Abstract
The pathogenesis of vesicular stomatitis virus (VSV) infection has not been investigated previously in native New World rodents that may have a role in the epidemiology of the disease. In the present study, 45 juvenile and 80 adult deer mice (Peromyscus maniculatus) were inoculated intranasally with VSV New Jersey serotype (VSV-NJ) and examined sequentially over a 7-day period. Virus was detected by means of immunohistochemistry and in situ hybridization in all tissues containing histologic lesions. Viral antigen and mRNA were observed initially in olfactory epithelium neurons, followed by olfactory bulbs and more caudal olfactory pathways in the brain. Virus also was detected throughout the ventricular system in the brain and central canal of the spinal cord. These results support both viral retrograde transneuronal transport and viral spread within the ventricular system. Other tissues containing viral antigen included airway epithelium and macrophages in the lungs, cardiac myocytes, and macrophages in cervical lymph nodes. In a second experiment, 15 adult, 20 juvenile, and 16 nestling deer mice were inoculated intradermally with VSV-NJ. Adults were refractory to infection by this route; however, nestlings and juveniles developed disseminated central nervous system infections. Viral antigen also was detected in cardiac myocytes and lymph node macrophages in these animals. Viremia was detected by virus isolation in 35/72 (49%) intranasally inoculated juvenile and adult mice and in 17/36 (47%) intradermally inoculated nestlings and juveniles from day 1 to day 3 postinoculation. The documentation of viremia in these animals suggests that they may have a role in the epidemiology of vectorborne vesicular stomatitis.
Keywords
Vesicular stomatitis (VS) is a disease of horses, cattle, and swine caused by related viruses in the genus Vesiculovirus of the family Rhabdoviridae. Disease most often is caused by one of two serotypes, VS virus (VSV) New Jersey (VSV-NJ) or VSV Indiana (VSV-I). Outbreaks of VS occur in the western United States, northern Mexico, and temperate regions of South America. Enzootic foci of VS exist in northern South America, throughout Central America, and at one site in the United States: Ossabaw Island, Georgia. 2,4,38,42 Clinical outbreaks of VS in livestock are associated with significant economic impact on producers and extensive regulatory responses by governmental agencies, including trade restrictions, market closures, and quarantines. 6,18 Such measures are mandated because of the clinical similarities between VS and foot-and-mouth disease and other foreign vesicular diseases in livestock species.
Despite these economic and regulatory impacts, little is known about the epidemiology and pathogenesis of VS in livestock or other animals. Viral reservoirs and amplification hosts are unknown, and routes of transmission in both enzootic and epizootic sites of VS are poorly defined. Insect biologic vectors capable of transmitting VSV-NJ and VSV-I have been identified, and transovarial transmission has been documented in some of these vectors. 12,39 However, in experimental insect infections, transovarial transmission was inefficient. 12 In addition, viremia associated with VSV-NJ or VSV-I infection has not been demonstrated in either domestic or wild ungulate species, 3,10,11 and currently it is not understood how insect vectors are initially infected with these viruses.
Rodents are the only known vertebrates that consistently develop a detectable viremia following VSV-NJ and VSV-I infection. Viremia associated with experimental infection with VSV-NJ or VSV-I has been demonstrated in deer mice (Peromyscus maniculatus), 41 laboratory mice (Mus musculus), 15 spiny rats (Proechimys semispinosus), 40 and Syrian hamsters (Mesocricetus auratus). 17 Furthermore, natural exposure to VSV-NJ and VSV-I has been demonstrated by detection of serum neutralizing antibodies in native New World rodents at both enzootic and epizootic sites of VS. 1,21,33,41
The pathogenesis of VSV-NJ and VSV-I infection in laboratory rodents has been examined. However, similar studies have not been performed with native New World rodents that are more likely to have a role in the epidemiology of VS. Most investigations in laboratory rodents have concentrated on the neuropathogenesis and immunopathogenesis of wild-type, temperature-sensitive mutant, and defective interfering particle viruses in mice. 5,13,14,31 Comprehensive studies examining the pathogenesis of infection with field isolates of VSV in laboratory rodents are few. 7,8,32 The present study was undertaken to investigate the pathogenesis of VSV-NJ infection in the deer mouse. Specific objectives of this research were to examine the pathogenesis of both intranasal and intradermal VSV-NJ infection. Virus isolation, histology, immunohistochemistry, and in situ hybridization were used to follow the course of VSV-NJ infection in deer mice over time.
Materials and Methods
Viruses
Two different field isolates of VSV-NJ were used. The first virus was isolated from a sand fly (Lutzomyia shannoni) from Ossabaw Island, Georgia. 9 This virus was passaged once in Vero cells, aliquoted, and stored at −70 C. The second virus was originally isolated in 1995 from a horse in Mesa County, Colorado, and was provided by the National Veterinary Services Laboratories (NVSL), US Department of Agriculture (NVSL accession No. 95-44625). This virus also was passaged once in Vero cells, aliquoted, and stored at −70 C in our laboratory. Before use, both viral isolates were quantified by end-point titration in Vero cells as previously described. 37
Animals
Juvenile (≤28 days of age) and adult (>42 days of age) deer mice were purchased from the Peromyscus Stock Center, University of South Carolina (Columbia, SC). When nestling (≤10 days of age) mice were required, 10 male/female pairs of deer mice were placed into breeding cages, and the offspring produced were used. Before any experimental work all mice were tested for serum neutralizing antibodies to VSV-NJ as previously described. 37
In the first experiment, 50 adult and 50 juvenile mice (sex matched) were used. Mice were anesthetized with ketamine (100 mg/kg) and xylazine (10 mg/kg), and 90 mice were inoculated intranasally with 1 × 106 median tissue culture infective doses (TCID50) of VSV-NJ Ossabaw in 50 µl of minimum essential medium (MEM). Ten mice served as controls and received MEM without virus. Groups of 10 mice (5 juveniles and 5 adults) were euthanatized by CO2 overdose at the following times postinoculation (PI): 6 hours, 12 hours, and days 1–7. Control mice were euthanatized on day 7 PI. Necropsies were performed, and serum, brain, cervical spinal cord, heart, lung, cervical lymph node, liver, spleen, kidney, adrenal gland, stomach, small intestine, cecum, colon, gonads, and nasal cavities were collected.
In a continuation of the first experiment, 35 adult deer mice were inoculated intranasally with 1 × 106 TCID50 of the Colorado VSV-NJ isolate in 50 µl of MEM. Five adult mice served as controls and received MEM without virus. Five mice were euthanatized daily on days 1–7 PI, and tissue samples were collected at necropsy as described above. Control mice were euthanatized on day 7 PI.
In the second experiment, 16 nestling, 20 juvenile, and 15 adult mice were inoculated intradermally following anesthesia. Each mouse received 1 × 106 TCID50 of VSV-NJ Colorado in 8 µl of MEM injected on the dorsal surface of the tail using a 10-µl glass syringe with supplied needles (TekLab, Baton Rouge, LA). Two juvenile and two adult mice served as controls and received MEM without virus intradermally. Four nestling mice were euthanatized daily on days 1–4 PI, four juvenile mice were euthanatized daily on days 1–5 PI, and tissues were collected at necropsy as described above. Blood was collected from five adult mice daily on days 1–3 PI to examine for viremia, and five adult mice were then euthanatized on days 4, 6, and 20 PI. Tissues were collected from the adults at necropsy as described above.
Virus isolation
Virus isolation attempts from serum were performed as previously described 36 using 25 µl of serum. Titration of positive virus isolations was performed by end-point titration as previously described. 37
Histology
Tissues were fixed in formalin for 24–48 hours and then embedded in paraffin. Nasal cavities were decalcified in 5% formic acid for 5 days prior to sectioning and embedment. Sections were cut at 3 µm and stained with hematoxylin and eosin.
Immunohistochemistry
Immunohistochemical examination of formalin-fixed, paraffin-embedded sections for VSV-NJ was performed using an alkaline phosphatase–conjugated streptavidin–biotin technique as previously described. 36 Tissues were sectioned at 3 µm, mounted on slides, and deparaffinized. Sections were blocked with 0.05% casein for 2 minutes, rinsed, and incubated for 1 hour at room temperature with a 1:1,500 dilution of anti–VSV-NJ hyperimmune mouse ascitic fluid (Dr. R. B. Tesh, Department of Pathology, University of Texas Medical Branch, Galveston, TX). Sections were washed, and biotinylated anti-immunoglobulin multilink and alkaline phosphatase–conjugated streptavidin label (Biogenex, San Ramon, CA) were used sequentially. Fast red (Biogenex) was used as the chromagen/substrate.
Double-label immunohistochemical examination of nasal epithelium for calretinin (an olfactory receptor neuron antigen) and VSV-NJ was performed, with modifications of techniques previously described. 22 Formalin-fixed, paraffin-embedded nasal cavities were sectioned at 3 µm, mounted on slides, deparaffinized, and flooded with 0.01 M citrate buffer (pH = 6.0) for 30 minutes in a steamer for antigen retrieval. Sections were rinsed and then incubated with a 1:1,000 dilution of rabbit anti-calretinin (Swiss Antibodies, Bellinzona, Switzerland) for 3 days at 4 C in a humid chamber. Following incubation, sections were washed, and biotinylated anti-immunoglobulin multilink (Biogenex) and peroxidase-conjugated streptavidin label (Vector Laboratories, Burlingame, CA) were added sequentially. Substrate was diaminobenzidine. Following development of desired staining, sections were washed and VSV-NJ immunohistochemistry was performed as described above.
Immunohistochemical examination was performed on lymph node and lung sections to aid in identification of cell types with positive VSV-NJ immunoreactivity. Sections were prepared as described above and treated with 0.01 M citrate buffer (pH 6.0) for 30 minutes in a steamer for antigen retrieval. Sections were rinsed and incubated with a 1:100 dilution of Mac-387 anti-myeloid/histiocyte antibody (DAKO, Carpinteria, CA) for 2 hours at room temperature. Following incubation, sections were washed, and biotinylated anti-immunoglobulin multilink (Biogenex) and peroxidase-conjugated streptavidin label (Vector Labs) were added sequentially. Diaminobenzidine was used as substrate. Negative controls for all immunohistochemical procedures consisted of exclusion of primary antibodies from tissue sections.
In situ hybridization
An 804-base pair segment of the VSV-NJ Ossabaw isolate nucleocapsid gene was generated using a reverse transcription polymerase chain reaction method as previously described. 36 This gene segment was cloned and amplified in a pGEM-4Z vector (Promega, Madison, WI) following the manufacturer's instructions. The vector was digested with the appropriate restriction enzyme, and digoxigenin-labeled nucleotides were added during transcription of the antisense (mRNA complementary) probe. Incorporation of digoxigenin was verified using dot blots. Formalin-fixed, paraffin-embedded tissue sections were cut at 3 µm, deparaffinized, rehydrated, and digested with 5 µg/ml Proteinase K for 15 minutes. Hybridization was performed overnight at 42 C with approximately 25 ng of probe per slide in a hybridization solution consisting of 5× standard saline citrate, 50% formamide, 5% blocking reagent (Boehringer Mannheim, Indianapolis, IN), 1% N-laurylsarcosine, and 0.02% sodium dodecyl sulfate. Stringent washes were performed with decreasing concentrations of standard saline citrate at 50 C and then at room temperature. Bound probe was detected using alkaline phosphatase–conjugated anti-digoxigenin antibody followed by chromagen/substrate nitroblue tetrazolium (NBT) and 5-bromo-4-chloro-3-indolylphosphate (BCIP).
Results
Virus isolations from serum
Virus isolations from serum for both experiments are summarized in Fig. 1. In the first experiment, viremia was detected in 35/72 (49%) juvenile and adult mice inoculated intranasally. Viremia was detected from day 1 to day 3 PI in these mice, and the mean serum titer for all mice inoculated intranasally was 2.4 TCID50/ml. Development of viremia was not dependent upon viral isolate: both the Ossabaw and Colorado viruses produced viremia in 49% of adults inoculated.
The number of deer mice becoming viremic over time following inoculation with VSV-NJ, by age and route of inoculation.
In the second experiment, viremia was not detected from day 1 to day 20 PI in any adult mice inoculated intradermally. In contrast, viremia was detected from day 1 to day 3 PI in 10/16 (63%) nestlings and 7/20 (35%) juveniles inoculated intradermally. The mean serum titer was 3.6 TCID50/ml for nestlings and 2.8 TCID50/ml for the juveniles.
Histology
Intranasal inoculation. Histologic lesions were consistently observed in the nasal turbinates of all juvenile and adult mice inoculated by the intranasal route, independent of viral isolate. Lesions were confined to regions of olfactory epithelium, sparing respiratory epithelium, and were first observed on day 2 PI. Initial lesions consisted of swelling and vacuolar change of olfactory epithelial cells followed shortly by necrosis of the olfactory epithelial cells with edema and minimal infiltrates of mononuclear cells and neutrophils in the mucosal propria. By days 3 and 4 PI, lesions had progressed to more intense necrotizing rhinitis involving olfactory epithelium and Bowman's glands, with exudation of neutrophils and fibrin into the nasal passages. By day 6 PI, many mice developed purulent rhinitis with erosion and ulceration of olfactory mucosa and filling of the nasal passages with degenerate neutrophils mixed with cellular debris. These lesions were occasionally unilateral but more commonly were observed bilaterally.
Bilateral histologic lesions were first observed in the olfactory bulbs on day 2 or day 3 PI and consisted of patchy necrosis of neurons in the glomerular, granular, and internal plexiform layers. By day 5 PI, there was more widespread neuronal necrosis within the glomerular and plexiform layers of the olfactory bulb, with multifocal microgliosis, astrocytosis, and reactive changes in the endothelium of small blood vessels. Necrosis of ependymal cells lining the olfactory ventricle was first observed on day 2 PI and was a consistent feature in all olfactory bulbs examined by day 4 PI.
Histologic lesions in the brain caudal to the olfactory bulbs were first observed on day 4 PI. The initial lesions consisted of patchy necrosis of the ependymal cells lining the third and lateral ventricles. By day 5 or day 6 PI, individual neuronal necrosis or necrosis of small groups of neurons was observed in many regions of the brain, including all periventricular neuropil surrounding the lateral, third, and fourth ventricles. Additionally, patchy neuronal necrosis was consistently observed in the anterior olfactory nuclei and olfactory tubercles. Less consistently, neuronal necrosis was observed in the pyriform cortex, the multifocal thalamic nuclei, the molecular and pyramidal layers of the hippocampus, the dentate gyrus, the multifocal hypothalamic nuclei, the septal complex, the amygdaloid nuclei, and various nuclei in the brain stem (pons), including the raphe nuclei, solitary tract nuclei, hypoglossal nuclei, and locus ceruleus.
In the cervical spinal cord, ependymal cell necrosis of the central canal was first observed on day 4 PI and was followed by necrosis of neurons in the adjacent neuropil by day 5 PI. By day 6 PI, necrosis of multifocal neurons in the dorsomedial and ventromedial ventral horns was observed, and by day 7 PI, necrosis of neurons throughout the gray matter of the dorsal and ventral horns was observed. A few mice had dense glial nodules within the white matter of the spinal cord adjacent to gray matter of the ventral horn containing necrotic neurons. Rare eosinophilic intracytoplasmic inclusion bodies, similar to Negri bodies, were observed in neuronal cell bodies of the ventral horn in several mice. Lesions observed in the central nervous system were similar for both VSV-NJ isolates (Ossabaw and Colorado).
Histologic lesions were observed in lungs of 31/90 (34%) mice inoculated intranasally with VSV-NJ Ossabaw and 14/35 (40%) mice inoculated intranasally with VSV-NJ Colorado. Lesions were observed as early as 12 hours PI and lasted throughout the course of the study (to day 7 PI). Most lesions consisted of mild focal suppurative bronchiolitis and bronchopneumonia. A few mice developed more intense bronchointerstitial pneumonia, with alveolar histiocytosis and rare type II pneumocyte hyperplasia.
Histologic lesions were observed in hearts of 7/90 (8%) mice intranasally inoculated with the Ossabaw isolate and 5/35 (14%) mice intranasally inoculated with the Colorado isolate. Lesions were mild and consisted of multifocal necrosis of cardiac muscle cells with minimal infiltration by macrophages. These lesions were observed from day 3 to day 7 PI.
Histologic lesions were not observed in the remainder of tissues examined from mice inoculated intranasally with either virus isolate, including cervical lymph node, spleen, liver, kidney, gastrointestinal tract, and gonad.
Intradermal inoculation. No histologic lesions were observed in any tissues from adult deer mice inoculated intradermally throughout the course of the study. In nestling and juvenile mice inoculated intradermally, significant lesions were confined to the brain, spinal cord, and heart.
Lesions were first observed in the spinal cord of intradermally inoculated young mice as early as day 2 PI. Necrosis of ependymal cells lining the central canal was observed on day 2 PI and was followed by patchy necrosis of neurons in the adjacent neuropil by day 3 PI. Necrosis of neurons became more widespread by day 4 PI, with both the dorsal and ventral horns affected.
The appearance of histologic lesions in the brain followed lesion development in the spinal cord by 1 or 2 days. Lesions were first observed in the ependyma lining the fourth, third, and lateral ventricles on day 3 PI. Necrosis of ependymal cells and rare necrosis of adjacent neurons were observed multifocally by this time. Many nestling mice had meningitis throughout the brain by day 2 or day 3 PI, with infiltration of the leptomeninges by small to moderate numbers of lymphocytes and macrophages. Most of these same mice also had perivascular cuffing of blood vessels with small numbers of lymphocytes and macrophages in the cerebrum and brain stem.
Multifocal necrotizing myocarditis was observed in 9/16 (56%) nestling and 8/20 (40%) juvenile mice inoculated intradermally by day 2 or day 3 PI. The lesions consisted of well-demarcated regions of cardiac muscle cell necrosis with infiltration by macrophages and rare neutrophils.
In 3/16 (19%) nestling mice inoculated intradermally, there were lesions in the olfactory epithelium by day 3 PI. These lesions were similar to those described for intranasally inoculated mice, i.e., necrosis and erosion of olfactory epithelium with mild edema and inflammatory infiltrates in the submucosal propria. Histologic lesions were not observed in remaining organs of nestling and juvenile mice.
Immunohistochemistry and in situ hybridization
Intranasal inoculation. For detection of VSV-NJ in intranasally inoculated juvenile and adult mice, both immunohistochemistry and in situ hybridization gave similar results (Table 1). Staining intensity and distribution of viral antigen and mRNA were similar for all tissues except lung and lymph node. Viral antigen and mRNA were detected in all tissues with histologic lesions in both age classes of mice; however, some tissues without lesions also displayed prominent antigen immunoreactivity but not mRNA hybridization signal.
Immunohistochemistry (IHC) and in situ hybridization (ISH) results from 90 juvenile and adult deer mice inoculated intranasally with vesicular stomatitis virus, New Jersey serotype.

∗ OLFEP = olfactory epithelium.
† OLB = olfactory bulb.
‡ CERLN = cervical lymph node.
In the nasal cavity of intranasally inoculated mice, virus was first detected by immunohistochemistry as early as day 1 PI when histologic lesions either were not apparent or were minimal. Using double-label immunohistochemistry for VSV-NJ and calretinin, virus was first observed in olfactory receptor neurons by day 1 PI (Fig. 2). By day 2 PI, virus was readily detected in olfactory epithelial cells and olfactory receptor neurons by means of both immunohistochemistry and in situ hybridization. By day 4 PI, immunohistochemistry revealed virus throughout olfactory epithelium and in Bowman's glands, neurons in the submucosal propria, and inflammatory cells in the mucosa and submucosa (Fig. 3).
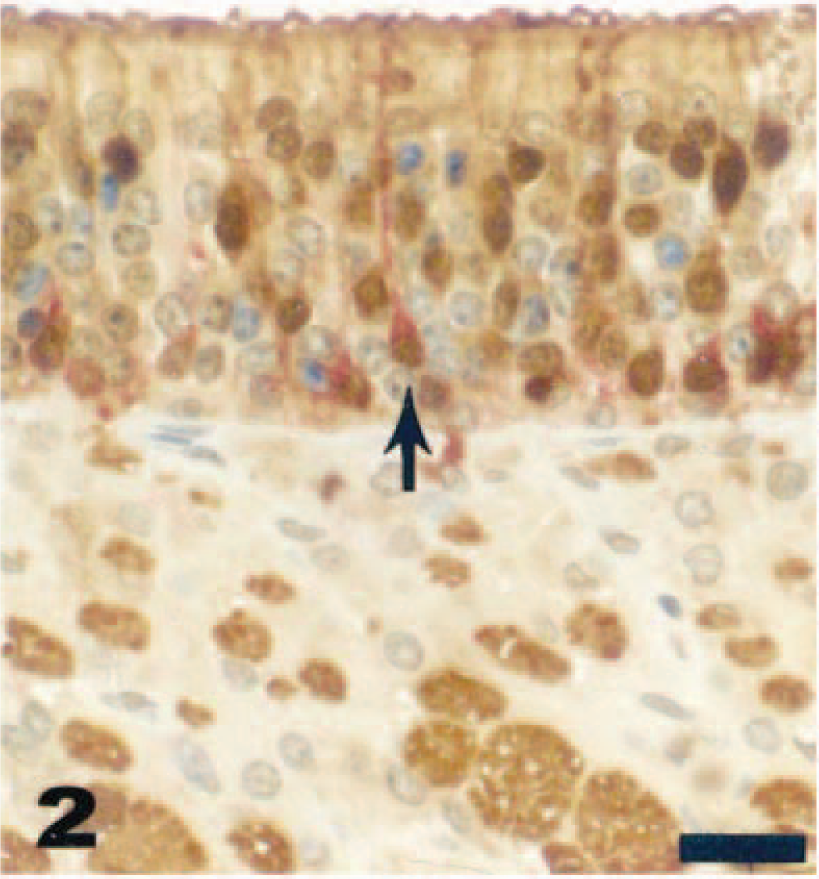
Olfactory epithelium; adult deer mouse, day 1 after intranasal inoculation with VSV-NJ. VSV-NJ antigen (fast red chromagen, arrow) in olfactory receptor neurons labeled with anti-calretinin (diaminobenzidine chromagen, brown). Double-label immunohistochemistry, streptavidin–biotin–peroxidase complex method followed by alkaline phosphatase technique, Mayer's hematoxylin counterstain. Bar = 15 µm.
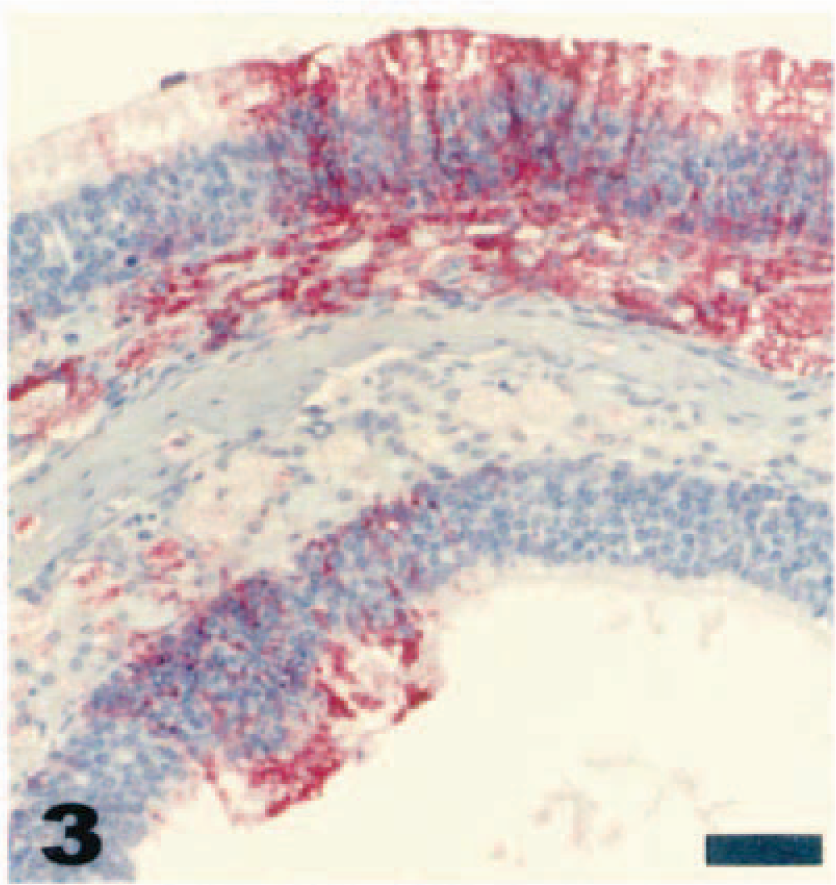
Olfactory epithelium; juvenile deer mouse, day 4 after intranasal inoculation with VSV-NJ. There is locally extensive VSV-NJ immunoreactivity in epithelial cells and in cells throughout the mucosal propria. Streptavidin–biotin alkaline phosphatase method, fast red chromagen, Mayer's hematoxylin counterstain. Bar = 100 µm.
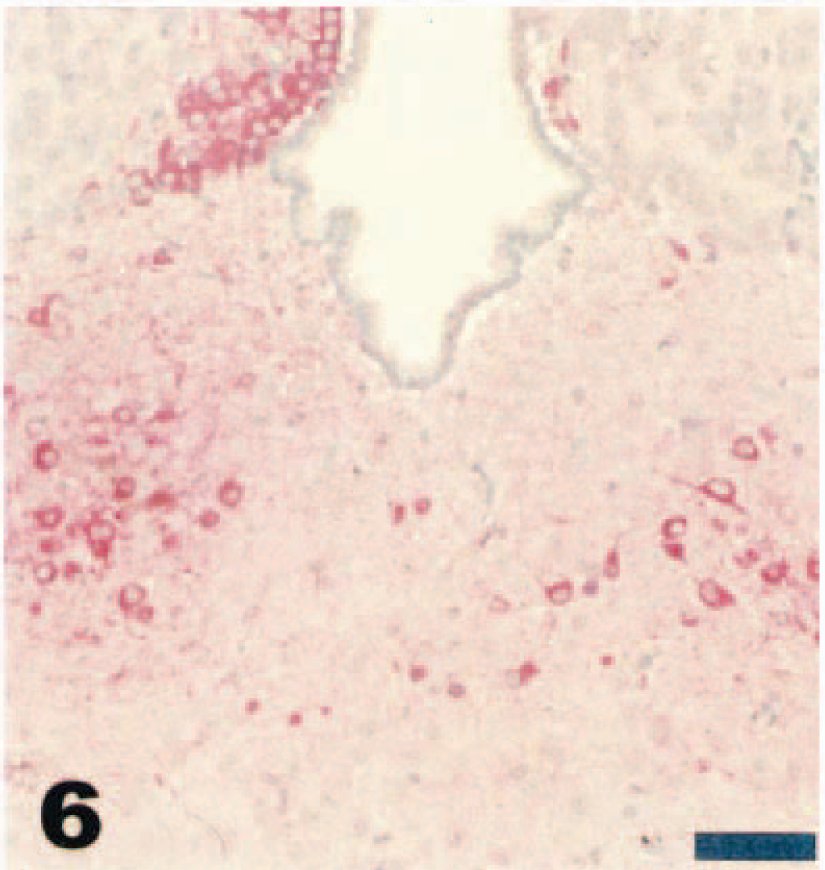
Virus was first detected in the olfactory bulbs by immunohistochemistry and in situ hybridization on day 2 PI. By day 3 PI, virus was present in most glomerular and plexiform layer cells in the olfactory bulbs (Fig. 4). Following infection of the olfactory bulbs, viral antigen and mRNA could be demonstrated in the anterior olfactory nuclei and olfactory tubercles by day 4 PI. By day 5 or day 6 PI, viral antigen and mRNA were present throughout widespread regions of the brain, including the amygdaloid nuclei, septal complex nuclei, thalamic and hypothalamic nuclei, hippocampus, pyriform cortex, and brain stem nuclei (Fig. 5). Viral antigen and mRNA were first detected in the olfactory ventricular ependyma by day 3 PI and spread rapidly to the lateral, third, and fourth ventricles, with detection in these structures by day 4 PI. By day 5 PI, many neurons in the periventricular neuropil stained strongly for viral antigen or mRNA throughout the brain (Fig. 6).
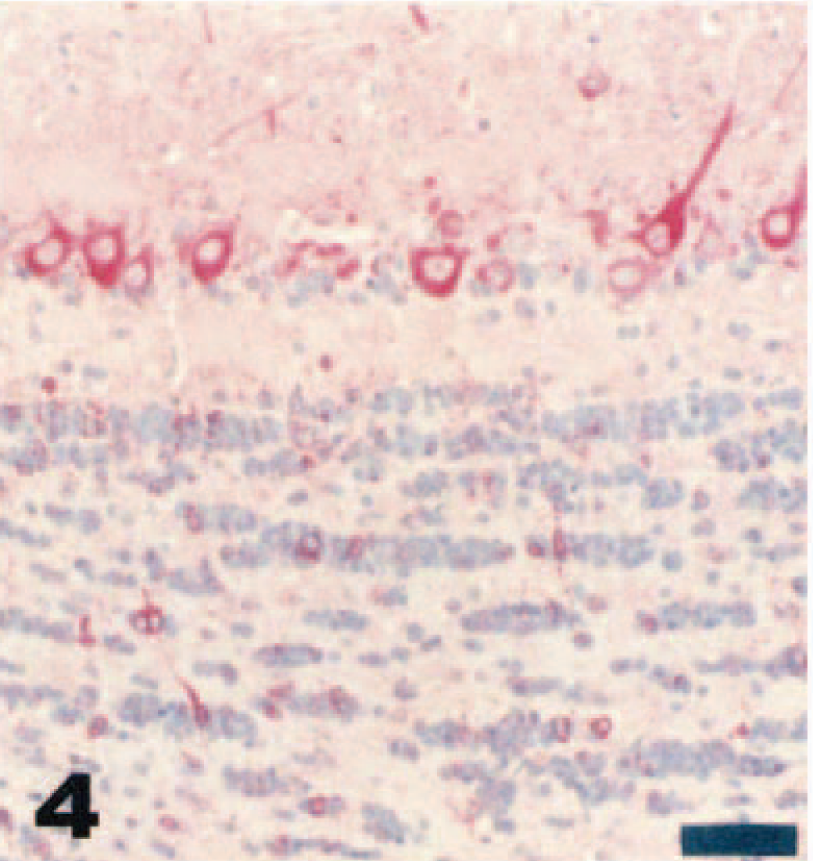
Olfactory bulb; juvenile deer mouse, day 3 after intranasal inoculation with VSV-NJ. Note diffuse VSV-NJ immunoreactivity in neurons of the glomerular and plexiform layers of the olfactory bulb. Streptavidin–biotin alkaline phosphatase method, fast red chromagen, Mayer's hematoxylin counterstain. Bar = 50 µm.
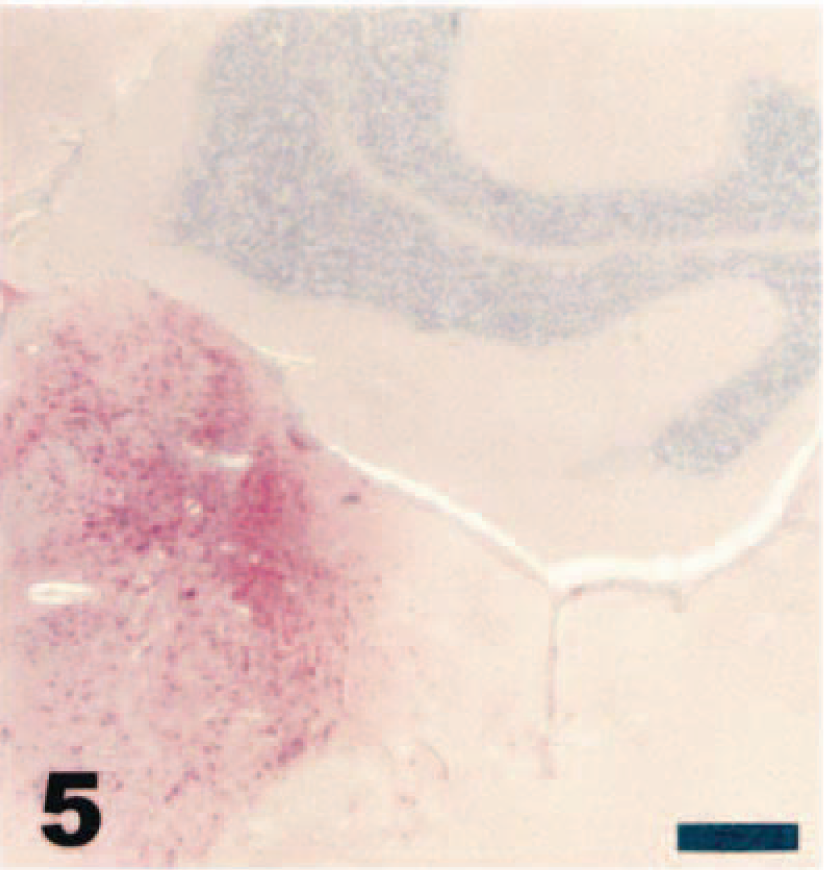
Brain; adult deer mouse, day 6 after intranasal inoculation with VSV-NJ. Note the unilateral intense immunoreactivity of the locus ceruleus and neighboring nuclei within the pons. Streptavidin–biotin alkaline phosphatase method, fast red chromagen, Mayer's hematoxylin counterstain. Bar = 250 µm.
Brain; adult deer mouse, day 5 after intranasal inoculation with VSV-NJ. Intense immunoreactivity of neurons in the periventricular habenular nuclei. Streptavidin–biotin alkaline phosphatase method, fast red chromagen, Mayer's hematoxylin counterstain. Bar = 100 µm.
Virus was first detected in the spinal cord of intranasally inoculated mice on day 5 PI. Using immunohistochemistry and in situ hybridization, viral antigen and mRNA were initially detected in ependymal cells of the central canal. By day 6 PI, viral antigen and mRNA were detected in neurons in the neuropil adjacent to the central canal and in neurons in both the dorsal and ventral horn gray matter distant from the central canal.
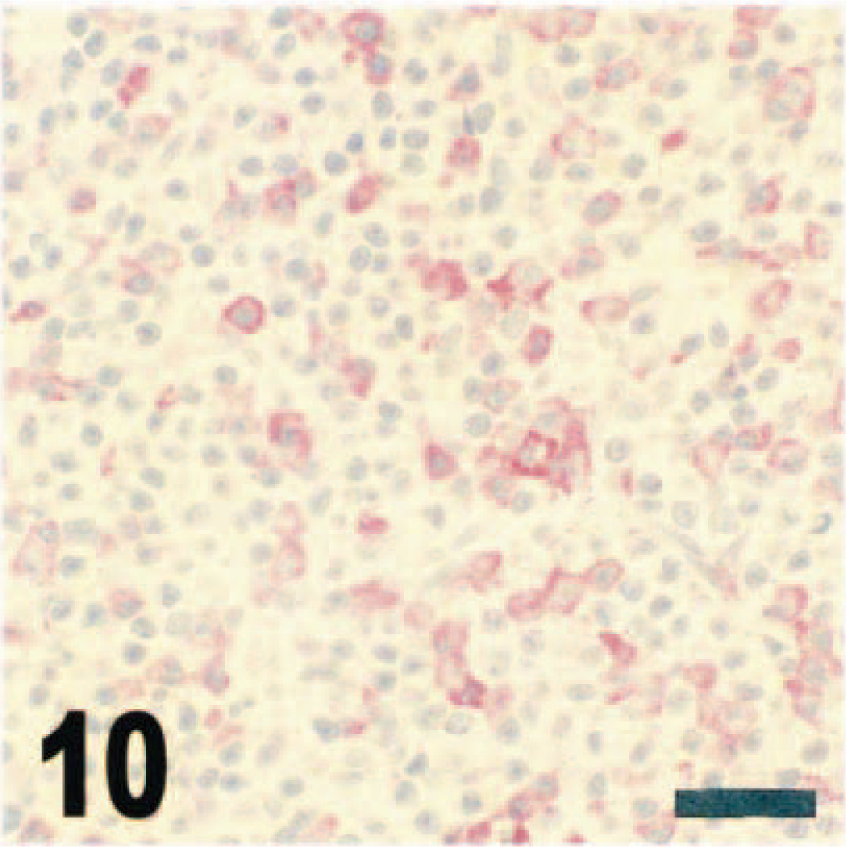
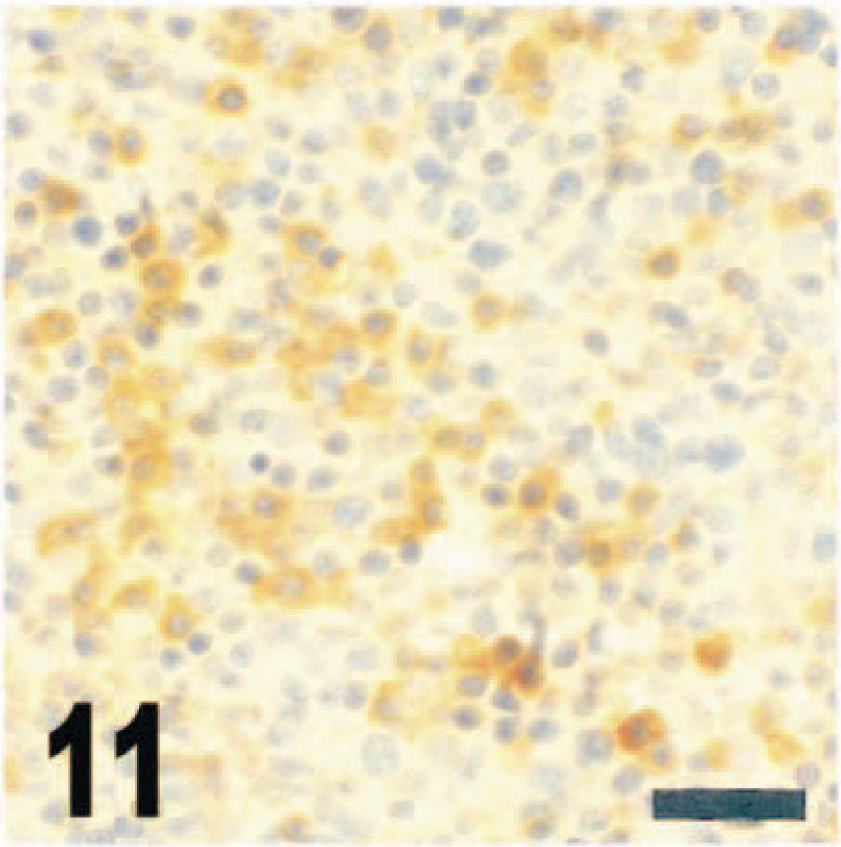
Viral antigen was detected in the lungs of intranasally inoculated mice from days 1 to day 5 PI; however, the degree of immunoreactivity was minimal. Virus was observed via immunohistochemistry in rare bronchiolar epithelial cells and occasionally in alveolar and interstitial macrophages in the lungs (Fig. 7). In contrast to observations in the central nervous system, where viral antigen and mRNA always were detected equally within lesions (Figs. 8,9), viral mRNA was not demonstrated by in situ hybridization in any cells in the lungs or in macrophages in lymph nodes. However, immunohistochemistry consistently revealed viral antigen in macrophages in cervical lymph nodes from day 2 to day 6 PI (Figs. 10,11).
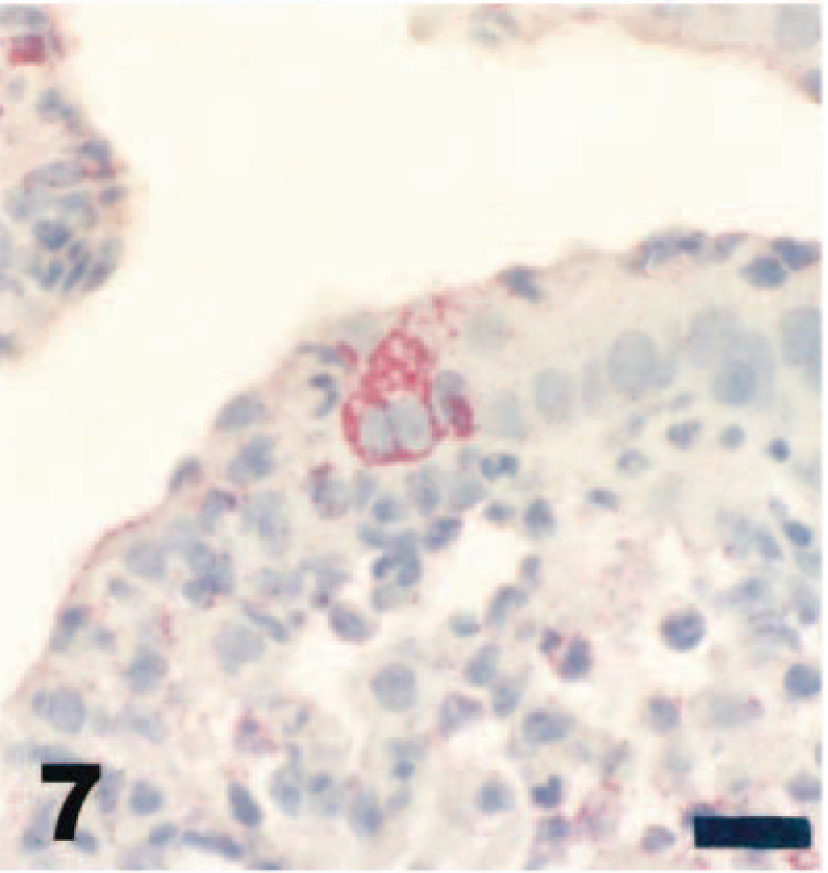
Lung; adult deer mouse, day 2 after intranasal inoculation with VSV-NJ. Note the few bronchiolar epithelial cells labeled for VSV-NJ antigen. Streptavidin–biotin alkaline phosphatase method, fast red chromagen, Mayer's hematoxylin counterstain. Bar = 15 µm.
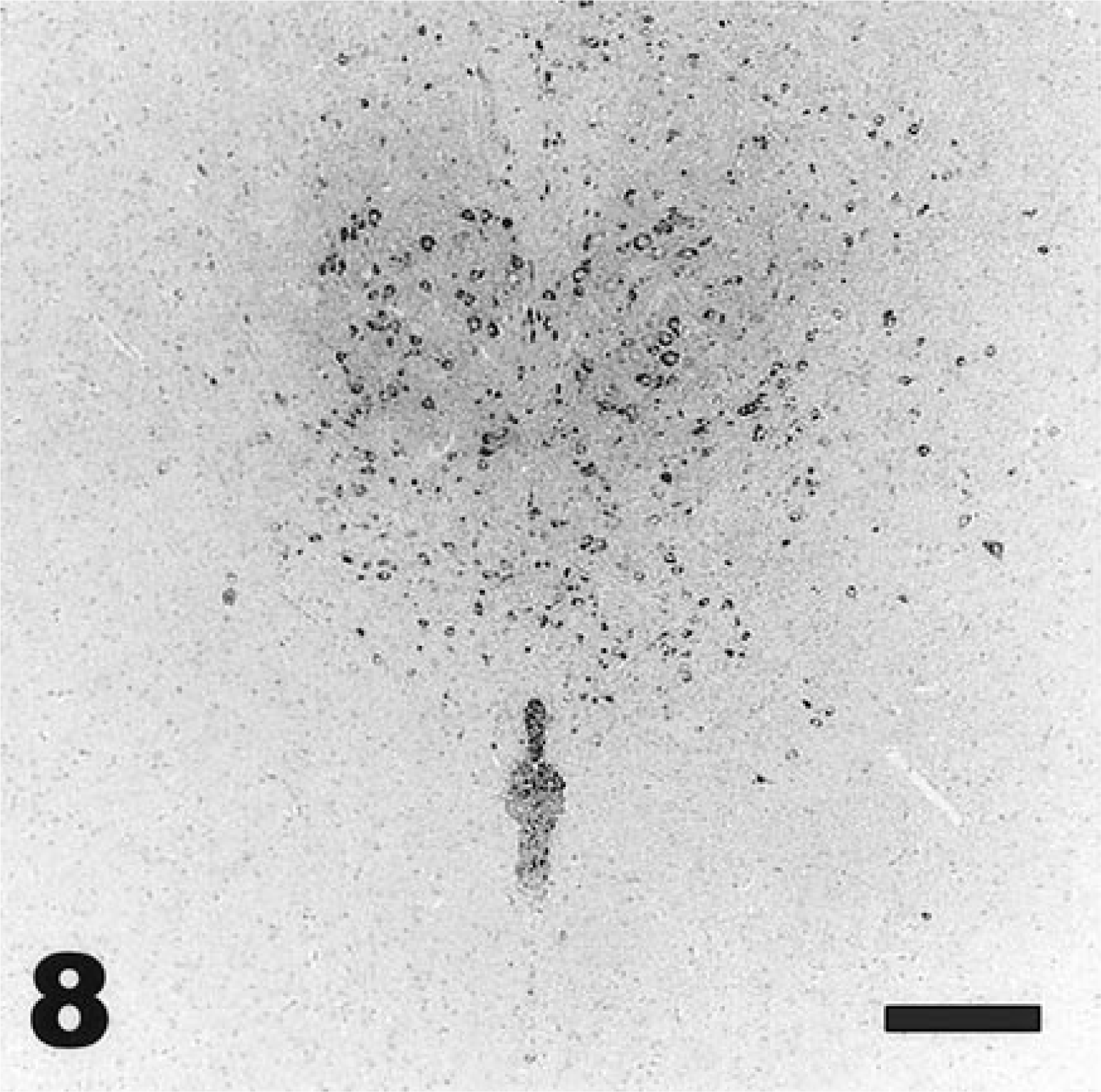
Brain; adult deer mouse, day 6 after intranasal inoculation with VSV-NJ. The ependymal cells of the aqueduct and neurons within the dorsal raphe nuclei show immunoreactivity for VSV-NJ. Streptavidin–biotin alkaline phosphatase technique, fast red chromagen, Mayer's hematoxylin counterstain. Bar = 100 µm.
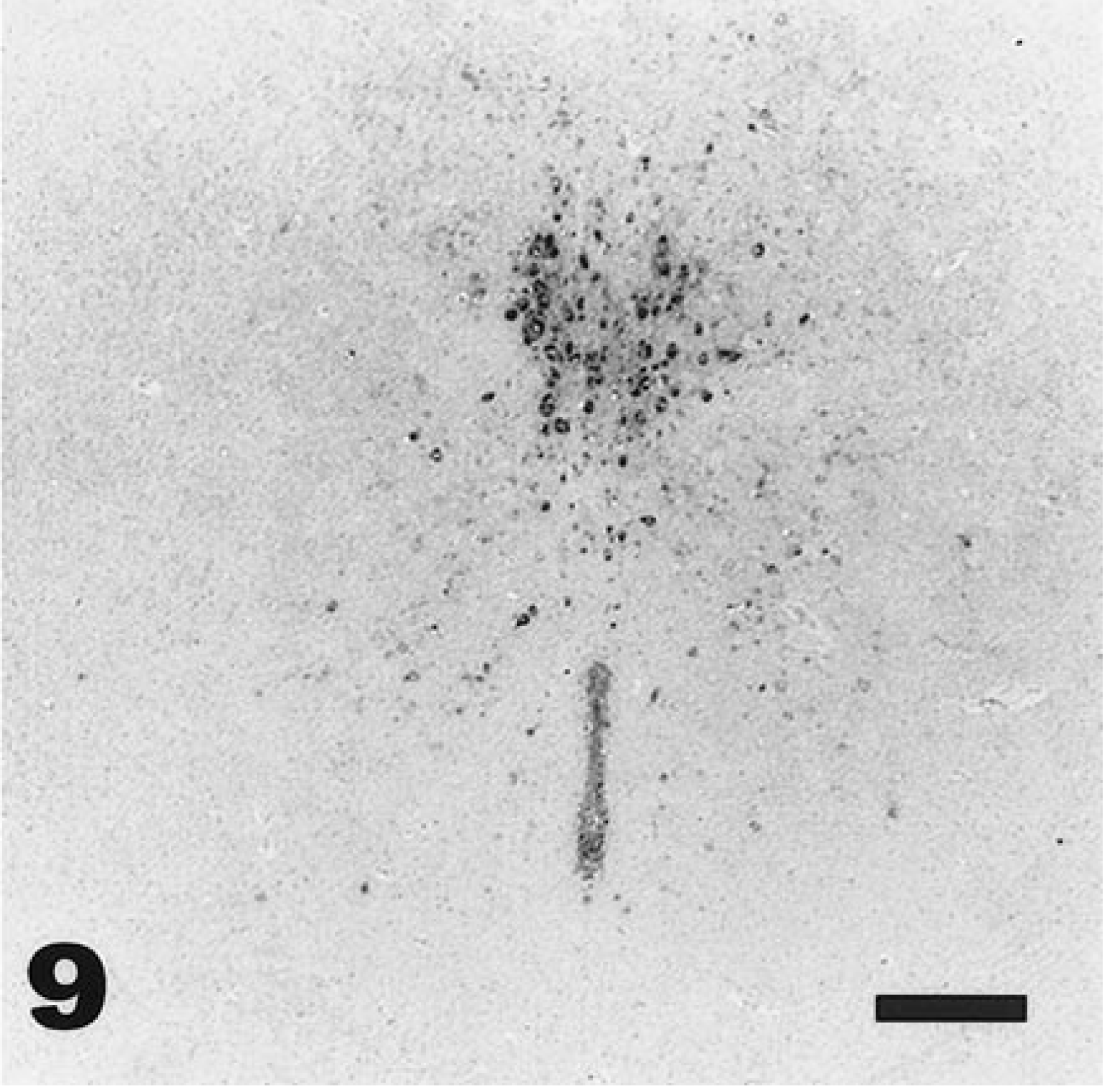
Brain; adult deer mouse, day 6 after intranasal inoculation with VSV-NJ. Hybridization signal is visible in ependymal cells of the aqueduct and neurons within the dorsal raphe nuclei. In situ hybridization, NBT/BCIP chromagen, Mayer's hematoxylin counterstain. Bar = 100 µm.
Cervical lymph node; juvenile deer mouse, day 3 after intranasal inoculation with VSV-NJ. There are multifocal cells morphologically resembling macrophages labeled for VSV-NJ antigen in the lymph node sinuses. Streptavidin–biotin alkaline phosphatase method, fast red chromagen, Mayer's hematoxylin counterstain. Bar = 50 µm.
Cervical lymph node; juvenile deer mouse, day 3 after intranasal inoculation with VSV-NJ. Note the macrophages throughout the sinus labeled with Mac-387 anti-myeloid/histiocyte antibody. Streptavidin–biotin peroxidase complex method, diaminobenzidine chromagen, Mayer's hematoxylin counterstain. Bar = 50 µm.
Occasional light immunohistochemical staining of cardiac muscle cells was observed following intranasal inoculation, usually on day 3 or day 4 PI. However, viral mRNA could not be demonstrated in these same cells with in situ hybridization. Virus was never detected in other organs by immunohistochemistry or in situ hybridization, including the spleen, liver, kidney, adrenal gland, gastrointestinal tract, and gonad.
Intradermal inoculation. In intradermally inoculated adult mice, viral antigen was not detected in any tissue at any time PI (Table 2), in agreement with the absence of histologic lesions in these animals. In nestling and juvenile mice inoculated intradermally, viral antigen and mRNA were readily detected in the spinal cord by day 2 PI, with intense immunoreactivity and hybridization signal in ependymal cells of the central canal and diffuse staining of neurons throughout the ventral and dorsal horn gray matter.
Immunohistochemistry results ∗ from deer mice of three age classes inoculated intradermally with vesicular stomatitis virus New Jersey serotype.
∗ Number of mice with positive antigen detection.
† N = nestling; J = juvenile; A = adult.
‡ OLFEP = olfactory epithelium; OLB = olfactory bulb; CERLN = cervical lymph node.
Viral antigen or mRNA were not detected in the brains of nestling and juvenile mice until day 3 PI, and then the pattern of immunoreactivity and hybridization signal was different when compared with that of mice inoculated by the intranasal route. Rather than the consistent and restricted staining observed in intranasally inoculated mice, nestlings and juveniles inoculated intradermally developed widespread staining of many regions of the brain. Most commonly stained regions included cerebellar folia (molecular, granular, and Purkinje cells), most of the neurons in the somatosensory cerebral cortex, much of the thalamus (ventral nuclei neurons especially), many neurons in the hippocampus (molecular and pyramidal cells), and neurons in the amygdaloid nuclei, the caudate putamen, the olfactory tubercle, and even the olfactory bulbs. Ventricular ependymal cells also were diffusely stained in most mice inoculated intradermally.
Intense VSV-NJ immunoreactivity of cardiac muscle cells was observed in hearts of intradermally inoculated nestling and juvenile mice by day 2 PI, corresponding to the significant histologic lesions observed in hearts of these young mice. In a few nestling mice, most of the myocardium was affected. Macrophages in cervical lymph nodes stained immunohistochemically for VSV-NJ by day 2 PI, and viral antigen was detected in these cells through day 4 PI in nestlings and day 5 PI in juveniles inoculated intradermally. However, in situ hybridization was not performed on hearts or lymph nodes to confirm the presence of mRNA. In one nestling mouse, there was intense VSV-NJ immunohistochemical staining of ganglion cells, inner neuronal layer cells, and outer neuronal layer cells of the retina on day 4 PI. Viral antigen was not detected in remaining tissues from intradermally inoculated nestlings and juveniles, including liver, spleen, kidney, adrenal gland, gastrointestinal tract, and gonad.
Discussion
Intranasal infection of laboratory mice with VSV provides a direct portal of entry for virus into the central nervous system. 16,19,20,23,25,30,34 Virus first infects neurons in the olfactory epithelium and reaches the olfactory bulbs via the olfactory nerves. 30 Dissemination of virus from the olfactory bulbs to other structures in the brain then occurs via retrograde and possibly anterograde transneuronal transport. 16,24,25,32 Similar retrograde and anterograde transneuronal transport of VSV was demonstrated within the brain following injection into the eye in NMRI strain mice. 24 A second route of viral dissemination in the brains of infected laboratory mice involves extensive spread through the ventricular system. 19,20,30
The results of the studies performed here with deer mice suggest that the pathogenesis of central nervous system infection is similar in this species following intranasal inoculation. Histologic lesions, immunohistochemistry, and in situ hybridization demonstrated a similar temporal pattern of infection, starting with neurons in the olfactory epithelium, progressing to infection of the olfactory bulbs, and terminating with dissemination of infection further into the brain via both the ventricular system and transneuronal spread. Based upon the histologic and immunohistochemical studies performed, virus appears to reach the spinal cord of intranasally inoculated mice by the central canal, probably via direct extension from the cerebral ventricular system.
As detected by immunohistochemistry, limited infection of the lungs occurs at the same time (day 1 PI) as infection of the olfactory epithelium in intranasally inoculated mice. Such infection is probably a result of aspiration of virus secondary to intranasal instillation. The onset of viremia also is day 1 PI, and viremia is followed a day later by detection of virus in macrophages in lymph nodes. Viral mRNA was not demonstrated in such macrophages using in situ hybridization, however, suggesting virus uptake without significant replication in these cells. In contrast to young laboratory mice and hamsters, 7,15,28 in juvenile or adult deer mice viremia did not lead to disseminated visceral infection; virus was not detected in the liver, spleen, kidney, or other organs.
Following intradermal inoculation, the course of infection in nestlings and juveniles differed from that of intranasally inoculated animals. Viremia was detected at a similar time in both inoculation groups; however, viral antigen and mRNA were detected in the central nervous system earlier in the intradermally inoculated animals. This earlier detection may have partly been due to age effects; younger rodents are more susceptible to central nervous system infection with VSVs. 7,8,25,27,34,35 The anatomic distribution of virus in the central nervous system of intradermally inoculated mice also differed from that in intranasally inoculated mice. Virus was first detected in the spinal cord, suggesting peripheral nerves as a portal of entry. This finding is supported by results of a preliminary study performed in our laboratory in which dorsal root ganglion neurons were labeled immunohistochemically for VSV-NJ before the spinal cord itself was labeled (E. W. Howerth, personal observation). Once virus reaches the spinal cord in these mice, dissemination occurs via the central canal and ventricular system. Such dissemination is supported by the initial detection of viral antigen and mRNA in spinal cord central canal ependymal cells followed a day later by similar demonstration of virus in ependymal cells of the cerebral fourth and third ventricles. Dissemination also possibly occurs by anterograde transneuronal transport, as suggested by the widespread labeling of neurons in the brains of intradermally inoculated young mice. A hematogenous route of infection cannot be ruled out, however, and may even be supported by the observations of meningitis, perivascular encephalitis, and occasional infection of olfactory epithelium in intradermally inoculated nestling mice. This finding may be related to an age effect caused by an immature blood–brain barrier or immune system; similar lesions were never observed in adult mice even during or following documented viremia. Infection of cardiac muscle cells in the heart and detection of viral antigen in macrophages in lymph nodes, first observed soon after the onset of viremia, probably occur as a result of viremia. Viral antigen in macrophages in lymph nodes and other tissues occurs in disseminated VSV infections in both young laboratory mice and young hamsters. 7
As with other neurotropic viruses, including Semliki Forest virus, 29 the ability of VSV to reach the central nervous system following intranasal inoculation has been ascribed, in part, to the age of mice infected. 7,34 Younger laboratory mice are particularly susceptible to intranasal inoculation, whereas older mice are resistant. The deer mouse differs from the laboratory mouse in this regard, with adult mice being equally susceptible to central nervous system infection following intranasal inoculation. Adult deer mice, however, are strongly resistant to intradermal inoculation, whereas nestling and juvenile deer mice are susceptible by this route. Infection in nestling and juvenile deer mice progresses to disseminated central nervous system disease following inoculation by this route.
These findings suggest a potential role for deer mice (and possibly other native rodents) in the epidemiology of VS. Previous work has demonstrated that at least one proposed VSV vector in the western United States, the black fly (Simulium vittatum), can be infected with VSV-NJ simply by cofeeding with experimentally infected flies on nonviremic deer mice. 26 The investigators performing this research were unable to document viremia in 6-week-old and 6-month-old deer mice following intramuscular inoculation with VSV-NJ and thus concluded that deer mice probably do not meet accepted criteria for a reservoir host for VSV. 26 The consistent viremia demonstrated here following intradermal inoculation (mimicking natural routes of exposure) in nestling and juvenile animals suggests that deer mice may in fact serve as amplifying or reservoir hosts for VSV-NJ, with viremia providing an initial means of infection for black flies and other vectors of VSV. Further laboratory and field research will be required to confirm any role that deer mice or other native rodents may have in the epidemiology of VS. Given the results produced here, emphasis may need to be placed on younger age classes, including nestling and juvenile animals. The deer mouse VSV-NJ viremia model established in this work should be valuable in future studies examining the interactions among insect vectors, VSVs, and potential amplifying or reservoir hosts.
Footnotes
Acknowledgements
We are grateful to Leslie Bauer, Deborah Perzak, Molly Murphy, and Melissa Scott for assistance with this research. This work was supported by the Veterinary Medical Experimental Station, University of Georgia.
