs that distinguish EEEV infection from WNV infection or any other encephalomyelitic disease, such as rabies, hepatic encephalopathy, or equine protozoal myeloencephalitis, antemortem diagnostics for EEEV and WNV are presumptive at best. 8 An accurate postmortem diagnostic test for identifying both EEEV and WNV in routinely formalin-fixed material from horses is critical to provide the correct diagnosis while minimizing the zoonotic risk to laboratory personnel. While RT-PCR is considered the gold standard for the confirmation of viral infection in tissue or body fluid,19,20 IHC and ISH both visualize the virus within the lesion of interest, which allows the diagnostician to correlate the microscopic lesions directly with the cause of disease.2,5,9 There are few reported studies that have evaluated IHC for detection of EEEV and WNV in FFPE tissues and, to the authors’ knowledge, only 1 report of ISH using DNA oligoprobes for detecting EEEV in FFPE equine tissues.2,5,9,12,18 The current study was designed to further evaluate the diagnostic utility for ISH and IHC in detecting EEEV and WNV in FFPE brain tissues of clinically affected horses.
Formalin-fixed brain tissues from 20 EEEV-positive horses and 16 WNV-positive horses confirmed by RT-PCR and virus isolation, either at the National Veterinary Services Laboratory (NVSL; Ames, Iowa) or the Diagnostic Center for Population and Animal Health (DCPAH; College of Veterinary Medicine, Michigan State University, Lansing, Michigan), were used in the present study. Tissues were routinely processed and paraffin embedded, with 5-μm sections either cut onto positively charged slides or separately submitted for RT-PCR detection. For all testing methods, 1 or more FFPE brain tissue sections were chosen for evaluation based on tissue availability for each animal and included sections of cerebrum, cerebellum, and/or brainstem. Only tissues with microscopic lesions were selected for the study, and serial sections of these tissues from all horses were tested by RT-PCR, IHC, and ISH for EEEV and WNV. The RT-PCR for EEEV was performeda,b as previously described, using EEEV primers that produce a 112–base pair (bp) amplicon from the viral capsid gene. 11 The WNV primers amplify a 155-bp region of the viral envelope glycoprotein gene. 18 Controls consisted of positive and negative EEEV or WNV FFPE equine brain tissue; all had been previously confirmed by RT-PCR on both the fresh and formalin-fixed tissue, as well as by virus isolation. The resulting RT-PCR findings included detection of EEEV in all 20 of the EEEV-positive cases (100%) and detection of WNV in all 16 of the WNV-positive cases (100%). All 20 EEEV cases were negative for WNV (100%) and all 16 WNV cases were negative for EEEV (100%).
For IHC, sections of equine brain tissue were evaluated using an antibody detection system utilizing alkaline phosphatasec,d as previously described.14,18 A mouse monoclonal anti-EEEV antibody e was used as the primary antibody for detection of EEEV (Fig. 3). 14 A mouse monoclonal anti-WNV antibody (clone 7H2), directed against an epitope on domain III of the E protein of WNV, f was used as the primary antibody for detection of WNV (Fig. 4). 18 Positive and negative control tissues (avian heart and brain tissue as WNV positive controls, equine brain tissue as EEEV positive control, and uninfected equine brain as negative controls) that had been previously confirmed by RT-PCR and virus isolation were run concurrently with the different IHC and ISH tests. Avian brain and heart tissue was used as a positive control due to difficulties identifying a good immunohistochemically WNV-positive equine brain sample. For negative reagent controls, the primary antibodies were replaced by homologous nonimmune sera.
Detection of hybridized viral RNA by ISH was performed using an automated labeling system.g,h The EEEV probe consisted of a 5’ fluorescein–labeled DNA sequence i complimentary to the viral E1 glycoprotein (Fig. 5). 9 The WNV probe consisted of a 5’ fluorescein–labeled DNA sequence j complimentary to the viral envelope protein (Fig. 6). Both sequences were previously designed by the Infectious Diseases Laboratory, Department of Pathology, College of Veterinary Medicine, The University of Georgia (Athens, Georgia).
The resulting IHC- and ISH-labeled slides were evaluated microscopically by a single ACVP board-certified pathologist (Kiupel). Cases were determined to be either positive or negative, with weak labeling included as positive, based on relative levels of specific cellular labeling when compared to negative and positive controls.
All 20 horses that had tested positive for EEEV by RT-PCR were also positive by IHC (Table 1). Immunohistochemistry labeling detected antigen in areas of necrosis and inflammation within the brain tissues of all EEEV cases (Fig. 3). Neurons and associated dendritic processes, along with fewer glial cells and astrocytes, were also positive. Some IHC cases demonstrated increased antigen reactivity, with stronger labeling, when compared to the same inflammatory regions in tissues that were EEEV positive using the ISH method. None of the horses that tested positive for WNV and negative for EEEV by RT-PCR were positive for EEEV by IHC. Immunohistochemistry failed to detect WNV antigen in any of the 16 horses that had been positive for WNV by RT-PCR and did not detect WNV antigen in any of the EEEV-positive brain tissue sections.
Comparison of immunohistochemistry and in situ hybridization for detecting Eastern equine encephalitis virus (EEEV) and West Nile virus (WNV) in formalin-fixed, paraffin-embedded equine brain tissue using reverse transcription polymerase chain reaction as the gold standard.
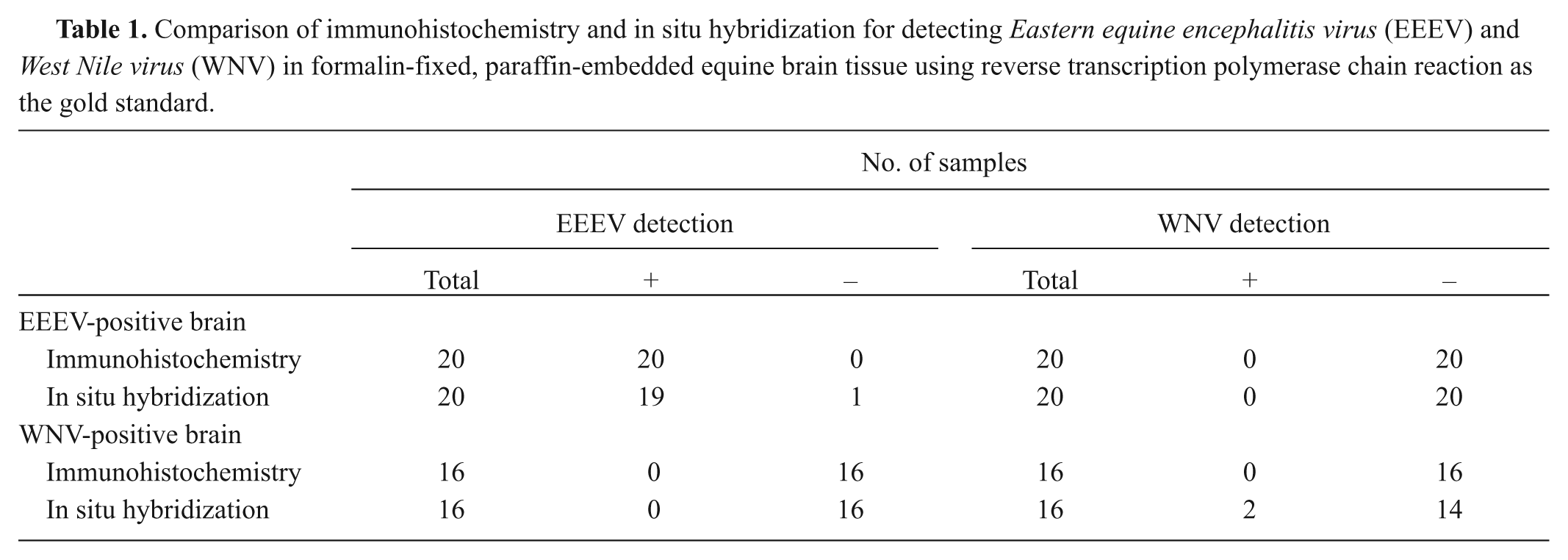
All but 1 of the 20 horses that tested positive for EEEV by RT-PCR were also positive by ISH (Table 1). Positive labeling was detected within areas of inflammation in the brain, specifically within the cytoplasm and dendritic processes of necrotic neurons (Fig. 5). Occasional normal appearing neurons were also positive, and associated axons, glial cells, and astrocytes stained positive with a more prominent, well-dispersed chromogen deposition. None of the horses that were WNV-positive and EEEV-negative by RT-PCR tested positive for EEEV by ISH. Two of the 16 horses that were positive for WNV by RT-PCR tested positive for WNV using ISH (Fig. 6). In the 2 positive cases, chromogen deposition was observed in the cytoplasm of a few neurons within inflammatory regions of the cerebrum. Several areas of inflammation were present in brain sections from all cases, but ISH reactivity was not observed within these areas in the other 14 samples. In situ hybridization did not detect WNV in any of the EEEV-positive brain tissue sections.
The goal of the current study was to evaluate the diagnostic sensitivity and specificity of automated ISH and IHC for detecting EEEV and WNV in FFPE equine brain tissues. For EEEV, IHC showed 100% diagnostic sensitivity and specificity, and ISH showed 95% diagnostic sensitivity and 100% diagnostic specificity when compared to the gold standard of RT-PCR. A 2-tiered Fisher exact test confirmed that there was no significant deviation between the ability for IHC and ISH to detect both EEEV and WNV antigens using RT-PCR as the gold standard for a positive result (P = 1.00 for EEEV, P = 0.48 for WNV). None of the EEEV cases were negative by IHC, and only 1 case tested negative for EEEV by ISH. Since only 2 of the 16 WNV cases were positive by ISH, and none of the cases were positive by IHC, these tests are minimally effective as diagnostic tools for detecting WNV in FFPE equine brain.
A DNA oligonucleotide probe, representing a conserved region of EEEV, appears to be highly diagnostically sensitive and specific for detecting EEEV in FFPE equine brain tissues using ISH methods. However, 1 tissue sample previously confirmed to be EEEV-positive by RT-PCR also tested positive by IHC, but did not demonstrate any reactivity using the ISH method. The inability to detect viral nucleic acid by ISH in this single sample may be due to variable lesion localization in serial sections or RNA degradation. 9 It is reasonable to assume that the viral messenger RNA and encoded protein concentrations vary in individual tissues.
In EEEV-positive tissues, there was a tendency for some of the IHC-labeled tissues to exhibit stronger, more intense labeling than their ISH counterparts. Neurons in an earlier stage of EEEV infection likely contain virions that are replicating at high enough rates to meet the threshold for detection required by the ISH probe targeting the envelope protein that was used in this experiment. Infected neurons that are in a later stage of virus replication, where more structural proteins are being created in preparation for budding, would also have more detectable E protein present. The probe for the present study is complimentary to the genomic positive-sense RNA, but should also be able to bind to the generated messenger RNA. If the sensitivity of the IHC antibody for detecting the envelope protein is high, then the concentration of envelope proteins per virion in an infected cell versus the amount of detectable RNA may explain the lack of reactivity observed in the single EEEV case that was ISH-negative but IHC-positive.
There is a strong possibility that prolonged fixation times resulted in stronger cross-linking and degradation of viral RNA within cells. Due to a scarcity of case material with confirmed RT-PCR positivity, some of the cases used in the current study were over 5 years old. This may explain why the false-negative ISH test was positive by IHC, as RNA is much more sensitive to degradation compared to protein. The intensity of labeling for ISH, and perhaps IHC, may have been affected by the extended storage time in paraffin.
Results of the current study indicate that both ISH and IHC are insufficient for detecting WNV in FFPE equine brain tissues. In situ hybridization had a diagnostic sensitivity of 12.5% and a diagnostic specificity of 100% when detecting WNV in FFPE tissue, while IHC failed to detect WNV in any of the FFPE equine brain tissues. The results were not unexpected, as WNV viral loads are reportedly low in equine neural tissues.2,15 Also, using neuronal inflammation and necrosis as morphological hallmarks to identify appropriate areas to test for viral antigen or nucleic acid presence may be inappropriate considering the apoptotic effects of cluster of differentiation (CD)8 host immune response following WNV infection.16,17,21 However, observation of microscopic lesions in neuronal tissues commonly results in request for additional diagnostic testing of such tissues.
Previous studies that demonstrated detection of WNV using IHC in brain sections of affected horses were designed to study the pathogenesis of the virus and its cellular localization and examined large numbers of tissue sections. 2 However, IHC lacks reliable diagnostic sensitivity for detecting WNV in FFPE equine brain tissue and therefore is not useful as a diagnostic test for WNV on single tissues with histopathologic lesions. For pathogenesis studies focused on detection of WNV nucleic acid in conjunction with histopathologic lesions, the ISH method could be used despite its low diagnostic sensitivity.
Footnotes
Acknowledgements
The authors would like to thank the Immunohistochemistry Laboratory and Virology Laboratory at the Diagnostic Center for Population and Animal Health, Michigan State University for their expertise in both Immunohistochemistry and in situ hybridization; the histopathology technicians at the Diagnostic Center for Population and Animal Health, Michigan State University for their technical assistance; the National Veterinary Services Laboratory in Ames, Iowa, for their work with polymerase chain reaction; and Dr. Christopher R. Gregory and the Infectious Disease Laboratory, Department of Pathology, College of Veterinary Medicine at the University of Georgia for their initial work developing the Eastern equine encephalitis virus ISH test.
