Abstract
Horses were inoculated with Vesicular stomatitis New Jersey and Indiana viruses by routes simulating contact and vector transmission. Clinical signs, lesions, antibody development, viral shedding and persistence, and viremia were monitored. Horses were infected with both viruses by all routes as confirmed by seroconversion. Salivation, primary lesions at inoculation sites, and secondary oral lesions were the most common clinical findings. Viral shedding was most often from the oral cavity, followed by the nasal cavity; titers were highest from oral cavity samples. Virus was rarely isolated from the conjunctival sac and never from feces or blood. Development of neutralizing antibody coincided with cessation of lesion development and detection of virus by isolation. Circulating virus-specific IgM, IgG, IgA, and neutralizing antibodies developed in most animals postinoculation (PI) days 6 to 12, depending on the route of inoculation. At postmortem (PI days 12 to 15), lesions were healing, were not vesicular, and did not contain detectable virus by isolation, reverse transcriptase polymerase chain reaction, or immunohistochemistry. Numerous infiltrating lymphocytes and plasma cells suggested that lesion resolution was partially due to local immunity. Detection of viral RNA from tonsil and lymph nodes of head at necropsy suggests that these tissues play a role in the pathogenesis of the disease; molecular techniques targeting these tissues may be useful for confirming infection in resolving stages of disease. The routes of inoculation used in this study reflect the diversity of transmission routes that may occur during outbreaks and can be used to further study contact and vector transmission, vaccine development, and clarify pathogenesis of the disease in horses.
Keywords
Introduction
Vesicular stomatitis (VS) is a disease of horses, cattle, and swine caused by related viruses in the genus Vesiculovirus of the family Rhabdoviridae. In the United States, outbreaks of VS have been associated with 2 genotypically and serologically distinct serotypes: Vesicular stomatitis New Jersey virus (VSNJV) and Vesicular stomatitis Indiana virus (VSIV). Of these, VSNJV has occurred most often and, in recent years (1995, 1997, 1998, 2004, and 2005), has been responsible for multiple outbreaks in the western United States. 1– 4, 11 The most recent outbreaks of VSIV occurred in the western United States during 1997 and 1998. 1 These and prior epidemics have resulted in substantial direct and indirect economic impact on the horse industry. 4 Furthermore, as an Office International Des Epizooties List A disease, VS outbreaks result in an extensive regulatory response, often resulting in quarantine and restricted domestic and international trade.
Elucidation of the pathogenesis and epidemiology of VS is complicated by a diversity of potential transmission routes, including biological vector, mechanical vector, and contact transmission. The relative importance of each of these potential transmission routes related to VS outbreaks in horses is unknown. Understanding the epidemiology of this disease is further complicated by the fact that vesicular stomatitis virus (VSV) infection in horses is often inapparent or subclinical 12 and that in many cases VS would not be detected under existing surveillance strategies that are based on the detection of vesicular lesions or related clinical signs. Without an understanding of viral transmission risks or development of effective VSV detection systems, efficient and cost-effective control and eradication strategies cannot be created to deal with these recurring outbreaks.
The overall objective of this study was to develop an experimental system that produces the various clinical responses associated with VS in the field in order to provide a reproducible system for future studies evaluating transmission efficiency, vaccine efficacy, and VS detection strategies. Specific objectives were to 1) determine if horses can be experimentally infected with recent western United States isolates of VSNJV and VSIV by 4 routes of inoculation simulating contact or vector transmission; 2) determine if development of clinical signs varies with inoculation route and serotype of virus; and 3) determine if the serologic response and source, duration, and viral titer associated with viral shedding vary with inoculation route and serotype.
Material and Methods
Experimental design
Twenty-four adult horses of varying age, sex, and breed were used (approved by the Animal Care and Use Committee, University of Georgia, IACUC# A1998-10153). Twelve horses were inoculated with VSNJV and 12 with VSIV. For each serotype, 3 were inoculated by intraepithelial/subepithelial injection of the left dorsolateral surface of the tongue (TONGUE), 3 were inoculated by intradermal injection of the haired skin of the chin (ID CHIN), 3 were inoculated by applying virus to a scarified area on the oral mucosa of the left lower lip (SCAR LIP), and 3 were inoculated per os (ORAL). For injection of the tongue and chin, 300 μl (VSNJV) or 100 μl (VSIV) of inoculum was injected in 3 places using a 25-gauge needle. Scarification of the oral mucosa of the lip was achieved by disrupting the epithelium 10 times with a dual-needle vaccine applicator (AirTite, Vineland, NJ) and smearing 300 μl (VSNJV) or 100 μl (VSIV) of viral inoculum over the scarified site. For oral inoculation, 1 ml of viral inoculum was deposited over the back of the tongue and the head held up for 1 minute. Animals were tranquilized with 0.3 to 0.4 mg/kg xylazine (Rompun, Bayer Animal Health, Shawnee Mission, KS) intravenously ± 0.01 to 0.02 mg butorphenol (Torbugesic, Fort Dodge Animal Health, Orange City, IA) intravenously for inoculation.
Animals were examined daily for clinical signs and the development of lesions. Blood was collected daily in heparinized tubes for virus isolation and reverse transcriptase polymerase chain reaction (RT-PCR) and in clot tubes for virus isolation, RT-PCR, and serology. Swabs of the buccal pouch (for saliva), the palatine and lingual tonsillar regions, the left nasal cavity, conjunctival sac, and rectal feces were obtained daily for virus isolation. Swabs were placed in 1 ml of viral transport medium (minimal essential medium, Sigma Chemical Co., St. Louis, MO) with 1,000 units penicillin G (Sigma), 1 mg streptomycin (Sigma), and 0.25 μg amphotericin B (Sigma)/ml and placed on ice.
Animals were euthanized via an overdose of sodium pentabarbitol (Beuthanasia-D Special, Schering-Plough Animal Health, Omaha, NE) on postinoculation (PI) days 12 to 15 and a necropsy performed. Tissue from residual lesions, palatine tonsil, and retropharyngeal and mandibular lymph nodes was collected at necropsy for virus isolation, RT-PCR, histopathology, and immunohistochemistry. Duplicate pieces of tissue were collected in 1.5-ml cryovials, snap frozen in liquid nitrogen, and stored at -70°C for RT-PCR or placed in 1 ml of viral transport medium on ice for virus isolation. A third sample of tissue was placed in 10% buffered formalin for histopathology and immunohistochemistry, allowed to fix for 24 to 48 hours, and then routinely embedded in paraffin.
Virus inoculum
Viruses used were 95-44620 VSNJV and 97-25323 VSIV obtained from the National Veterinary Services Laboratories, APHIS, USDA, Ames, Iowa (NVSL). To prepare inoculums, 10 μl of virus stock was inoculated into 75 cm2 flasks of Vero cells and supernatant harvested at 24 hours and frozen at -70°C until use. For dose estimation, endpoint titration of a frozen aliquot of each viral stock was performed in Vero cells prior to starting the experiment. Frozen virus stocks were diluted at the time of animal inoculation, with titers confirmed by endpoint titration in Vero cells. All animals inoculated with VSNJV received 107.1 median tissue culture infective doses (TCID50)/animal); animals inoculated with VSIV by scarification or needle inoculation received 106.4 TCID50/animal; and animals inoculated per os with VSIV received 107.1 TCID50/animal.
Immunohistochemistry
Formalin-fixed tissue from residual lesions, palatine tonsil, retropharyngeal lymph node, and mandibular lymph node were embedded in paraffin and sectioned at 3-μm thickness. Sections were deparaffinized and stained for viral antigen using an alkaline phosphatase-conjugated streptavidin-biotin technique (Biogenex, San Ramon, CA) as previously described. 6 For horses infected with VSIV, anti-VSIV mouse ascites fluid (American Type Culture Collection, Manassas, VA) was used as the primary antibody. Deparaffinized sections were also stained using the following primary antibodies: rabbit anti-CD3 (DAKO;1°50 dilution), mouse anti-BLA.36 (Biogenex;1°50 dilution), mouse anti-CD79a (DAKO;1°30 dilution), and mouse anti- MAC 387 (DAKO;1°100) using Citra solution (Biogenex) with steam for antigen retrieval and appropriate secondary biotinylated antibodies (Vector Laboratories, Burlingame, CA). The immunologic reaction was visualized with a peroxidase-conjugated avidin-biotin complex system (Vector Laboratories) with 3,3′-diaminobenzidine as chromagen (Vector Laboratories).
Antibody determinations
Sera were assayed for neutralizing antibodies to VSNJV and VSIV as previously described. 5 Values are reported as the reciprocal of the highest dilution neutralizing 1,000 TCID50 of virus, and an antibody titer of ≥8 was considered indicative of seroconversion.
Sera were also tested for IgM, IgA, and IgG antibodies to VSNJV and VSIV. A capture enzyme-linked immunosorbent assay (ELISA) was used to detect IgM antibodies. Sera from all animals of each inoculation group were assayed on the same plate. Briefly, 96-well ELISA plates were incubated for 48 hours at 4°C with sheep anti-horse IgM-mu chain-specific antibody (Bethyl Laboratories, Inc.; diluted 1°200 in 0.015 M carbonate-0.035 M bicarbonate buffer pH 9.6), then blocked with 5% non-fat dried milk (NFM) in phosphate-buffered saline (PBS) for 15 minutes at room temperature. After washing with 0.05% Tween in PBS (PBST), each test sera (diluted 1°10 in S/A diluent [1% NFM in PBS]) was added to 4 wells and incubated at 37°C for 30 minutes. Plates were then washed; for each test sera either positive antigen (VSNJV or VSIV IgM capture [ELISA] antigen; NVSL) or negative antigen (VS [NORMAL] IgM capture (ELISA) antigen; NVSL) diluted 1°60 in S/A diluent was added to duplicate wells and incubated for 45 minutes at 37°C. After washing, either anti-VSNJV or VSIV mouse ascites fluid, as appropriate, was added (diluted 1°750 in ACA diluent [1% NFM and 0.95% NaCl in PBS]) and incubated for 30 minutes at 37°C, followed by washing and incubation with horseradish peroxidase (HRP)–conjugated rabbit anti-mouse antibody (Vector; diluted 1°400 in ACA diluent) for 30 minutes at 37°C. Finally, after washing, 2,2′ Azino-bis (3-ethylbenzthiazoline-6-sulfonic acid) liquid substrate (Sigma) was added and color development recorded using a 405-nm filter in an automated microplate reader. Positive and negative controls were included on each plate (PI days 0 and 8 serum from the same VSNJV- or VSIV-infected horse and duplicate wells receiving either positive or negative antigen). Duplicate wells were averaged, and for each test sera the final optical density (OD) was calculated by subtracting the mean absorbance of the negative wells from the mean absorbance of the positive wells. An OD increase >0.2 was considered significant.
For IgG and IgA, an indirect ELISA was used and sera from all animals of each inoculation group were assayed on the same plate. Briefly, wells of 96-well ELISA plates were incubated overnight at 4°C with antigen (VSNJV ELISA antigen #2-NVSL 3195 or VSIV ELISA antigen #4- NVSL 3195) diluted 1°2000 in carbonate-bicarbonate buffer and then blocked with 10% NFM in PBS (30 minutes at room temperature). Test sera diluted in 1% NFM in PBS (1°50 for VSNJV-infected horses or 1°20 for VSIV-infected horses) were added to duplicate wells. Positive and negative control sera were included on each plate: positive controls were 5 2-fold dilutions of PI day 12 serum from 1 horse infected with either VSNJV (neutralizing titer: 8,192) or VSIV (neutralizing titer: 32,768), as appropriate; PI day 0 serum from the same horses was used as negative control serum. Additional controls included wells receiving no serum, no antibody, or no serum or antibody. All controls were done in duplicate. Plates were incubated for 30 minutes at 37°C and washed with PBST, and then HRP-conjugated sheep anti-horse IgG heavy- and light-chain or HRP-conjugated sheep anti-horse IgA-alpha-chain–specific (Bethyl; diluted 1°1,000 in 1% NFM in PBS) was added to all wells except those used for blanking or “no antibody” control wells, which received PBS instead. After a 30-minute incubation at 37°C, plates were washed and 2,2′ Azino-bis (3-ethylbenzthiazoline-6-sulfonic acid) liquid substrate was added. Plates were read on a microplate reader using a 405-nm filter and results recorded when an average OD of 1.0 (VSNJV) or 0.6 (VSIV) for the duplicate positive control wells was reached. Results of duplicate wells were averaged and then values normalized by dividing the mean test sample OD by the mean OD of the day 0 sample. Increases in OD >0.25 and >0.35 for IgG and IgA, respectively, were considered significant.
Virus isolation
Virus isolation was done on the day of sample collection. Virus isolation from swabs, blood, and plasma was attempted using Vero cells as previously described. 16 Virus isolation was also attempted from tissues obtained at necropsy (retropharyngeal and mandibular lymph nodes, tonsil, and residual lesions) as previously described. 5 Titration of positive virus isolations was performed via end-point titration, as previously described, 14 and all isolates were confirmed as VSNJV or VSIV through immunocytochemistry using VSNJV or VSIV hyperimmune mouse ascites fluid.
RT-PCR
Total RNA was extracted from 1 ml of heparinized whole blood (days 1–4 PI) or 140 μl of serum (from positive whole blood samples only) using a kit (blood-QIAmpRNA blood mini kit; serum-QIAmp Viral RNA mini kit; QIAGEN, Valencia, CA). Total RNA was extracted from approximately 75 mg of tonsil and retropharyngeal lymph node tissue as previously described. 15 Viral RNA from horses infected with VSNJV was amplified by a nested RT-PCR using primers complementary to the N gene of an Ossabaw Island VSNJV isolate and an RNA PCR kit (GeneAmp RNA PCR kit; Perkin Elmer, Roche Molecular Systems, Inc., Branchburg, NJ) to amplify a 138-bp product as previously described. 15 Viral RNA from horses infected with VSIV was amplified by a semi-nested RT-PCR using an RNA PCR kit (GeneAmp RNA PCR kit) and primers designed (courtesy of L. L. Rodriguez) to amplify an 804-bp product of the N gene of VSIV (IN-N612F 5′-TGT GGG GAA ATG ACA GTA ATT-3′ and IN-N1155R 5′-CAA TCC TCC GGT ACT ATC AT-3′). An internal primer (IN-N969F 5′-ATT GAC AGC TCT TCT GCT CA-3′) was used with IN-N1155R in the second nested reaction to amplify a 186-bp product. Amplification products were analyzed by agarose gel electrophoresis.
Results
Clinical findings
Other than excessive salivation in horses with oral lesions and vague signs of depression, clinical signs were not readily apparent. Horses never stopped eating, and rectal temperatures were rarely elevated above 102°F and never above 103.5°F. In horses infected with VSNJV, only 3 horses had rectal temperatures higher than 102°F: 2 ID CHIN (PI days 2 and 4 in 1 horse; PI day 2 in the other) and TONGUE (days 1–4 PI). Only 2 horses infected with VSIV had rectal temperatures higher than 102°F, and both were in the TONGUE group (PI days 6, 7, 10, 12 in 1; PI day 2 in the other).
In all, 18/24 (75%) of the horses infected with VSNJV and VSIV developed lesions (Table 1); all animals that developed lesions seroconverted. Five of 6 horses that failed to develop lesions were in the ORAL group; 4 of these horses seroconverted. In VSNJV-inoculated horses, only those in the SCAR LIP and ID CHIN groups developed secondary lesions, whereas in horses inoculated with VSIV, secondary lesions developed in horses in the SCAR LIP, ID CHIN, and TONGUE groups.
Virus isolation results for horses challenged with VSNJV and VSIV by four different routes.
Number of horses per inoculation route.
Number of horses that developed lesions.
Virus isolation results from swabs (saliva, tonsil, nasal, conjunctiva, and feces) are based on 30 virus isolation attempts (10/horse from PID 1–8, 10, and 12) for each virus/route trial. Virus isolation results from blood and plasma are based on 12 isolation attempts (4/horse from PID 1–4) for each virus/route trial.
Number of horses seroconverting (range of maximum neutralizing antibody titers observed).
Horses inoculated with VSNJV developed primary lesions at the site of inoculation as early as PI day 1 (swelling and blanching of the area due to early vesicle formation; Fig. 1) in the TONGUE group and as early as PI day 2 in the SCAR LIP (reddening, superficial necrosis, and erosion) and ID CHIN groups (serous crusting). By PI day 2, horses inoculated in the TONGUE group had large ruptured vesicles extending around the region of inoculation in which the dorsal tongue epithelium was separated from the underlying submucosa and eventually sloughed, leaving a reddened superficial ulcer with ragged margins (Fig. 2). These lesions had evidence of healing as early as PI day 4 and by PI day 12, although still reddened and eroded, were contracting and had smooth margins (Fig. 3). In 2 horses in the SCAR LIP group (Fig. 4), the primary lesion remained limited to the site of inoculation (Fig. 5), and the area was starting to heal by PI day 5; in the third horse, spreading of the primary lesion resulted in large areas of shallow ulceration on the lower lip mucosa. All 3 of these horses developed secondary lesions (vesicles that proceeded to shallow ulceration): on the mucocutaneous junction of the lower (PI day 4) and upper (PI day 8) lip of 1 horse, mucocutaneous junction of lower and upper lip (PI day 8) of another horse, and tongue (PI day 4) of the third horse (Fig. 6). All lesions had evidence of healing by PI day 12. Crusting of the skin of 2 animals in the ID CHIN group spread to involve the lip with secondary vesicular lesions developing at the mucocutaneous junction (PI day 5 1 horse; PI day 7 1 horse) (Figs. 7, 8). These lesions were healing by PI day 12. On PI day 8, 1 horse in the ORAL group developed tongue vesiculation similar to horses in the TONGUE group and was recorded as a primary lesion in Table 1; no other lesions developed in this animal.
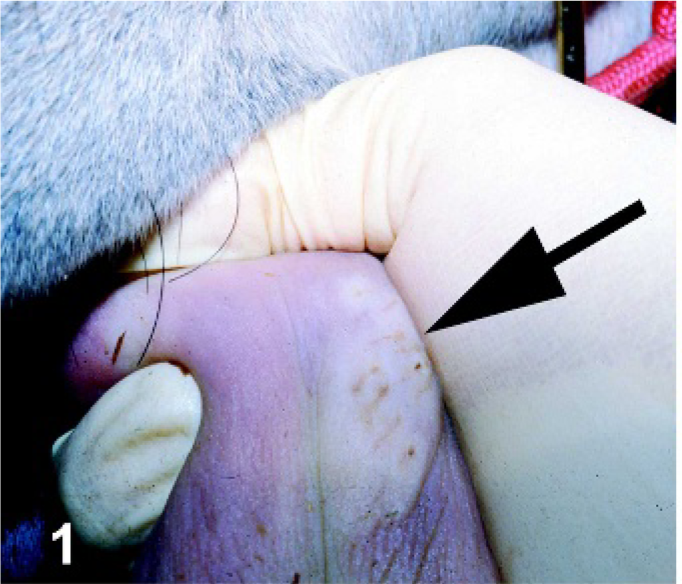
Tongue. Horse experimentally infected with VSNJV via tongue inoculation, PI day 1. Vesicle forming on the dorsal surface of the tongue at the site of inoculation.
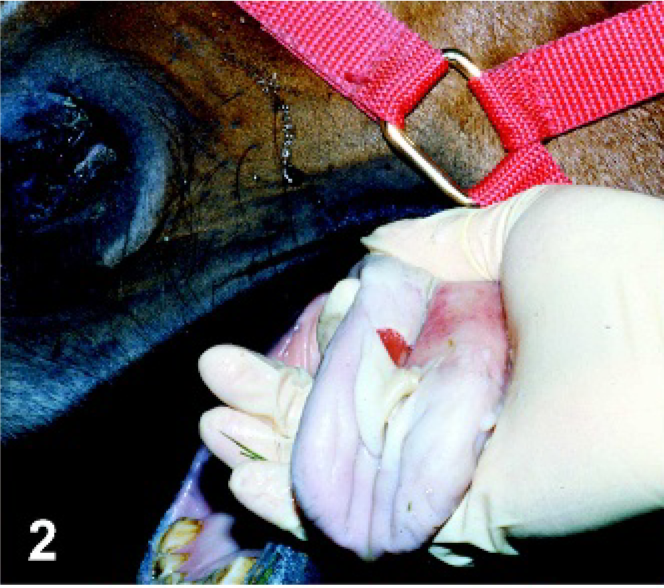
Tongue. Horse experimentally infected with VSNJV via tongue inoculation, PI day 2. Large ruptured vesicle resulting in extensive slough of the dorsal epithelium of the tongue.
Tongue. Horse experimentally infected with VSNJV via tongue inoculation, PI day 12. Note the pallor of the epithelium distal to the arrows. This area had sloughed due to extensive vesiculation and now is almost completely reepithelialized. A remnant ulcer with contracting, healing edges remains.
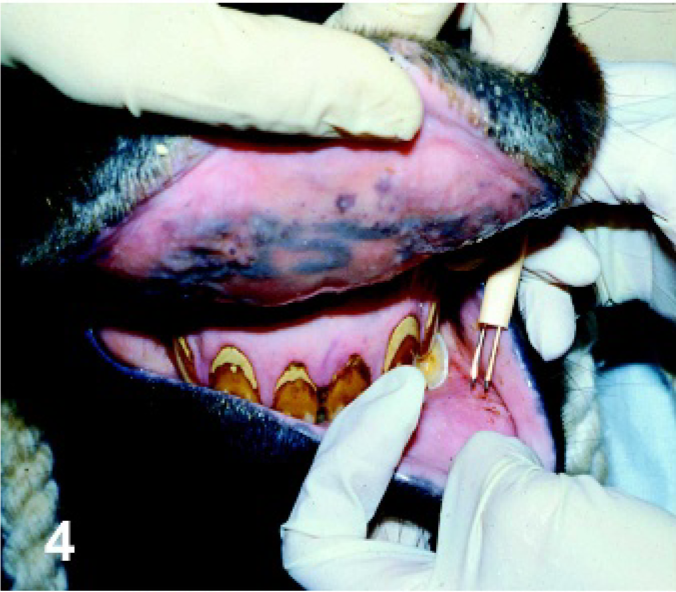
Lip. Horse. For 1 route of inoculation, a dual-needle vaccine applicator was used to scarify the mucosal surface of the lip, and virus was applied to the area.
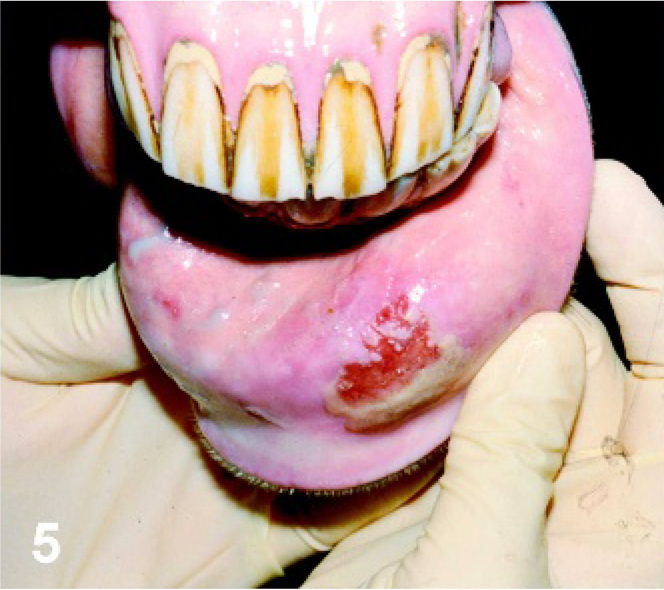
Lip. Horse experimentally infected with VSNJV via lip scarification, PI day 4. Typical eroded lesion observed at the site of scarification.
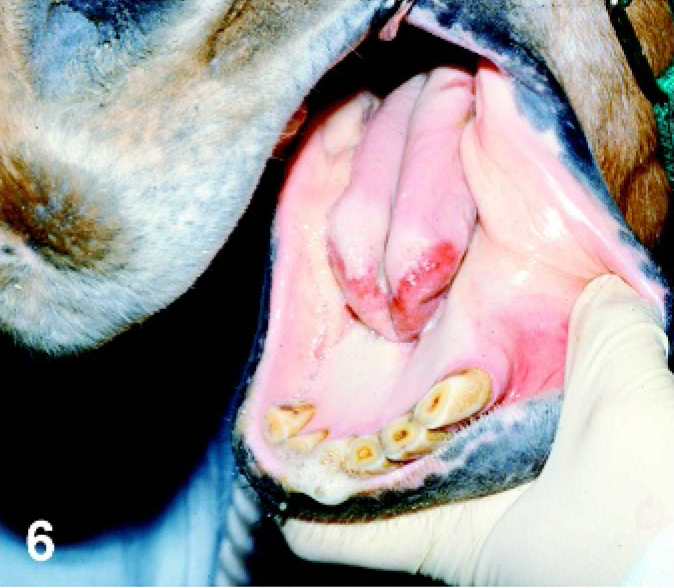
Tongue. Horse experimentally infected with VSNJV via lip scarification, PI day 4. Ruptured secondary vesicle with the loss of epithelium on the tip of the tongue.
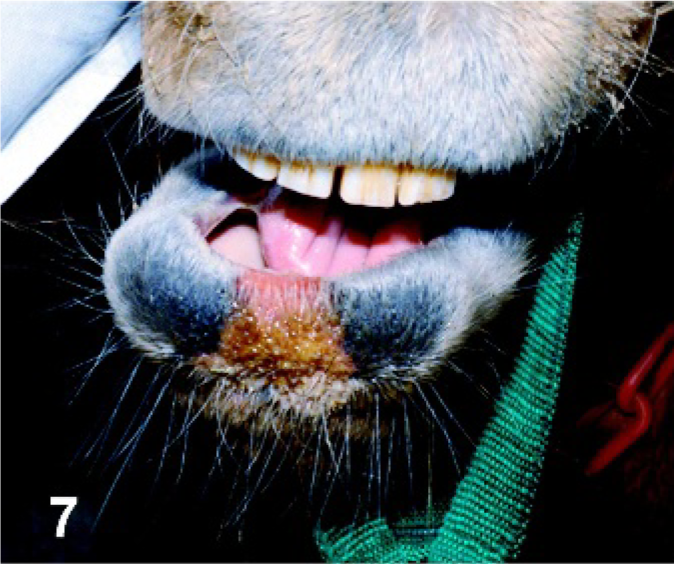
Lip. Horse experimentally infected with VSNJV via intradermal inoculation in the haired skin of the chin, PI day 6. Crusty lesion at the site of inoculation has spread to the lip.
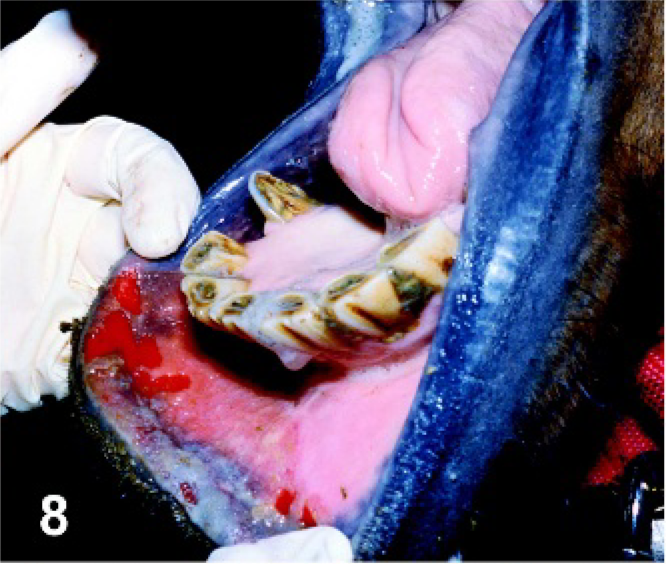
Lip. Horse experimentally infected with VSNJV via intradermal inoculation in the haired skin of the chin, PI day 8. Severe swelling of the lower lip with multiple ruptured mucosal vesicles along the mucocutaneous junction.
In VSIV-inoculated animals, lesion development was temporally similar to those infected with VSNJV although slightly different in appearance. All horses in the TONGUE group had blanched, raised areas consistent with early vesiculation in the area of inoculation on PI day 1. In 2 horses, this advanced to severe swelling and hardening of the tongue on PI day 3 and large areas of shallow ulceration on PI day 4 that did not seem as deep as those in the VSNJV-infected horses. In the third horse, vesiculation led to extensive sloughing of the dorsal mucosa of the tongue with deeper ulceration, similar to horses inoculated with VSNJV, on PI day 3 (Fig. 9). Tongue lesions started to heal on PI days 6 and 7. One horse developed a secondary lesion on the mucocutaneous junction of the left upper lip (PI day 7). Two horses in the SCAR LIP group developed lesions in the area of inoculation on PI day 2 (red or blanched raised areas suggestive of early vesiculation), which advanced to erosion on PI day 4; 1 had a secondary vesicular lesion develop on the tongue (PI day 5), and the other had secondary vesicular lesions develop on the left side of the tongue (PI day 5), mucocutaneous junction of the lower lip (PI day 7), and mucocutaneous junction of the left upper lip corresponding to the tongue lesion (PI day 8). The third horse had reddening of the site of inoculation on PI day 1 that did not develop further and had no other lesions develop. There was crusting of the area of inoculation in the ID CHIN horses by PI day 3 (Fig. 10). Secondarily, this lesion expanded to involve the lower lip in 2 horses, resulting in severe swelling and hardness (PI days 3 and 4; Fig. 11), and 1 horse developed a secondary swallow, although large, ulcer on the tip of the tongue (PI day 5). No lesions developed in the ORAL group. Lesions in all horses were healing at the time of necropsy (Fig. 12).
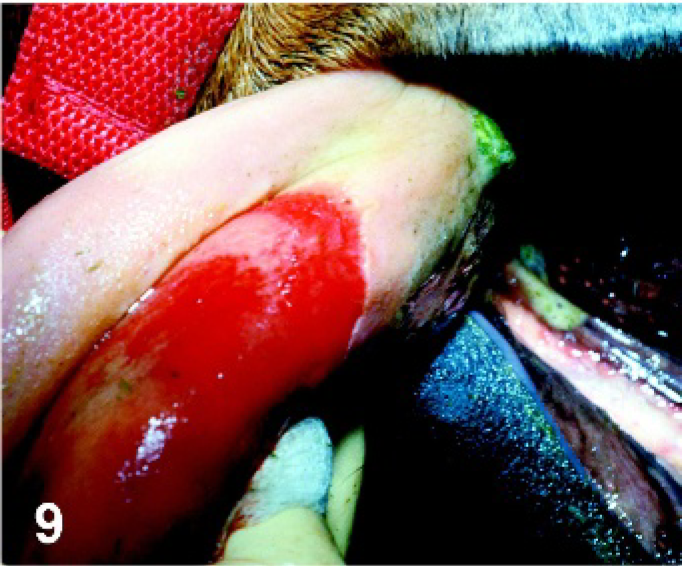
Tongue. Horse experimentally infected with VSIV via tongue inoculation, PI day 3. Ruptured vesicle with sloughing of epithelium leaving eroded base at the site of inoculation on the dorsal surface of the tongue.
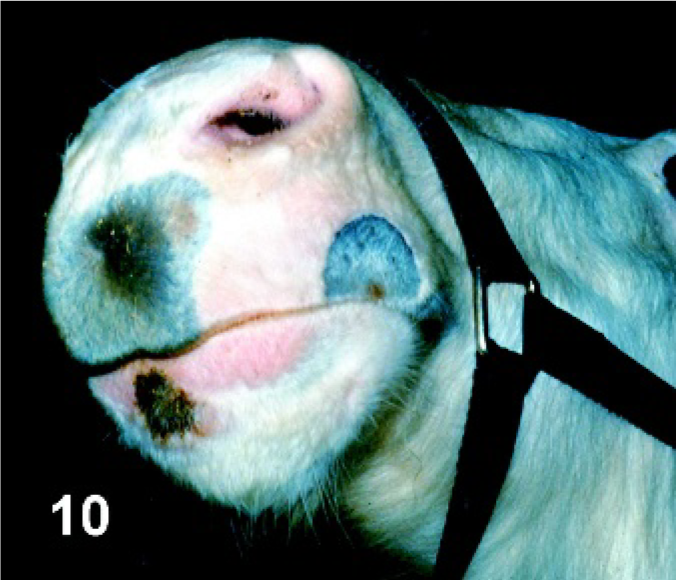
Chin. Horse experimentally infected with VSIV via intradermal inoculation in the haired skin of the chin, PI day 3. Crusty lesion at the site of inoculation on the haired skin of the chin.
Lip. Horse experimentally infected with VSIV via intradermal inoculation in the haired skin of the chin, PI day 6. Spread of the lesion to the lower lip, which is swollen and crusty (although not shown, the mucosal surface of the lip had multiple eroded areas to secondary vesiculation).
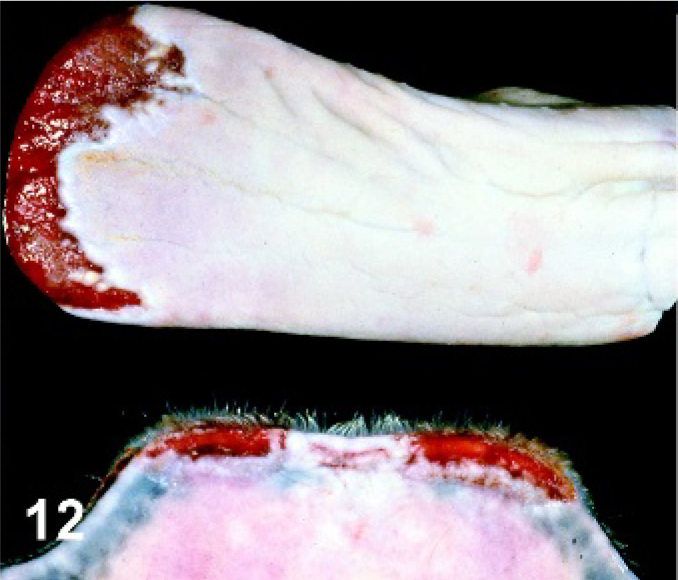
Tongue and lip. Horse experimentally infected with VSIV via scarification of lip, PI day 13. Healing secondary lesions on the mucosal surface at the mucocutaneous junction of the lower lip and along the tip of the tongue.
Necropsy findings
Horses infected with VSNJV and VSIV had similar findings at necropsy (PI days 12–15). Lesions in the area of oral mucosa scarification were healed, although the area was discolored in some animals. Lesions on the tongue (primary or secondary) or secondary ones that developed on the mucocutaneous junction of the lip, although still eroded and reddened, were healing and contracting with reduction in size and smooth margins (Figs. 3, 12). Lesions on the haired skin of the chin were hairless and depigmented, crusty, or no longer apparent. Palatine tonsils appeared enlarged in 3 animals (1 VSNJV tongue inoculated; 2 VSNJV oral scarification animals).
Histopathology and immunohistochemistry
Microscopic lesions were similar in both VSNJV- and VSIV-infected horses. Tongue lesions were fully to partially reepithelialized (primary lesions) (Fig. 13a) or superficially ulcerated (secondary lesions) (Fig. 13b) with a narrow underlying bed of neovascularization (compare to normal tongue; Fig. 13c). Submucosa underlying these areas had mild (primary lesions) to moderate (secondary lesions), mostly perivascular infiltrations of mononuclear cells that were predominantly plasma cells and lymphocytes, admixed with lesser numbers of neutrophils. Immediately adjacent to these areas, epithelium had evidence of regeneration and for at least several centimeters the submucosa had moderate, superficial, and deep perivascular infiltrations of lymphocytes and plasma cells. Underlying reepithelializing or ulcerated areas, infiltrating mononuclear cells were a mixture of cells positive for BLA.36 (Fig. 13d), CD3 (Fig. 13e), and MAC 387, while in adjacent submucosa, infiltrating cells were of approximately equal numbers of CD3- and BLA.36-positive cells. A few individual BLA.36-positive cells (Fig. 13f), some appearing to be lymphocytes and others dendritic macrophages, as well as individual and small clusters of CD3-positive cells (Fig. 13g), were present in the intact epithelium adjacent to reepithelializing or ulcerated areas. There were no CD79 staining lymphocytes in the lesions despite the presence of CD79-positive cells in the lymph node on the same slide.
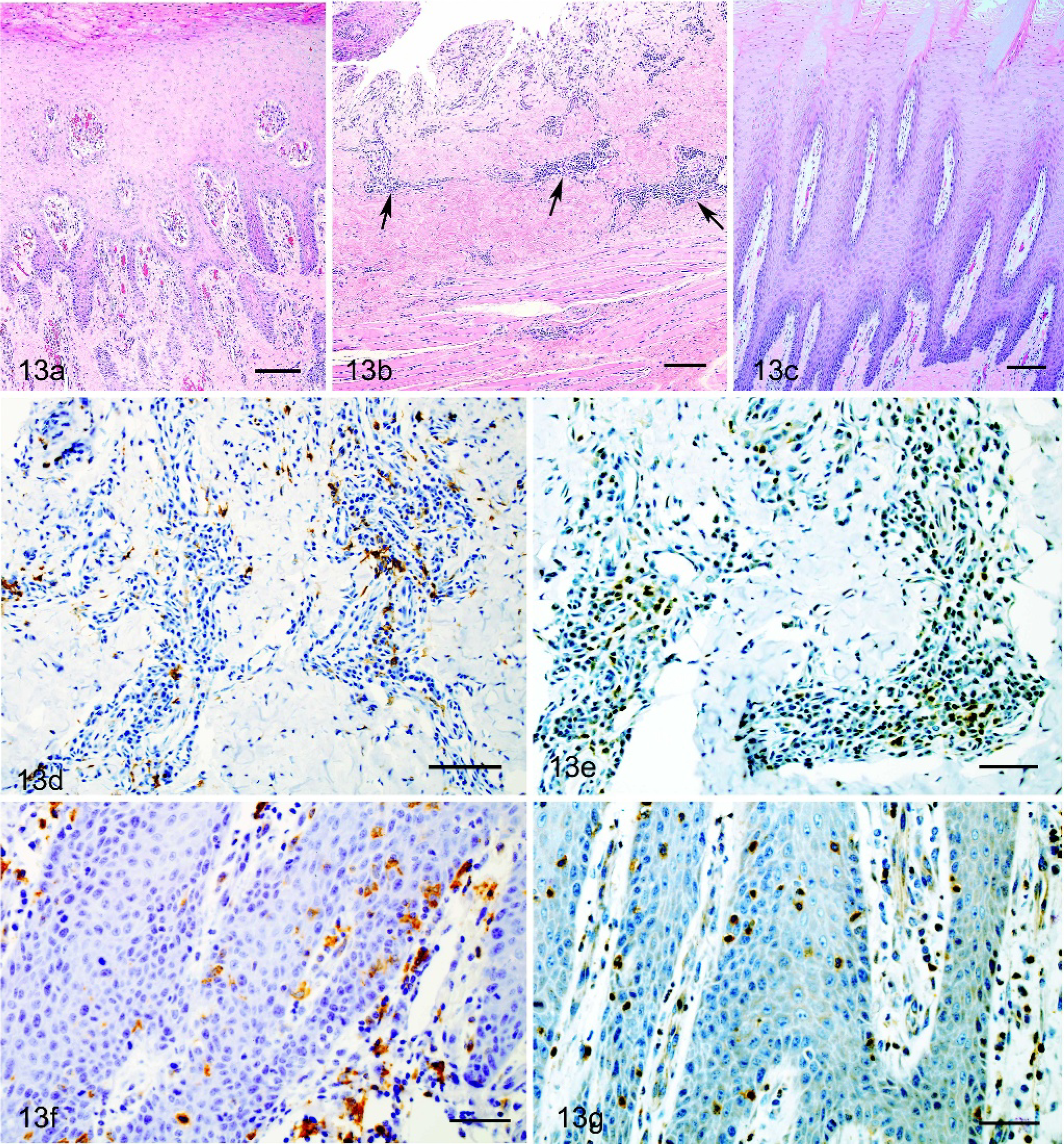
Lesions at the mucocutaneous junction of lip were similar in character and type of infiltrating cells as the tongue lesions, although ulceration was confined to the mucosa. As in the tongue, CD3- and BLA.36-positive lymphocytes were seen in adjacent intact mucosal epithelium and epidermis.
In the chin skin of animals either inoculated in the chin or in whom oral lesions spread to involve the lower lip, there was serocellular crusting and erosion that never breeched the epidermal basement membrane. Ruptured inflamed follicles and moderate perivascular infiltrations of plasma cells and mononuclear cells that were predominantly CD3- and BLA.36-positive were also present.
In horses that did not become infected or develop lesions, tongue, mucocutaneous junction of lip, and chin skin had normal epithelium with minimal perivascular infiltrations of CD3- and BLA.36-positive lymphocytes and plasma cells and no CD3- or BLA.36-positive cells were seen in the epithelium. Tonsil and lymph nodes of both horses that became infected and those that did not were similar; all animals had large secondary lymphoid follicles in these tissues. VSV antigen was not detected in lesions, tonsil, or retropharyngeal or mandibular lymph nodes of any horse.
Antibody response
Neutralizing antibodies developed in 11 of 12 horses inoculated with VSNJV and 11 of 12 horses inoculated with VSIV; both horses that failed to seroconvert were in the ORAL group (Tables 1, 2). In general, maximum neutralizing antibody titers were highest in the SCAR LIP and TONGUE horses. However, 1 ID CHIN horse (VSIV) developed the highest neutralizing titer (32,768) of all horses, probably because of extensive secondary lesions that developed in the mouth. Horses in the ORAL groups also had much lower maximum neutralizing titers than those in horses inoculated by other routes. This was most likely related to a lack of lesion development in this group because the only ORAL horse (VSNJV) that developed lesions had the highest neutralizing titer (512) of the group. Serum-neutralizing antibodies were first detected on PI day 6, 7, 8, or 12, depending on the virus and inoculation group, and increased over time (Fig. 14). For both VSNJV and VSIV, animals inoculated by needle or scarification developed antibodies sooner than did those inoculated per os. In general, for all routes of inoculation, animals inoculated with VSNJV developed antibodies prior to animals inoculated with VSIV. By PI day 15, ORAL animals still had titers that were minimal compared with those on PI day 12 in animals inoculated by other routes.
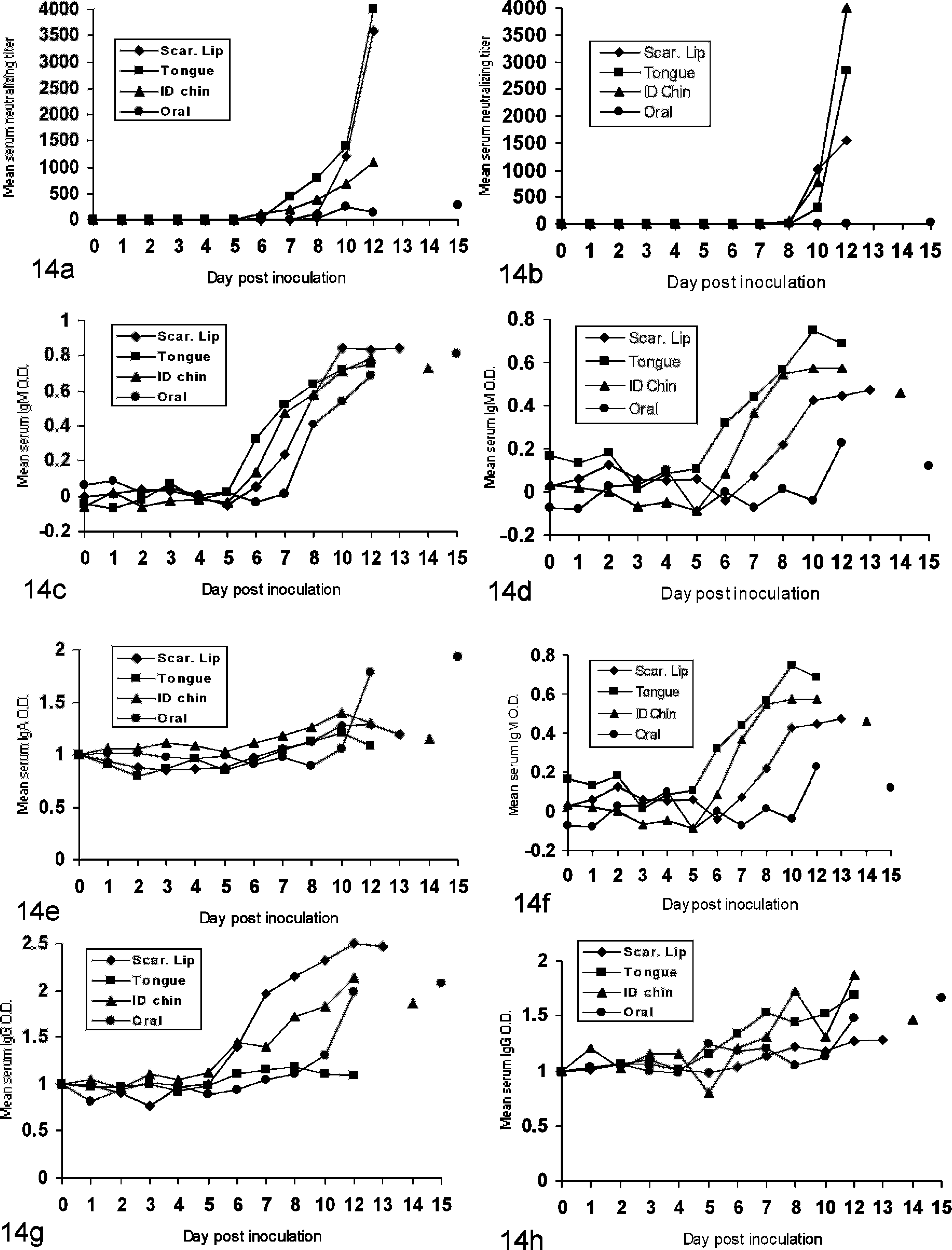
Mean serum-neutralizing antibody titers and mean serum IgM, IgG, and IgA optical densities by serotype and route of inoculation. Horses inoculated with VSNJV
Duration of viral shedding and viral titers observed from swab samples collected from horses infected with VSNJV and VSIV.
Duration of viral shedding based on consecutive days of virus isolation for each horse (number of horses from which virus was detected).
Viral titers expressed as log10 TCID50 /swab.
Mean serum ODs of virus-specific IgM and IgG in horses that developed neutralizing antibody, by inoculation group and serotype, are given in Fig. 14. In horses inoculated with both VSNJV and VSIV, significant increases in the mean IgM and IgG ODs occurred in all inoculation groups and roughly coincided with the development of neutralizing antibody. Mean OD increases for IgM and IgG were temporally similar, but the rapidity and magnitude of increase were faster and greater, respectively, for IgM. For both viruses, initial increases in mean ODs varied slightly between inoculation groups, with significant increases occurring later for the ORAL groups. Only horses that had neutralizing antibody developed virus-specific IgM and IgG. Of these, only 1 failed to develop detectable IgM; all developed detectable IgG. The horse that failed to develop detectable IgM was in the VSNJV ORAL group and had developed only a very low neutralizing titer (32) by PI day 15.
Mean serum ODs of virus-specific IgA in horses that developed neutralizing antibody are given in Fig. 14 by inoculation group and serotype. For both viruses, detectable virus-specific circulating IgA developed in all inoculation groups, but only horses that developed neutralizing antibody had detectable virus-specific IgA. Of the 11 seropositive VSNJV-infected animals, only 7 (TONGUE 1/3; SCAR LIP 2/3; ID CHIN 2/3; ORAL 2/2) developed detectable IgA, with significant increases first seen on PI day 6 (TONGUE), PI day 10 (SCAR LIP), PI days 7 and 10 (ID CHIN), and PI day 12 (ORAL). Eight of 11 seropositive VSIV-infected animals (TONGUE 1/3; SCAR LIP 3/3; ID CHIN 2/3; ORAL 2/2) developed detectable IgA, with significant increases first seen on day 10 (TONGUE), PI days 8 and 10 (SCAR LIP), PI days 10 and 12 (ID CHIN), and PI days 12 and 15 (ORAL). The SCAR LIP and ID CHIN animals that failed to develop circulating IgA had mild or no oral lesions and low neutralizing titers.
Virus isolation
Virus shedding occurred in all inoculation groups (Tables 1, 2). In both VSNJV- and VSIV-infected horses, and for all routes of inoculation, virus was most frequently isolated and in highest titer from swabs of saliva from the oral cavity or from swabs taken from the tonsillar region (Tables 1, 2). Virus was less frequently isolated from nasal cavity and only from horses with oral lesions. Isolation from the conjunctival sac was rare and only from horses inoculated with VSNJV. Virus was never isolated from feces, blood, or plasma. In both VSNJV- and VSIV-infected horses, virus shedding was most frequent, more protracted, and typically of higher titer in TONGUE and SCAR LIP groups (Tables 1, 2). Virus was detected, albeit in low titer (<2.9), in swabs of the tonsillar area in 5 horses that did not develop lesions (1 ORAL, VSNJV; 3 ORAL, VSIV; 1 ID CHIN, VSIV) and from 2 horses without oral lesions (both ID CHIN, 1 VSNJV and 1 VSIV).
Viral shedding occurred as early as PI day 1 or 2 in both VSNJV- and VSIV-infected animals (Fig. 15). For all inoculation groups, most viral shedding occurred during the first 6 days PI and had ceased by PI day 10 in all animals (Fig. 15). Development of serum-neutralizing antibodies coincided with the cessation of virus shedding (Fig. 16). In VSNJV-inoculated horses, virus was isolated from 110 of 400 virus isolation attempts (from saliva, tonsil, nasal, conjunctival, and fecal swabs) preseroconversion but only 13 of 200 virus isolation attempts postseroconversion. Of these 13, 11 isolations were made on the same day as seroconversion and in the remaining 2 on the day after detection of seroconversion. For VSIV, virus was isolated from 105 of 485 virus isolation attempts preseroconversion and 0 of 115 virus isolation attempts postseroconversion. Virus was not isolated from retropharyngeal lymph node, mandibular lymph node, tonsil, or residual lesion tissue obtained at necropsy on PI days 12 to 15.
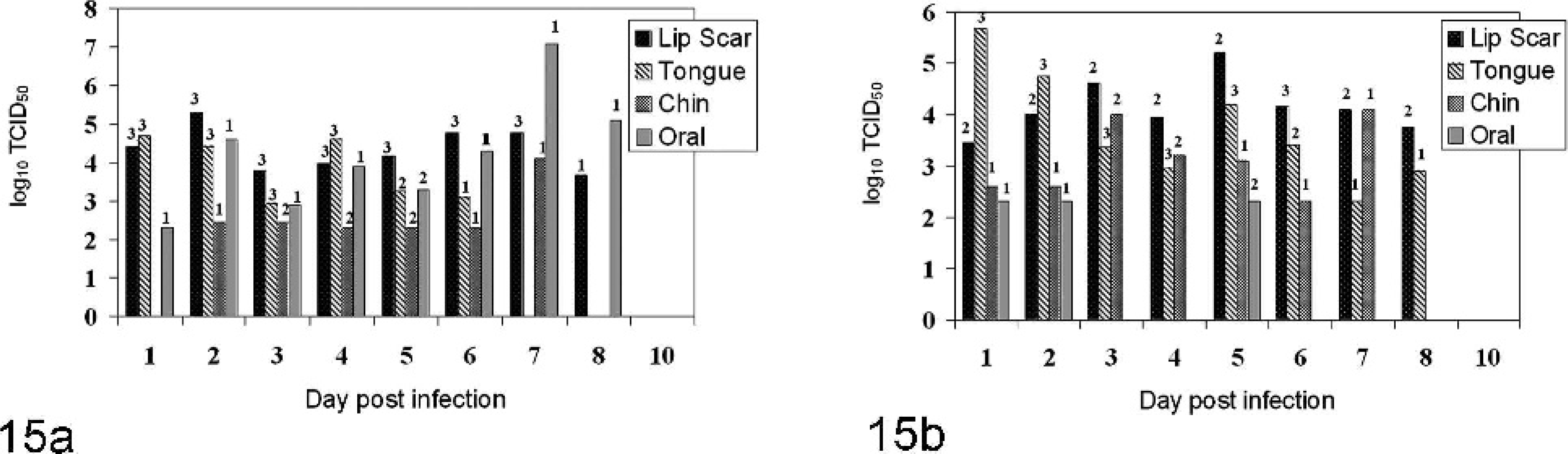
Average maximum viral titers observed by postinoculation day and route of infection for horses inoculated with VSNJV
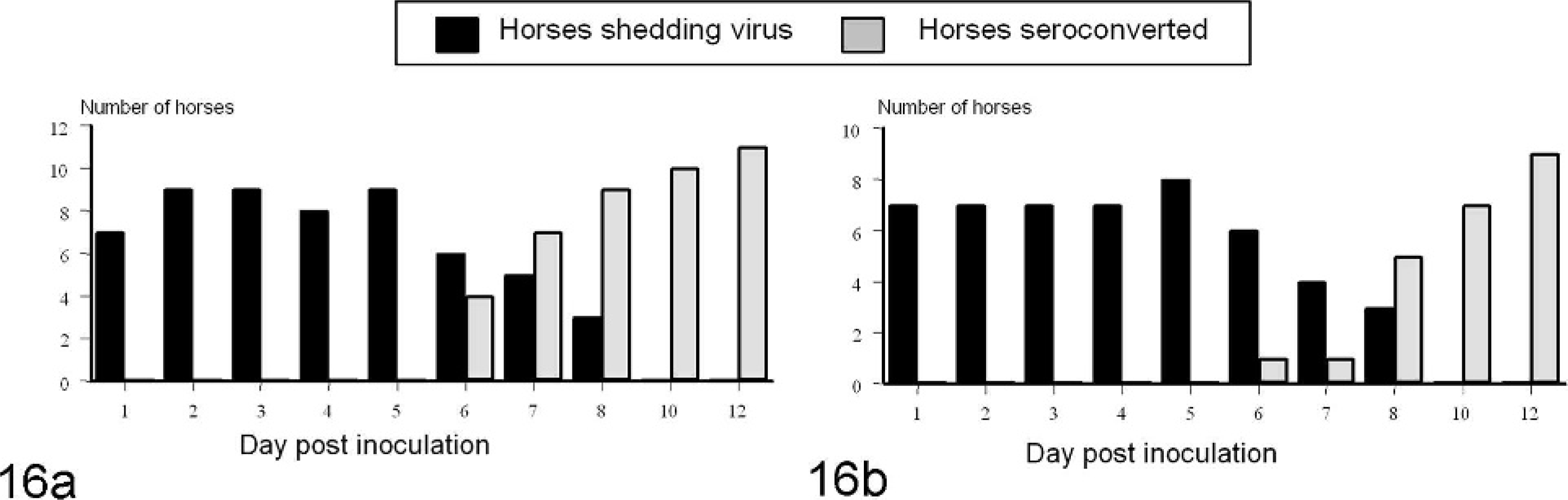
Temporal development of viral shedding and seroconversion for horses infected with VSNJV
RT-PCR
Viral RNA was not detected in the whole blood of any animal infected with VSNJV. Viral RNA was detected in the blood of 4 horses inoculated with VSIV, 3 TONGUE (PI days 2, 3; PI days 2–4; PI day 4) and 1 SCAR LIP (PI day 3), and was detected in 3 of 7 corresponding serum samples. Most VSNJV- and VSIV-infected horses had detectable viral RNA in tonsil and/or retropharyngeal lymph node collected at necropsy (Table 3); no animal that failed to seroconvert or shed virus had positive RT-PCR results. Not all animals with RT-PCR–positive tonsils had positive lymph nodes and vice versa.<1?tlsb?>
RT-PCR results for horses challenged with VSNJV and VSIV by 4 different routes.
Number of horses per inoculation route.
Discussion
We were able to infect horses with VSNJV and VSIV by routes simulating contact (TONGUE, SCAR LIP, ORAL) or vector transmission (ID CHIN); findings for each route were similar between the 2 viruses. Primary lesions at the site of inoculation, or secondary oral lesions, and increased salivation, which has often been associated with VS in horses, 7 were the principal clinical findings. Primary lesions developed rapidly at the site of inoculation, but true vesicular lesions only developed in the oral cavity. Secondary lesions also developed rapidly (within 8 days PI) and were often more obvious than the primary lesion but were confined to the lips and oral cavity. Because significant lesions were confined to the mouth, VS may go undetected in horses infected under natural conditions who may then serve as an undetected source of virus for contact transmission. This could also help explain reports of the apparently subclinical cases. 10, 12 However, it is important to note that true subclinical cases did occur in this study, because only 82% of horses that became infected developed lesions. Subclinical cases also occurred in experimentally infected pigs, especially when virus was inoculated away from the oral cavity or coronary band. 8, 15 Thus, in a natural situation in which viral inoculation may occur at sites other than in or around the oral cavity, the number of subclinical cases could potentially be higher than in this study.
Applying virus to superficial breaks or direct viral inoculation of oral mucous membranes was most effective in producing lesions and was associated with more frequent and higher titered viral shedding, most likely due to the development of vesicular lesions, similar to what is seen in experimental studies in swine. 8, 15 Only 1 horse inoculated per os developed lesions (vesicular lesions of the tongue). This particular horse was aged and had severe dental attrition and enamel points that could have caused lacerations of the tongue, potentiating the development of oral lesions. Thus, during outbreaks, trauma to the oral mucosa caused by poor roughage and tooth care could increase the possibility of infection.
As seen in experimentally infected swine, vesicular lesions did not develop at the site of inoculation in haired skin. 8, 15 However, in these horses, when the infection spread from the haired skin of the chin to the nonhaired areas of the lip, vesiculation and quite extensive lesions developed in the mucosa of the lip. Thus, there appear to be unique features of nonhaired skin that allow the development of vesicles.
Both primary and secondary lesions healed rapidly and within 2 weeks of infection were reduced to healing erosions and ulcers that did not contain virus or viral antigen. Thus, virus isolation and immunohistochemistry would not be rewarding in confirming a diagnosis at this phase of the disease. However, tonsil and lymph nodes from the head contained viral RNA at this time, and RT-PCR might be useful in making a diagnosis.
Virus shedding was common from the oral cavity and reached titers that have been shown to orally infect swine 5 and were very close to the titers shown by this study to infect horses per os. This coupled with excessive salivation suggests that during an outbreak contact transmission via contaminated food and water is likely partially responsible for the spread of disease. Although in many horses this oral shedding can be attributed to oral lesions, shedding also occurred in animals without detectable oral lesions, and, because viral RNA was found in palatine tonsil without oral lesions, we speculate that replication in tonsils is the source of some viral shedding, similar to what has been shown in pigs. 15 Horses have abundant tonsillar tissue surrounding the oral cavity, including the palatine tonsil (lateral to the tongue and behind the palatoglossal arch), lingual tonsil (at the base of the tongue), tubular tonsil (around the auditory tube), and pharyngeal tonsil (on the dorsal wall of the pharynx), all of which might contribute to oral shedding of the virus.
Shedding from the nasal cavity could also contribute to environmental contamination. Shedding could indicate replication in the nasal mucosa; however, because titers were relatively low, we believe this virus originated from either the oral cavity or replication in the tonsils.
Fecal shedding has been occasionally documented in swine experimentally infected with VSV 15 but was not seen in these horses. These viruses apparently do not replicate in the intestinal mucosa of horses, or swallowed virus from the oral cavity is inactivated or diluted beyond detection in the equine digestive tract. Thus, virus contamination of the environment via feces is unlikely to occur.
Although viral RNA was detected in the blood of horses infected with VSIV, virus was never detected in blood by virus isolation. Failure to detect virus in blood via isolation is consistent with experimental studies in swine 5, 8, 13, 15 , and, to date, viremia sufficient to infect biological insect vectors has not been documented in any domestic animal infected with VSNJV or VSIV.
Failure to detect viral RNA in the blood of VSNJV-infected horses may be due to differences in sensitivity of the RT-PCR assays for the 2 viruses rather than differences in viremia. Although the presence of detectable viral RNA in the blood of VSIV-infected horses in this study does not necessarily indicate the presence of infectious virus, it could indicate that viremia below levels detectable by virus isolation occurs in VS. Although low-level viremia probably is not directly involved in the transmission of disease, this finding coupled with RT-PCR detection of viral nuclei acid in tonsil and oral shedding of virus in ID CHIN horses that failed to develop oral lesions suggests that virus can spread from primary sites of inoculation in the skin to tonsil via low-level viremia with subsequent tonsillar replication, resulting in oral shedding without the development of oral lesions. Such a pathogenesis would also explain why swine experimentally inoculated in haired skin of the ear shed virus from the oral cavity without the development of oral lesions. 8, 15
Neutralizing antibodies developed rapidly after inoculation via routes simulating both contact and vector transmission and coincided with the rapid cessation of virus shedding. Similar results are reported for experimental studies in swine 13, 15 and suggest that transmission risks associated with seropositive animals are minimal. The risk of contact transmission in swine was dependent on the presence of vesicular lesions. 15, 16 Results from horses are consistent with this possibility; highest viral titers were associated with horses with vesicular lesions. These potential relationships have important implications to control strategies and should be further investigated.
VSV-specific IgM, IgG, and IgA antibodies developed in both VSNJV- and VSIV-infected animals. There was a rapid specific IgM and IgG response in all animals that developed neutralizing antibody, in animals with and without lesions. Temporal development was similar to other reports in experimentally infected animals. 9, 17, 18 Specific IgA antibodies also rapidly developed but not in all animals. In some animals, failure to develop IgA appeared to be related to lack of oral lesion development. On the other hand, very few of the TONGUE group animals developed IgA, although they developed extensive vesicles on the tongue and shed virus from the oral cavity. Thus, it appears that route and/or location of inoculation determines the development of IgA. The role of both circulating and secretory IgA in the pathogenesis of this disease warrants further study.
Little is known about the pathogenesis of VS in horses. Although animals were not sequentially killed and examined during the first week of this study, we can offer a partial pathogenesis based on our findings. We hypothesize that primary replication occurs in tonsils after oral inoculation or within the epithelium at the site of inoculation with subsequent spread, via low-titer viremia or via saliva if oral lesions develop, to tonsils. Primary or secondary replication in tonsils results in viral shedding into the oral cavity, where secondary lesions may develop if there are breaks in the oral mucosa. There is subsequent drainage to or infection of lymph nodes of the head from oral lesions, and rapid resolution of the lesions probably occurs due to both the rapid development of neutralizing antibodies and local immunity, as evidenced by the appearance of lymphocytes (both T and B cells) and plasma cells in the epithelium and submucosa/dermis surrounding lesions.
In conclusion, the routes of inoculation used in this study can be used to further study contact and vector transmission and vaccine development and to clarify the pathogenesis of the disease in horses.
Footnotes
Acknowledgement
Supported in part by funds from the Grayson-Jockey Club Research Foundation.
