Abstract
Devil facial tumor disease (DFTD) is an emergent transmissible cancer exclusive to Tasmanian devils (Sarcophilus harrisii) and threatening the species with extinction in the wild. Research on DFTD began 10 years ago, when nothing was known about the tumor and little about the devils. The depth of knowledge gained since then is impressive, with research having addressed significant aspects of the disease and the devils’ responses to it. These include the cause and pathogenesis of DFTD, the immune response of the devils and the immune evasion mechanisms of the tumor, the transmission patterns of DFTD, and the impacts of DFTD on the ecosystem. This review aims to collate this information and put it into the context of conservation strategies designed to mitigate the impacts of DFTD on the devil and the Tasmanian ecosystem.
Keywords
The Tasmanian devil (Sarcophilus harrisii) is threatened with extinction in the wild by an aggressive, transmissible, and invariably fatal cancer known as devil facial tumor disease (DFTD). The devil is the world’s largest extant carnivorous marsupial and unique to the island state of Tasmania. It is listed as endangered by the International Union for the Conservation of Nature as a result of the DFTD epidemic. 15
The extinction in the wild of the Tasmanian devil would have profound impacts. It is an internationally recognized species with an iconic status and inhabits a unique ecologic niche. As devil populations decline, this niche is at risk of being filled by feral cats and potentially foxes, the consequences of which would prove disastrous for native species. 18,20
DFTD is a transmissible tumor, passed between devils by biting. A viral etiology was initially suspected when the transmission pattern became obvious 37 ; however, it is now clear that the tumor cells are the sole etiologic agent. 56 As we discuss below, this is a rare event in nature, as the tumor must not only find a way to infect the new host but also evade the host’s immune mechanisms to colonize the tissues. This form of tumor transmission in vertebrates is apparent in only 1 other disease—the canine transmissible venereal tumor (CTVT), 62,74 which shares some similarities to DFTD. Interestingly, a new transmissible cancer has been recently described in soft-shell clams, suggesting that transmissible cancers might be more common in nature than originally thought. 43
Measures to conserve the Tasmanian devil include the maintenance of a genetically sustainable captive insurance population, the translocation of healthy devils to disease-free areas, and research aimed at developing a protective DFTD vaccine. Nature has also responded to the dramatic decline in the devil population with reproductive compensation, seeing an increase in precocial breeding and possibly an increase in female young born to diseased mothers. 25,35 Time will tell how successful these measures are in countering the predicted extinction of the wild Tasmanian devil.
Origins of DFTD
Cancer is the result of uncontrolled cell division that evades the host’s immune surveillance function. Cancer cells typically die with their host, but DFTD has the remarkable feature of being a clonally transmissible cancer. 50,56 It thus spreads from one individual to the next and outlives its host in the process. CTVT—the only other naturally occurring transmissible tumor of vertebrates—is a sexually transmitted cancer of dogs, with a worldwide distribution, 62 and it is discussed later in this review.
Carcinogens, infectious agents, and genetic predisposition are the usual inciting causes of cancer. It is unknown what gave rise to the first devil facial tumor (DFT), but genomic analysis demonstrated that this primary tumor appeared in a female devil <20 years ago. 49 DFTD was first observed in 1996 in the far northeast of Tasmania 19 and has since spread to affect the majority of the species’ geographic range, up to 90% of individuals within certain locations. 41 It causes mortality in all affected animals, seemingly within 6 months of the tumor’s appearance. 18 Death results from starvation, depending on the size and location of the tumors, or from metastases and subsequent organ failure.
An independent review of chemical residues found in healthy devils and those affected with DFTD was commissioned by the Save the Tasmanian Devil Program in 2008. 63 The chemicals selected for investigation included heavy metals, herbicides, and pesticides. Residues of dioxins, dibenzofurans, polychlorinated biphenyls, brominated diphenyl ethers, arsenic, cadmium, and lead were detected in the fat and/or liver of most animals but at levels similar to those found in other species at the top of the food chain, including humans. There were no significant differences in residue levels between the healthy and diseased devils, suggesting that there is no link between chemicals and DFTD. A viral etiology was also explored, and although the results have not been formally published, the electron microscopy investigation of 29 DFTs found no evidence for a causal or associated virus. 61
Pathology of DFTD
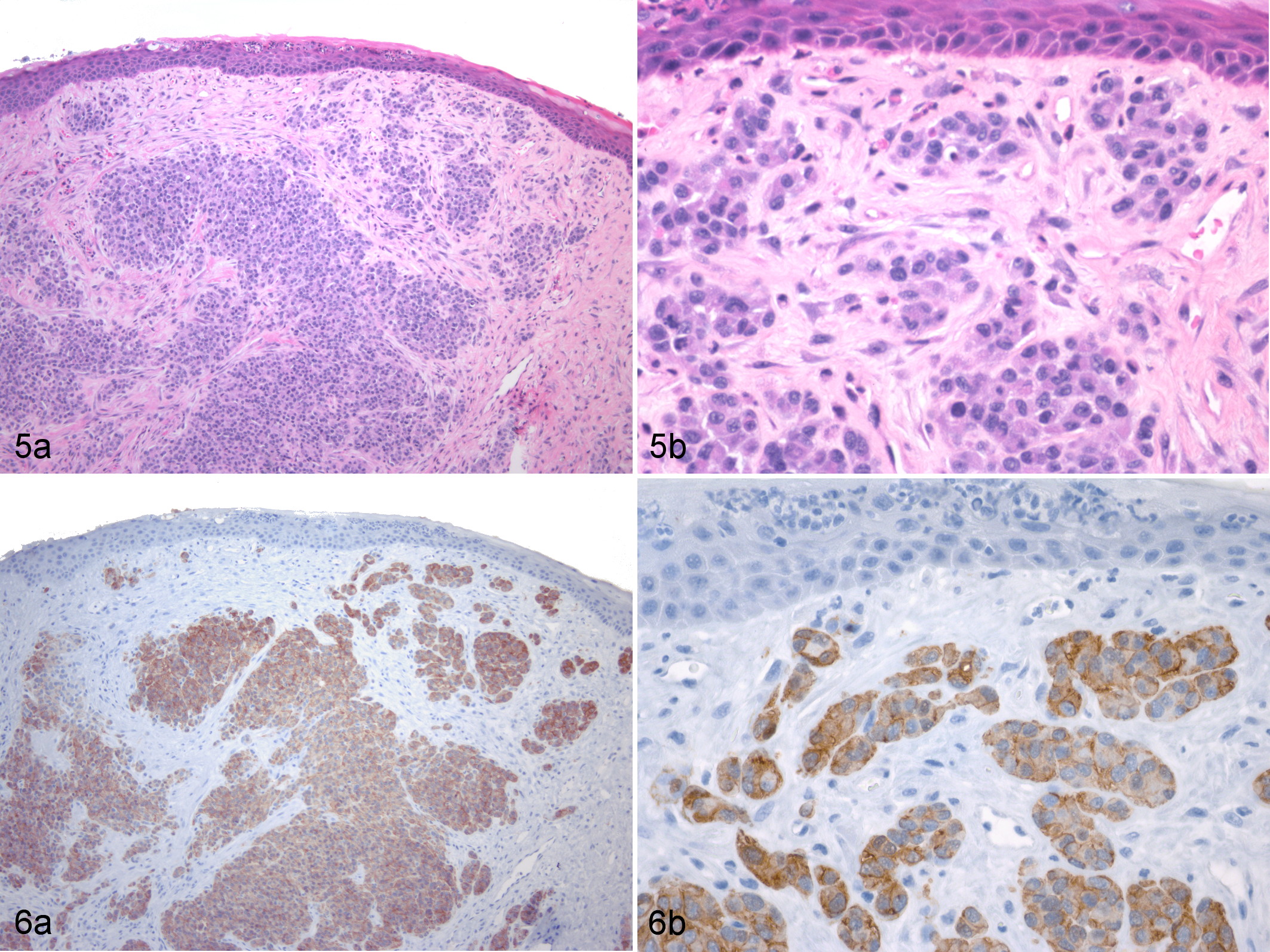
The prevalent characteristic of DFTD, as the name suggests, is the presence of locally aggressive tumors on the facial area. The tumors occur primarily inside the mouth (gingival mucosa, hard palate, lips) and on the head (cheeks, lips, muzzle) and neck. More than 1 tumor can be present and have considerable variation in size and external appearance. The majority of tumors are well circumscribed, >3 cm, and ulcerated. The tumors often show disruption of the epithelium, necrosis, exudation, and bacterial contamination (Figs. 1–3). Metastases are a common finding (65% of cases in 1 study) 38 and occur primarily in the draining lymph nodes, lungs, and kidneys (Fig. 4). The neoplastic cells are arranged in nodules or bundles and enclosed by a thin pseudo-capsule, and the cells have been described as pleomorphic, round to spindle shaped, and with large central nuclei. 38 Variable levels of mitoses, necrosis, and a paucity of inflammatory cells were also described (Fig. 5). The anaplasia exhibited by the DFTD cells 38 is consistent with the highly malignant nature of the tumor. 10
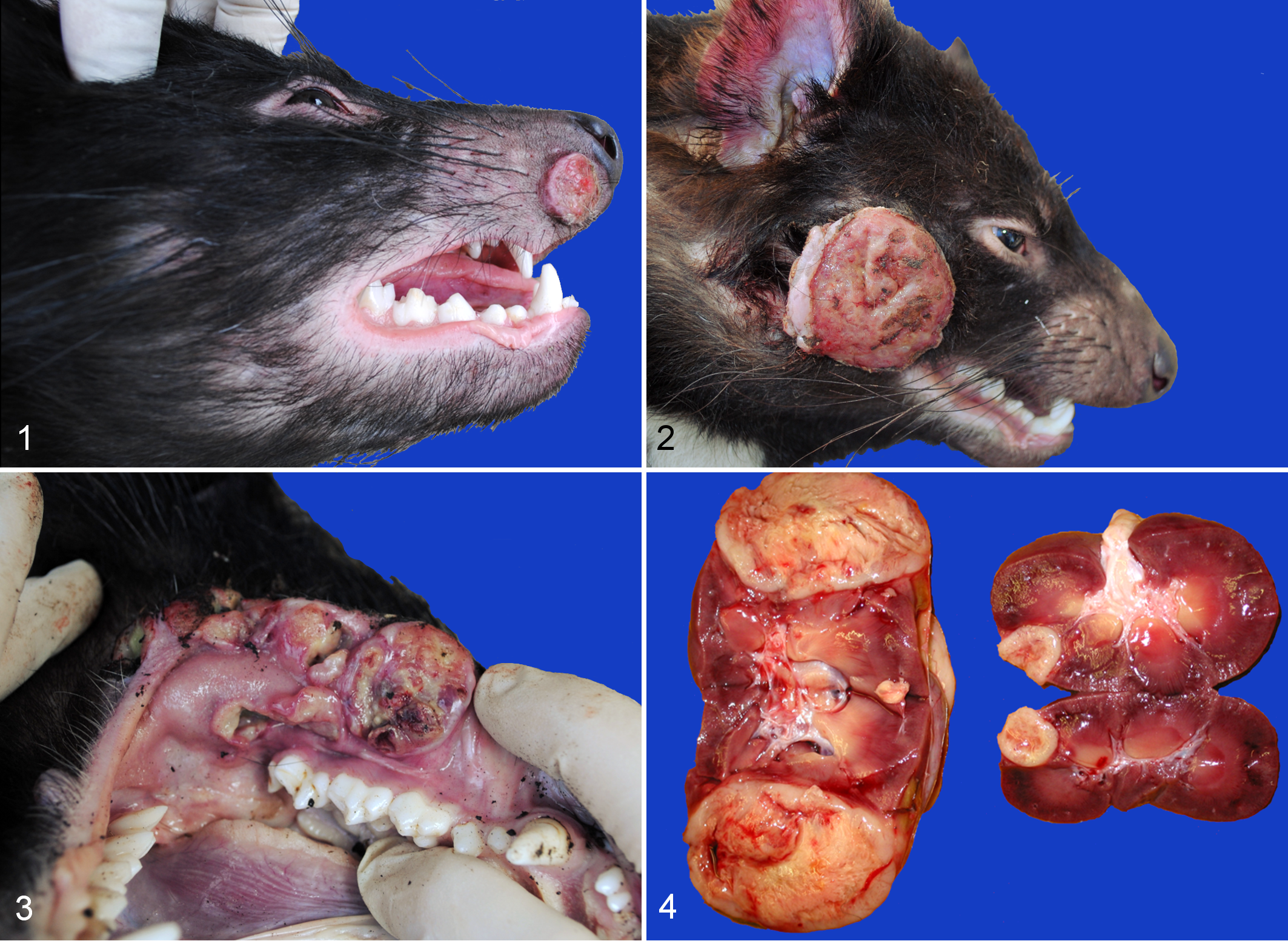
Devil facial tumor disease, Tasmanian devil.
Devil facial tumor disease (DFTD), skin, Tasmanian devil.
Based on immunohistochemical findings, DFTD has been classified as a sarcoma, since it is negative for epithelial markers such as cytokeratin, epithelial membrane antigen, and von Willebrand factor and positive for S-100 and vimentin. DFTs were initially found positive for neuroendocrine markers (neuron-specific enolase, chromogranin A, and synaptophysin), which prompted the authors to suggest that DFTD was of neuroendocrine origin. 39 Further research, however, found DFTs to be negative for chromogranin A and identified expression of proteins associated with the peripheral nervous system.
Deep sequencing of the DFTD transcriptome revealed the tumor to be of Schwann cell origin. 50 Several genes involved in the myelination of axons are upregulated in DFTD cells when compared with control tissues. The gene that encodes for myelin basic protein had the highest expression in DFTD when compared with the housekeeping gene. Other structural myelin genes apparent in the DFTD transcriptome include periaxin, myelin protein zero, and peripheral myelin protein 22. 50 Protein expression as determined by immunohistochemical analyses found 100% of primary DFTD tumors, DFTD metastases, DFTD cultured cells, and mouse xenografts to be strongly positive for a Schwann cell marker, periaxin (Fig. 6). 76 Other immunohistochemistry marker proteins—including S100, peripheral myelin protein 22, nerve growth factor receptor, nestin, and myelin basic protein—did not identify all DFTD cells, and some showed significant levels of background/nonspecific staining.
The genetic and protein analysis provided convincing evidence for the Schwann cell origin of DFTD. 76 The ability of Schwann cells to instigate and modulate an immune response 82 and the plasticity retained by mature Schwann cells 24 may have contributed to DFTD evolution as a transmissible cancer. 50
Allograft Theory of DFTD Transmission
Transmissible cancers are an extremely rare occurrence in nature, but evidence for the clonality of DFTD is conclusive, from both karyotypic and genetic perspectives. Pioneering research demonstrated that the karyotypic rearrangement of 11 DFTs was complex and identical. G-banding of DFT chromosomes showed several abnormalities, including the loss of both chromosomes 2, 1 chromosome 6, the long arm of chromosome 1, and both sex chromosomes, with the addition of 4 new unidentified markers. Since it is impossible that each DFT acquired the same complex rearrangement by chance, it was concluded that DFTs are clones derived from the same original tumor, and so the allograft theory of disease transmission was put forward. In addition, the authors identified 1 host with a pericentric inversion of 1 of the chromosomes 5, but its DFT contained no such inversion, demonstrating that its DFT could not have arisen from that host. 56 More recent research based on chromosome painting and gene mapping further characterized the DFT chromosomes and suggested that chromosomes 1 and 2 and chromosomes 5 and 6 were mislabeled in the original G-banding. Accordingly, the karyotype of DFT cells is described as having a deletion of both chromosomes 1, a deletion of 1 pair of chromosome 5, and an addition to the short arm of chromosome 2 (which originated from chromosomes 1 and X). The origin of the 4 marker chromosomes was elucidated as being derived from chromosomes 1, 5, and X.9
The genetic evidence supporting DFT clonal origin has been acquired from microsatellite and major histocompatibility complex (MHC) genotyping as well as whole-genome analysis. Matched tumor and host samples from 15 devils and blood samples from 11 nondiseased individuals were genotyped at 4 polymorphic microsatellite loci and MHC class I and II loci. While 90% of sampled devils had unique genotypes, all the tumors were identical at multiple microsatellite and MHC loci, supporting the tumor’s clonal nature. 70 A larger study of 25 matched tumor and host samples and 10 samples from nondiseased devils were acquired from 16 locations across Tasmania. Fourteen microsatellite loci were genotyped, and all tumors shared a comparable genotype across all loci, independent of location, sex, or age of the devil. 50 Both these studies found that the tumor genotype was distinct from that of the host devils, showing it to be impossible for DFTs to have arisen from the host’s own tissues and consequently supporting the tumor allograft theory.
It is relevant to note here the contribution of telomere length and telomerase activity to the continued proliferation of DFT cells. It has been suggested that the cells monitor and regulate the length of individual telomeres to favor their genomic stability and possibly increased proliferation. 78
Finally, whole genome analysis 44,49 further substantiated the tumor’s clonal origin and allograft theory of transmission by demonstrating that DFTs share structural variants and copy number changes distinct from their hosts.
Evidence for Direct Tumor Cell Transmission
The successful experimental induction of DFTD in naïve devils by the transfer of cultured tumor cells and/or cells from primary DFTs took place several years ago, although this has not been formally described. 61 More recently, 2 captive devils were challenged with live DFTD cells following an immunization trial. Both devils developed palpable tumors, confirmed as DFTs by histopathology, at the site of a subcutaneous injection of 25 000 live DFTD cells. Although not designed to prove Koch postulates, these experiments did so incidentally and thus supported the theory that DFTD cells can be transferred from an infected devil to another. 29 The identification of viable tumor cells on the canine teeth of DFTD-affected devils provided evidence for the natural mechanism of DFTD transmission. 53
The closest relatives of the Tasmanian devil are other members of the dasyurid family, including the spotted tailed quoll (Dasyurus maculatus) and the eastern quoll (Dasyurus viverrinus), both present in Tasmania. There is no evidence of DFTD occurring naturally in these species, 18 although transmission trials have not been performed. DFTD transmission trials were carried out on mice, but only severely immunodeficient mouse strains developed tumors after implantation. 32,59
These independent lines of evidence convincingly characterize DFTD as a transmissible tumor that acts as an allograft and evades the host immune system. There are 3 possible explanations for how a tumor allograft could establish in the devil population: (1) devils have a poor immune response; (2) devils are genetically identical at the MHC; or (3) the tumor cells evolved to evade the host’s immune system.
Each theory has been explored and is summarized below.
The Devil’s Immune Response
The early consensus that marsupials have a primitive immune system has been overturned, with current research showing their immune response to be closely akin to that of eutherian mammals. 3 The overall similarity of the mammalian immune systems with respect to development, components, and complexity is noteworthy given that divergence of marsupials from their eutherian counterparts occurred nearly 150 million years ago. 4
Innate Immune Response of the Tasmanian Devil
Tasmanian devils are carnivores, specialized scavengers, and opportunistic predators. Their diet and biting behavior expose them to a wide range and high level of bacteria and parasites, yet there is little evidence that wild devils succumb to disease of significance from such pathogens. 52 It is thus logical to assume that devils have fully functional innate immune systems, since these provide primary protection against bacterial and parasitic pathogens.
Neutrophils are a key component of the innate immune system. The efficiency of devil neutrophils was established with functional assays that demonstrated phagocytic uptake of Escherichia coli. This, combined with the nitro blue tetrazolium assay for the presence of oxidizing compounds, indicated that this aspect of their innate immune response was proficient. 31
Humoral Immune Response of the Tasmanian Devil
Devils immunized with horse red blood cells had elevated titers of immunoglobulin (IgG) antibody against horse red blood cells 1 week after the first injection, indicating a rapid response. The relatively high titers were maintained throughout the 8-month period of testing, with a subsequent booster 6 months later resulting in rapid and significant secondary responses. These results provided evidence that the devil is capable of a competent humoral immune response, including a memory response. 33 Further research demonstrated the devil’s ability to mount an antibody response against xenogeneic tumor cells. Tasmanian devils were immunized with the K562 human leukemia cell line, and all developed high IgG antibody titers against the K562 cells following their second immunization. 5
Cell-Mediated Immune Response of the Tasmanian Devil
Tasmanian devils resist most bacterial and parasitic insults; however, they and other dasyurids succumb to a high incidence of neoplasias. 6 Common tumors in devils include, but are not limited to, mammary and perianal adenocarcinomas, squamous cell carcinomas, and cutaneous lymphomas. 6,14,66 Cell-mediated immunity plays a crucial role in both tumor and allograft rejection, and in light of the devil’s susceptibility to tumors and the allograft nature of DFTD, a thorough understanding of the devil’s cell-mediated immune response is required. Detailed investigation and clarification of immune cell function, however, are significantly hampered by the lack of species-specific reagents. 19,54 For example, antibodies against CD4 and CD8 have only recently been developed and only for use in immunohistochemistry. 21
Results of T-cell proliferation assays revealed that devils have a relatively robust mitogen-induced response. 31,72 Of particular note was the finding that devils affected with DFTD had similar mitogen-induced responses as their disease-free counterparts, suggesting that immune suppression, at least at the level of lymphocyte proliferation, does not explain susceptibility to DFTD.
The study referred to previously, in which immunized devils showed an antibody response to K562 cells, 5 also demonstrated the presence of natural killer (NK) cells in the peripheral blood and provided evidence of strong cytotoxic responses against the K562 cells in the presence of immune serum. Antibody-dependent cell-mediated cytotoxicity carried out by NK cells may provide an explanation for this, implying a possible route for inducing an anti-DFT response. It is unclear whether NK cells can be activated against DFT cells in the presence of anti-DFT antibodies. While there are gaps in the knowledge, all research to date suggests that devils have a fully functional immune system comparable to that of other mammals, and a limited immune response does not explain the transmission of DFTD.
Genetic Diversity and the MHC
Low genetic diversity of Tasmanian devils is expected, given that they are an island species 12 ; indeed, devils have been shown to exhibit low heterozygosity and allelic diversity at microsatellite loci. 26 However, these results give information on population history (eg, relatedness and previous population bottlenecks), whereas analysis of MHC diversity better represents a population’s “fitness” and ability to counter disease challenges.
The MHC is a cluster of genes occurring in all vertebrates and is the most polymorphic portion of the mammalian genome. These genes were first associated with foreign tissue (allograft) transplantation but are now known to be essential in the immune recognition of pathogens and tumor cells. In addition, MHC molecules provoke vigorous T-cell responses against incompatible cells and regulate the immunologic mechanisms of tissue graft rejection. 13,34
The diversity of MHC genes (ie, polymorphism) provides the foundation for specific immune responses against infectious agents, such as bacteria and viruses. A diversity of MHC molecules allows presentation of different antigen fragments from the infectious agents to a diverse range of T cells. This produces a stronger immune response than that when only a few antigen fragments are presented to T cells. Thus, MHC diversity contributes to disease resistance within a species. However, it was less the contagious nature of DFTD and more so its similarity to an allograft and consequent failure to be rejected by the Tasmanian devil’s immune system that put the spotlight on the devil’s MHC.
Low diversity of MHC class I in Tasmanian devils has been verified by sequence analysis. 70 An exploration of the MHC diversity in historical and ancient devil samples showed very similar levels to those of the modern devil population, suggesting that low MHC diversity has been a feature of the species for the last 10 000 years. 45
This research therefore supported the intuitive explanation that low genetic diversity in devils may be a contributing factor that makes devils more susceptible to allograft acceptance. This was, however, countered by subsequent skin graft transplant experiments. 30 Cheetahs are another example of a wild species recognized as having extremely low genetic diversity, and allogeneic skin grafts between cheetahs were performed to test MHC variation. Of the 14 cheetahs, only 3 showed signs of rejection, which took at least 40 days. Monomorphism at the MHC complex was suggested as the probable cause. 51,64 In contrast, allogeneic skin graft experiments performed among Tasmanian devils 30 found that all 7 recipient devils rejected the grafts within 14 days. The immunologic mechanism of the rejection was confirmed by the characteristic infiltration of CD3-positive lymphocytes. This suggests competent T-cell activity against the allografts and implies that the immune system of individual devils can recognize foreign MHC and should therefore have the potential to mount a response against DFTD.
The research demonstrating a functional immune system and sufficient genetic diversity to reject allografts (other than DFT) suggested that the tumor itself is responsible for escaping the devil’s immune response.
Immune Escape Mechanisms of DFTD
Overview of Tumor Immune Escape Mechanisms
Immune escape mechanisms have been recognized in a wide variety of tumors, including melanoma, mammary carcinoma, and various adenocarcinomas. 28,67 These include downregulation or defective expression of MHC class I, downregulation or defective mechanisms of antigen processing, and secretion of immunosuppressive cytokines. Inhibition of antigen-presenting cells, in particular dendritic cells, is recognized as a significant immune escape mechanism of tumors in people and mice. 75
Downregulation of MHC
Since 2007 DFTD cells have been known to possess MHC class I transcripts. 70 The presumption that MHC was expressed on the DFTD cell surface supported the hypothesis that the devils, with their reduced MHC diversity, did not identify the tumor cells’ foreign MHC, thus allowing survival of the tumor. It has recently been demonstrated that DFTD cells downregulate expression of their MHC genes, and this represents a key mechanism by which the tumor evades immune detection. 71 Of great significance is that expression of MHC class I molecules can be restored on DFTD cells both in vitro and in vivo. The former has been demonstrated by treating cells with the cytokine interferon γ and the latter (albeit rarely) on DFTD biopsies, which have positive staining via immunohistochemistry for β2 microglobulin (a component of MHC class I). This finding has implications for vaccine development and is discussed later in this review.
Immunosuppressive Cytokines
Secretion of immunosuppressive cytokines by tumors of Schwann cell origin has been demonstrated as a mechanism for escaping immune detection. 79 The tumor-promoting role of transforming growth factor β (TGFβ) in the microenvironment of human cancers has been reviewed, 23,27,47 and the significant role of TGFβ in the progression and regression of experimentally induced CTVT has been explored. 22 The immunosuppressive cytokine interleukin 10 (IL-10) has a complex and vast array of functions, but its significance with respect to immune escape lies in its ability to suppress T-cell proliferation and the production of certain inflammatory cytokines, such as interleukin 1 and tissue necrosis factor. 65
Synthesis of cytokines by DFTD cells as a method to evade the devil’s immune response and enhance its growth was suggested by the identification of IL-10 and TGFβ transcripts in the DFTD transcriptome. This was supported by immunohistochemistry on DFTD biopsies that detected IL-10 and TGFβ in the DFTD tumor cells. 21 Quantitative polymerase chain reaction results did not show upregulation of these cytokines nor of vascular endothelial growth factor A or interleukin 6 when compared with normal devil tissue. 46 Because tumor biopsies are composed of a mixture of different cells and because it is unknown whether these transcripts were translated into protein, detecting protein expression would be valuable as a complementary method to quantify these cytokines. 69,80
In summary, evidence demonstrates that devils have an effective immune response and adequate MHC diversity to recognize and reject allografts, suggesting that the tumor cells have evolved mechanisms to escape the immune response. Downregulation of MHC by DFT cells is possibly the most important immune escape mechanism that allows DFTD transmission between devils.
Evolution of DFTD, Stability of the DFTD Cell Line, and Significance of Different Strains
Four strains of DFTD have been identified on the basis of their different karyotypes. 57 These authors suggest that the variants are likely to have different biological characteristics with respect to factors such as transmission and virulence, although there are few corroborating data of this. Based on chromosome painting and gene mapping, it has been suggested that the minimal cytogenetic differences among tumor strains may not have clinical implications. 9 An increase in tetraploidy was observed in DFTs in devils from the Forestier Peninsula where disease suppression by selective culling was being trialled, 77 and since polyploid cells are often larger and may be slower to divide, 55 this could have consequences for tumor growth. Because devils were removed from that site, long-term effects of tetraploidy in that population could not be assessed. The influence of ploidy was observed in a longitudinal study in northwest Tasmania where a high initial prevalence of tetraploid tumors in the study site was associated with low DFTD infection rates and limited host population effects. When the diploid DFTD variant reached the site, it replaced the tetraploid variant, causing disease prevalence and population effects to rapidly increase. 17
Canine Transmissible Venereal Tumor
Given that DFTD and CTVT are the only naturally occurring transmissible tumors known to exist in vertebrates, it is relevant to compare the 2 diseases. The following is a brief summary of 3 reviews comparing CTVT and DFTD. 2 ,48,68
The malignant and fatal nature of DFTD is the most pronounced difference between it and CTVT. Typical cases of CTVT rarely metastasize or cause fatality in immunocompetent hosts. Interestingly, the tumors share the same immune escape mechanism of MHC class I downregulation as they establish themselves in the host. This “progressive phase” of CTVT whereby only 3% of tumor cells express MHC class I is followed by the stationary or regressive phase, characterized by cessation of tumor growth. This phase is associated with increased expression of cell surface MHC class I, lymphocyte infiltration, and increased levels of the host-derived interferon γ. 22 The fact that MHC expression can be restored on both CTVT and DFTD cells (ie, lack of expression is due to regulatory mechanisms rather than structural defects) has obvious implications for tumor recognition by the host immune system. Restoration of MHC class I expression occurs during the natural course of CTVT infection, suggesting that this tumor has reached an equilibrium with its host, allowing the survival of both.
Serum IgG antibody against CTVT cells has been demonstrated in CTVT-affected dogs, suggesting a humoral immune response to the disease. 7,8 A humoral immune response in the form of IgG antibodies against DFTD cells was also evident in devils undergoing a DFTD immunization trial. 29 While it is cell-mediated immunity that is primarily responsible for antitumor activity, IgG antibody production against DFTD is a significant finding in a disease characterized by powerful immune escape mechanisms.
Epidemiology and Compensatory Responses to Population Decline
DFTD was first observed in 1996 at Mount William National Park in the state’s far northeast and has since spread west and south. Figure 7 shows DFTD distribution from 1996 to 2015.
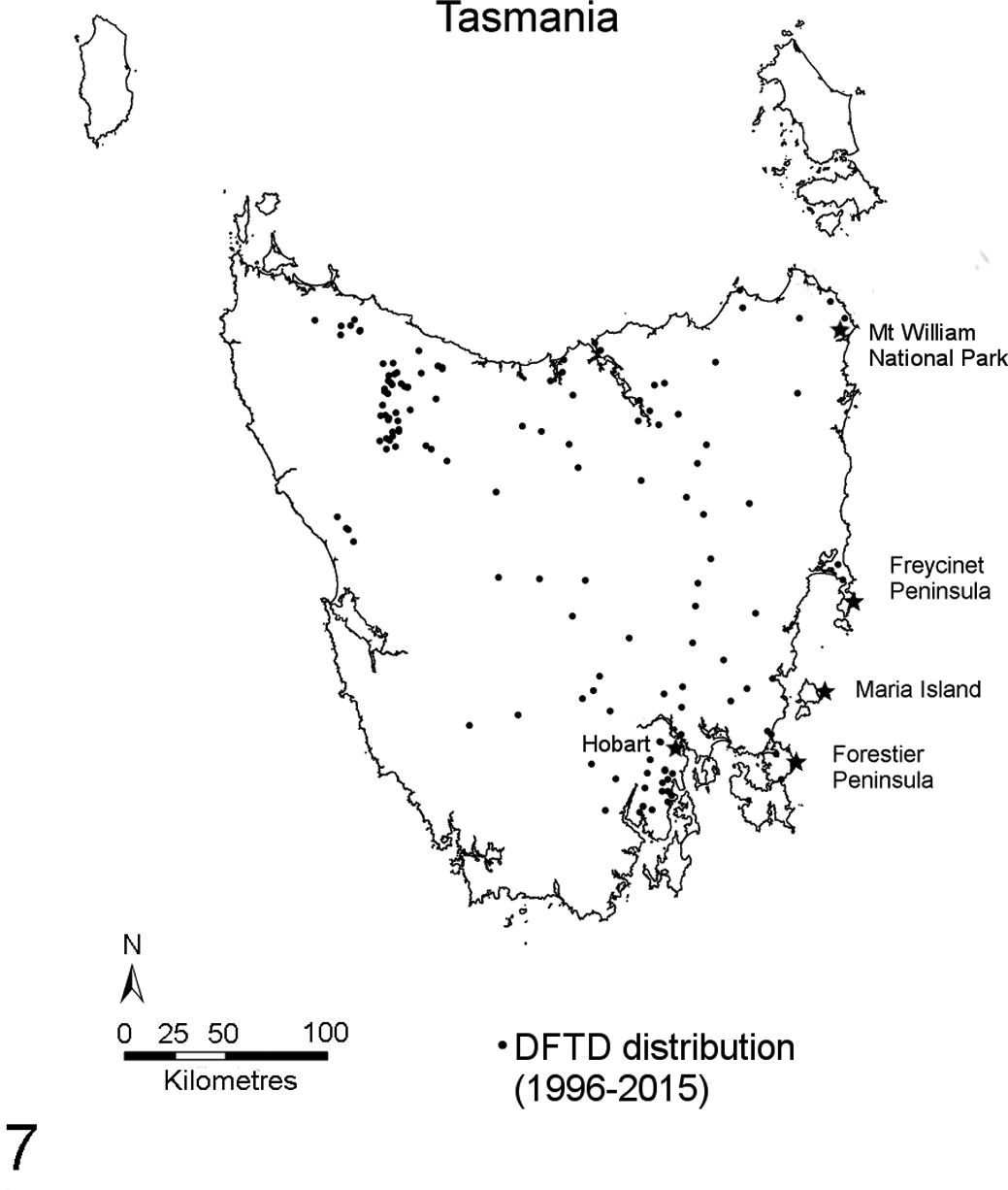
Map of Tasmania showing the distribution of devil facial tumor disease (DFTD) from 1996 to 2015. Each dot represents 1 case of DFTD confirmed by histology; stars represent locations mentioned in the text.
Most contagious diseases are dependent on the density of their host populations, so when a population falls below a certain level, the causal pathogen dies out. However, DFTD prevalence is maintained in devil populations that have suffered significant (up to 90%) declines, which strongly suggests that DFTD follows a frequency-dependent pattern of transmission. 40 This transmission pattern is typical of sexually transmitted diseases, and field studies indicate that bite wounds peak during the mating season, giving DFTD epidemiologic characteristics of a sexually transmitted disease. 16
There is a high DFTD prevalence (at least 50%) among adult devils in populations where disease is well established, 40 and it is rare to find devils >3 years of age in these areas despite a natural life expectancy of up to 6 years in the wild. There is no consistent evidence to show a difference between the sexes in DFTD prevalence. 40
A rise in precocial breeding by female devils in diseased populations, explained by increased food availability and growth rates resulting in earlier sexual maturity, demonstrates reproductive compensation for population decline. 35 These authors also noted that diseased females showed a propensity for producing female-biased litters when compared with their healthy sisters, suggesting that sex allocation bias is a result of DFTD infection.
Ecologic Impacts
The devil is Tasmania’s top-order land predator and as such is a highly interactive keystone species. Consequently, its decline is expected to have significant deleterious impacts on Tasmania’s ecosystem. The decline is likely to advantage feral predators, with reported changes in feral cat behavior and possibly increased abundance. 11,20 Devils may have prevented fox incursions in Tasmania from establishing in the past and thus protected the state’s native fauna from the devastating effects that foxes have had on the Australian mainland. 18 Devils are specialized scavengers, and the expected increase in carrion in the environment due to declining devil numbers could favor alternative scavengers (eg, forest ravens), further disrupting the ecosystem balance. 42
Management Options Available to Save the Species
A range of management options has been considered to save the Tasmanian devil from extinction, and these are summarized in Table 1, along with their advantages and disadvantages. Some options, such as captive breeding and translocation, have already been effectively implemented, whereas others have either proved unsuccessful (eg, disease suppression) or are a work in progress (eg, vaccine development).
Proposed Management Tools to Save the Tasmanian Devil from Extinction.

Abbreviation: DFTD, devil facial tumor disease.
This review alludes to DFTD evolution, and certainly the development of a less aggressive disease would favor survival of the host and, by consequence, the disease. It is possible that a more benign form of DFTD might evolve given enough time and devil hosts, and it is conceivable that implementation of certain management options would interfere with this evolutionary process. However, given the rapid and devastating effects of DFTD to date with no apparent emergence of reduced tumor aggression or host resistance, it would be irresponsible to neglect intervention and let the disease take its course knowing the implications of devil extinction.
Final Remarks
DFTD is a unique cancer that has developed strategies to avoid the host’s immune response and has capitalized on the biting behavior of devils to allow transmission between individuals. This unfortunate situation has provided unique opportunities to study transmissible cancers, including mechanisms of cancer cell transfer and immune escape. It has also highlighted the possibility that transmissible cancers in wild animals could be more common than originally considered. Protection of Tasmanian devils from DFTD has, and will continue, to require a coordinated approach from various governmental and nongovernmental bodies. The successful establishment of an insurance population of healthy captive devils and research toward a vaccine will facilitate the reintroduction of these animals into the wild. This will provide a new era in wildlife management and research.
Footnotes
Acknowledgements
We thank Samantha Fox and Jim Richley for providing data for Figure 7 and David Pemberton and Samantha Fox for their insightful comments on ![]() . We are grateful for the support received from the Save the Tasmanian Devil Program and funding received from the Save the Tasmanian Devil Appeal.
. We are grateful for the support received from the Save the Tasmanian Devil Program and funding received from the Save the Tasmanian Devil Appeal.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
