Abstract
Immunohistochemical techniques were used to characterize the disfiguring and debilitating fatal neoplastic disease, devil facial tumor disease (DFTD), which has recently affected a significant proportion of the wild population of Tasmanian Devils (Sarcophilus harrisii). The diagnostic values of a number of immunohistochemical stains were employed to further characterize 50 representative cases. The neoplasms were negative for cytokeratin (0/48), epithelial membrane antigen (0/42), von Willebrand factor (vWF) (0/11), smooth muscle actin (SMA) (0/26), desmin (0/47), glial fibrillary acid protein (0/13), CD16 (0/13), CD57 (0/43), CD3 (0/18), and LSP1 (0/16). DFTD cells were positive for vimentin (50/50), S-100 (41/48), melan A (11/39), neuron specific enolase (35/35), chromogranin A (12/12) and synaptophysin (29/30). The cells were negative for amyloid (0/30) and stained negatively with Singh's silver (0/34) but were weakly argyrophilic (3/40) using Grimelius histochemical stain. These staining characteristics are consistent with cells of neuroectodermal origin.
Keywords
The condition known as devil facial tumor disease (DFTD) has arisen within the last decade affecting wild populations of Tasmanian Devils (Sarcophilus harrisii) in the north, central, east, and south of Tasmania. A variety of neoplasms have been described in Tasmanian Devils in previous publications, 9 but never has a disease been reported to occur with such high frequency and of a single morphologic type.
Traditional methods of light microscopy and transmission electron microscopy (TEM) were unable to identify possible histiogenic origins of DFTD cells; and they were identified as malignant undifferentiated soft tissue neoplasms. Immunohistochemistry (IHC) was the next step in an effort to further classify the neoplasm. IHC tests work on the premise that immunophenotypes are preserved even in poorly differentiated neoplasms and can be used as markers to determine the class of cell origin. There are few publications reporting the results of cross-species reactivities 19, 28 and there have been no studies of immunohistochemistry applied to Tasmanian Devil tissue. In this paper, a suite of immunostains were applied to normal Tasmanian Devil tissues and DFTD neoplasms.
Materials and Methods
To characterize the immunohistochemical properties of the devil facial tumor disease neoplasms, field samples were collected from wild populations of Tasmanian Devils from different geographic locations throughout Tasmania.
Animals and sampling
Sample collections were undertaken in the period June 2001 to December 2004. Routine pathological methods were employed to examine 91 DFTD cases, 33 suspect, and 131 non-DFTD cases. Specimens were collected from live (65) and deceased (157) animals. Live animals were sampled in the field. Surgical biopsies of all grossly visible tumors were taken from Tasmanian Devils sedated either with a combination of detomidine (50 μg/kg of Domitor®) and butorphanol (0.4 mg/kg of Torbugesic®) or anesthetized with Isoflurane® (induction rate was 5% for 3–5 minutes and then maintained at 1–3%. Selected animals were euthanatized with an overdose of intravenous sodium pentobarbitone (Lethobarb®, Virbac [Australia] Pty Ltd).
Histology and immunohistochemistry
All tissue samples were collected either by surgical punch biopsy, incisional biopsy, or postmortem sample. Neoplastic tissues obtained from Tasmanian Devils were fixed in 10% neutral buffered formalin (NBF) for up to 7 days and then routinely processed into paraffin blocks from which 3-μm sections were cut and stained with hematoxylin and eosin. Selected cases were also stained using the Grimelius technique, Singh's silver, and Congo red as described in Bancroft. 3
All sections were examined using routine light microscopy and morphologic diagnoses of the tumor types were made. We considered that any Tasmanian Devil with a neoplasm composed of round to spindle-shaped cells arising in the dermis or subcutaneous tissue had DFTD, in accordance with criteria identified in an earlier paper. Those neoplasms that did not fit these criteria were excluded from this study.
After the diagnosis of DFTD, samples were then prepared for IHC. The area of interest on selected hematoxylin and eosin–stained sections of DFTD were identified on the corresponding paraffin block and dissected free using a 5-mm-diameter stainless steel corer. These biopsies were then re-embedded as composite paraffin blocks from which 3-μm sections were cut and placed on sialinized slides.
Positive control tissues from nondiseased Tasmanian Devils, prepared in the same way, were tested with a panel of immunohistochemical stains to gain consistent results before tests were conducted on DFTD neoplasms.
Following deparaffinization, rehydration, antigen retrieval, and quenching of endogenous peroxidase activity, polyclonal and monoclonal primary antibodies were applied. The antigen retrieval method and media, the optimal antibody titers (diluted with Dako S0809), and functional avidities (incubation periods) were determined for the primary antibodies used in this study. They include mouse monoclonal antibodies against CD16 (1°20, Dako), CD57 (1°100, Zymed), cytokeratin AE1/AE3 (CK) (1°100, Dako), desmin (1°100, Dako), epithelial membrane antigen (EMA) (1°100, Dako), leucocyte specific antigen (LSP1) (1°40, Oxford), melan A (1°50, Dako), neuron specific enolase (NSE) (1°250, Dako), smooth muscle actin (SMA) (1°200, Dako), and vimentin (1°100, Dako) and rabbit polyclonal antibodies against CD3 (1°150, Dako), chromogranin A (CgA) (1°600, Dako), glial fibrillary acid protein (GFAP) (1°2000, Dako), S-100 (1°250, Dako), synaptophysin (1°50, Dako) and von Willebrand factor (vWF) (1°200, Dako).
The sections for CD3, CgA, CK, NSE, S-100, synaptophysin, and vimentin were pretreated by microwaving (Kambrook KER-686LE, 700 W) on reheat twice for 4-minute periods and another 2 more times on low for 4-minute periods in TES pH 9 buffer solution. The sections for CD57, desmin, melan A, and vWF were similarly treated but in sucrose/citric pH 6.5 solution. The sections for CD16 were pretreated by steaming within a receptacle containing EDTA pH 8 inside a rice cooker (Black & Decker BRC-100) containing deionized water for 20 minutes. The sections for EMA and GFAP were pretreated with proteinase K/Tris for 3 minutes; and for LSP1, the sections were pretreated by microwaving on high in 0.01 M Tris-sodium citrate pH 6.0 for three 6-minute periods. No pretreatment was required for SMA. The optimal antibody incubation period was determined to be 10 minutes for CD3, CK, desmin, melan A, SMA, S-100, vimentin, and vWF; 15 minutes for GFAP; 30 minutes for CgA, EMA, NSE, and synaptophysin; and 60 minutes for CD16, CD57, and LSP1.
Following washing, the labelled avidin-biotinylated enzyme complex (ABC) technique was employed for amplification of primary antibody tissue binding with CgA, CK, desmin, GFAP, melan A, NSE, SMA, S-100, synaptophysin, vimentin, and vWF; the Envision (Dako, product code K4001) method was used with CD3, CD16, CD57, and EMA; and alkaline phosphatase/anti–alkaline phosphatase (APAAP) method was used for LSP1.
Following washing, LSP1 sections were visualized using the chromogenic solution NBT/BCIP (p-nitroblue tetrazolium chloride/5-bromo-4-chloro-3-indolyl) and counterstained with nuclear fast red. For all other sections, the chromogenic solution, 3,3-diaminobenzidine (DAB) was applied and counterstained with hematoxylin (two dips), washed in tap water, then immersed in bluing solution for 30 seconds and washed again in tap water. The sections were then dehydrated through graded alcohols (95%, 100%) and left in xylene until coverslips were mounted with DPX.
In many tumors, the surrounding normal tissue provided both positive and negative internal controls. Further external positive and negative controls were prepared with formalin-fixed, paraffin-embedded (FFPE) normal Tasmanian Devil tissues, which included cerebrum, trigeminal nerve, pituitary gland, adrenal gland, pancreas, liver, intestine, kidney, bladder, skin, skeletal muscle, heart, spleen, lymph node, tonsil, and bone marrow. Immunostains were run on their respective positive controls and only when these returned positive staining reactions localized to the appropriate cellular compartments (as outlined by the respective manufacturers' product information leaflets) were the IHCs performed on DFTD cells. Negative controls were obtained by omission of the primary antibody with substitution by buffer solution.
Selected areas were photographed using a Leica DFC 320 (Leica Microsystems DI) digital camera.
Quantification of cells showing antigen expression
All sections were examined using routine light microscopy and the stain distribution and intensity were tabulated into a Microsoft™ Excel Spreadsheet.
The reactivity index was calculated from 100 cells randomly selected on oil immersion (100×). The immunoreactivity was scored on the following aspects including staining intensity (L = low, M = moderate, H = high), percentage of cells stained (NR = no reaction in controls; 0 = reaction in controls, but no reaction in DFTD; + = 01 to 30% positive; ++ = 31 to 65% positive; +++ = 66 to 100% positive), staining pattern (Di = homogenous, Gr = granular, Ve = vesicular), and staining distribution (Nu = nucleus, Cy = cytoplasm).
Only those cells recognizable histologically as DFTD-type cells were counted and they were considered to be positive if the staining reaction was localized to the cytoplasm.
Results
DFTD cells were negative for amyloid (0/30) and stained negatively with Singh's argentaffin silver stain (0/34) but were argyrophilic using Grimelius' stain in 8% of cases (3/40). In two of these cases, the cytoplasmic granularity was sparse and in another, it was present only in the peripheral cells.
CD11d, CD18, CD20, CD45, CD45RA, CD79a, CD79b, and MAC387 failed to produce results on an array of normal Tasmanian Devil tissues (cerebrum, trigeminal nerve, pituitary gland, adrenal gland, pancreas, liver, intestine, kidney, bladder, skin, skeletal muscle, heart, spleen, lymph node, tonsil, bone marrow, and DFTD neoplasms) and so were not used further on case material.
Using the antigen retrieval methods as described earlier, there were negligible differences in staining results for tissues fixed for up to 7 days. The results for IHC stains are tabulated in Table 1. DFTD sections were negative for cytokeratin (0/48) (Fig. 1), epithelial membrane antigen (0/42), vWF (0/11), SMA (0/26), desmin (0/47) (Fig. 2), glial fibrillary acid protein (0/13) (Fig. 3), CD16 (0/13), CD57 (0/43), CD3 (0/18), and LSP1 (0/16). There was a scattered positive reaction for CD3 (8/18) and the occasional LSP1 positive cells infiltrating the neoplasms, but these were identified as lymphocytes. Nonneoplastic cells were distinguished from DFTD cells by their size and morphology.
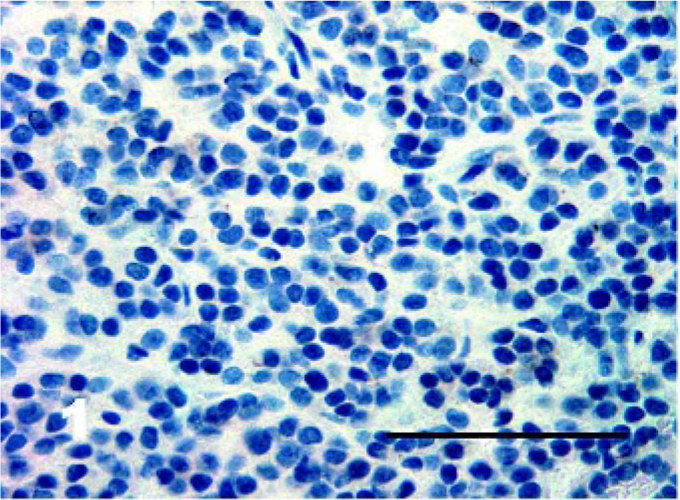
Tasmanian Devil, DFTD neoplasm. All DFTD cells stained negative for cytokeratin. Immunostaining for cytokeratin with hematoxylin counterstain. Bar = 50 μm.
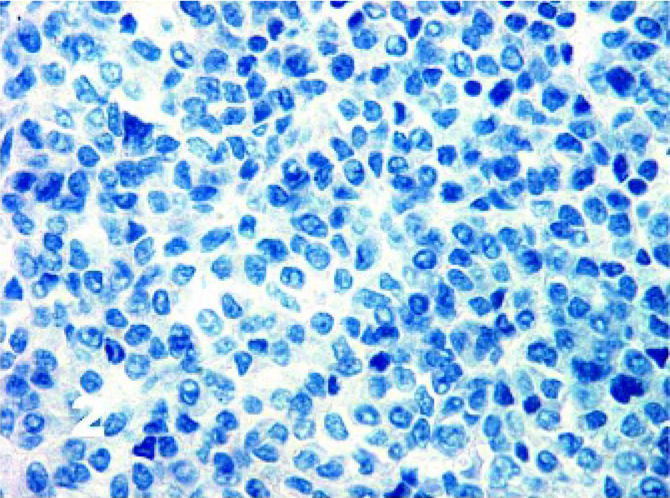
Tasmanian Devil, DFTD neoplasm. Negative staining in all DFTD cells for desmin. Immunostaining for desmin with hematoxylin counterstain.
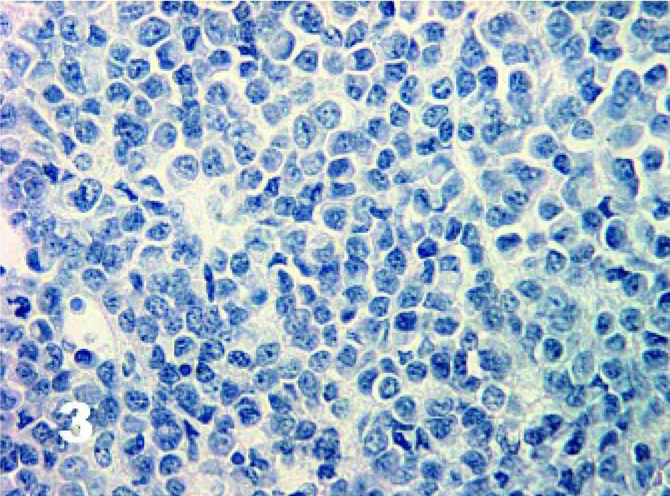
Tasmanian Devil, DFTD neoplasm. Negative staining in all DFTD cells for glial fibrillary acid protein. Immunostaining for GFAP with hematoxylin counterstain.
Selected antigen expression in devil facial tumor disease neoplasms immunostained with a panel of antibodies.
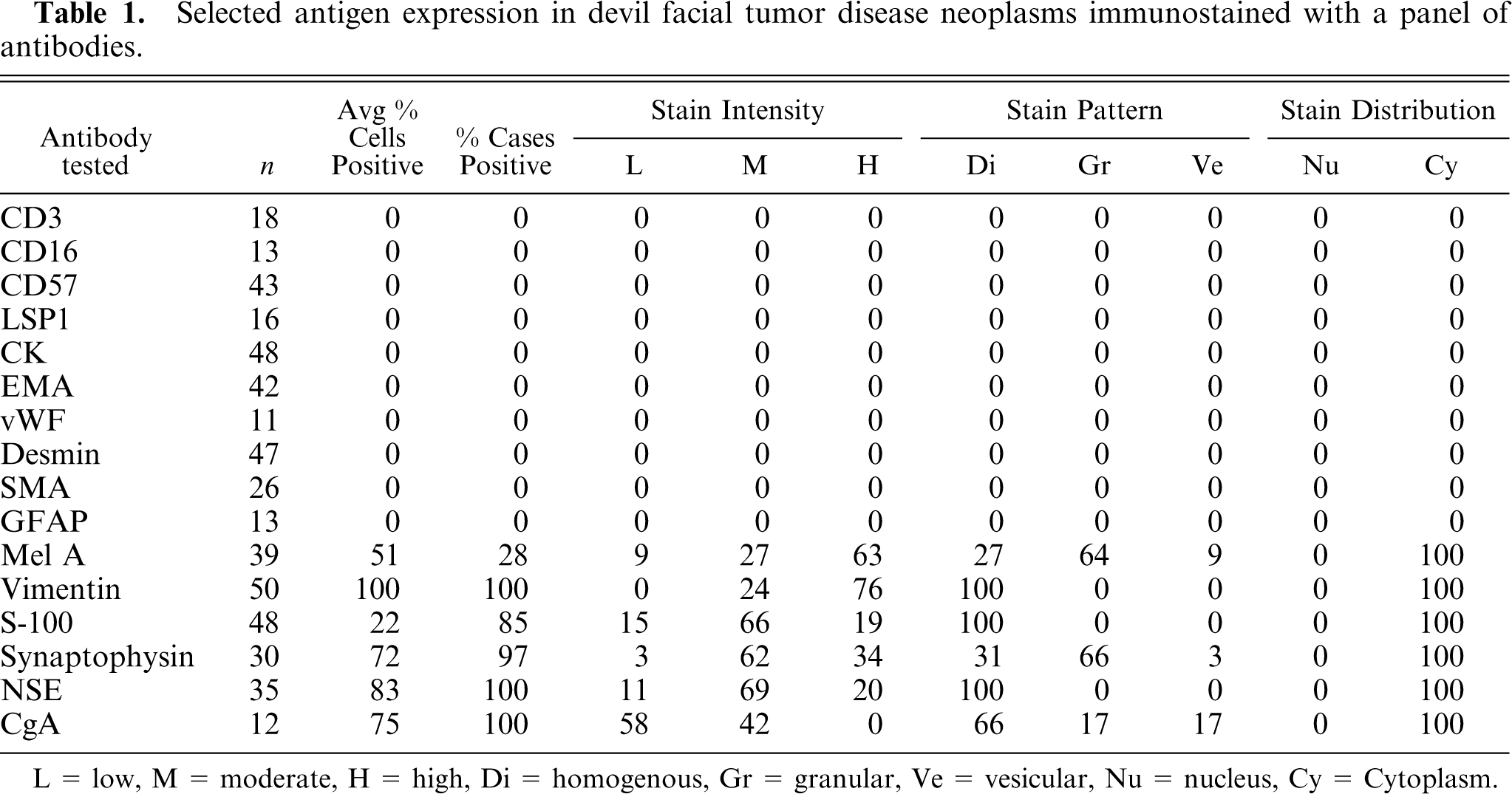
L = low, M = moderate, H = high, Di = homogenous, Gr = granular, Ve = vesicular, Nu = nucleus, Cy = Cytoplasm.
There was homogenous moderate to high intensity cytoplasmic staining in all DFTD cells for vimentin (50/50) with 42% (21/50) having brown granularity around the cytoplasmic borders (Fig. 4). Eighty-five percent (41/48) of DFTD cases stained positive for S-100 where an average of 22% of cells in each case were positive (Fig. 5). The staining pattern was homogenous and was contained within the cytoplasm (41/41). Staining intensity was variable and was generally moderate (27/41) to high (8/41) intensity. Twenty-eight percent (11/39) of DFTD cases stained for melan A where an average of 51% of cells in each case were positive (Fig. 6). The staining pattern was variable ranging from granular (7/11) to homogenous (3/11) to vesicular (1/11). Staining intensity was variable and was generally moderate (3/11) to high (8/11) intensity. There was homogenous moderate to high intensity cytoplasmic staining in all DFTD cases for NSE (35/35) with an average of 83% of cells staining per case (Fig. 7). In 20% (7/35) of cases, there was some granular cytoplasmic staining. All DFTD cases expressed low intensity positivity in their cytoplasm for CgA (12/12) with an average of 75% of cells per case staining (Fig. 8). Staining ranged from homogenous (8/12) to granular (2/12) to vesicular (2/12). In 97% (29/30) of DFTD cases, there was moderate (18/29) to high (10/29) intensity cytoplasmic staining, which was homogenous (11/29) or granular (18/29) for synaptophysin, with an average of 72% of cells in each case staining positive (Fig. 9).
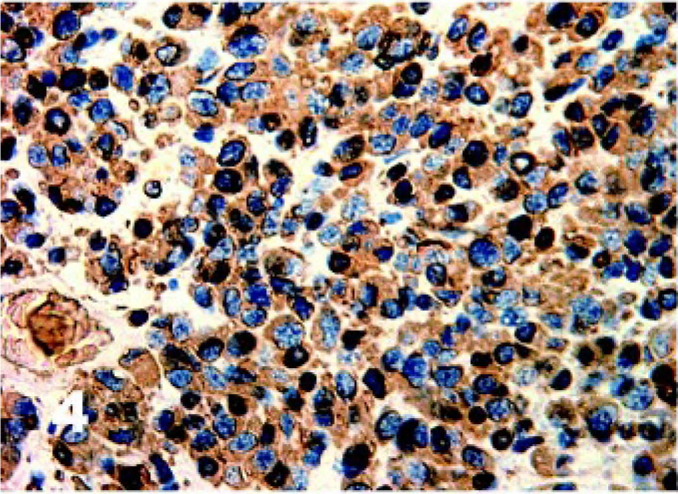
Tasmanian Devil, DFTD neoplasm. Homogenous moderate to high intensity cytoplasmic staining in all DFTD cells for vimentin. Immunostaining for vimentin with hematoxylin counterstain.
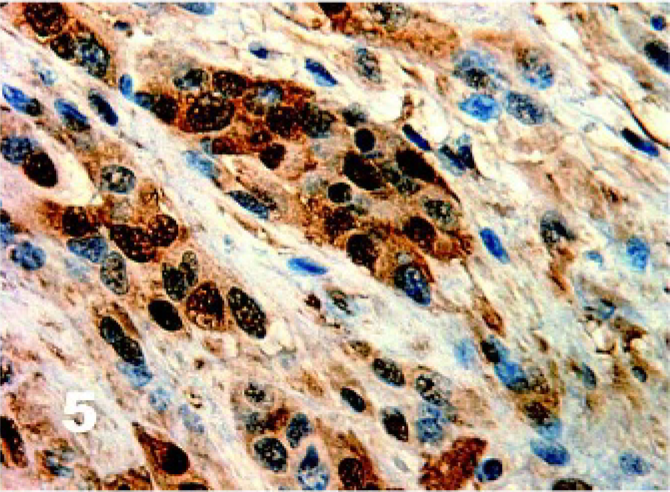
Tasmanian Devil, DFTD neoplasm. Patchy homogenous moderate cytoplasmic staining of DFTD cells for S-100. Immunostaining for S-100 with hematoxylin counterstain.
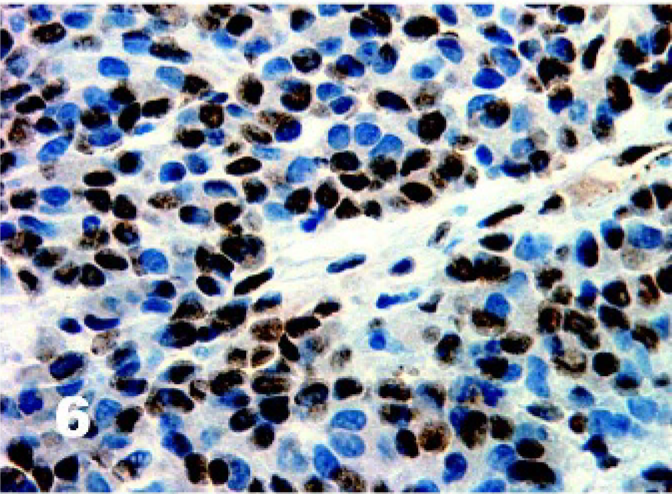
Tasmanian Devil, DFTD neoplasm. Twenty-eight percent of DFTD cases stained for melan A where an average of 51% of cells in each case were positive. Immunostaining for melan A with hematoxylin counterstain.
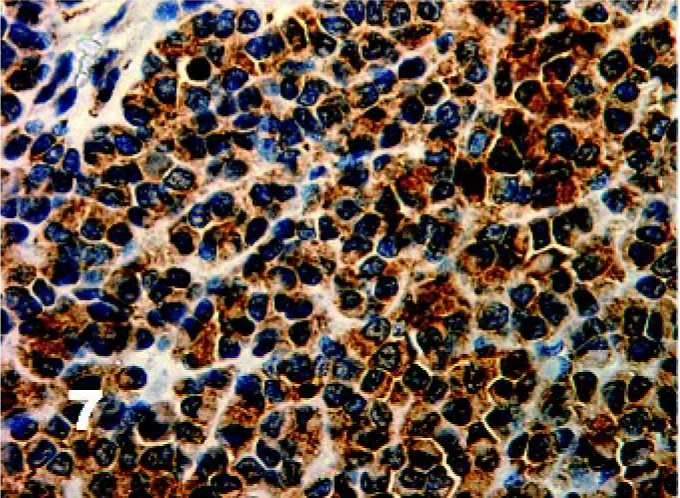
Tasmanian Devil, DFTD neoplasm. Diffuse homogenous expression of neuron specific enolase in all DFTD cases. Immunostaining for NSE with hematoxylin counterstain.
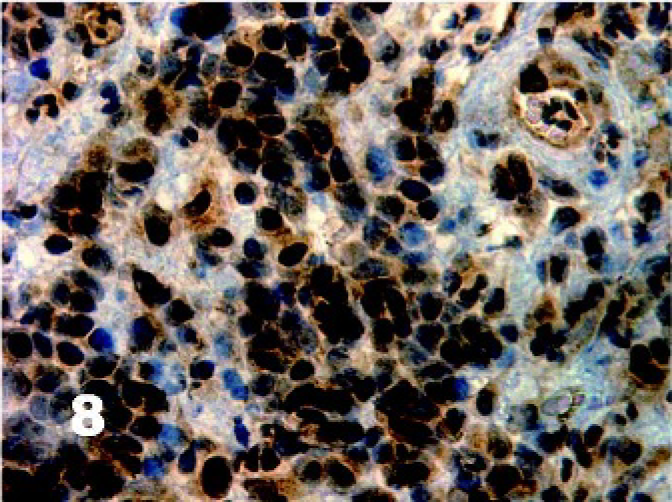
Tasmanian Devil, DFTD neoplasm. All DFTD cases expressed low intensity positivity in their cytoplasm for chromogranin A. Immunostaining for CgA with hematoxylin counterstain.
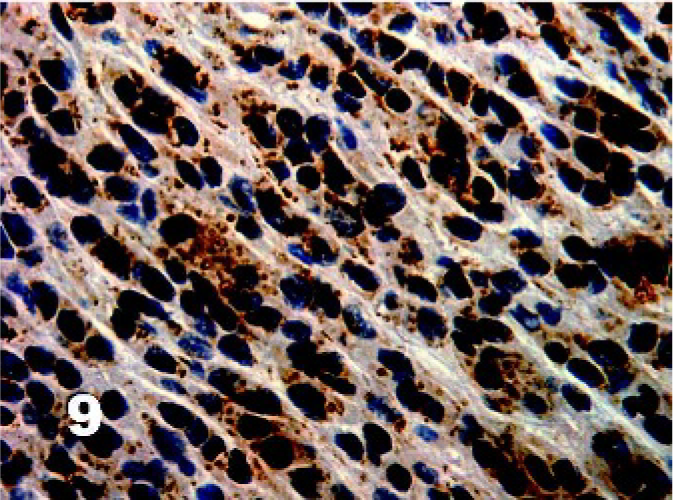
Tasmanian Devil, DFTD neoplasm. High intensity granular staining of DFTD cell cytoplasm for synaptophysin. Immunostaining for synaptophysin with hematoxylin counterstain.
Discussion
There has been little agreement on the cell type and classification of the neoplasm of DFTD. 16 DFTD cells are pleomorphic, anaplastic, and have no distinctive ultrastructural features. Differential diagnoses have included nonepitheliotropic cutaneous lymphosarcoma, poorly differentiated mast cell tumor, canine cutaneous histiocytoma, Ewing's sarcoma, and a neoplasm similar to canine transmissible venereal tumor (TVT). The results of the present study using a range of IHCs applied to normal Tasmanian Devil tissues and DFTD lesions indicate that the DFTD cells are most likely to be of neuroendocrine origin. The consistency of the immunoreactivity results in DFTD neoplasms from animals from different geographic locations and in different Tasmanian Devil populations is indicative that the animals are all suffering from the one disease; a neoplasm that has never been described before in an animal or human.
According to literature, the most useful markers in routine diagnosis are intermediate filaments because they are preserved even in poorly differentiated neoplasms to maintain their three-dimensional shape and can subsequently be used as markers to determine the class of the cell of origin. There are 6 main types of intermediate filaments that are tissue specific, which include cytokeratin, vimentin, desmin, glial fibrillary acid protein (GFAP), neurofilament proteins, and nestin. Cytokeratins exist in all epithelial cell types. 6 Vimentin is found in most mesenchymal cells and mesothelium. 35 Desmin is present in skeletal, cardiac, and visceral smooth muscle cells and some vascular smooth muscle cells. 23, 35 GFAP occurs in astrocytes, Schwann cells, and enteric glial cells. 21 Neurofilament proteins exist in all neuronal neurofilaments in the central and peripheral nervous systems. Nestins are intermediate filaments of the neural stem and progenitor cells. 17, 21 With the appropriate clinical and histologic features, the presence of such markers can be diagnostically helpful, particularly for round-cell tumors. 6
DFTD is unlikely to be of muscle or neural origin because all tumors we tested were negative for desmin, SMA, and GFAP, respectively. 8, 35 Immunoreactivity to CD3 was detected only as a scattering throughout the tumor and associated with lymphocyte infiltrates. LSP1 was once named lymphocyte specific antigen but their ability to cross-react with several other leucocytes has caused their name to be changed to leucocyte specific antigen. 18 Thus negative staining for CD16, CD57, and LSP1 confirms DFTD to be unrelated to leucocytes, including B cells, monocytes, granulocytes, plasma cells, dendritic cells, Langerhans cells, neutrophils, and myeloid cell lines. 14, 18, 26 Von Willebrand factor immunonegativity supports the nonendothelial nature of DFTD origins. Negativity for epithelial markers such as CK, EMA, and vWF and positivity for S-100 and vimentin could be proposed as evidence for DFTD being classified as a sarcoma, which is consistent with some of its histologic features. 22, 35 The homogenous and intense immunoreactivity for S-100 is suggestive for, among other things, neural crest–derived neoplasia. 35
DFTD was once hypothesized to be a variant of lymphosarcoma 16 and as such, a retroviral etiologic agent was suspected. However, our present study using specific neuroendocrine IHC stains (NSE, CgA, and synaptophysin) all produced positive results, which provides strong evidence to refute the original hypothesis. These results along with evidence of weak argyrophilic differentiation, morphologic and ultrastructural characteristics, are not inconsistent with DFTD being of neuroectodermal origin. DFTD cells are round to polygonal and arranged in a similar architectural pattern as neuroendocrine organs (e.g., islets of Langerhans, adrenal gland, thyroid, and pituitary gland), interspersed with delicate fibrous stroma. 22 Neuroendocrine tissues are derived from the embryonic neural crest, are widely dispersed throughout the body where they do not form discrete organs, and they are in especially high concentrations in tactile tissues such as the fingertips, the lips, 22 and in the whisker bed. 11 These latter sites are the locations where DFTD neoplasms most commonly originate.
The differential diagnoses based on IHC expression are summarized in Table 2.
Immunohistochemical expression of various neoplasms with a panel of immunostains. Devil Facial Tumor Disease(DFTD), Ewing's sarfcoma, 1, 30 canine transmissible venereal tumor, 22 Merkel cell tumor, 2, 15, 24– 27, 31 melanoma, 1, 12 primitive neuroectodermal neoplasia, 33 and neuroblastoma. 1, 25, 30
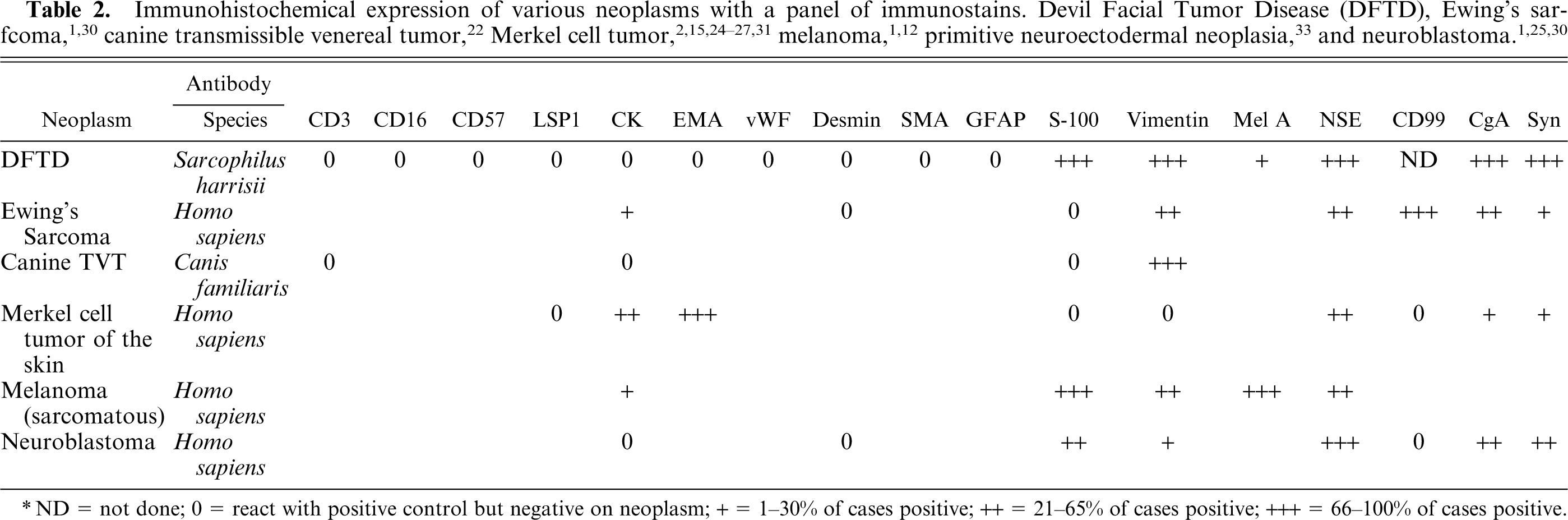
ND = not done; 0 = react with positive control but negative on neoplasm; + = 1–30% of cases positive; ++ = 21–65% of cases positive; +++ = 66–100% of cases positive.
DFTD shares some morphologic features with Ewing's sarcoma (ES) and ES shares some similar IHC staining characteristics with DFTD by staining negative for desmin and positive for vimentin, NSE, CgA, and synaptophysin. 1, 30 However, ES expresses CK and is negative for S-100, 30 and there are ultrastructural differences.
DFTD also shares some morphologic, immunohistochemical staining, 22 and possibly epidemiologic features with canine TVT, which is a round-cell tumor of the skin. However, TVT is negative for S-100. 22 Karyotyping by cytogenetic analysis has revealed complex chromosomal rearrangements in DFTD cells but the nature of the aneuploidy differed from that found in TVT: DFTD cells were hypodiploid and contained chromosomal deletions and 4 complex marker chromosomes whose derivation was uncertain (A. Pearse, personal communication).
Merkel cell tumors have rarely been reported in mammals, but the head and whiskers are the sites most commonly affected. 2, 11 Merkel cells are also argyrophilic 5 and staining with methyl green pyronin demonstrated a faintly staining accumulation of ribonucleic acid. Merkel cell tumors, like DFTD, share similar growth architecture 27 and positive immunostaining with NSE 15, 31 and are negative to LSP1 31 and high-molecular-weight CK (AE1/AE3). 24, 27 However, Merkel cell tumors differ from DFTD as they are positive for EMA, 31 variably positive for low-molecular-weight CK, 15, 27, 31 and negative for S-100 31 and vimentin. 27 There were mixed reports of reactivity to synaptophysin 15, 24, 27 and CgA. 2, 15, 27 In veterinary medicine, such neoplasms have since been reclassified as extramedullary plasmacytoma after immunoglobulins were found in the cytoplasm. 20, 22 Amyloid deposition has been reported to occur in some plasma cell tumors of canines, 22 but was not found in DFTD. Also, the ultrastructural studies of DFTD showed few differentiated organelles and lacked characteristics of Merkel cells such as neuroendocrine granules, intercellular junctions, rigid spinous processes, and a paranuclear collection of intermediate filaments. 27 In DFTD, small desmosome-like elements were reported, but these were too primitive.
Twenty-eight percent of DFTD cases were positive to melan A which provided further evidence that DFTD is of neural crest origin. Neural crest cells are pleuripotent 22 and have diverse differentiation capabilities, including differentiation into melanocytes. 4 This raises the following question. Without pigmentation, could DFTD be an amelanotic melanoma? The major diagnostic criteria for amelanotic melanomas is that they contain premelanosomes, mature melanosomes, or intracytoplasmic myelinoid figures (aberrant premelanosomes). 34 There was no ultrastructural evidence of such organelles within DFTD cells from the cases examined. Furthermore, Masson's-Fontana histochemical staining, which stained positively 27% of amelanotic melanomas in one study, 7 was consistently negative in all DFTD cases examined. Also, unlike DFTD cells, melanomas typically express cytokeratin, 1, 12 so it is unlikely that DFTD is a variant of melanoma.
It could be argued that DFTD IHC staining pattern is consistent with neuroblastoma, with both being consistently negative for CK and desmin and positive for vimentin, S-100, NSE, CgA, and synaptophysin. 1, 22, 25, 30 However, unlike neuroblastomas, DFTD cells are strongly positive for vimentin in all cases and lack ultrastructural evidence of neural differentiation such as cytoplasmic extensions or dendritic processes. Also, the anatomic distribution of the two differ, as neuroblastomas occur mainly in the adrenal medulla or in the retroperitoneum 22, 32 and these neoplasms are generally diagnosed in juveniles rather than sexually mature adults. 32
The immunohistochemical characteristics reported in this present paper, along with morphologic evidence refutes the original diagnosis of DFTD being a variant lymphosarcoma. 16 We have provided strong evidence for classifying DFTD as an undifferentiated neuroendocrine tumor (NET) which is unlike any other seen in humans or animals. There are many types of NETs and DFTD is a malignant NET as evidenced by the high occurrence of metastases (65%), large amount of necrosis, cellular pleomorphism and anaplasticity, rapid tumor growth, and apparent high mortality rate.
Comparative studies in humans indicate that genetic factors play a major role in the development of NETs. Further investigations are required to explore this in DFTD. Tasmanian Devils have moderately low genetic diversity, consistent with an island founder effect. 13 This along with their habit of engaging in “jaw wrestling” 29 and the propensity for tumors to arise in the lips, oral mucosa, or the face of Tasmanian Devils could be consistent with the tumors being spread as allografts amongst closely related family members. 10 Tasmanian Devils do have genetically differentiated eastern and northwestern subpopulations 13 and this could be one reason why the northwestern population remains so far unaffected.
In most DFTD tumors there is little evidence of a cell-mediated immunologic reaction with only 7% containing any evidence of lymphocyte infiltration in HE sections. This also could be consistent with an “allograft theory,” and further studies on the immune system of Tasmanian Devils including individuals with DFTD are warranted.
An alternative explanation for the sudden occurrence of DFTD in multiple geographic locations across Tasmania could be the occurrence of multiple concurrent epidemics owing to an unknown etiology. An epidemiologic analysis of DFTD should clarify this and may shed insights into the possible etiopathogenesis of the disease.
Transmission trials to test Koch's postulates are also required to investigate the allograft theory.
Footnotes
Acknowledgements
This work has been supported by the Tasmanian State Government. Testing was performed in the diagnostic laboratories of DPIW Tasmania; Murdoch University, Western Australia; and University of Sydney, New South Wales. We would like to thank Gerard Spoelstra and Michael Slaven for their untiring support with histologic preparations and help with the immunohistochemistry. We also thank Majid Ghoddusi, Susan Hemsley, Mark Krockenberger, Parameswaran Venkat, and Philip Nicholls with help on the choices of and reference materials for immunostaining. The project has also been strongly supported by our colleagues at Resource Management and Conservation Branch of DPIW for the collection of samples. All this work could not have been accomplished without the great technical assistance from the Diagnostic Services team within the Animal Health Laboratories of DPIW. All the work was conducted under the guidance of the DFTD steering committee.
