Abstract
The myosin heavy chain (MHC) composition of a given muscle determines the contractile properties and, therefore, the fiber type distribution of the muscle. MHC isoform expression in the laryngeal muscle is modulated by neural input and function, and it represents the cellular level changes that occur with denervation and reinnervation of skeletal muscle. The objective of this study was to evaluate the pattern of MHC isoform expression in laryngeal muscle harvested from normal cadavers and cadavers with naturally occurring left laryngeal hemiplegia secondary to recurrent laryngeal neuropathy. Left and right thyroarytenoideus (TA) and cricoarytenoideus dorsalis (CAD) were obtained from 7 horses affected with left-sided intrinsic laryngeal muscle atrophy and from 2 normal horses. Frozen sections were evaluated histologically for degree of atrophy and fiber type composition. MHC isoform expression was determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) of muscle protein. Histologic atrophy was seen in all atrophic muscles and some right-sided muscles of 3 affected horses, as well as the left TA of 1 normal horse. Fiber type grouping or loss of type I muscle fibers was observed in the left-sided laryngeal muscles in all but 1 affected horse, as well as in the right muscles of 2 affected horses, and the left TA of 1 normal horse. SDS-PAGE showed 2 bands corresponding to the type I and type IIB myosin isoforms in the CAD and TA of the 2 normal horses. Affected horses demonstrated a trend toward increased expression of the type IIB isoform and decreased expression of the type I isoform in atrophic muscles. This study confirmed the presence of histologic abnormalities in grossly normal equine laryngeal muscle, and it demonstrated an increased expression of type IIB MHC with a concurrent decreased expression of type I MHC in affected muscles. Evaluation of muscle fiber changes at the cellular level under denervated and reinnervated conditions may aid in assessing future strategies for reinnervation or regeneration of atrophic laryngeal muscle.
Keywords
Introduction
Recurrent laryngeal neuropathy (RLN) is a common problem in the equine athlete, 35 causing clinical signs due to incomplete arytenoid abduction in up to 15% of horses examined endoscopically. 1, 10, 13, 26, 31 RLN is more commonly known as idiopathic laryngeal hemiplegia. Evidence of subclinical disease, including axonopathy of the recurrent laryngeal nerve and neurogenic atrophy of the intrinsic laryngeal musculature, is present in nearly twice as many horses. 2, 5, 6, 9 The clinical effects of RLN and the histologic changes in the affected laryngeal muscles have been well characterized; 15– 17, 25, 33 however, little is known about the cellular and molecular level events that occur in the laryngeal muscle of horses affected by RLN.
Myosin is the most abundant protein in skeletal muscle, and the myosin heavy chain (MHC) molecule contains the adenosine triphosphatase (ATPase) activity that transduces chemical energy into mechanical force generation. 24 The MHC isoform composition of a given muscle fiber is modulated by developmental, hormonal, mechanical, and neural factors. 4, 37, 39, 42, 43 Each MHC isoform has distinct ATPase and shortening velocity properties and represents the molecular basis for muscle fiber diversity. Three MHC isoforms with fast contractile properties (types IIa, IIb, and IIx) and 1 isoform with slow contractile properties (type I) have been identified. In addition, in the laryngeal muscle of rats and rabbits a unique MHC isoform (type IIl) has been identified, which may represent a molecular level adaptation to distinctive physiologic demands, such as the need for a high contractile velocity. 11, 34, 40 Although this unique MHC isoform is absent from human laryngeal muscle, 44 it is not known whether this isoform is expressed in equine laryngeal muscle.
The effects of denervation on the MHC composition of skeletal muscle in laboratory animal species have been well studied. In rat limb and diaphragm muscle, there is a general trend toward an increase in expression of type II fibers and a decrease in type I fibers. 22, 27, 30 Similarly, rat laryngeal muscle also demonstrates decreased expression of type I MHC following denervation, 12 as well as an increase in expression of types IIa and IIx and a decrease in type IIb. 38, 41 Shiotani et al. 43 found that when the thyroarytenoideus muscle of rats was denervated by a crush injury to the recurrent laryngeal nerve, the muscle had an MHC expression pattern consistent with denervation, but when vocal fold movement returned 30 days later, the pattern of MHC expression in the thyroarytenoideus (TA) muscle was similar to that in control rats with intact vocal fold function. Therefore, restoration of function appears to correlate with restoration of a normal MHC phenotype. Addition of neurotrophic factors via gene transfer also appears to restore normal MHC phenotype following denervation in the rat larynx. 21
Evaluation of muscle fiber changes at the level of MHC expression under conditions of atrophy, as seen with RLN, may aid in assessing future strategies for reinnervation of equine laryngeal muscle, particularly since normal MHC isoform expression may indicate appropriate and functional reinnervation. In addition, evaluation of these changes may aid in understanding the pathogenesis of RLN. The objective of our study was to evaluate the pattern of MHC isoform expression in equine laryngeal muscle harvested from normal larynges and from those with gross evidence of RLN. Since RLN affects both adductors and abductors of the larynx, we chose to evaluate the TA and the cricoarytenoideus dorsalis (CAD) muscles, both of which are innervated by the recurrent laryngeal nerve.
Materials and Methods
Horses
Larynges were harvested within 24 hours of euthanasia from 9 horses euthanatized for reasons unrelated to the upper respiratory tract. Horses ranged in age from 3 to 20 years old (mean, 10.9 ± 4.6 years) and included 7 geldings and 2 intact males. Breeds included 7 Thoroughbreds, 1 Quarter Horse, and 1 Warmblood. Seven horses had gross evidence of atrophy of the left intrinsic laryngeal musculature (designated “affected”), and 2 horses had grossly normal larynges (designated “normal”). Four muscles were collected from each horse: the left and right TA muscles (LTA, RTA), and the left and right CAD muscles (LCAD, RCAD).
Tissue preparation
After each muscle was dissected free of its cartilaginous attachments, a rectangular section of muscle measuring 0.5 cm × 0.5 cm × 2.0 cm was cut such that the long axis of the rectangle was parallel with the muscle fibers. The section was then stretched, frozen in liquid nitrogen, and stored at -80°C. One gram of frozen muscle tissue was stored in a microcentrifuge tube at -80°C for protein extraction. The remainder of the frozen muscle sample was mounted on a stainless steel specimen disc using optimal cutting temperature (OCT) media and sectioned at 20 μm using a cryostat (Microm HM 500 M, Waldorf, Germany). The muscle sample was mounted such that cross-sectional slices of the muscle fibers were obtained. Frozen sections were then mounted on glass slides and stored at -20°C until staining.
Histochemistry
One half of the slides of frozen sections were stained for routine histopathologic analysis using hematoxylin and eosin (HE), 20 and the other half were stained for myofibrillar ATPase activity after preincubation at pH 9.4. 3 All slides were then dried, cover slipped, and stored at room temperature. For both staining methods, muscle sections were evaluated using light microscopy.
HE-stained sections were used to describe the degree of histologically apparent muscle atrophy present for each muscle sample. The following 3-point grading scale was used:
Normal histologic appearance.
Mild atrophy, characterized by muscle fiber diameter disparity.
Marked atrophy, characterized by muscle fiber atrophy in multiple sites.
All HE-stained muscle sections were reviewed and graded by a board-certified veterinary pathologist (PLH) who was blinded as to the gross appearance of each muscle.
Sections stained for myofibrillar ATPase were evaluated for the percentage of type I and type II fibers in each section. Type I fibers have a low ATPase activity at pH 9.4, and, therefore, these fibers stained lightly, whereas type II fibers have high ATPase activity at pH 9.4 and stained dark. ATPase-stained sections also were evaluated for patterns of distribution of the type I and type II fibers. Normal distribution of fiber types in equine laryngeal muscle is a mosaic pattern, 15, 33 whereas denervation atrophy can result in fiber type grouping or a marked loss of a given fiber type. 5 Therefore, sections were characterized as having either a normal mosaic pattern or an abnormal pattern. Those with an abnormal pattern were further subdivided into those having either fiber type grouping or a marked loss of a given fiber type. ATPase-stained sections were reviewed by 2 of the authors (CMA and ML) in a blinded fashion.
Protein extraction and SDS-PAGE
Tissue from the 36 muscle samples (bilateral CAD and bilateral TA for all 9 horses), as well as 2 control protein samples with known MHC isoform compositions (rat TA muscle and mouse diaphragm) were processed separately. Muscle tissue was homogenized in ice-cold wash buffer (40 mM NaCl, 1.4 mM NaH2PO4, 3.6 mM Na2HPO4; pH 7.2) using a sonicator. Samples were sonicated while being maintained in an ice bath to prevent excessive heating of the muscle tissue. The homogenate was centrifuged at 6000 × g for 15 minutes at 4°C. The pellet was suspended in extraction buffer (100 mM Na4P2O7, 5 mM EGTA, 5 mM DTT; pH 8.6) for 1 hour at 4°C. The sample was then centrifuged at 20,000 rpm for 20 minutes at 4°C. Supernatant was mixed with an equal volume of cold 100% glycerol and stored at -20°C. The protein concentration was determined using a colorimetric assay (Bio-Rad, Hercules, CA), and standardized to 0.4 mg/ml prior to gel electrophoresis.
Protein was heated for 5 minutes at 100°C in 2× sample buffer (100 mM Tris, 25% glycerol, 2% sodium dodecyl sulfate [SDS], 0.01% bromphenol blue), separated on an 8% acrylamide gel with 0.8 μg of protein in each lane, and run overnight at 70 V and 4°C. These electrophoresis parameters have previously been shown to provide optimal separation and imaging of the bands representing myosin isoforms. 8, 32
Western blot
An acrylamide gel containing separated protein from the laryngeal muscles of a normal horse was transferred onto a nitrocellulose membrane at 100 mA overnight at 4°C. The membrane was blocked and incubated with a primary monoclonal antibody against all MHC isoforms (mouse anti-human MHC; Developmental Studies Hybridoma Bank, University of Iowa, Iowa City, Iowa) for 1 hour (1°1000). The membrane then was incubated with a peroxidase-labeled secondary antibody (1°50,000). MHC bands were identified using the enhanced chemiluminescence immunodetection method. Western blot was only performed on laryngeal muscle from 1 normal horse in order to identify the bands that represented MHC isoforms. For all other horses, the bands were identified based on their migration patterns on the silver-stained acrylamide gel. The bands were easily identified in this fashion, as MHC is a very large and abundant protein which migrates through acrylamide gel much more slowly than any other muscle protein. 4
MHC isoform imaging
Acrylamide gels were fixed in 50% methanol/10% acetic acid to prepare them for silver staining to image the protein bands. Silver staining was performed according to the method published by Oakley et al. 36 Silver-stained gels then were rinsed with distilled water, and the images were digitized using a digital camera (Nikon D100, Tokyo, Japan). Densitometry of the bands representing the myosin isoforms then was performed using Image J Java image processing software (National Institutes of Health, Bethesda, Maryland). The density of the individual MHC bands was expressed as a percentage of the total measured density of MHC bands for each muscle.
Statistical analysis
Data were analyzed using a mixed-model analysis of variance (PROC MIXED, Statistical Analysis Systems, SAS Institute Inc., Cary, NC). The class (independent) variables were: muscle (LCAD, RCAD, LTA, RTA), category (normal vs. affected), and the interaction between muscle and category. Horse number (1 through 9) was the random variable. The dependent variables were the percentage of type II muscle fibers and the relative density of the type II MHC bands. A Tukey-Kramer adjustment was used for comparison of the different means. The level of significance was P < 0.05.
Results
Histopathology
Of the affected horses, 1 horse was graded histologically as having mild atrophy of both the LTA and LCAD; the remaining 6 horses had marked histologic atrophy of both of these muscles. In addition, mild histopathologic changes were observed in the RCAD of 2 affected horses, the RTA of 3 affected horses, and the LTA of 1 normal horse. Figures 1, 2, and 3 depict examples of HE-stained sections from horses with normal muscle, mild atrophy, and marked atrophy, respectively. Results from all 9 horses studied are summarized in Table 1.
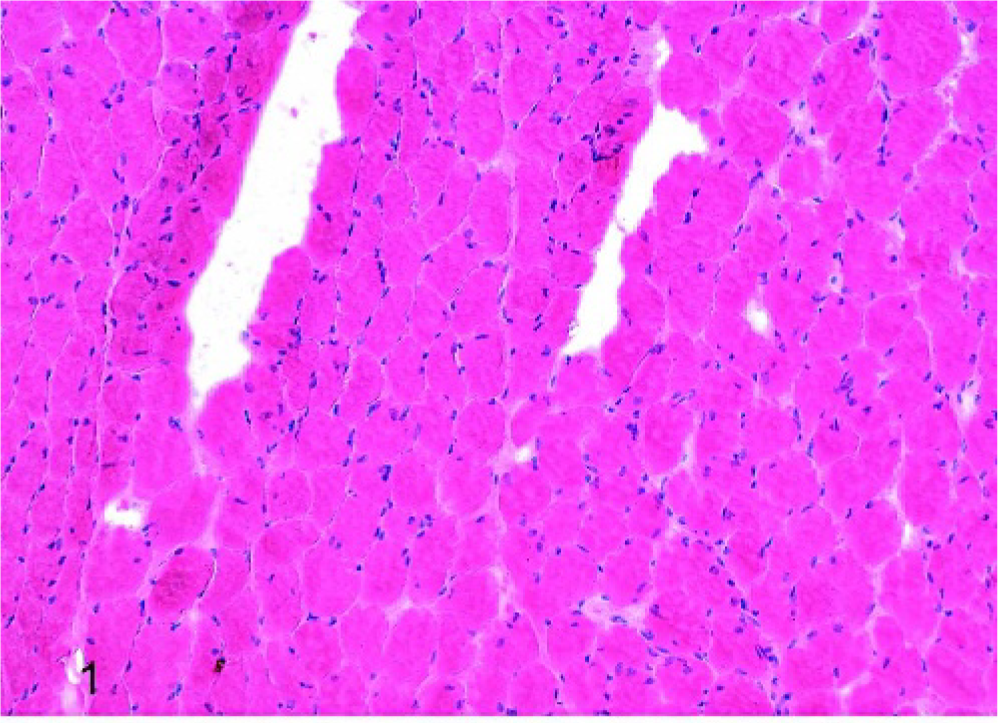
Laryngeal muscle, left CAD, horse No. 9 (normal); normal histologic appearance, HE stain. Scale bar = 50 μm.
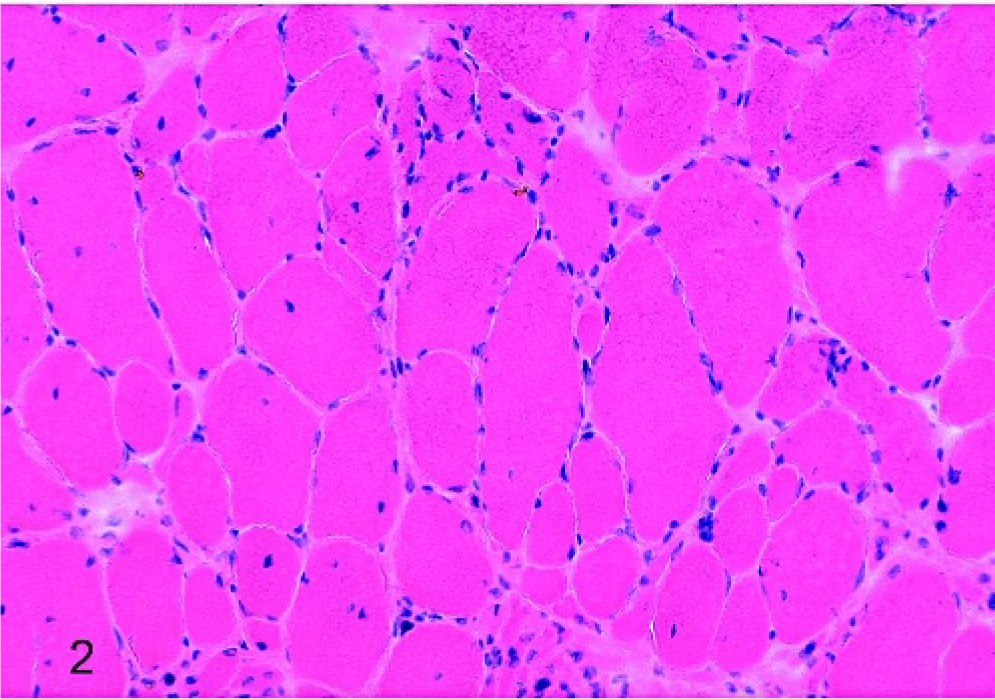
Laryngeal muscle, left CAD, horse No. 4 (affected); mild histologic atrophy, characterized by classic small group atrophy, HE stain. Scale bar = 50 μm.
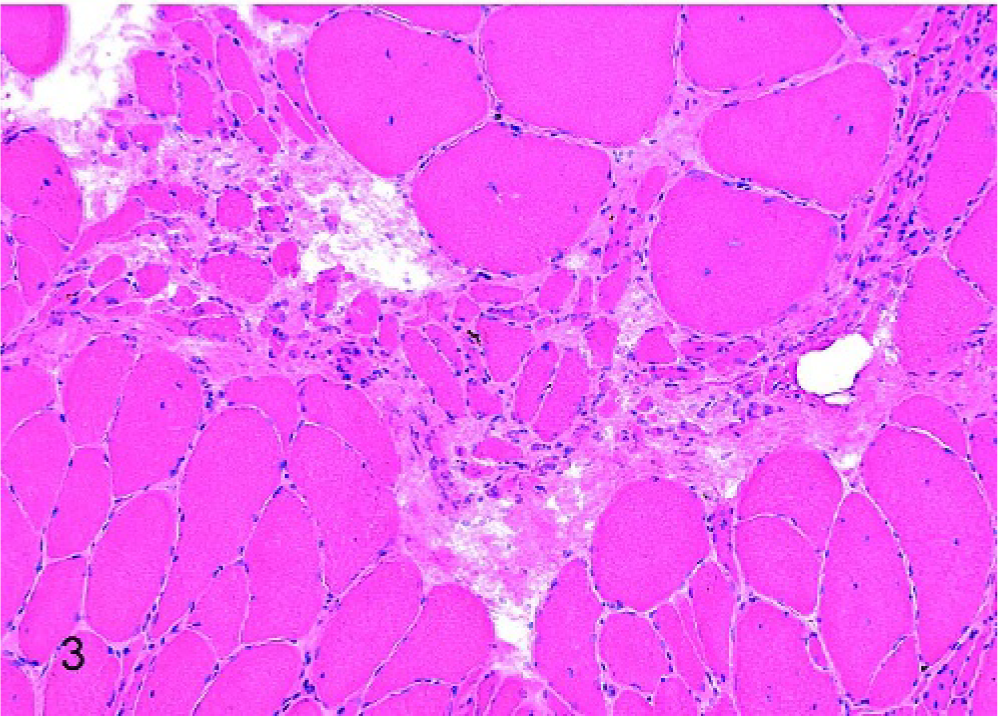
Laryngeal muscle, left CAD, horse No. 1 (affected); marked histologic atrophy, characterized by classic large group atrophy and internal nuclei in fibers of normal to increased diameter, HE stain. Scale bar = 50 μm.
Summary of histologic results and MHC expression pattern for laryngeal muscle from all 9 horses studied.

ATPase staining: percentage type I vs. type II muscle fibers
The results are expressed as percentage type II fibers relative to the total number of both type I and type II fibers. The percentages of type II fibers in the 2 normal horses in the LCAD, RCAD, LTA, and RTA, respectively, were: 60.5 ± 2.1, 62.0 ± 1.4, 67.5 ± 2.1, and 59.0 ± 1.4. The percentages of type II fibers in the 7 affected horses in the LCAD, RCAD, LTA, and RTA, respectively, were: 86.9 ± 9.3, 63 ± 5.4, 89.3 ± 12.9, and 65.3 ± 6.2. There was a statistically significant increase in the percentage of type II muscle fibers in the LCAD and LTA muscles of affected horses when compared to all other muscles.
ATPase staining: fiber pattern distribution
Abnormal fiber distribution, including fiber type grouping or marked loss of type I muscle fibers, was present in the left-sided laryngeal muscles in all but 1 affected horse, as well as in the RTA of 2 affected horses. One of the 2 affected horses with abnormal fiber distribution in the RTA also had abnormal fiber distribution in the RCAD. Abnormal fiber distribution was present in the LTA of the normal horse, which had histologic evidence of mild atrophy on HE-stained sections. The further characterization of abnormal fiber patterns into fiber type grouping or loss of type I fibers is shown in Table 1. Figures 4, 5, and 6 show the normal mosaic patterns of type I and type II muscle fibers seen in a grossly normal laryngeal muscle, fiber type grouping in the left CAD muscle of an affected horse, and marked loss of type I fibers in the left CAD of another affected horse, respectively.
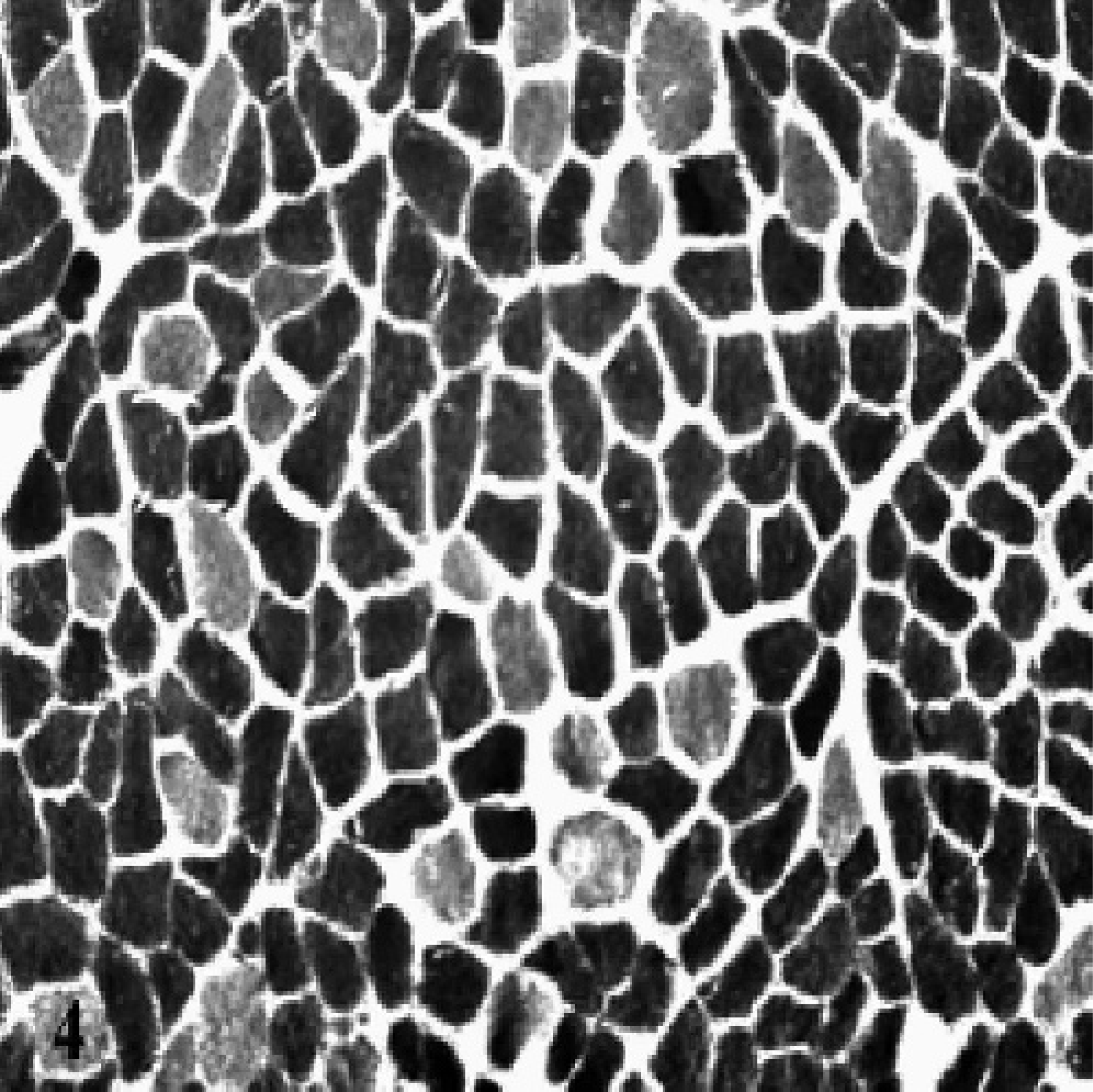
Grossly normal laryngeal muscle, RCAD, horse No. 2. A normal mosaic fiber pattern distribution of type I and type II muscle fibers is demonstrated. ATPase stain at pH 9.4. Scale bar = 50 μm.
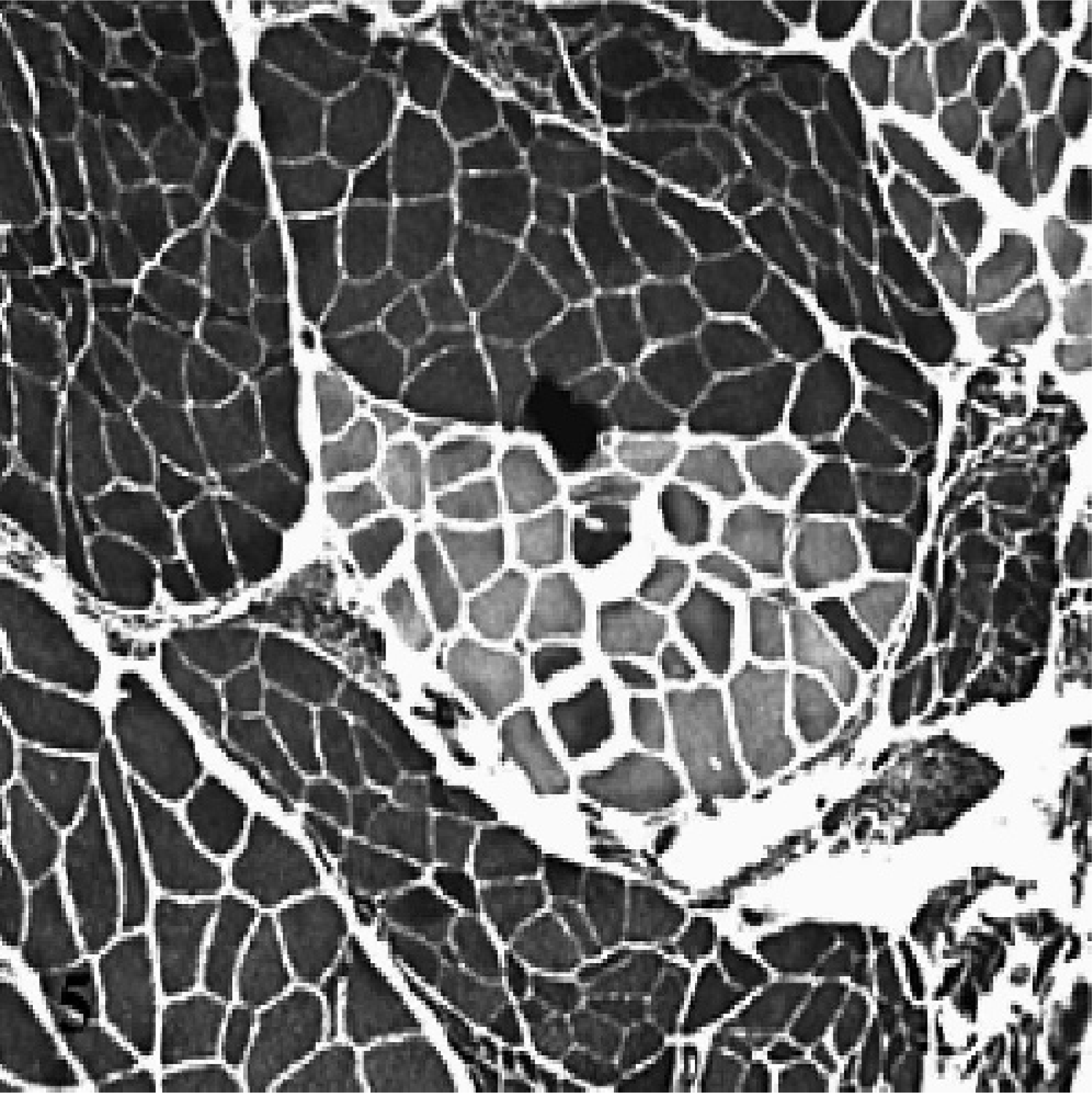
Atrophic laryngeal muscle, LCAD, horse No. 4. An example of fiber type grouping of both type I and type II fibers is depicted. There is also evidence of small and large group atrophy in adjacent fascicles, consistent with ongoing denervation or failure to reinnervate. ATPase stain at pH 9.4. Scale bar = 100 μm.
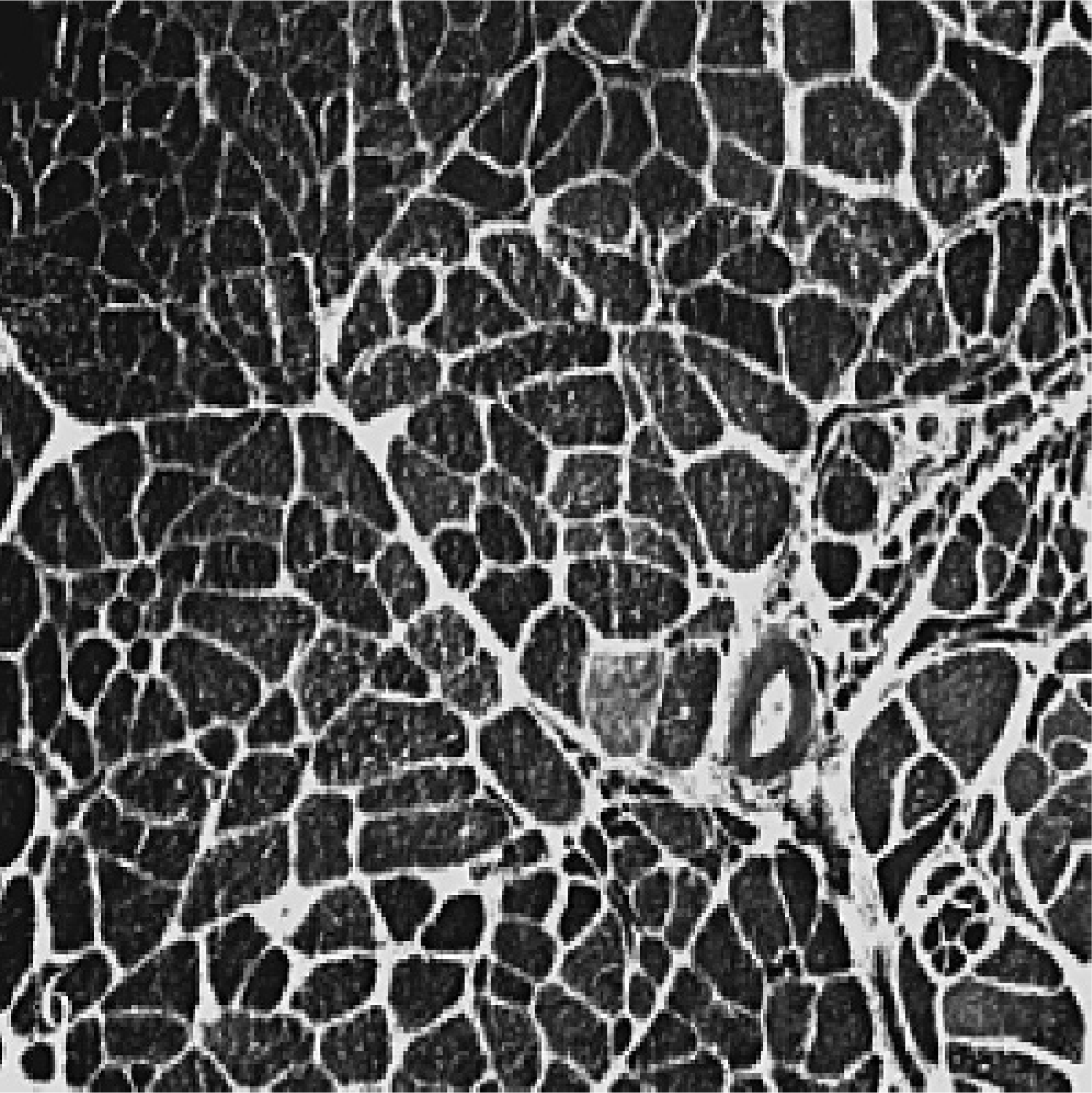
Atrophic laryngeal muscle, LCAD, horse No. 3. A marked loss of type I fibers is shown. In this field of view only a single type I fiber is seen in cross-section (stained lighter) among numerous type II fibers. Evidence of small and large group atrophy also is present. ATPase stain at pH 9.4. Scale bar = 75 μm.
MHC isoform expression: normal horses
Sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) of the laryngeal muscles in the 2 normal horses showed 2 bands that reacted with a nonspecific anti-MHC antibody on Western blot (Fig. 7, top panel). A silver-stained gel comparing the migration pattern of the MHC bands in the equine CAD to those in the rat TA muscle and mouse diaphragm is shown in the bottom panel of Fig. 7. From this migration pattern it appears that the 2 MHC isoforms expressed in equine laryngeal muscle are type I and type IIb. The unique laryngeal muscle MHC isoform found in the rat TA (type IIl) does not appear to be present in the laryngeal muscle of horses.
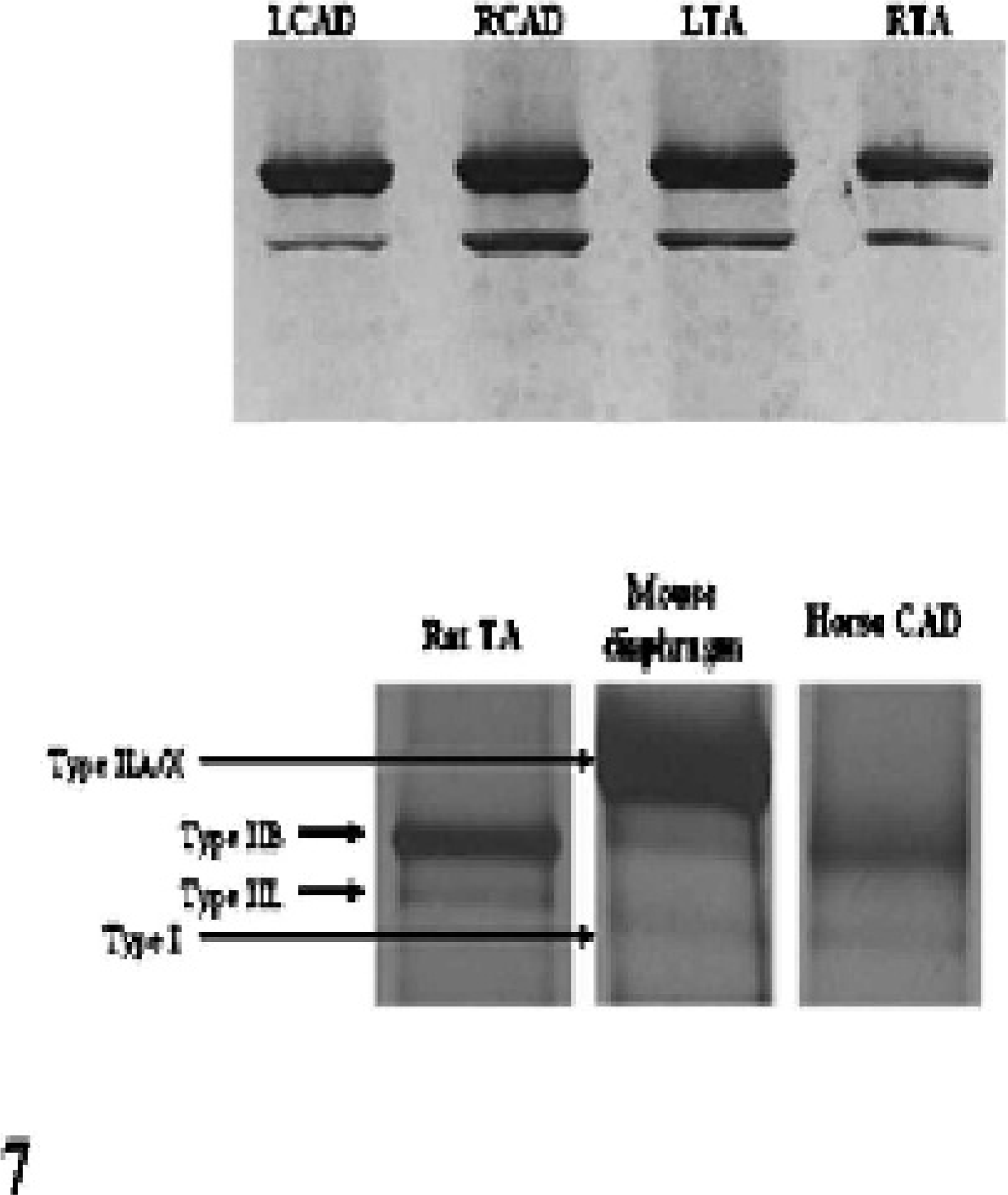
Top. Western blot of protein isolated from normal laryngeal muscles using a nonspecific mouse anti-human MHC antibody. Bottom. Silver-stained polyacrylamide gel demonstrating the migration pattern of myosin isoforms in a normal equine laryngeal muscle vs. 2 controls (rat thyroarytenoideus and mouse diaphragm).
MHC isoform expression: affected horses
SDS-PAGE of the laryngeal muscle of 2 of the affected horses showed an MHC expression pattern similar to that in the normal horses. Three affected horses had a shift in isoform expression in the left TA muscle only, such that a single isoform is expressed (Fig. 8, top panel). One affected horse had a shift toward a single band in both the left TA and the left CAD (Fig. 8, middle panel), and one affected horse had a shift toward a single band in all muscles except the right CAD (Fig. 8, bottom panel). One affected horse with a normal MHC expression pattern had only mild histologic evidence of atrophy, while the 5 horses with altered MHC expression patterns had marked histologic atrophy in the left-sided laryngeal muscles studied.
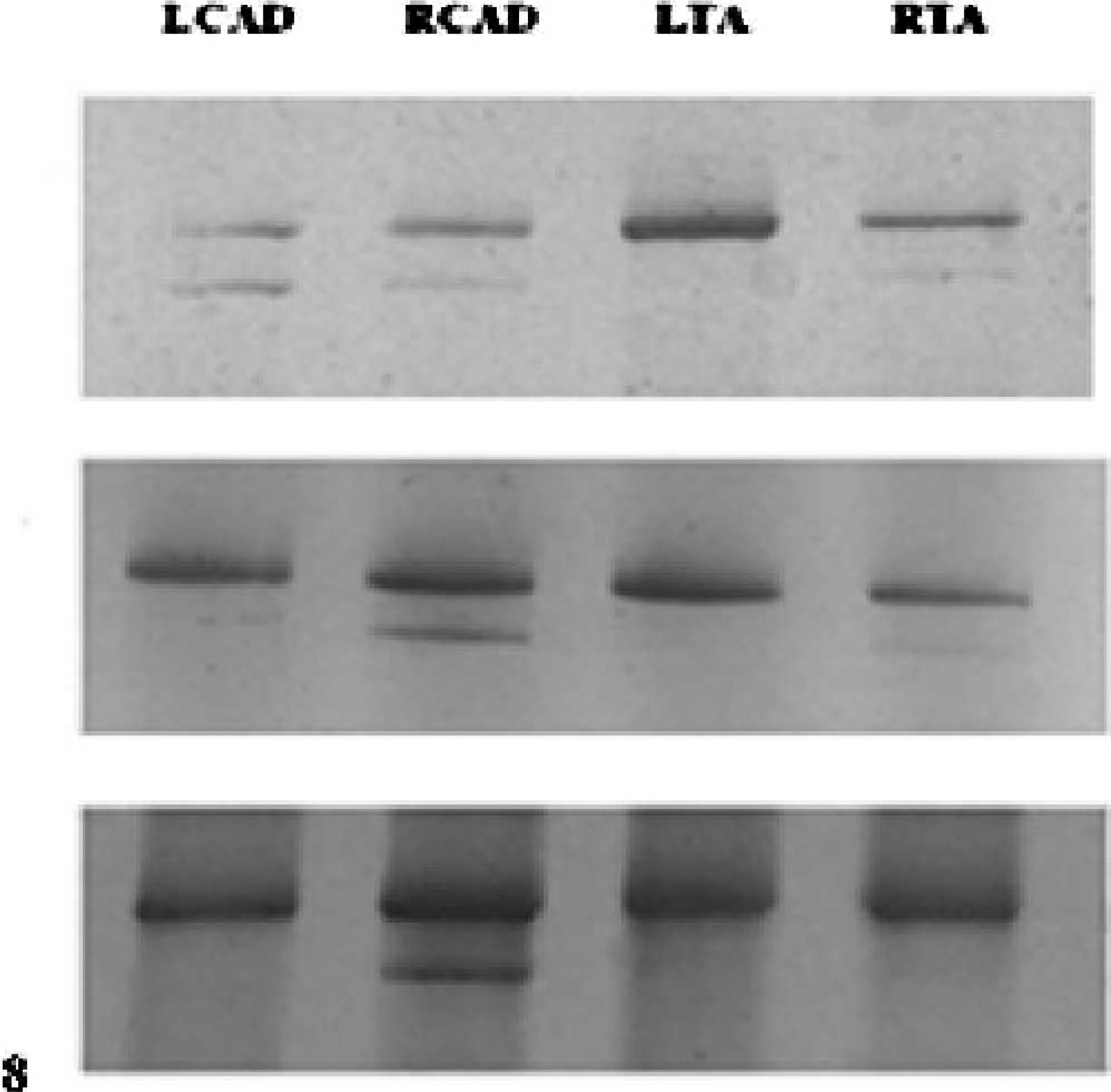
Top. Silver-stained polyacrylamide gel showing MHC isoform expression in an affected horse (No. 1), which had a shift toward a single MHC band in the LTA muscle. Middle. Silver-stained gel showing MHC isoform expression in an affected horse (No. 6) with a shift in MHC expression to a single band in the LCAD and the LTA. Bottom. Silver-stained gel showing MHC isoform expression in an affected horse (No. 7) with a shift in MHC expression to a single band in all muscles except the right CAD (bottom panel).
When densitometry was used to quantify MHC isoform expression, the contributions of the presumed MHC type IIb band to the total MHC density (i.e., percent total MHC density) for the 2 normal horses in the LCAD, RCAD, LTA, and RTA, respectively, were: 72.5 ± 4.9, 79.0 ± 2.8, 77.5 ± 0.7, and 82.5 ± 2.1. The percentages of total MHC density for the 7 affected horses in the LCAD, RCAD, LTA, and RTA, respectively, were: 78.0 ± 17.1, 77.0 ± 9.8, 93.7 ± 11.1, and 84 ± 14.2. Affected horses demonstrated a trend toward increased relative density of the type IIb band in the left TA muscle (i.e., increased expression of the type IIb MHC and decreased expression of type I MHC); however, this increase in density was not statistically significant.
Discussion
Recurrent laryngeal neuropathy is a common clinical problem in athletic horses, causing paralysis or paresis of the intrinsic laryngeal muscles innervated by the recurrent laryngeal nerve. Paralysis or paresis of these muscles produces airway obstruction during exercise, which results in respiratory noise and decreased exercise tolerance. Although the pathogenesis of RLN is not completely clear, most evidence points to a distal axonopathy of the recurrent laryngeal nerve and subsequent neurogenic atrophy of the muscles supplied by this nerve. 5, 6, 15– 17 Studies on the histologic changes that occur in laryngeal muscles of affected horses have aided in understanding the progression and clinical presentation of the disease, but the molecular level events that occur in RLN have not been studied. An understanding of the changes in protein expression in affected muscles may help clarify the pathogenesis of this disease and develop future strategies for treatment. The objective of the present study was to describe the changes in MHC expression in laryngeal muscle that occur in horses with evidence of RLN on postmortem evaluation. We found that atrophic equine laryngeal muscle demonstrated an increase in the percentage of type II muscle fibers in ATPase-stained frozen sections, which may be due to a shift toward type IIb and away from type I in MHC isoform expression.
The shift toward expression of a single MHC band was most apparent in the left TA muscle of affected horses, but it also was observed in the left CAD and the right TA of affected horses. This is consistent with previous studies that have shown that laryngeal adductors are affected more severely and earlier in the course of the disease of RLN. 5, 33 When the MHC band densities of all horses were grouped together, the increased relative density of the presumed type IIb MHC band in the left TA muscle was not statistically significant. This may be due to the variation in the course of the disease that would likely be found in a population of affected horses selected at postmortem, since RLN has been shown to be a progressive disease. 10, 13, 23 Studies have shown histologic evidence of ongoing demyelination and remyelination of the recurrent laryngeal nerve as a result of a continual injury and attempts at nerve repair and, therefore, likely ongoing axonal injury as well. 6, 16 A population of affected horses selected at one given timepoint would have varying degrees of muscle atrophy as an endstage event with ongoing nerve injury. Future studies to confirm the altered MHC composition in affected horses may be performed on horses with experimentally induced RLN, which would eliminate the variation in the course of the disease at the time of sample collection.
While the shift in MHC isoform expression toward the presumed type IIb isoform was not significant in affected horses, there was a significant increase in the percentage of type II fibers on ATPase-stained frozen sections. This apparent discrepancy may be due to the presence of hybrid muscle fibers expressing both type I and type II MHC within the same fiber. The development of hybrid fibers expressing more than one MHC isoform has been shown to be a phase in the transition of the myocyte from one fiber type to another. 37, 45, 46 As hybrid fibers are not detected on ATPase-stained sections, it is possible that fibers in the process of transition to a type II phenotype are detected as darkly stained type II fibers on frozen sections.
The altered MHC expression in the right TA of 1 of the affected horses, as well as histologic evidence of atrophy in the right TA of 2 affected horses, confirms previous reports that RLN causes disease, at least at the microscopic level, in the right-sided laryngeal muscles of affected horses, even if the disease is clinically more common on the left side. 5, 15, 16, 23 In addition, the right TA with altered MHC expression was grossly normal, suggesting that protein level changes may occur before grossly apparent atrophy, and perhaps clinical signs, occur. Likewise, histologic evidence of muscle atrophy was observed in the left TA muscle of a normal horse with no grossly apparent atrophy of the left-sided laryngeal muscle. It is unclear why this muscle had a normal MHC expression pattern despite the histologic changes, but this may also represent an effect of the variation in the time course of the disease seen in our sample population.
Altered expression of muscle fiber types and MHC content following denervation has been well studied in rat limb and diaphragm muscle. 22, 28, 30, 45 A shift away from a slow phenotype with denervation appears to occur at the transcriptional level in rodent limb muscle. 27, 28 An apparent mechanism may be a decrease in the in vivo activity of the MHC type I gene promoter under denervated conditions. 29 Another possible mechanism may be an increased gene expression of the cell cycle inhibitors p21 and GADD45 under denervated conditions in rat laryngeal muscle. 7 Although the current study only evaluated changes in MHC expression at the protein level, future investigations examining MHC gene expression in normal and affected equine laryngeal muscle may help to further clarify the molecular events that occur with RLN. In addition, the current study evaluated the overall pattern of MHC expression. Additional investigation using monoclonal antibodies against each MHC isoform (types I, IIa, IIb, IIl, and IIx) may be beneficial.
A unique MHC isoform has been identified by several different investigators in the laryngeal muscle of laboratory animal species. 11, 34, 38, 40 This MHC isoform, termed “type IIl,” has also been referred to as “type IIeo,” as it has been shown to be identical to an MHC isoform with a high contractile velocity found only in extraocular muscle. This unique isoform is not found in human laryngeal muscle 32, 44 and does not appear to be present in equine laryngeal muscle based on the results of the present study.
Studies investigating the MHC isoform composition of equine limb muscle during development and with increased exercise training have noted a lack of expression of MHC type IIb in these muscles. 18, 47 Nevertheless, it was not surprising to find this isoform in equine laryngeal muscle, as expression of MHC type IIb in humans is limited to the craniofacial muscles and is not found in limb muscles. 19 While type IIb is expressed in rodent limb muscle, 28 it appears that the differential expression of MHC isoforms in different muscle groups is similar between horses and humans, and differs from that in rodents.
One obvious limitation of the present study is the lack of available information on the clinical status of the horses from which samples were obtained. As the samples were taken from horses without a detailed history related to upper airway function prior to death, we are unable to conclude whether the postmortem findings correlate with upper airway function prior to death. Future studies using laryngeal muscle from horses that have had upper airway endoscopic examinations performed prior to euthanasia would be useful.
In conclusion, the results of the present study show that, based on patterns of MHC isoform expression, laryngeal adductors are affected more severely and earlier in the course of the disease of RLN, and that RLN is a bilateral disease. Evaluation of muscle fiber changes at the protein level under denervated and reinnervated conditions may aid in understanding the cellular events that occur with the ongoing nerve injury associated with RLN. In addition, these experiments may be useful in assessing future strategies for reinnervation or regeneration of atrophic laryngeal muscle.
Footnotes
Acknowledgement
This study was supported by the Raymond Firestone Research Fund.
