Abstract
Lipin-1 (Lpin1)–deficient lipodystrophic mice have scant and immature adipocytes and develop transient fatty liver early in life. Unlike normal mice, these mice cannot rely on stored triglycerides to generate adenosine triphosphate (ATP) from the β-oxidation of fatty acids during periods of fasting. To compensate, these mice store much higher amounts of glycogen in skeletal muscle and liver than wild-type mice in order to support energy needs during periods of fasting. Our studies demonstrated that there are phenotypic changes in skeletal muscle fibers that reflect an adaptation to this unique metabolic situation. The phenotype of skeletal muscle (soleus, gastrocnemius, plantaris, and extensor digitorum longus [EDL]) from Lpin1-/- was evaluated using various methods including immunohistochemistry for myosin heavy chains (Myh) 1, 2, 2a, 2b, and 2x; enzyme histochemistry for myosin ATPase, cytochrome-c oxidase (COX), and succinyl dehydrogenase (SDH); periodic acid–Schiff; and transmission electron microscopy. Fiber-type changes in the soleus muscle of Lpin1-/- mice were prominent and included decreased Myh1 expression with concomitant increases in Myh2 expression and myosin-ATPase activity; this change was associated with an increase in the presence of Myh1/2a or Myh1/2x hybrid fibers. Alterations in mitochondrial enzyme activity (COX and SDH) were apparent in the myofibers in the soleus, gastrocnemius, plantaris, and EDL muscles. Electron microscopy revealed increases in the subsarcolemmal mitochondrial mass in the muscles of Lpin1-/- mice. These data demonstrate that lipin-1 deficiency results in phenotypic fiber-specific modulation of skeletal muscle necessary for compensatory fuel utilization adaptations in lipodystrophy.
Lipin family members (Lipin-1, 2, and 3) are phosphatidate phosphatase (PAP) enzymes that are critical in normal lipid biosynthesis and storage and play a role in lipid metabolism through transcriptional regulation of peroxisome proliferator-activated receptor α (PPARα) and PPARγ coactivator 1α (PGC-1α). 6 –8,10 –12,15 Lipin-1 is highly expressed in adipose, liver, skeletal muscle, and testes in mice and in adipose and skeletal muscle in humans. 10,29 –31 Lipin-1 was originally identified as the gene that was mutated in the lipodystrophic fatty liver dystrophy (fld; Lpin1-/-) mouse 29 and plays an essential role in adipocyte growth and differentiation. These mice are typically very thin, have low circulating leptin concentrations, and develop insulin resistance. 19,29,32,40 The morphological phenotype of this mouse is evident only in the homozygous state and has been characterized (reviewed in 10 ). The most notable findings in these mice are that they develop fatty liver early in life (which resolves prior to weaning), have scant and immature adipocytes, and develop a peripheral neuropathy (demyelination), which has been reported to be mediated by the PAP deficiency. 23,24,27 Humans with LPIN1 mutations are not lipodystrophic, although some individuals with LPIN1 mutations may have reduced adiposity. 12,44 The lack of lipodystrophy in humans may be the result of expression of the functionally similar LPIN2 gene product in adipose. 10 –12
Lpin1-/- lipodystrophic mice cannot rely on stored triglycerides to generate adenosine triphosphate (ATP) from the β-oxidation of fatty acids during periods of fasting. Instead, these mice store much higher amounts of glycogen in skeletal muscle and liver than wild-type mice to support energy needs during periods of fasting. 43 Normally during the feeding phase, mice use glucose that is not directed to glycogen storage for energy. However, to direct glucose toward a compensatory increase in glycogen storage during the fasting period, Lpin1-/- mice have increased hepatic fatty acid synthesis (∼30-fold) to provide fuel for β-oxidation by mitochondria in muscle during the fed state. 43 These liver/muscle flux interactions form an unique reversed diurnal metabolic cycle, in which glucose from glycogen in muscle is available for muscle utilization in the fasted state, and fatty acids from the liver are available for muscle utilization in the fed state. 43 The balance between the increases in hepatic fatty acid synthesis and muscle utilization in the fed state provides a mechanism to ameliorate fatty liver in the Lpin1-/- mouse that would otherwise occur as a consequence of adipose tissue dysfunction, as well as to satisfy energy needs of skeletal muscle while maximizing glycogen storage.
Given the metabolic changes in fuel utilization in Lpin1-/- mice, we wanted to investigate how this change affected muscle fiber type phenotype (Table 1) and to identify any similarities between the skeletal muscle changes in humans and LPIN1 mutations. Skeletal muscle fibers are generally categorized into fast twitch, typically glycolytic fibers (myosin heavy chain 2; Myh2), or slow twitch, typically oxidative fibers (myosin heavy chain 1; Myh1). There is a misconception that skeletal muscle fibers with high oxidative capacity are type 1 fibers; in fact, there is a wide variation in the oxidative capacity of type 2 fibers, and fast-twitch muscles may be subdivided based on the degree of oxidative capacity and fiber-shortening velocity. Fast-twitch fatigue-resistant (FR) fibers have high oxidative capacity (in mouse, Myh2a) and fast-fatigable (FF) fibers have low oxidative capacity (in mouse, Myh2b). 38 There are also fast-twitch fibers with intermediate shortening velocities between FR and FF fiber types (in mouse, Myh2x). 4,5 Differentiation between the oxidative capacity of fiber types can be evaluated by measuring mitochondrial enzyme activity histochemically and include succinate dehydrogenase (SDH) and cytochrome-c oxidase (COX) activity (Table 1). 38 These activity assays, as well as myosin ATPase assays, are commonly used to classify muscle fibers into type 2 (fast twitch) or type 1 (slow twitch). 42 Myh1 slow-twitch motor units stain strongly for SDH and COX compared with Myh2 FF fibers, although Myh2 FR fibers may have greater SDH and COX activity than Myh1 fibers. 38 Myh2x fibers (in rat skeletal muscle) have moderate to strong SDH and COX staining. 25,36 This complexity allows for flexibility in how muscle fibers can generate ATP under a wide assortment of metabolic conditions. 1 In this study, we demonstrate that the skeletal muscle fiber phenotype in Lpin1-/- mice is notably altered, presumably to adapt to metabolic perturbations. These changes include fiber-type switching, hybrid fibers, and increased oxidative capacity.
Summary of Phenotypic and Biochemical Characteristics of Skeletal Muscle.
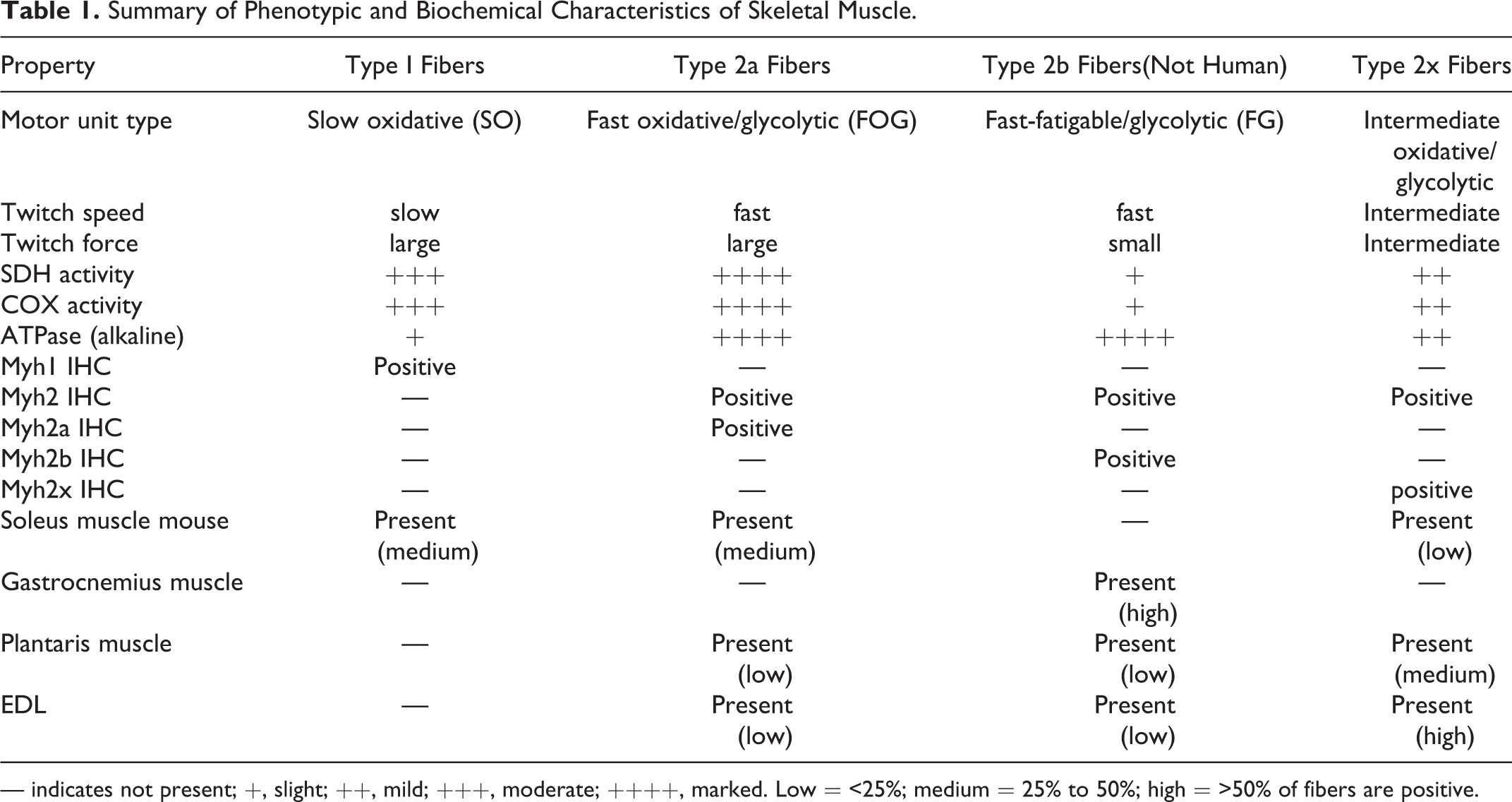
— indicates not present; +, slight; ++, mild; +++, moderate; ++++, marked. Low = <25%; medium = 25% to 50%; high = >50% of fibers are positive.
Materials and Methods
Animals
Male and female Lpin1-deficient (BALB/cByJ fld/J; Lpin1-/-; Jackson Laboratory, Bar Harbor, ME) and wild-type littermates (Lpin1+/+) were evaluated at 5 months of age. Genotype of mice was confirmed by TransnetYX, Inc (Cordova, TN). Mice were maintained under SPF barrier conditions at the Albert Einstein College of Medicine (AECOM) Barrier Facility, and all studies were carried out under an AECOM Institutional Animal Care and Use Committee–approved protocol. Bedding and in-cage sentinels were negative for all tested pathogens. Mice were fed autoclaved Purina Rodent Chow 5058 ad libitum, were on Bed-o’cobs 1/4-inch bedding (Andersons, Inc, Maumee, OH), and received autoclaved water via water bottles. Mice were humanely euthanized using isoflurane anesthesia followed by exsanguination. Evaluations were carried out on mice fasted for 5.5 hours (ad libitum access to water) prior to euthanasia.
Muscle Enzyme Histochemistry and Immunohistochemistry
The gastrocnemius (with plantaris), soleus, and extensor digitorum longus muscles (EDL) were removed through careful dissection to prevent stretching or crushing damage to the muscles. The muscles were affixed to cork and immediately frozen directly in liquid nitrogen or in 2-methylbutane (Fisher Scientific, Pittsburgh, PA) chilled in liquid nitrogen and then stored at –80°C until cryosectioning. Either method resulted in excellent tissue morphology.
Muscles were cryosectioned into 5-μm cross sections and placed on microscope slides (Superfrost Plus, Fisher Scientific, Hampton, NH) and briefly air dried. Histologic sections were stained for glycogen using the periodic acid–Schiff (PAS) reaction and for routine histology by hematoxylin and eosin. All samples were evaluated using enzyme histochemistry for the mitochondrial enzyme activities of myosin-ATPase (pH 9.6: inhibits slow, type 1 fiber ATPase activity only; type 1 fibers are light, type 2 fibers are dark), COX, and SDH using standard protocols. 34 The myosin-ATPase assay was carried out only under alkaline conditions because one of the reagents for the acidic assay was not available for purchase (sodium barbital). Immunohistochemistry was performed on frozen sections postfixed in acetone. Sections were stained for slow (Myh1; 1:40 dilution, mouse monoclonal IgG1, Vector Laboratories, Burlingame, CA) and fast (Myh2; 1:40, mouse monoclonal IgG1, Vector Laboratories), Myh2a (SC-71; 1:50 dilution, Developmental Studies Hybridoma Bank [DSHB], University of Iowa, Iowa City, IA), Myh2b (BF-FB; 1:50 dilution, DSHB), and Myh 2x (6H1; 1:50 dilution, DSHB) myosin heavy chains. Endogenous peroxidase activity was blocked using 0.3% hydrogen peroxide in water. The sections were stained by routine immunohistochemistry (IHC) methods, using the avidin-biotin-horseradish peroxidase complex to localize the antibody bound to antigen, with diaminobenzidine as the final chromogen. All immunostained sections were lightly counterstained with hematoxylin. The appropriate immunoglobulin isotype control was substituted for the primary antibody to serve as a negative control. Slides were evaluated by light microscopy. For gastrocnemius, plantaris, and soleus, COX and ATPase staining, 13 Lpin1+/+ and 15 Lpin1-/- mice were evaluated; for Myh1 and Myh2 IHC, SDH enzyme histochemistry, and PAS histochemical staining, at least 10 mice of each genotype were evaluated. For the IHC for Myh2 (Myh2a, Myh2b, Myh2x) subtypes, 7 Lpin1+/+ and 10 Lpin1-/- mice were evaluated. For EDL, COX and ATPase enzyme histochemistry and Myh1 and Myh2 IHC, 3 Lpin1+/+ and 5 Lpin1-/- mice were evaluated. A single section of muscle was evaluated for each stain.
Transmission Electron Microscopy
The soleus and EDL muscles from Lpin1+/+ (n = 3) and Lpin1-/- (n = 6 and 9, respectively) mice were evaluated by transmission electron microscopy (TEM). Muscle pieces were fixed with 2.0% paraformaldehyde and 2.5% glutaraldehyde in 0.1 M sodium cacodylate buffer, pH 7.4, for 60 minutes at room temperature, postfixed with 1% osmium tetroxide followed by 1% uranyl acetate, dehydrated through a graded series of ethanol, and embedded in LX112 resin (LADD Research Industries, Burlington, VT). Ultrathin sections of 80 nm were cut with a Reichert Ultracut UCT on to 200 mesh copper grids, poststained with uranyl acetate followed by lead citrate, and viewed on a JEOL 1200EX TEM at 80 kv.
Results
Slow-Twitch Type 1 Muscle Fibers in the Soleus of Lpin1-/- Mice Undergo Fiber-Type Switching to Fast-Twitch–Oxidative Type-2 Fibers
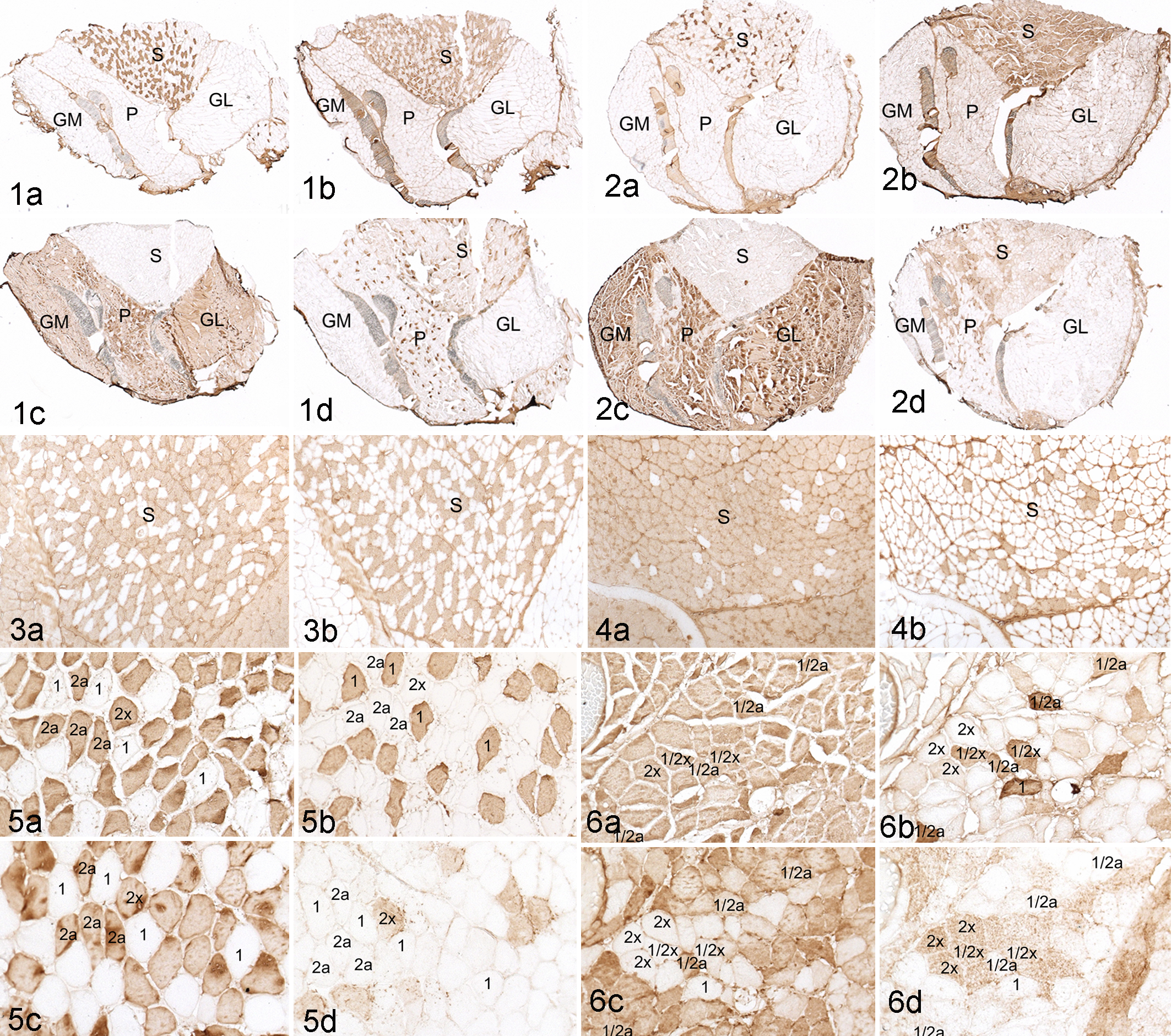
Changes in the skeletal muscle fiber type in Lpin1-/- mice were only in the soleus (Fig. 2a–d). The soleus muscle is a “red muscle,” which is characterized in the mouse as having both slow-twitch oxidative muscle fibers (Myh1) and fast-twitch oxidative-glycolytic fibers (Myh2a and Myh2x; Table 1; Fig. 1a–d). This is in contrast to the gastrocnemius muscle, which is composed entirely of Myh2b fibers and, the plantaris muscle, which is primarily Myh2b with scattered Myh2x and rare Myh2a fibers (Figure 1a–d). While the soleus muscles of Lpin1+/+ mice appeared to have only slightly more Myh2 than Myh1 fibers (Figs. 1, 3), the soleus of the Lpin1-/- mice was comprised primarily of Myh2a type myofibers with a dramatic reduction in Myh1 myofiber staining (Figs. 2, 4). There were no Myh fiber-type differences in the gastrocnemius muscles in Lpin1-/- mice as compared with Lpin1+/+ mice. The EDL muscle fibers were Myh2 but were not evaluated in more detail.
Soleus Muscle in Lpin1-/- Mice Has Increased Numbers of Myh1/Myh2 Hybrid Fibers
In addition to having more Myh2 immunopositive fibers, there was an increased presence of hybrid fibers in the soleus of Lpin1-/- mice as compared with Lpin1+/+ mice (Figs. 5, 6). Hybrid fibers are defined as those that express two or more Myh isoforms. While Myh1/2a hybrid fibers, Myh2a/2x, and occasionally Myh1/2x hybrid fibers are identified in normal skeletal muscle, they are uncommon. However, in the soleus of the Lpin1-/- mice, Myh2 expression frequently overlapped Myh1 expression (hybrid Myh1/2; Fig. 6), with the presence of Myh1/2a or Myh1/2x hybrid fibers variable between Lpin1-/- mice. Our studies did reveal type Myh1/2a hybrid fibers in the soleus of Lpin1+/+, but these were very uncommon.
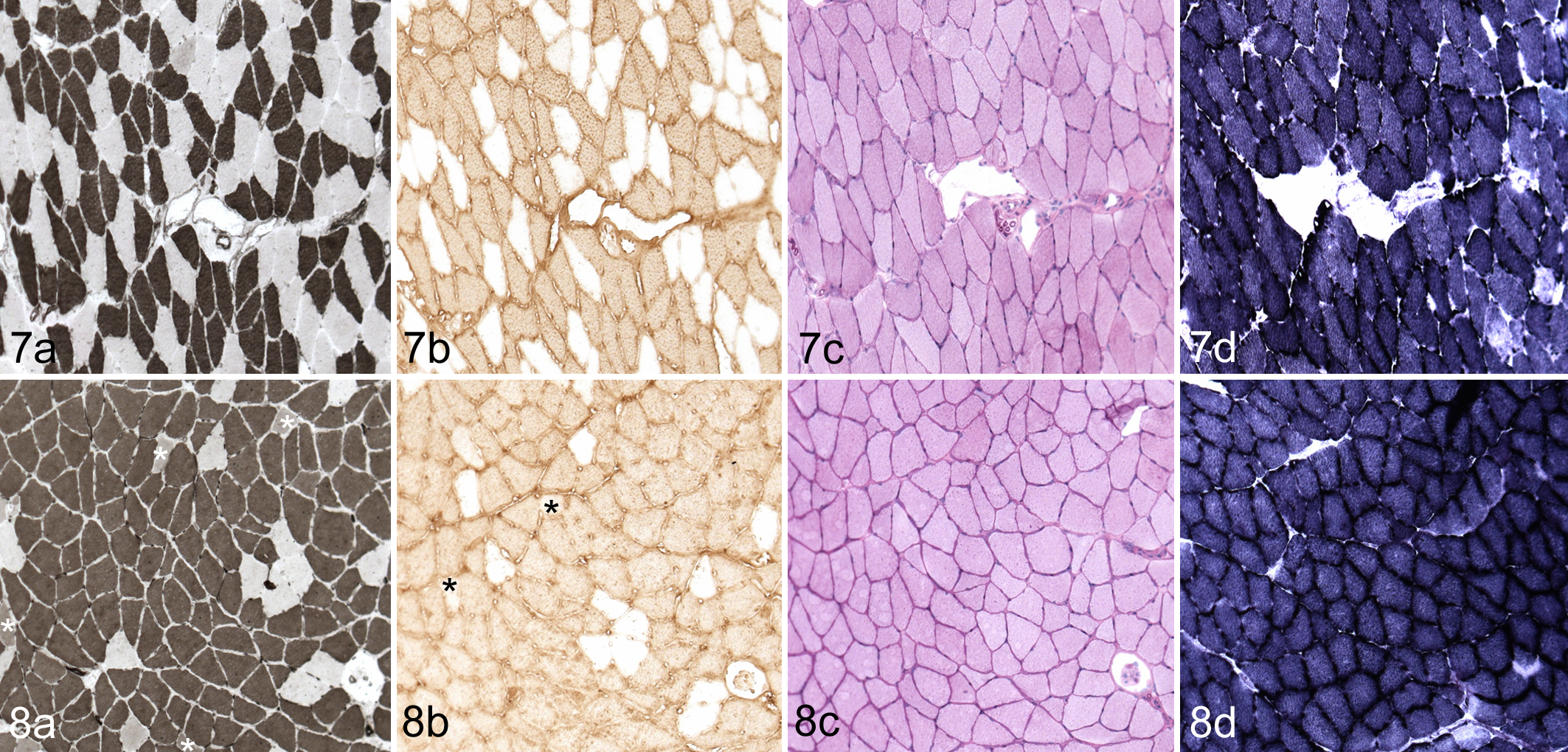
Myosin-ATPase enzyme histochemistry corroborated the IHC findings for fiber-type changes in the soleus. Under alkaline conditions, myosin-ATPase activity associated with slow-twitch fibers is inhibited (Myh1); only fast-twitch fibers stain darkly (Myh2). Typically, myosin-ATPase staining in type Myh2a fibers is more intense than type Myh2b fibers. The soleus in Lpin1+/+ mice had myosin-ATPase staining, which correlated with Myh2 immunohistochemistry in all muscles evaluated (Fig. 7). The soleus fibers of the Lpin1-/- mice were often nearly entirely myosin-ATPase positive (Fig. 8), consistent with the decrease in Myh1 and the increase in Myh2-positive fibers by IHC. However, myosin-ATPase staining was lighter in Myh2a fibers in Lpin1-/- than in Lpin1+/+ mice (Figs. 7, 8). Myosin-ATPase activity in Myh1/2 hybrid fibers of the soleus of Lpin1-/- mice was notably less dark than in adjacent Myh2a fibers (likely reflective of its partial Myh1 phenotype; Fig. 8). Myosin-ATPase staining in the gastrocnemius and plantaris muscles, which are comprised nearly entirely of Myh2b fibers, was not clearly different between Lpin1-/- and Lpin1+/+ mice (data not shown).
Mitochondrial Enzyme Activity and Glycogen Staining Are Altered in Lpin1-/- Mouse Soleus, Gastrocneumius, Plantaris, and EDL Muscles
In the soleus muscles of Lpin1+/+ mice, mitochondrial COX and SDH activity manifested as a “checkerboard” pattern of dark and light staining of muscle fibers (Figs. 7d, 9). Myh2a fast-twitch glycolytic-oxidative fibers have greater COX and SDH activity than the Myh1 slow-twitch oxidative fibers and thus stain more darkly. In the Lpin1-/- mice, the checkerboard pattern of light and dark staining was lost (Figs. 8d, 10), consistent with the replacement of Myh1 myofibers with Myh2 myofibers. The staining was also relatively uniform and of intermediate intensity (ie, darker than Myh1 and lighter than Myh2 fibers in the wild-type mouse soleus; Figs. 8d, 10).
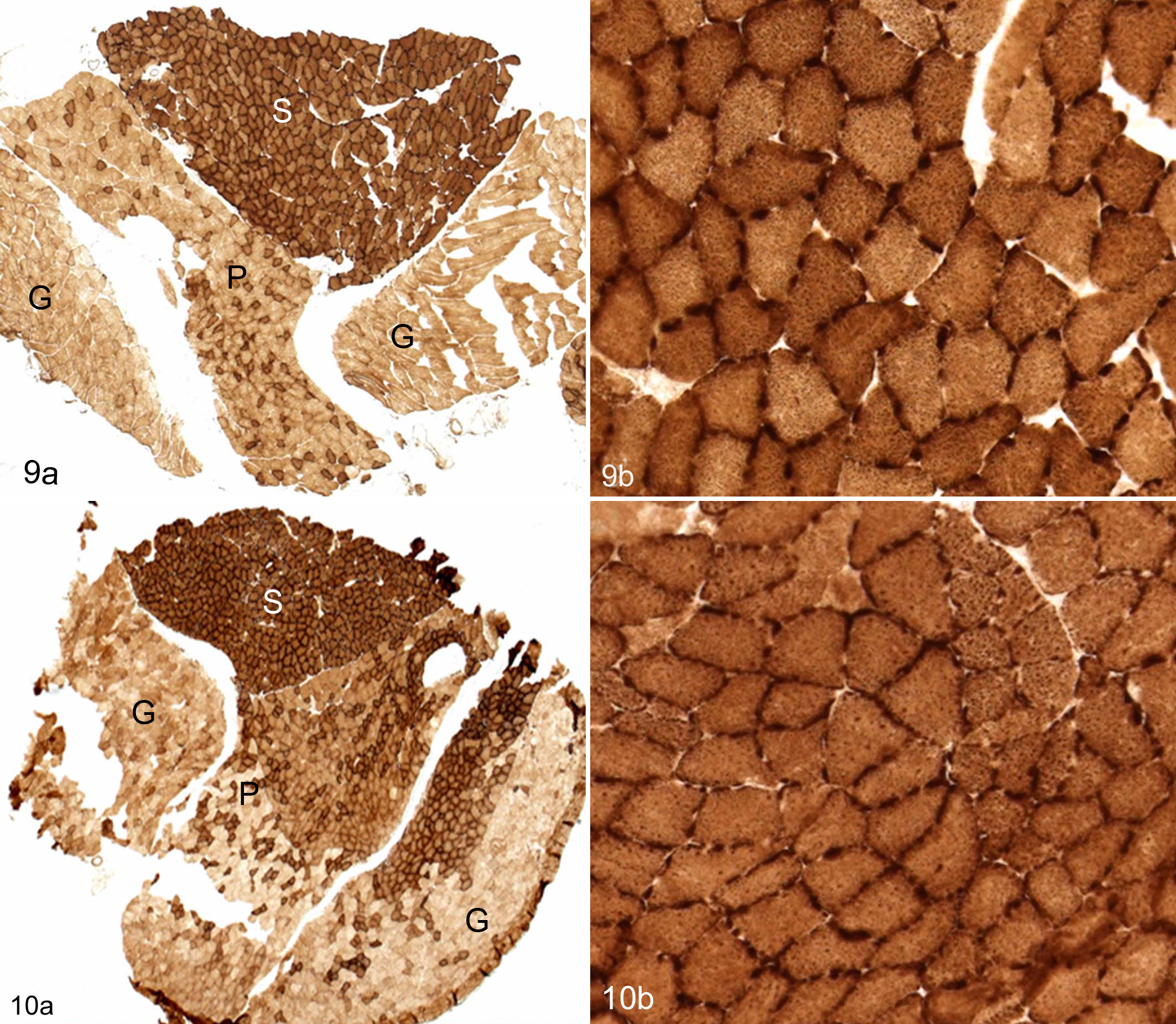
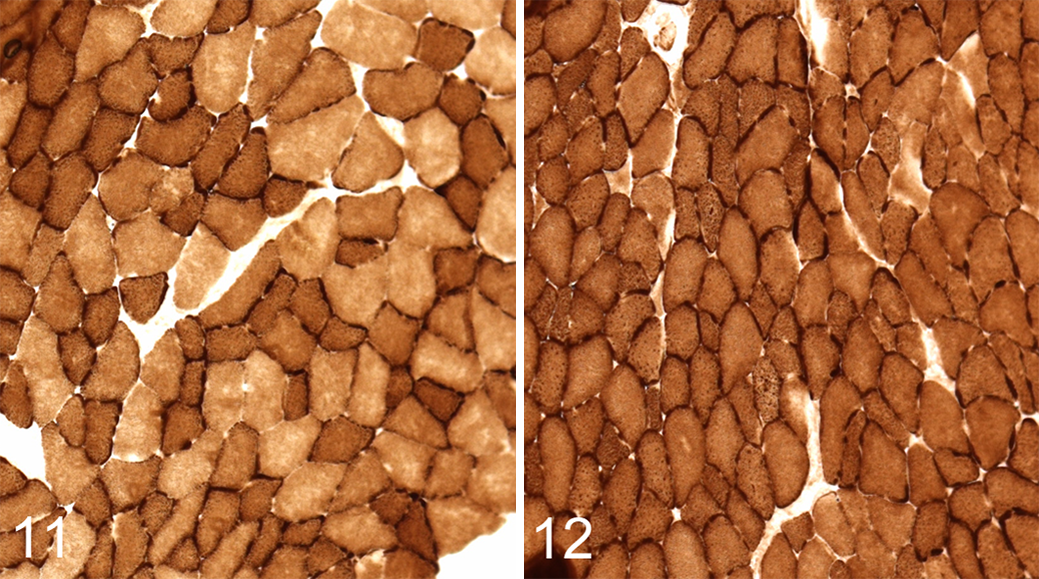
While there was no alteration in Myh fiber-type expression in the gastrocnemius, plantaris, and EDL muscles of Lpin1-/- mice, the myofibers had notably increased COX and SDH activity by enzyme histochemistry (Figs. 10, 12), in contrast to the mitochondrial activity of the Myh2 isotypes in Lpin1+/+ mice (Figs. 9, 11). The distribution of COX staining in the gastrocnemius and plantaris muscles was often darker and more extensive in Lpin1-/- mice as compared with Lpin+/+ mice (Figs. 9 vs 10); rather than scattered throughout the red portion of the muscle as in wild-type mice, the darkly staining COX fibers were irregularly distributed and clustered, sometimes over a large area in the Lpin1-/- mice (Figs. 9 vs 10). The dark staining often was at the cell membrane of the muscle fiber, most probably representative of subsarcolemmal mitochondrial activity, 22 rather than uniformly throughout the cell cytoplasm. The EDL muscle, which is also Myh2 fiber type, also had increased oxidative enzyme activity by COX as compared with Lpin1+/+ mice (Fig. 11 vs. Fig. 12), which was notable in the subsarcolemmal regions of the cells.
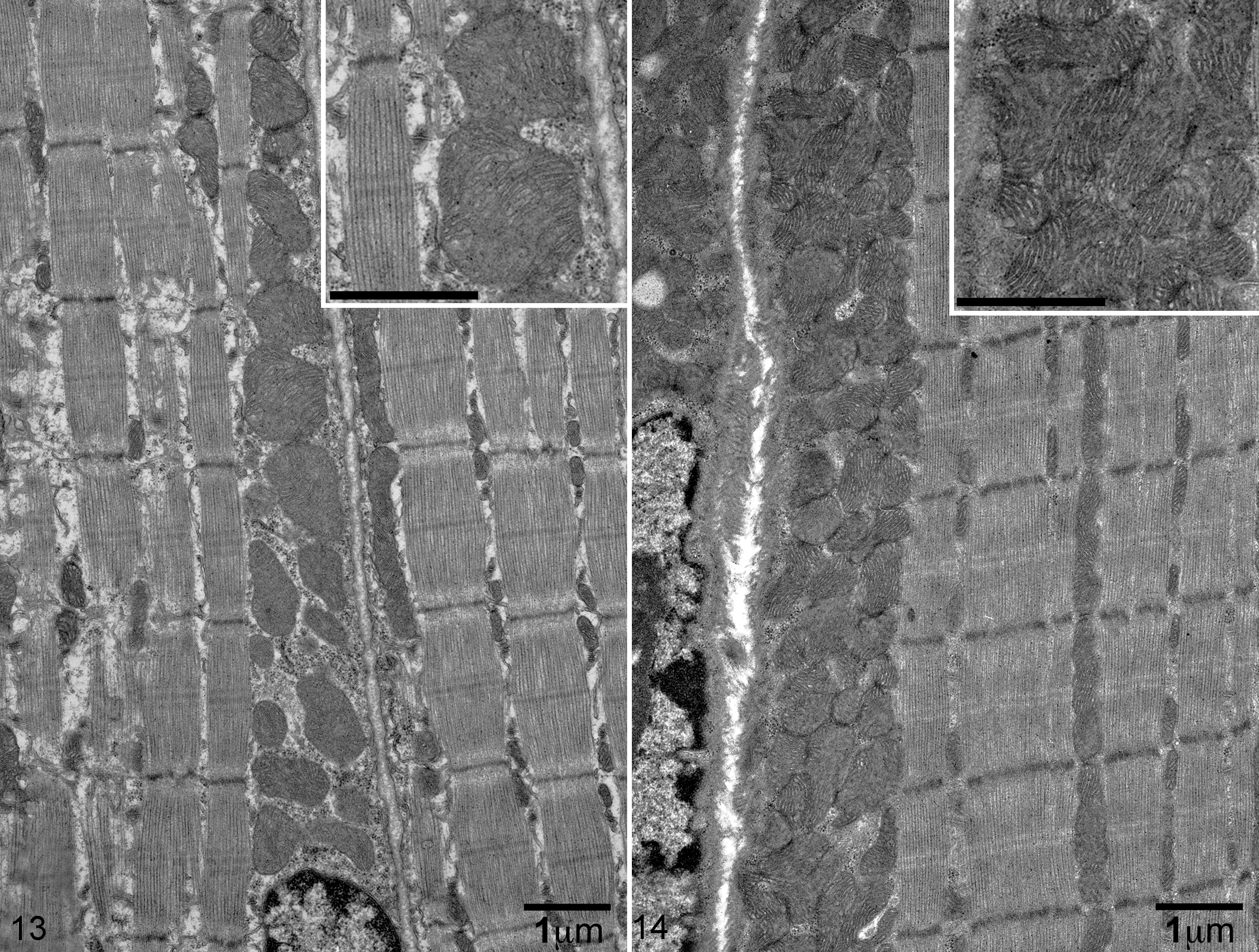
Mitochondrial Mass Is Increased in the Skeletal Muscle of Lpin1-/- Mice as Compared With Lpin1+/+ Mice by TEM
Consistent with the increased subsarcolemmal mitochondrial enzyme activity staining (COX and SDH) in the muscles of Lpin-/- mice, the number of mitochondria within the subsarcolemmal area on electron microscopy for Lpin1-/- mice was expanded as compared with Lpin1+/+ mice (Figs. 13 and 14). Subsarcolemmal mitochondria in the Lpin1+/+ mice were typically 1 to 2 deep (Fig. 13), whereas in the Lpin1-/- mice, subsarcolemmal mitochondrial densities increased to 5 to 6 deep (Fig. 14) and appeared smaller and more irregular in diameter. Similar changes in subsarcolemmal mitochondria mass was also seen between the Lpin1+/+ EDL and the Lpin1-/- EDL (data not shown). Lipid droplets were rarely evident by TEM, which was similar between the Lpin1+/+ and the Lpin1-/- mice.
PAS Staining in Skeletal Muscle Is More Uniform but Less Intense in Lpin1-/- Mice
The darkest (most intense) PAS staining (reflective of glycogen content) in the soleus muscle of Lpin+/+ mice was in the Myh2a-positive fibers, with lighter staining in the Myh1-positive cells. In Lpin1-/- mice, the muscle fibers were stained somewhat uniformly by PAS throughout the soleus (Fig. 8c), unlike in the Lpin1+/+ mice, which maintained a checkerboard pattern of staining (Fig. 7c). PAS staining was typically less intense in the skeletal muscles in Lpin1-/- mice as compared with the Lpin1+/+ mice.
Discussion
Lipodystrophic fatty liver dystrophy mice, which have a genetic deletion resulting in the absence of the Lpin1 gene product, have been shown to have altered fuel utilization 43 as a result of underdeveloped adipocytes and the inability to store triglycerides in adipose for energy during periods of fasting. Our studies demonstrated phenotypic differences in the skeletal muscle of the Lpin1-/- mice as compared with the wild-type control mice, which reflect adaptations to its unique metabolic situation. In particular, these adaptations expand the fuel utilization options of the skeletal muscle fibers by allowing oxidative fibers to become more glycolytic and glycolytic fibers to become more oxidative (Table 2). These changes include fiber-type switching from Myh1-type fibers to Myh2a or hybrid Myh1/2a or Myh1/2x fibers in the soleus muscle, an increase in mitochondrial enzyme activity with greater subsarcolemmal mitochondria density, and a more uniform storage of glycogen (by PAS staining) for all soleus muscle fibers. These changes are consistent with modifications to support flexibility in fuel utilization by skeletal muscle in the face of severely limited fatty acid storage capacity. 43
Summary of Skeletal Muscle Phenotypic and Biochemical Changes in Lpin1-/- Mice Compared With Wild-Type Controls.
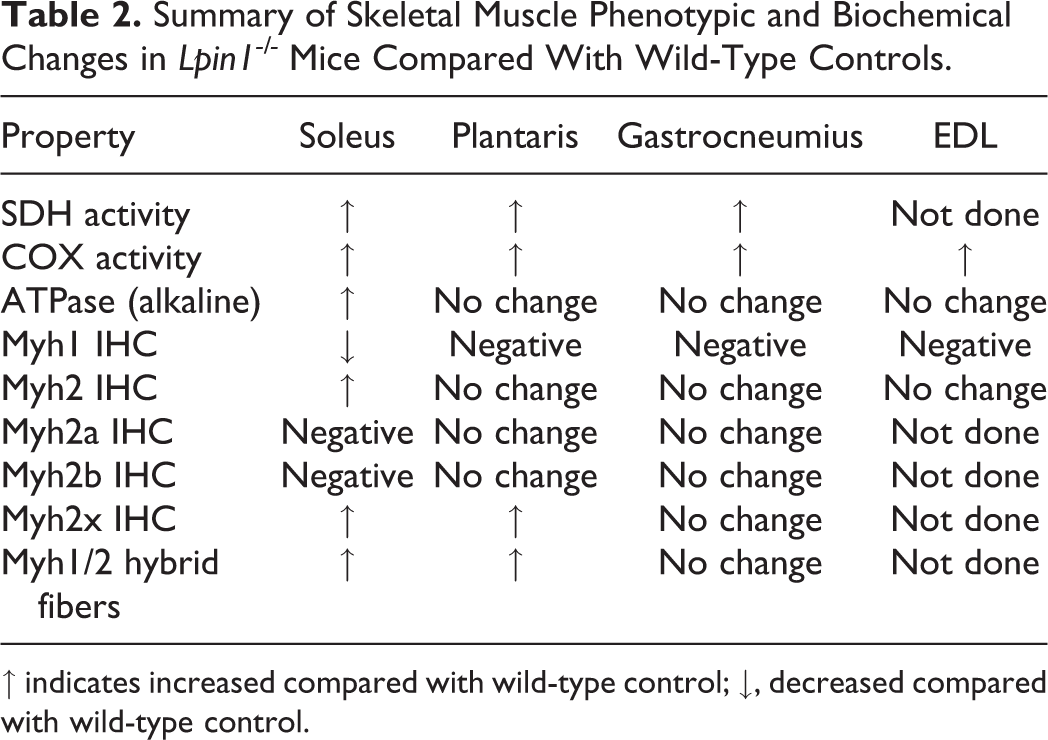
↑ indicates increased compared with wild-type control; ↓, decreased compared with wild-type control.
Fiber-type switching was readily apparent in the soleus muscle of Lpin1-/- mice, as this was the only muscle evaluated with Myh1 fibers. This muscle in mice is composed of approximately equal numbers of Myh1 (slow-twitch) and Myh2a (fast-twitch myofibers) myofibers. 3 In the Lpin1-/- mice, the Myh1 myofibers transitioned to Myh2a and had increased numbers of hybrid Myh1/2a or Myh1/2x myofibers as compared with wild-type mice. While we believe that the Myh1 to Myh2a fiber-type switching is a metabolic adaptation, demyelination is also reported in Lpin1-/- mice, and its contribution to this change is uncertain. There were no apparent increases in hybrid fibers in the other muscles examined, although rarely there were Myh2b/Myh2a hybrid fibers in the plantaris muscle.
Some of these changes may reflect a loss of PAP activity in the skeletal muscles, as is the case with LPIN1 mutations in humans. Humans with LPIN1 mutations typically present with symptoms related to rhabdomyolysis. 26,39 Lpin1-/- mice also have rhabdomyolysis, typically with metabolic stress, such as fasting. 46 The incidence may be underestimated in the mouse, however, as in vivo diagnosis is mainly limited to plasma creatine kinase assessment. The cause for rhabdomyolysis in humans has been suggested to be an accumulation of lysophospholipids, which may act as detergents and may disturb normal signal transduction. 45 Phenotypic changes in the skeletal muscle of humans with LPIN1 mutations, other than rhabdomyolysis, have not been well described. One case report indicated that there were increased numbers of hybrid muscle fibers (Myh2c) in a child with LPIN1 mutation–associated rhabdomyolysis, which has some comparability with the Lpin1-/- mice. 2,39 Data in humans suggest, however, that once the rhabdomyolysis resolves, muscle fiber-type changes return to normal, which is different from the mouse. 45 In addition, Dubowitz and Coraline 13 stated that skeletal muscle from humans with LPIN1 mutations appears to transition from Myh2 to Myh1 myofiber type; however, if the fiber type was inferred from the enzyme histochemistry, it may instead reflect increased oxidative capacity in Myh2 fibers, which would could be misinterpreted as an increase in Myh1 fibers. Ultimately, the type of LPIN1 mutation(s) may dictate some phenotypic changes within the skeletal muscle of humans, which may be unrelated to global metabolic disturbances. Work is in progress developing mice with skeletal muscle–specific Lpin1 deletion to specifically address the contribution of skeletal muscle Lpin1 loss to the changes observed in the study.
Because there are differences in the skeletal muscle phenotype of Lpin1-/- mice as compared with humans with LPIN1 mutations, the changes described in our studies reflect the plasticity of skeletal muscles to allow for a greater range of options for ATP generation. The presence of hybrid fibers and fiber-type switching reflects an adaptive response to metabolic needs and has been reported in humans in relation to metabolic stress. 14,16,20,37 Examples include Myh fiber-type alterations in obese individuals and those with insulin resistance (type 2 diabetes) 28,41 as well as in athletes. 35 Increases in skeletal muscle mitochondrial enzyme activity and distribution were also evident in Lpin1-/- mice, which was presumably an adaptive response to meet the metabolic demands of the tissue. Mitochondrial enzyme activity was evaluated by COX and SDH enzyme histochemistry. In the soleus, the typical checkerboard pattern of COX and SDH staining was lost, with all fibers staining intermediate between Myh2a and Myh1 fiber types with retention of sarcolemmal staining. This change in mitochondrial enzyme activity correlated with the presence of hybrid fibers and the transition to predominately Myh2a fiber type (Myh2a fibers have greater oxidative activity than Myh1 fibers in the mouse). The gastrocnemius and EDL muscles in Lpin1-/- mice also had increased COX and SDH staining intensity and distribution. In the gastrocnemius, which in Lpin1+/+ mice had only scattered oxidative-glycolytic Myh2b fibers, Lpin1-/- mice often had large clusters of intensely staining fibers, with notable subsarcolemmal staining. Increases in mitochondrial enzyme activity in Lpin1-/- mice correlated with TEM findings of increased subsarcolemmal mitochondrial density as compared with Lpin1+/+ mice (soleus and EDL muscles; gastrocnemius was not evaluated by TEM). Increased mitochondrial density likely reflects mitochondrial biogenesis. Increased metabolic demands, such as in athletes, have been shown to drive mitochondrial biogenesis. 18 It has been postulated that the functional capacity of subsarcolemmal mitochondria may be crucial for fatty acid oxidation, glucose transport, and propagation of insulin signaling to generate ATP for energy for protein synthesis and other cell processes. 9,21 Unlike at the subsarcolemmal location, mitochondria in the intermyofibrillar location generate energy to support muscle contraction 9,17,33 ; there was no apparent difference between Lpin1-/- and Lpin1+/+ mice in the density or size of these mitochondria. Together, these data show that there are increased skeletal muscle mitochondria in Lpin1-/- mice, which specifically supports the capacity to use fatty acids as a source of energy.
Previous work in our lab demonstrated that the skeletal muscle of Lpin1-/- mice increased glycogen storage, presumably to maintain energetics during fasting. 43 Evaluation of glycogen by PAS staining revealed that in these mice, glycogen was more uniformly distributed across muscle cells in the Lpin1-/- mice but was slightly less intense than in Myh2 fibers in the Lpin1+/+ mice. This is consistent with the findings by Xu et al, 43 which identified more glycogen overall in the muscles of Lpin1-/- mice than their wild-type counterparts.
Our data demonstrate that Lpin1-/- mice have some phenotypic similarities to humans with LPIN1 mutations and may have value as a model for the human disease. However, the phenotypic changes appear to be more dramatic in mice than in humans. This difference reflects skeletal muscle compensation to allow for use of a greater range of fuels in the face of severe metabolic disruptions and, as such, may serve as a model for studying skeletal muscle plasticity in metabolic disease.
Footnotes
Acknowledgements
Special thanks go to the members of the Histology and Comparative Pathology Facility at the Albert Einstein College of Medicine, in particular Hong Zhang for enzyme- and immuno-histochemistry, Jeffrey Harding and Victor Nieves for muscle cryosectioning, and Hillary Guzik for assistance with the EM figures.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: R.S.S., S.R.M., G.S.P., and F.P.M. were supported by the National Cancer Institute cancer center support grant (P30CA013330), and I.J.K. was supported by the Einstein-Mount Sinai Diabetes Research Center grant P60DK020541.
