Abstract
Devil facial tumor disease (DFTD) is a transmissible neoplasm that is threatening the survival of the Tasmanian devil. Genetic analyses have indicated that the disease is a peripheral nerve sheath neoplasm of Schwann cell origin. DFTD cells express genes characteristic of myelinating Schwann cells, and periaxin, a Schwann cell protein, has been proposed as a marker for the disease. Diagnosis of DFTD is currently based on histopathology, cytogenetics, and clinical appearance of the disease in affected animals. As devils are susceptible to a variety of neoplastic processes, a specific diagnostic test is required to differentiate DFTD from cancers of similar morphological appearance. This study presents a thorough examination of the expression of a set of Schwann cell and other neural crest markers in DFTD tumors and normal devil tissues. Samples from 20 primary DFTD tumors and 10 DFTD metastases were evaluated by immunohistochemistry for the expression of periaxin, S100 protein, peripheral myelin protein 22, nerve growth factor receptor, nestin, neuron specific enolase, chromogranin A, and myelin basic protein. Of these, periaxin was confirmed as the most sensitive and specific marker, labeling the majority of DFTD cells in 100% of primary DFTD tumors and DFTD metastases. In normal tissues, periaxin showed specificity for Schwann cells in peripheral nerve bundles. This marker was then evaluated in cultured devil Schwann cells, DFTD cell lines, and xenografted DFTD tumors. Periaxin expression was maintained in all these models, validating its utility as a diagnostic marker for the disease.
Devil facial tumor disease (DFTD) is a recently emerged infectious disease that is threatening the survival of the Tasmanian devil (Sarcophilus harrisii), the world’s largest carnivorous marsupial.13,25 This cancer is characterized by primary tumors located around the mouth and face that grow to in excess of 10 cm in diameter and often ulcerate. Karyotypic analysis of DFTD has revealed a substantial and consistent pattern of chromosomal abnormalities among the tumors. This similarity has led to the proposal that DFTD is transmitted as an allograft. 33 Further support for clonal transmission and therefore the contagious nature of this tumor is provided by genetic analysis of DFTD tumors at the DNA, microsatellite, and major histocompatibility complex loci.28,39 Facial biting with the transfer of viable tumor cells dislodged from ulcerated tumors in close proximity to the canine teeth is considered the most likely means of DFTD transmission. 32 There is no natural immunity to the establishment and growth of the tumors, and affected animals die within months of tumor appearance.13,18,41 At the current rate of spread of disease, the species could face extinction within 25 to 35 years. 26
Conservation management trials have begun. These include establishment of captive populations of healthy devils in mainland Australia and the removal of DFTD-affected devils from relatively isolated local populations in Tasmania. The diagnosis of DFTD is based on histopathology, clinical appearance of the disease in affected animals, and cytogenetics. Karyotyping is the most accurate method for tumor diagnosis, and classical analysis by chromosomal banding requires collection of fresh tumor biopsies and establishment of cell cultures in vitro. This is not only time-consuming but also labor intensive. As Tasmanian devils are also susceptible to other types of neoplastic diseases, a specific diagnostic test that can be carried out rapidly on fixed samples is required to differentiate DFTD tumors from other cancers of similar morphological appearance.2,10,16
DFTD was initially described as a malignant neuroendocrine neoplasm. 22 Our recent studies on the Tasmanian devil transcriptome revealed that DFTD expresses a set of genes related to the myelination pathway in the peripheral nervous system (PNS). 28 We also detected protein expression of myelin and other peripheral nerve markers including myelin basic protein (MBP), peripheral myelin protein 22 (PMP22), myelin protein zero (MPZ), nerve growth factor receptor (NGFR), and periaxin (PRX). On the basis of these results, we proposed that DFTD is of Schwann cell origin and presented periaxin as a potential diagnostic marker for DFTD. In this study, we present a semi-quantitative assessment of the pattern of immunohistochemical expression of myelin and other neuronal proteins in DFTD tumors and Tasmanian devil tissues. Additionally, we further evaluate the utility of periaxin as a diagnostic marker in alternative models of the disease such as DFTD cell lines and murine xenografted DFTD tumors as well as for devil Schwann cells in primary culture.
Methods
Tissue Samples
A total of 20 primary DFTD tumors and 10 DFTD metastases samples were used in this study. The samples were supplied by the tissue bank held at the Menzies Research Institute, University of Tasmania, and the Mount Pleasant Laboratories from the Tasmanian Department of Primary Industries, Parks, Water and Environment (DPIPWE).
Histology and Immunohistochemistry
Three-micrometer-thick paraffin sections were prepared from DFTD tumors, and normal devil tissues previously fixed in 10% buffered formalin and placed onto 3-aminotriethoxysilane-coated slides. Standard hematoxylin and eosin (HE) staining was performed at the Anatomical Pathology Department of the Royal Hobart Hospital. For immunohistochemistry, sections were initially deparaffinized in a histology oven at 60°C for 15 minutes followed by two 5-minute washes in xylene and then rehydrated through successive graded ethanol solutions and washed for 5 minutes in distilled water. Heat-induced antigen retrieval was performed in citrate buffer, pH 6 (Dako, Carpinteria, CA) using an electric pressure cooker at medium heat for 10 minutes, followed by a 20-minute cooling period at room temperature.
The slides were preincubated with serum-free blocking solution (Dako) for 30 minutes and then incubated with the primary antibody for 1 hour (see Table 1 for a complete list of antibodies). Endogenous peroxidase activity was then quenched by treating the slides in 3% hydrogen peroxide in phosphate-buffered saline (PBS) for 10 minutes at room temperature. Signal detection was carried out using the anti-mouse (K4007) or anti-rabbit (K4003) Dako EnVision System, HRP, (Dako). The sections were counterstained with hematoxylin for 40 seconds and mounted.
List of Antibodies Tested by Immunohistochemistry (IHC) and Immunofluorescence (IF)
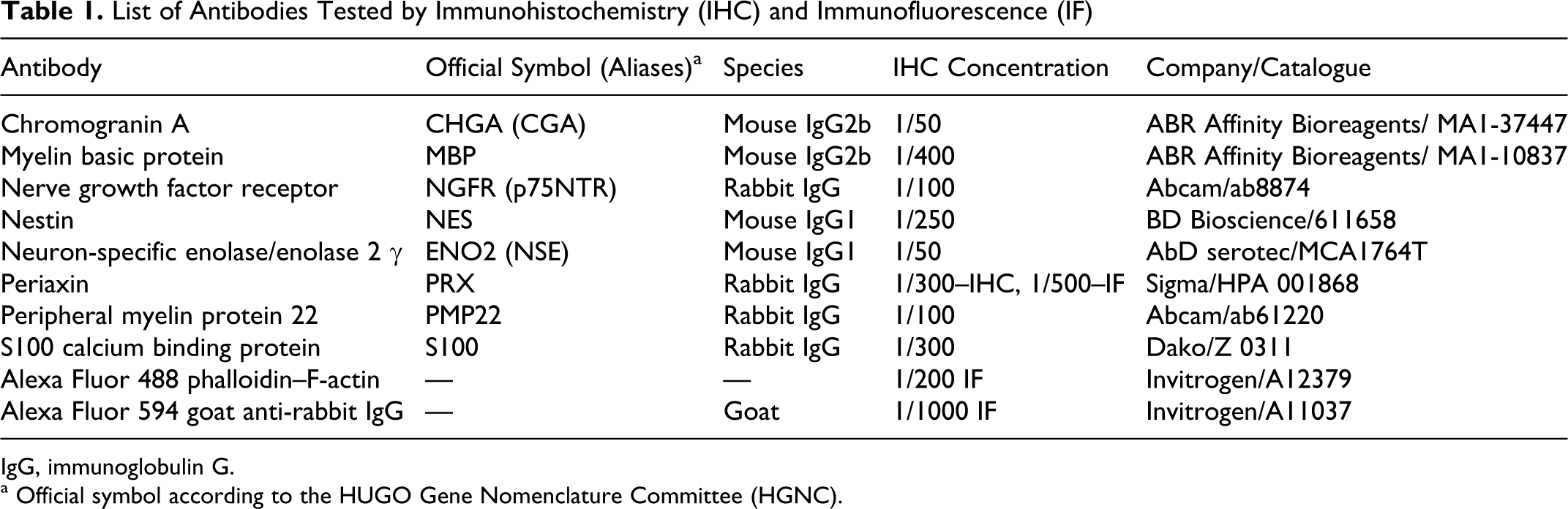
IgG, immunoglobulin G.
a Official symbol according to the HUGO Gene Nomenclature Committee (HGNC).
The specificity of the primary antibodies was validated with recognized positive controls using appropriate normal devil tissues and negative controls by omitting the primary antibody for each immunohistochemistry run. Peripheral nerves bundles, usually located in tissues adjacent to tumor nests, were used as a positive control for MBP, NGFR, NSE, PMP22, periaxin, and S100; spinal cord was used as control for nestin (NES) and pituitary gland for CGA.
Annotation and Interpretation of Protein Expression in Tissues
A light microscope (Olympus-BX50) coupled with a camera (Leica-DFC320) was used for visualization and acquisition of the images. Labeling reactions, analyzed semiquantitatively by the principal author and a trained veterinary pathologist, were assessed according to the following criteria: (1) immunolabeling intensity (negative, weak, moderate, or strong) compared with that of the positive control; positive labeling was defined by the presence of 10% or more immunoreactive cells in tumor sections; (2) fraction of tumor cells labeled (10–25%, 25–50%, 50–75%, or >75%); and (3) subcellular localization (nucleus, cytoplasmic or associated with the cell membrane) combined with parameters describing the labeling characteristics (ie, smooth, granular, or fine granular).
Primary Schwann Cell Culture
The following protocol was modified from Mauritz et al. 24 Sciatic nerves were collected into Dulbecco’s modified Eagle medium (DMEM; Gibco, Auckland, New Zealand) during autopsy of an adult male devil, and the epineurium was dissected with micro scissors and forceps. The epineurium-free tissue was incubated for 14 days at 37°C and 5% CO2 in predegeneration solution that contained DMEM, 10% fetal calf serum (FCS; Gibco), 1% penicillin/streptomycin, and 0.002 μM forskolin (Sigma, St. Louis, MO). Nerve fascicles were washed gently with DMEM and then incubated with DMEM-FCS supplemented with 10% FCS and containing 50 U of penicillin and 50 μg of streptomycin (penicillin–streptomycin liquid, Sigma) and an enzyme mixture consisting of 0.15 U/ml collagenase and 0.24 U/ml dispase (Invitrogen, Carlsbad, CA) at 37°C and 5% CO2 for 20 hours. After dissociation through glass pipettes, the homogenous cell suspension was centrifuged at 235 × g for 5 minutes at 21°C. Following resuspension, the cells were filtered through 80-μm mesh to remove undigested tissue. The cell filtrate was plated into a tissue culture flask and incubated at 37°C and 5% CO2 in DMEM culture media (DMEM, 0.02 μM forskolin, 10 ng/ml fibroblast growth factor (Promega, Madison, WI), 5 μg/ml bovine pituitary extract (Sigma), and 1% penicillin–streptomycin liquid (Invitrogen). Two weeks later the cells were harvested for immunofluorescence analysis.
Immunofluorescence
Cultured DFTD tumor cells and primary cultured devil Schwann cells were plated onto poly-L-lysine (Sigma)–coated coverslips and incubated to semiconfluence at 37°C and 5% CO2. DFTD tumor cells were incubated in complete RPMI medium (RPMI 1640 [Gibco]; 10% FCS; 2 mM L-glutamine [Sigma], and 40 mg/ml of gentamicin [Pfizer, Sydney, Australia]). Schwann cells were incubated in the DMEM culture medium described above. The cells were fixed in 4% paraformaldehyde-glucose (Sigma) for 30 minutes followed by 3 washes with PBS. Cells were then incubated in 10% bovine serum albumin for an hour to block nonspecific binding. Primary antibody was incubated in 0.3% Triton X100 overnight at 4°C. After washing with PBS, the cells were incubated in the dark with a fluorescently labeled secondary antibody in PBS for an hour at room temperature. Actin stain (phalloidin) was added and incubated for an additional 30 minutes. The coverslips were washed with PBS and rinsed with distilled water prior to mounting onto glass slides using fluorescent mounting medium (Dako). Labeling was visualized under a confocal microscope (Zeiss LSM 510 Meta). Isotype control experiments for all the antibodies were also performed. The antibodies used for immunofluorescence are showed in Table 1.
Murine Xenografted DFTD Tumors
We have described a DFTD xenograft model in immunocompromised NOD/SCID mice. 19 In brief, NOD/SCID mice were injected subcutaneously with viable DFTD tumor cells. All inoculated mice developed tumors. Additionally, cells derived from these tumors were successfully passaged into other NOD/SCID mice.
Results
Histological Features of DFTD
The histological appearance of DFTD tumors was identical to previous descriptions in both primary tumors and metastases. 21 Briefly, tumor cells formed large nodular aggregates, often within a thin, fibrous pseudo-capsule and composed predominantly of neoplastic cells supported by minimal fibrovascular stroma. Cells could also be arranged in bundles, cords, or streams separated by fine strands of collagen fibers. Cells were polygonal to round with a high nuclear to cytoplasm ratio. Mitotic figures were commonly observed with up to 7 figures at high magnification (40×) in some cases. Most of the large tumor masses contained central areas of necrosis. Inflammatory cells were rare and when present were mostly confined to the periphery of the tumor.
Patterns of Expression of Schwann Cell Markers in DFTD and Devil Tissues
We have previously demonstrated that a number of Schwann cell markers are expressed in DFTD, including PRX, S100 protein, PMP22, NGFR, NES, and MBP. 28 From this work we confirmed that periaxin expression was observed in 100% of DFTD primary tumors (n = 20) and 100% of DFTD metastases (n = 10). Moderate immunoreactivity for S100 protein was also observed in 100% of primary tumor samples (n = 20) and 100% of DFTD metastases (n = 10). PMP22 expression was detected in 95% of primary tumors (19/20) and 90% of metastases (9/10). As well, 65% of primary DFTD tumors (13/20) and 80% of metastases (8/10) were positive for NGFR. Immunoreactivity for NES was found in 45% of primary tumors (9/20) and 40% of DFTD metastases (4/10), and positivity for myelin basic protein (MBP) was only observed in 2/20 DFTD primary tumors.
In this article, we present a detailed analysis of the immunohistochemical labeling patterns of periaxin, S100 protein, PMP22, NGFR, NES, and MBP. We also include the neuroendocrine markers neuron-specific enolase (NSE) and chromogranin A (CGA). The semiquantitative evaluation of the labeling includes fraction of labeled cells, cellular localization, and description of the characteristics of the stain (Table 2). We also present the pattern of expression of the markers in normal devil tissues (Table 3).
Expression of Myelin and Neuronal Markers in DFTD Primary Tumors and Metastases
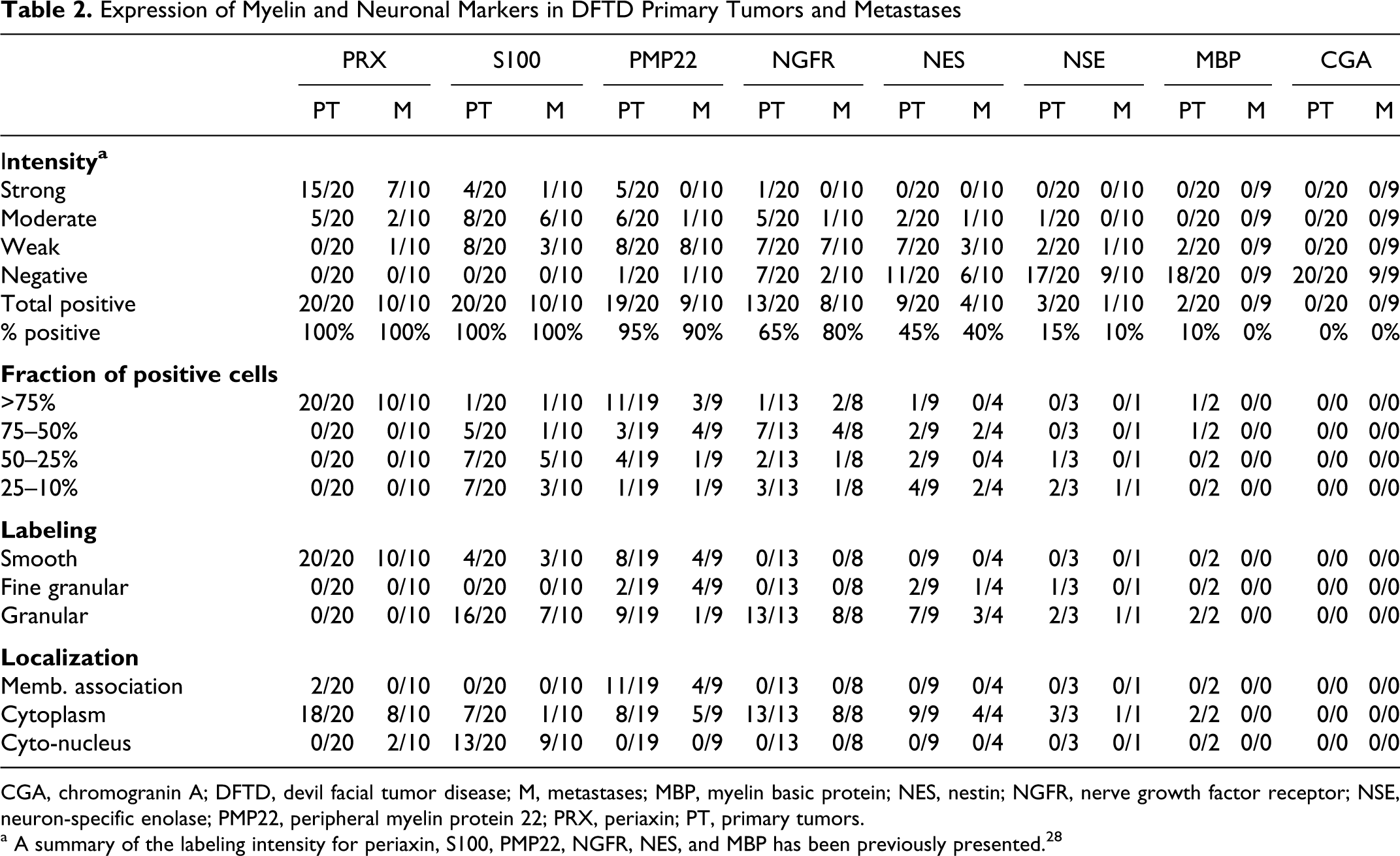
CGA, chromogranin A; DFTD, devil facial tumor disease; M, metastases; MBP, myelin basic protein; NES, nestin; NGFR, nerve growth factor receptor; NSE, neuron-specific enolase; PMP22, peripheral myelin protein 22; PRX, periaxin; PT, primary tumors.
a A summary of the labeling intensity for periaxin, S100, PMP22, NGFR, NES, and MBP has been previously presented. 28
Pattern of Expression of Selected Markers in Normal Devil Tissues
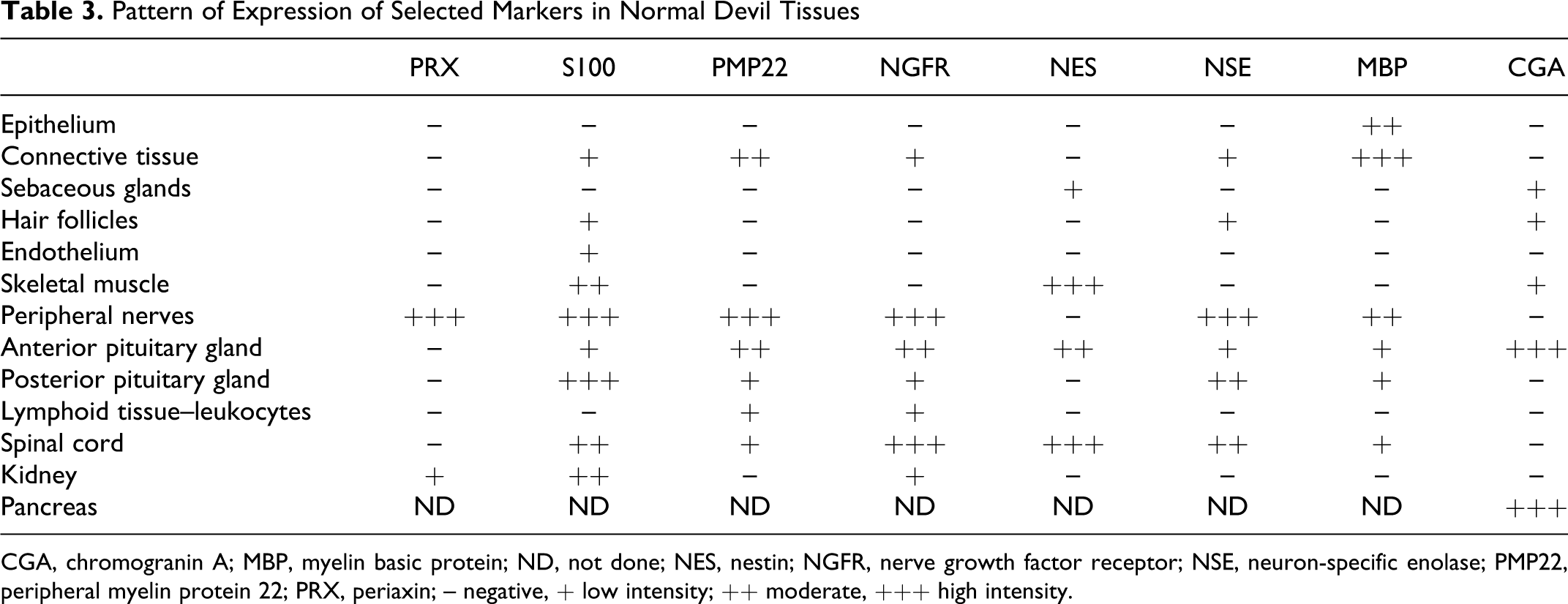
CGA, chromogranin A; MBP, myelin basic protein; ND, not done; NES, nestin; NGFR, nerve growth factor receptor; NSE, neuron-specific enolase; PMP22, peripheral myelin protein 22; PRX, periaxin; – negative, + low intensity; ++ moderate, +++ high intensity.
Of the 8 markers assessed, periaxin, a marker of myelinating Schwann cells, 9 gave the most consistent labeling of tumor cells (Table 2). In all cases the labeling was moderate to strong, cytoplasmic and observed in more than 75% of the tumor cells. Periaxin expression, as previously indicated, 28 was also observed in all DFTD primary tumors and metastases. Stronger immunoreactivity was occasionally observed at the edges of the tumor fascicles. One particular feature of periaxin was its strong specificity for DFTD tumor cells and the lack of background labeling in other tissues (Figs. 1, 2 ). Peripheral nerves within these DFTD tumor sections were clearly identified by their strong and specific immunoreactivity for periaxin (Table 3).
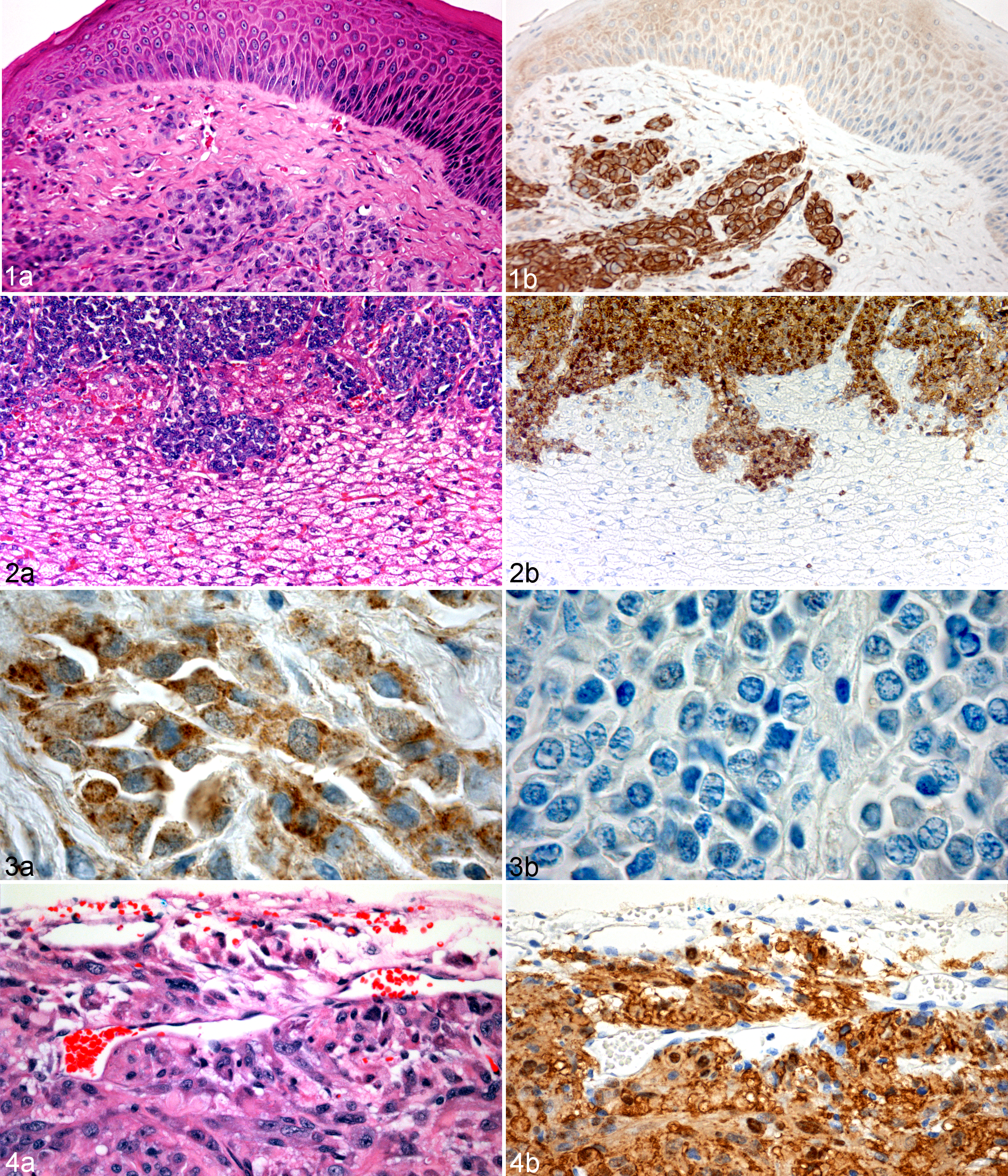
S100 protein is a commonly used marker for peripheral nerve sheath tumors. 6 S100 was previously observed in all DFTD samples. 28 However, the intensity of the labeling was weak to moderate and detected only in a small proportion of tumor cells; background labeling was also observed in other tissues (Tables 2 and 3). In DFTD tumor cells, labeling with S100 was consistently granular and located in both cytoplasmic and nuclear compartments. Similar to periaxin, the level of S100 expression varied across tumor nodules (ie, the cells at the periphery of the tumor exhibited stronger immunoreactivity compared to cells in the center). Strong S100 immunoreactivity was also observed in peripheral nerves and in the nucleus of neurons and oligodendrocytes located in devil spinal cord.
Strong immunoreactivity for PMP22, a major component of myelin in the peripheral nervous system, was observed at the outer margins of the tumor nodules and in neoplastic cells at the advancing edge of the tumor. PMP22 expression was smooth to fine granular. The labeling was predominantly cytoplasmic and frequently enhanced at the cell membrane (Table 2). Necrotic areas also showed strong and specific granular labeling. Peripheral nerve bundles were positive for PMP22. Intense nonspecific background of the connective tissue was frequently observed (Table 3).
The labeling pattern for NGFR was cytoplasmic and granular, and it was observed in more than 50% of the DFTD tumor cells (Table 2). Some populations of cells, usually located to the edges of the tumor fascicles, were strongly positive for NGFR. Immunoreactivity to NGFR was observed in peripheral nerves, spinal cord, and anterior and posterior pituitary gland. Occasional nonspecific labeling was observed in connective tissues and in renal tubular epithelium (Table 3).
The labeling expression of NES was weak to moderate and very variable. Positive cells showed granular cytoplasmic labeling. A particular feature of NES labeling (not observed with the other markers) was its tendency to form coarse granules or clusters within the cytoplasm of the cell. Skeletal muscle cells were strongly positive for NES, whereas sebaceous glands showed weak immunoreactivity (Tables 2 and 3).
As indicated previously, positivity for MBP was observed in only 2 of the 20 DFTD primary tumors. 28 In these 2 samples, more than 50% of the cells showed immunoreactivity. The labeling pattern was diffuse, granular, and cytoplasmic. MBP expression was observed in peripheral nerves. Significant patchy background labeling was present in many samples, making overall interpretation difficult. For instance, nonspecific and strong labeling was observed in connective tissues, epidermis and dermis. Necrotic foci within the tumor also showed nonspecific immunoreactivity.
Weak to moderate granular and cytoplasmic labeling for NSE was observed in 13% of samples (4/30) with less than 50% of cells in each sample being positive (Fig. 3a ). Notably, cells at the periphery of the tumor nests showed stronger immunoreactivity. Immunoreactivity for NSE was detected in peripheral nerves, spinal cord, and pituitary gland. Weak background labeling was observed in connective tissues and hair follicles.
No immunoreactivity was detected for CGA in DFTD viable tumor cells (Fig. 3b). However, strong granular and cytoplasmic immunoreactivity for this protein, which is normally found in endocrine cells, 14 was observed in pituitary gland and cells of the islets of Langerhans in the pancreas. Background labeling was observed in hair follicles and muscle cells.
Periaxin Expression in Xenografted Tumors and Cultured Schwann Cells
We observed strong periaxin labeling in all neoplastic cells within xenografted DFTD tumors. The periaxin labeling was maintained in tumors from more than 2 passages (Fig. 4).
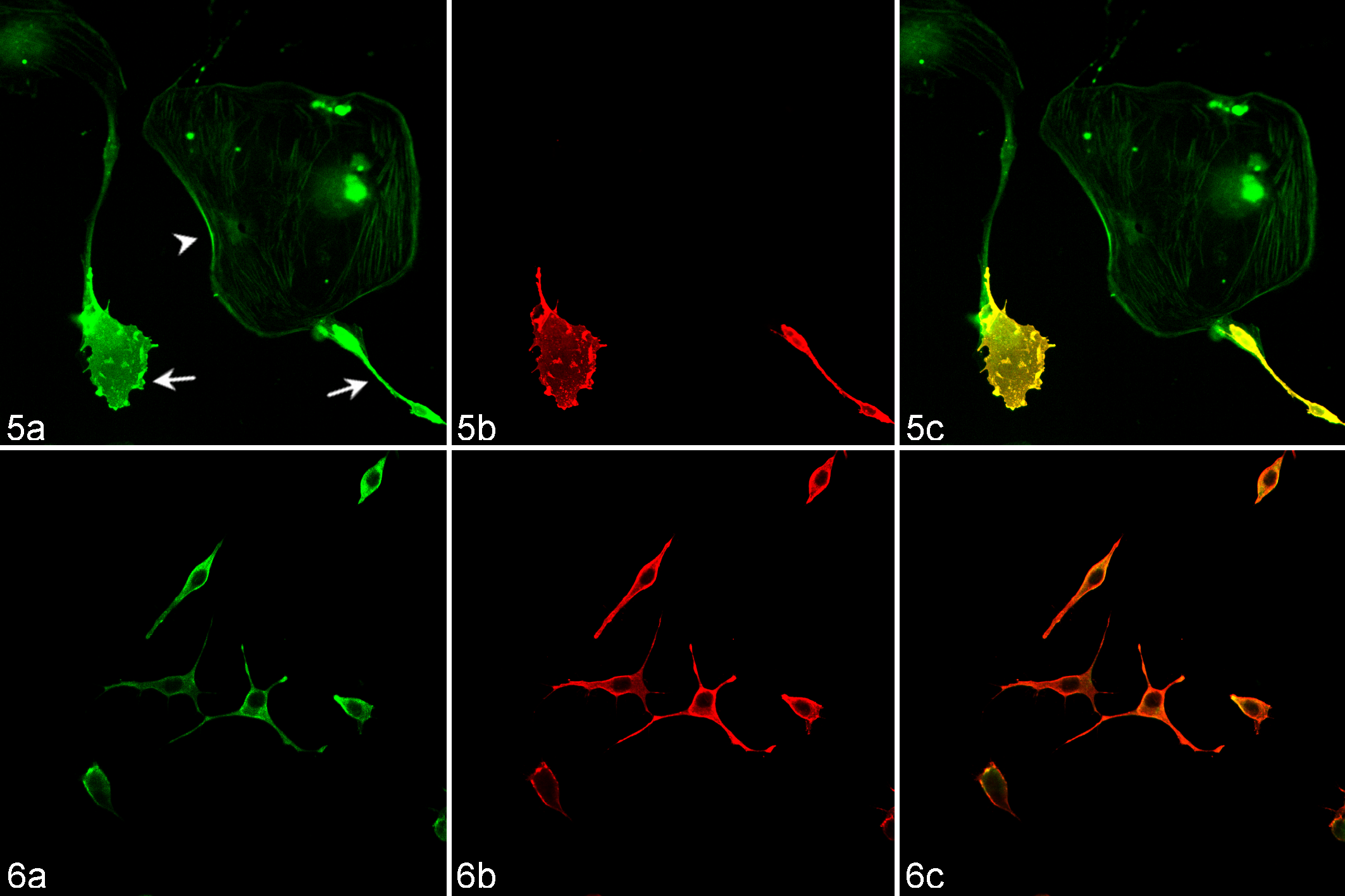
Finally we used immunofluorescence techniques to confirm the expression of periaxin in primary cultured Schwann cells from an adult Tasmanian devil and DFTD cultured cell lines. Periaxin expression was conserved in normal Schwann cells and was a useful marker to differentiate these cells from cocultured fibroblasts (Fig. 5). Periaxin also consistently labeled all DFTD tumor cells obtained from tissue culture (Fig. 6).
Discussion
Applying immunochemistry techniques to the study of protein expression in marsupial tissues is challenging.3,22 One of the major constraints is the limited availability of commercial antibodies targeted to marsupial tissues. Application of sensitive and specific immunohistochemical markers for peripheral nerve sheath tumors has also been a diagnostic challenge in human oncology. 34 This is especially relevant for antibodies against antigens not widely studied such as myelin proteins. In this study of devil tissue, we found that some antibodies, in particular polyclonal antibodies, could produce relatively high levels of nonspecific labeling in connective and other tissues. Moreover, secondary avidin or biotin conjugated antibodies produced high background. This nonspecific labeling was notably reduced with a biotin-free visualizing system and by selecting monoclonal antibodies. However, although there have been important advances revealing the composition of the marsupial genome (eg, the first complete genome sequence for a marsupial was recently presented), 27 investigations that confirm protein expression are limited. 3 In this sense, the results of immunohistological studies, such as the one presented here, are still descriptive rather than explanatory. Molecular and genetic research studies on the Tasmanian devil and DFTD are in progress, and they will complement and improve our understanding of protein expression, not only in this species but in marsupials in general.
The first immunohistological study of DFTD revealed labeling characteristics consistent with cells of neuroendocrine origin. 22 The neural crest, which arises from the neuroectoderm, gives rise to a number of cell types during vertebrate embryogenesis. These neural crest–derived cells include neurons and glial cells from the peripheral nervous system, pigment cells in the skin (melanocytes), endocrine cells, and a variety of mesenchymal cell types.8,17,20 Our study provided additional support to the neuroectodermal origin of DFTD by detecting immunoreactivity to NSE, a protein commonly found in neuronal and neuroendocrine cells.23,35 However, we were unable to detect CGA, a glycoprotein that is found in secretory vesicles of neurons and endocrine cells. 14 The presence of neuronal and Schwann cell markers in DFTD and the lack of CGA expression support the recent finding that DFTD tumors originated from cells of the peripheral nervous system lineage rather than neuroendocrine as originally proposed. 22
Tumors of the PNS have been described in humans, dogs, cattle, and other animals.1,5,30,36,37 Furthermore, the Schwann cell is thought to be the major contributor to the formation of both benign and malignant peripheral nerve sheath neoplasms.4,7,11 Diagnosis of these tumors is a diagnostic challenge given the lack of specific immunohistochemical markers of neural differentiation. 34 Although labeling for S100 protein shows some nonspecific activity, it is the most commonly used marker to identify peripheral nerve sheath tumors (PNST) of various types.6,42 In this study, S100 was identified in all DFTD tumor samples. The intensity of labeling and fraction of immunoreactive cells varied considerably among samples, which has been a common finding in other studies.31,40 We observed S100 labeling in other tissues such as endothelium, skeletal muscle, hair follicles, kidney, and connective tissue, and so it is not a particularly cell-specific marker.
Other markers normally tested in PNST with different levels of reliability include MBP, NGFR, and NES.515,38,40 We found NGFR positivity in 70% of our samples. However, intensity of labeling was highly variable. Nestin and MBP stained only a small fraction of samples. Interpretation of results was also difficult because of the high level of nonspecific labeling with MBP.
We have also tested periaxin, a recently described marker of Schwann cell lineage that was found to be expressed in the devil transcriptome.9,28 We found that labeling for periaxin was intense and specific in DFTD tumors. Periaxin strongly labeled almost 100% of the neoplastic cells in all the DFTD primary tumors and metastases with minimal nonspecific labeling. Our study confirmed that the expression of periaxin at the protein level is preserved after the xeno-transplantation and passage of DFTD tumor cells into NOD-SCID mice, including tumors from the second generation. Similarly, cultured devil Schwann cells and DFTD tumor cell lines labeled positive for periaxin. Lacerations and scars due to biting are common in devils, 12 and in some cases small lesions can be misdiagnosed as DFTD. Periaxin has been a useful marker for identifying tumor cells in punch biopsy of early DFTD tumors. Furthermore, 9 non-DFTD Tasmanian devil tissues were previously evaluated to be negative for periaxin. 28 Samples included 3 mammary adenocarcinomas, 3 histiocytomas, 1 cystic papillary adenocarcinoma, 1 squamous cell carcinoma, and 1 hepatic fibrosarcoma.
Therefore, we confirm periaxin as a reliable, sensitive, and specific marker for DFTD. It has been suggested that the most valuable immunohistologic markers are those that are linked to the cell of origin or those that reflect the genetic signature of the tumor. 29 As shown here, periaxin certainly meets these 2 criteria. Our study also suggests that periaxin could be a potential maker for the diagnosis of tumors of Schwann cell origin in other species.
An interesting aspect of our findings is that despite their similar morphology, the tumor cells surrounding the tumor nests have higher levels of protein expression, especially in the areas where the cells are in close contact with the surrounding normal connective tissue. The variability of expression of the markers between and within a tumor could reflect different stages of tumor development. This is revealed by the pattern of growth of the tumor fascicles or nodules, which is clearly concentric and layered with centralized areas of tumor cell necrosis. These findings suggest an important role of the cellular contacts and tumor microenvironment for proliferation and migration of the tumor cells. The interaction of tumor cells with the extracellular matrix and its function for tumor invasion are important aspects that require further investigation.
In summary, our study confirmed periaxin as a new sensitive marker for DFTD that will greatly facilitate the diagnosis of the disease in histological and cytological samples. The improvement of the diagnosis of DFTD and the finding that overexpressed proteins in the tumor can be recognized with specific markers open new opportunities for the understanding and management of this unusual disease.
Footnotes
Acknowledgements
We are grateful to Colette Harmsen, Sarah Peck, and Stephen Pyecroft (DPIPWE) for providing many of the samples. We thank Narelle Phillips and Steve Weston for invaluable help with histological preparations and the staff from the Neurorepair Group at Menzies Research Institute.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: a Dr Eric Guiler Tasmanian Devil Research Grant and support from the Turner Foundation.
