Abstract
Canine rhabdomyosarcoma (RMS) presents a diagnostic challenge due to its overlapping histologic features with other soft tissue sarcomas. The diagnosis of RMS currently relies on positive immunohistochemical (IHC) labeling for desmin; however, desmin expression is also observed in non-RMS tumors. Myogenin and MyoD1 are transcription factors reported to be sensitive and specific IHC markers for human RMS, but they are not widely used in veterinary oncology. The goals of this study were to develop an IHC protocol for myogenin and MyoD1, evaluate myogenin and MyoD1 labeling in canine RMS, and report clinical outcomes. Sixteen cases of possible RMS were retrospectively evaluated. A diagnosis of RMS was confirmed in 13 cases based on histological features and immunolabeling for myogenin and MyoD1, with the aid of electron microscopy in 2 cases. Desmin was negative in 3 cases of RMS. Two cases were of the sclerosing variant. The median age of dogs with RMS was 7.2 years. Anatomic tumor locations included previously reported sites such as bladder, larynx, heart, and orbit, as well as other locations typical of soft tissue sarcomas. Survival ranged from 47 to 1480 days for 5 dogs with available data. This study demonstrated that MyoD1 and myogenin should be included with desmin as part of a diagnostic IHC panel for canine RMS. Utilization of these antibodies to improve the accuracy of canine RMS diagnosis will ultimately allow for better characterization of the biological behavior and clinical outcomes of this disease, providing the groundwork for future comparative investigations in canine RMS.
Canine rhabdomyosarcoma (RMS) is an uncommon tumor that arises from skeletal muscle and exhibits a high degree of variability in gross and histologic morphology. In humans, RMS is the most common soft tissue tumor of young adults and children. 24 A number of classification schemes are available for human RMS: the Horn-Enterline Classification, the International Classification of RMS, the Cytohistological (Palmer) Classification, the International Society of Pediatric Oncology Classification, the National Cancer Institute Classification, and the World Health Organization Classification that was recently modified. 24,27 The World Health Organization Classification categorizes human RMS as follows: embryonal (including the botryoid subtype), alveolar, pleomorphic, and spindle cell/sclerosing. 24 These categories and subtypes of human RMS carry varying prognoses, with the botryoid subtypes potentially associated with a favorable prognosis compared to the poor prognosis associated with alveolar RMS. 22,24
Canine RMS is classified similarly to human RMS using the World Health Organization Classification, but the prognostic significance of the different RMS subtypes is undetermined in dogs. 3 Diagnosis of canine RMS has been challenging, potentially due to its low frequency of occurrence and high level of variability in histomorphology. Cytology has not been a reliable way of diagnosing canine RMS, as the tumor cells can range in appearance from undifferentiated round cells to mature myoblasts or rhabdomyoblasts. 21 Although histologic evaluation of canine RMS is sometimes diagnostic, RMS cells can have widely variable histologic morphology, for example, appearing as round cells, or well-differentiated or undifferentiated mesenchymal cells. 3,21 As a result, canine RMS may be inaccurately diagnosed. 3 In human RMS, immunohistochemistry (IHC) is an accepted diagnostic tool to distinguish RMS from other mesenchymal or embryonic tumors and to aid in subtyping RMS. 3,32 Subclassifying human RMS is clinically important due to the varying prognoses associated with different subtypes. 32 Historically, the lack of specificity of IHC markers for human RMS presented a diagnostic challenge, as myogenic markers such as desmin and actin can be expressed in RMS as well as in normal skeletal muscle and non-myogenic sarcomas. 3,4 MyoD1 and myogenin are regulatory transcriptional factors that are expressed in the early stages of skeletal muscle differentiation and have been reported as sensitive and specific IHC markers for human RMS diagnosis. 4,30 However, the use of myogenin and MyoD1 to assist in the diagnosis of canine RMS is uncommonly reported. 5,8,13,14,21,23 Instead, veterinary diagnostic pathology continues to rely on less specific markers such as muscle-specific actin, desmin, and vimentin to diagnose RMS, which can potentially result in inaccurate diagnoses. 5
Thus, the small repository of information that is currently available for canine RMS needs to be expanded in order to improve diagnosis and understanding of the disease and its biological behavior, which in turn will advance treatment approaches. The goals of this study were to develop an IHC protocol for myogenin and MyoD1 in order to evaluate the expression and utility of these markers in the diagnosis of canine RMS arising from various anatomical locations, as well as describe the clinical outcomes of this case series of canine RMS.
Materials and Methods
Case Selection
A search of the database of the Veterinary Diagnostic Laboratory (VDL) at Colorado State University was done between July 1, 2010, and March 1, 2015, for canine tumors that were diagnosed as RMS or soft tissue sarcomas (STS) with RMS listed as a differential diagnosis. Both external and internal university submissions were included. Out of 122 732 canine samples submitted for histopathology, 145 cases were identified and those with available paraffin blocks, good-quality histologic slides, and patient-related information such as signalment and tumor location were selected for inclusion in this study. As a result, 16 archived cases from various anatomic locations were selected based on the original final diagnosis being RMS or STS that was undifferentiated, poorly differentiated, or anaplastic. All samples were from client-owned animals and had been submitted for routine histopathological evaluation by their attending veterinarian.
Histopathological and Immunohistochemical Analysis
Archived, formalin-fixed paraffin-embedded tumor samples were routinely sectioned and stained with hematoxylin and eosin (HE). A board-certified veterinary pathologist (DPR) reviewed all HE-stained slides to determine whether the tumors had features consistent with RMS and the slides had sufficient tumor tissue for IHC. Unaware of the original Colorado State University-VDL diagnosis, this pathologist blindly evaluated the histomorphological features of the tumors and IHC labeling, and evaluated ultrastructural features (where applicable) to arrive at a final diagnosis. Histological diagnosis and classification was based on those morphological features previously reported by Caserto for the diagnosis of RMS, in conjunction with reference to the human RMS classification literature from which these canine features were adapted. 3,10,12,24,25,27 A light microscopic diagnosis of RMS was based on the presence of any combination or amount of these previously described features of RMS, including (1) large round to polygonal, brightly eosinophilic rhabdomyoblast cells; (2) multinucleated myotubular “strap” or “racquet” cells with or without the presence of cytoplasmic cross-striations; (3) a pleomorphic mixture of polygonal to stellate to spindle cells displaying varying degrees of these 2 aforementioned morphologies; or (4) small, densely packed round cells with scant cytoplasm and uniformly round hyperchromatic nuclei, in conjunction with the absence of a histological features suggestive of other soft tissue sarcomas, such as lipid-like vacuoles (liposarcoma, hibernoma) or prominent and interlacing fascicles of monomorphic spindle cells (fibrosarcoma, leiomyosarcoma). When distinct histomorphological features of tumors consistent with RMS were observed, histologic subtyping was performed according to a canine-adapted human RMS classification scheme. 3
Immunohistochemical labeling with myogenin and MyoD1 was performed using the following primary antibodies: monoclonal mouse anti-human myogenin (ThermoFisher, clone F5D, 1:800 dilution) and monoclonal mouse anti-human MyoD1 (ThermoFisher, clone 5.8A, 1:50 dilution). The genes for myogenin (MYOG) and MyoD1 (MYOD1) are conserved in different species including the dog and mouse. The protein BLAST analysis using the mouse myogenin sequence against the canine database demonstrated 93% identity (209/224 amino acids) with zero gaps (0%) between the mouse and dog myogenin sequences. Similarly, the protein BLAST analysis using the mouse MyoD1 sequence against the canine database demonstrated 90% identity (286/319 amino acids) with one gap (0%) between the mouse and dog MyoD1 sequences. These results are highly predictive of antibody recognition between the mouse and dog. The vendor datasheet for the myogenin antibody states a cross-reactivity with the dog. 28 Additionally, reports in the literature for the myogenin and MyoD1 antibodies demonstrate cross-reactivity with canine tissue, and a cross-reactivity profile for both antibodies is available from the CiteAb database. 1,6 –8,14,23,26 Desmin IHC had been performed in 11 of the tumors by the Colorado State University-VDL, and the authors of this study performed desmin IHC for the 5 samples that did not receive initial desmin IHC. Immunohistochemistry for desmin was performed using a monoclonal mouse anti-human desmin ready-to-use primary antibody (Leica Biosystems Inc, clone DE-R-11). Immunohistochemical labeling with all 3 primary antibodies was performed using a Leica Bond III autostainer platform. Briefly, slides were de-paraffinized in xylene and rehydrated using a series of graded alcohols. Epitope retrieval was performed using pH 9.0 tris-EDTA buffer at 95 °C for 20 minutes (epitope retrieval 2 setting). Detection for all antibodies was performed using an alkaline phosphatase polymer conjugated rabbit anti-mouse secondary antibody (Leica Inc), and positive labeling was visualized using the Fast Red chromogen substrate. A Bond ready-to-use universal negative control was used (Leica Inc), which served as the isotype control. Additionally, many of the tumors had adjacent normal tissues that served as internal negative controls. A RMS previously determined to express MyoD1 and myogenin served as the positive control (RMS positive control provided by Dr Beth Valentine, Oregon State University; expression data not shown).
Semiquantitative scoring of both percentage of neoplastic cells that were immunolabeled and the intensity of positive tumor cell immunolabeling for myogenin and MyoD1 was performed in a blinded fashion as follows: 0 = no positivity; 1+ = rare to <25% positivity; 2+ = 25% to 50% positivity; 3+ = 50% to 75% positivity; 4+ = >75% positivity, while intensity was scored as follows: 0 = no intensity; 1+ = weak intensity; 2+ = moderate intensity; 3+ = strong intensity. Tumor samples were interpreted as positive for MyoD1 and myogenin only when nuclear labeling was observed, regardless of the presence of cytoplasmic (nonspecific) labeling, a diagnostic criterion previously utilized in the validation of these markers in human formalin-fixed paraffin-embedded RMS cases. 19
Transmission Electron Microscopy
Transmission electron microscopy (TEM) evaluation was performed in 4 cases (cases 6, 10, 12, 15) that had equivocal diagnoses based on histopathology and IHC labeling. Selected areas of tissue were punched out of paraffin blocks previously examined by light microscopy. Following removal of the paraffin using xylene, the samples were rehydrated through graded ethanol and washed with 0.1 M sodium cacodylate buffer (pH 7.4). The tissues were re-fixed in 2.5% glutaraldehyde and 2% formaldehyde in the same buffer, washed, and post-fixed with buffered 1% osmium tetroxide. After final buffer washing and dehydration to 100% ethanol, the tissue samples were passed through propylene oxide and infiltrated with Eponate 12 epoxy resin (Ted Pella, Inc). Resin-infiltrated samples were transferred to an embedding mold and polymerized at 60 °C. Ultrathin sections (70-90 nm) were cut from the polymerized blocks using a diamond knife on a Reichert Ultracut E ultramicrotome, mounted on copper grids, and stained with uranyl acetate and lead citrate. Grids were examined and photographed using a JEOL JEM-1400 transmission electron microscope equipped with an Orius SC 1000 CCD camera (Gatan, Inc).
Patient Profile and Outcome
Case information including signalment, age at diagnosis, tumor location, and survival time were obtained from review of medical records and from follow-up communications with the referring veterinarian or owners. Age at diagnosis was calculated based on time of initial diagnosis of the primary tumor, whether or not it was diagnosed as RMS on initial evaluation. Survival was calculated as number of days from time of diagnosis to time of death. Dogs whose lesions were diagnosed at the time of necropsy were excluded from survival time reporting. Dogs that were lost to follow-up had follow-up times calculated as number of days from time of diagnosis to date of last follow-up.
Results
A total of 16 cases of suspected or diagnosed as canine RMS submitted to the Colorado State University-VDL were included in the study. Patient signalment, local recurrence, locations of the primary and metastatic tumors of all dogs initially included are listed in Table 1.
Clinical Findings and Final Diagnoses.
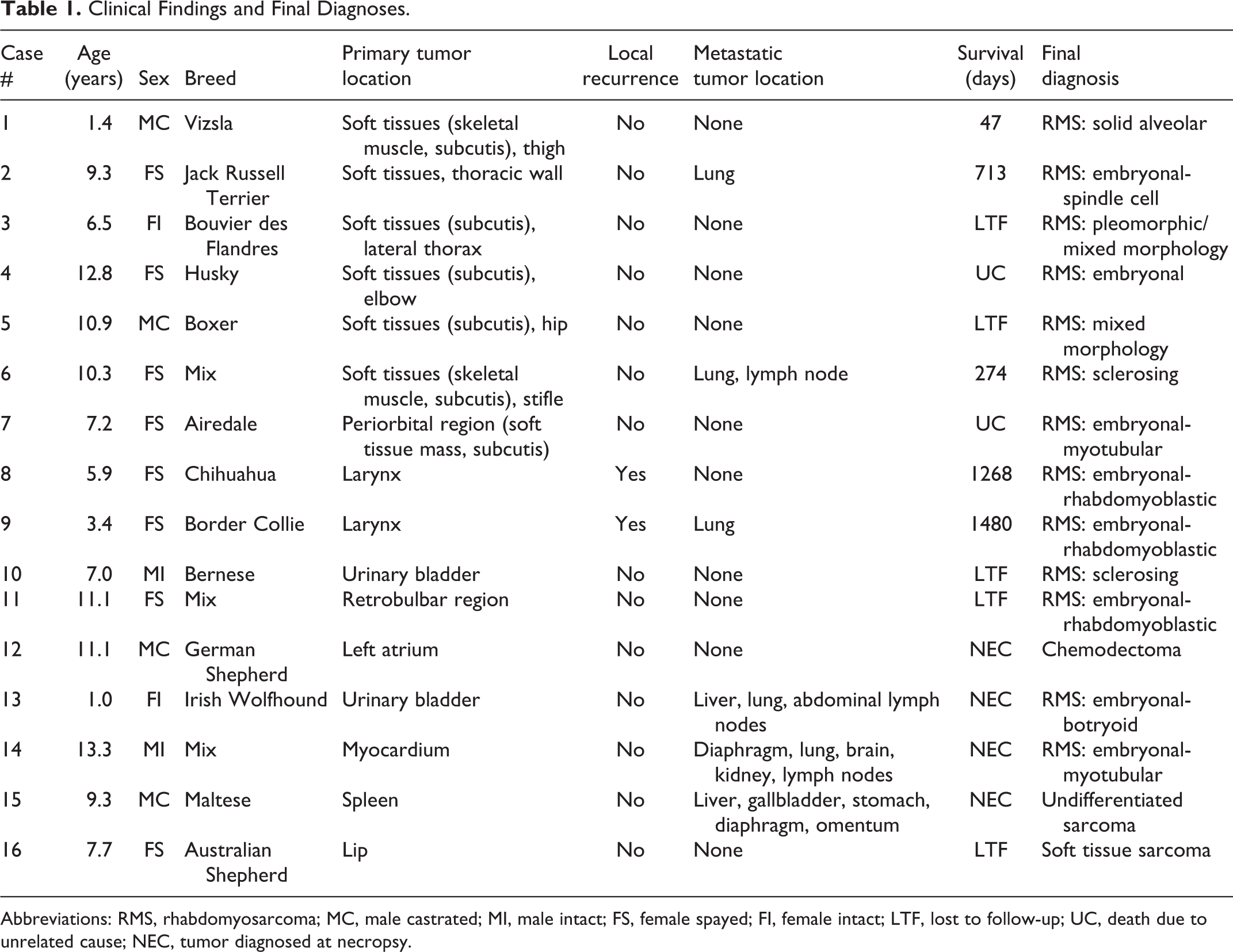
Abbreviations: RMS, rhabdomyosarcoma; MC, male castrated; MI, male intact; FS, female spayed; FI, female intact; LTF, lost to follow-up; UC, death due to unrelated cause; NEC, tumor diagnosed at necropsy.
Of the 16 cases, 12 tumors had been originally diagnosed as RMS, 3 as STS/possible RMS, and as anaplastic sarcoma (Table 2). Mitotic count was variable, with 7 of 16 cases exhibiting between 25 and 40 mitoses per ten 40× fields (2.37 mm2 area). The remaining 9 cases exhibited 2 to 16 mitoses per ten 40× fields.
Immunohistochemical Findings and Diagnoses.
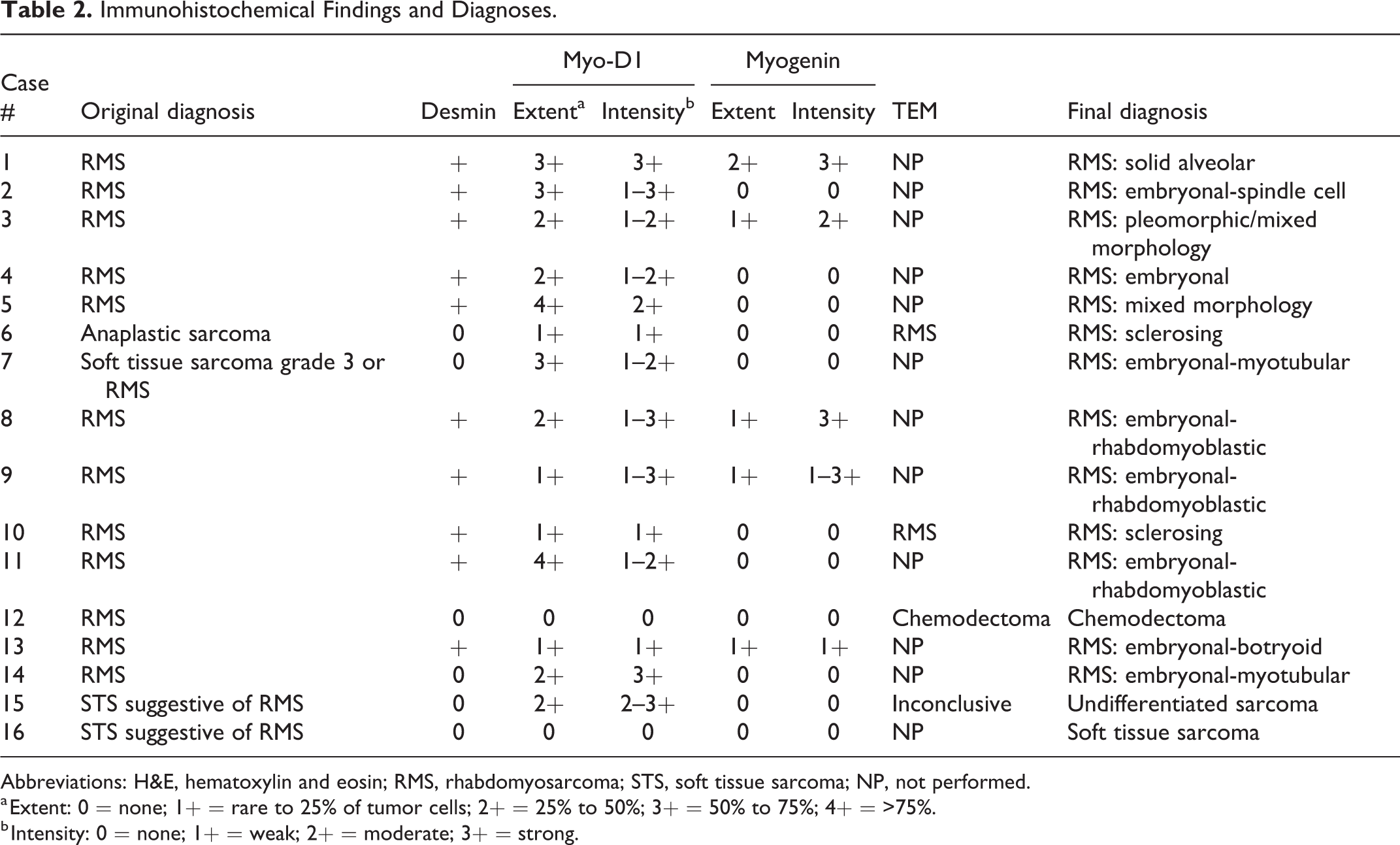
Abbreviations: H&E, hematoxylin and eosin; RMS, rhabdomyosarcoma; STS, soft tissue sarcoma; NP, not performed.
a Extent: 0 = none; 1+ = rare to 25% of tumor cells; 2+ = 25% to 50%; 3+ = 50% to 75%; 4+ = >75%.
b Intensity: 0 = none; 1+ = weak; 2+ = moderate; 3+ = strong.
On subsequent evaluation of HE-stained slides, 10 of 16 tumors had definitive histologic features characteristic of RMS, and were classified into one of the defined RMS histologic subtypes. 3 The most common was the rhabdomyoblastic variant of embryonal RMS (Fig. 1). The other 6 cases did not display typical histological features diagnostic of RMS and were diagnosed as undifferentiated/pleomorphic sarcoma based on histopathology alone.
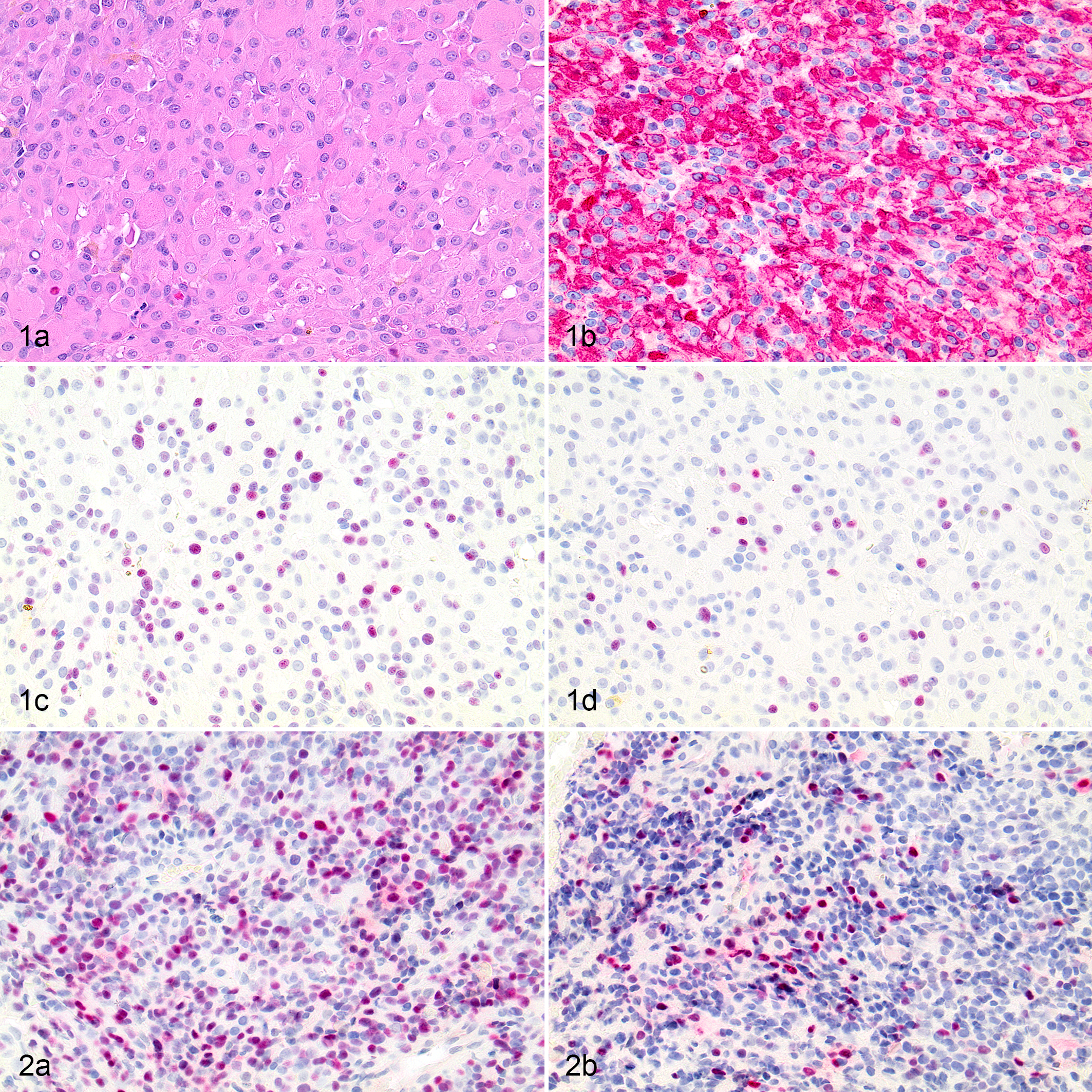
Embryonal rhabdomyosarcoma, rhabdomyoblastic variant, larynx, dog, case 8. (a) Neoplastic cells are uniformly round to slightly polygonal with abundant eosinophilic homogeneous to finely granular cytoplasm. Hematoxylin and eosin (HE). (b) Neoplastic cells have diffuse, strong, cytoplasmic immunolabeling for desmin. Positive nuclear labeling for MyoD1 (c) and myogenin (d) is present in less than 50% of tumor cells.
Immunohistochemical reactivity to myogenin, MyoD1, and desmin was then used to complement the histopathologic diagnoses. The utility of the presence or absence of positive IHC labeling with MyoD1 and/or myogenin antibodies to further support or refute the histopathologic diagnosis of RMS, based on the reported sensitivity and specificity of these markers, was evaluated. 4,15,19,31 Fourteen of 16 tumors were positive for MyoD1. Both the intensity of MyoD1 labeling and extent of tumor cell labeling were highly variable across different tumors, ranging from weak to strong, and from rare expression to expression in >75% of neoplastic cells (Table 2). Of the 4 cases with low (1+) extent of tumor cell positivity, 3 displayed very rare MyoD1 immunolabeling. Localization of MyoD1 immunoreactivity was nuclear in 6 samples (Fig. 2a), and both nuclear and cytoplasmic in 8 samples. Five of 16 tumors displayed positive immunoreactivity for myogenin. In contrast to MyoD1, the intensity and degree of positive tumor cell myogenin labeling were less variable, and was usually moderate to strong in ≤50% of tumor cells (Table 2). Myogenin immunoreactivity was nuclear for all 5 cases (Fig. 2b). Ten of 16 tumors were immunoreactive for desmin. Three of the desmin-negative tumors (cases 6, 7, 14) were given a final diagnosis of RMS in this study based on a combination of histopathology, IHC, and, where available, TEM findings.
Of the 6 cases that were not diagnosed as RMS based on histopathology alone, 4 were found to be positive for myogenin and/or MyoD1 (but only 3 were finally diagnosed as RMS). Specifically, 1 case (case 1) had MyoD1 and myogenin labeling (Fig. 2a and b), while 3 cases had only MyoD1 labeling. However, 1 of these 3 cases (15), although demonstrating rare positive nuclear reactivity for MyoD1, could not be confirmed as RMS with TEM evaluation (see below). Additionally, HE-stained sections of this tumor had features which were not suggestive of RMS, with a complete lack of any cells resembling rhabdomyoblasts (ie, large, round, eosinophilic; or small, primitive forms). Instead the neoplastic cells had pale, eosinophilic to amphophilic, fibrillar to sometimes vacuolated cytoplasm embedded in a collagenous stroma and arranged in interlacing bundles. The remaining 2 cases with histological features not consistent with RMS were both negative for MyoD1, myogenin, and desmin. One was diagnosed as STS (16) and the other as a chemodectoma based on TEM features (12, see below).
TEM was performed for 4 cases (cases 6, 10, 12, 15). Case 6 had a morphology in HE-stained sections consistent with a sclerosing variant of RMS (Fig. 3a), a rare subtype in humans. 33 This tumor was desmin and myogenin negative but displayed rare 1+ nuclear labeling for MyoD1 (Fig. 3b). TEM evaluation of case 6 revealed individual polygonal cells reminiscent of the rhabdomyoblastic morphology observed on light microscopy with extensive poorly developed cytoplasmic myofilaments arranged in a tangled, haphazard fashion, and associated with numerous polyribosomes and short electron-dense bands or plaques, consistent with a diagnosis of RMS (Figs. 4, 5). Similar to case 6, case 10 had a morphology in HE-stained sections consistent with the sclerosing variant of RMS (Fig. 6a). This tumor also displayed rare 1+ nuclear labeling for MyoD1 (Fig. 6b), but in contrast to case 6, was desmin-positive (Fig. 6c). TEM evaluation of case 10 revealed similar ultrastructural findings of individual polygonal cells with haphazardly arranged, poorly developed myofilament tangles occasionally associated with polyribosomes and short electron dense bands/plaques, and abundant extracellular collagen. However, myofibrils or z bands were not conclusively demonstrated by TEM. Despite this, a diagnosis of a sclerosing variant of RMS was made based on the presence of myofilament tangles and polyribosomes in conjunction with the dual MyoD1 and desmin-positive labeling. Case 12 did not have any TEM features consistent with RMS, such as myofilament tangles and polyribosomes, but instead had abundant cytoplasmic electron-dense granules consistent with neuro-secretory granules. The presence of these granules in conjunction with a heart-base location of the mass and negative immunolabeling for desmin, myogenin, and MyoD1 was consistent with a diagnosis of chemodectoma (Suppl. Fig. S1A-D). TEM evaluation of case 15 was extremely confounded by poor preservation of ultrastructural morphology. TEM demonstrated some cytoplasmic filaments suggestive of RMS, but the tumor had other nuclear and cytoplasmic features that were suggestive of leiomyosarcoma such as nuclear indentation and thin filaments with periodic dense bodies. 11,18 Compared to cases 6 and 10, the cytoplasmic filaments in case 15 were less frequent, and not arranged in tangles or associated with polyribosomes. Additionally, nuclei from these cells appeared elongated with rounded ends, and poorly defined external lamina with adjacent sub-plasma membrane densities were present, features consistent with leiomyosarcoma. 11,18 Case 15 was desmin- and myogenin-negative. While this case did display some nuclear labeling of MyoD1, the degree of cytoplasmic labeling greatly outweighed the nuclear reactivity, suggesting this labeling was nonspecific. This lack of immunoreactivity for myogenic markers, in conjunction with inconsistent morphology in HE-stained sections and ultrastructural features more consistent with leiomyocarcoma, resulted in a final diagnosis of undifferentiated sarcoma, presumably leiomyosarcoma.
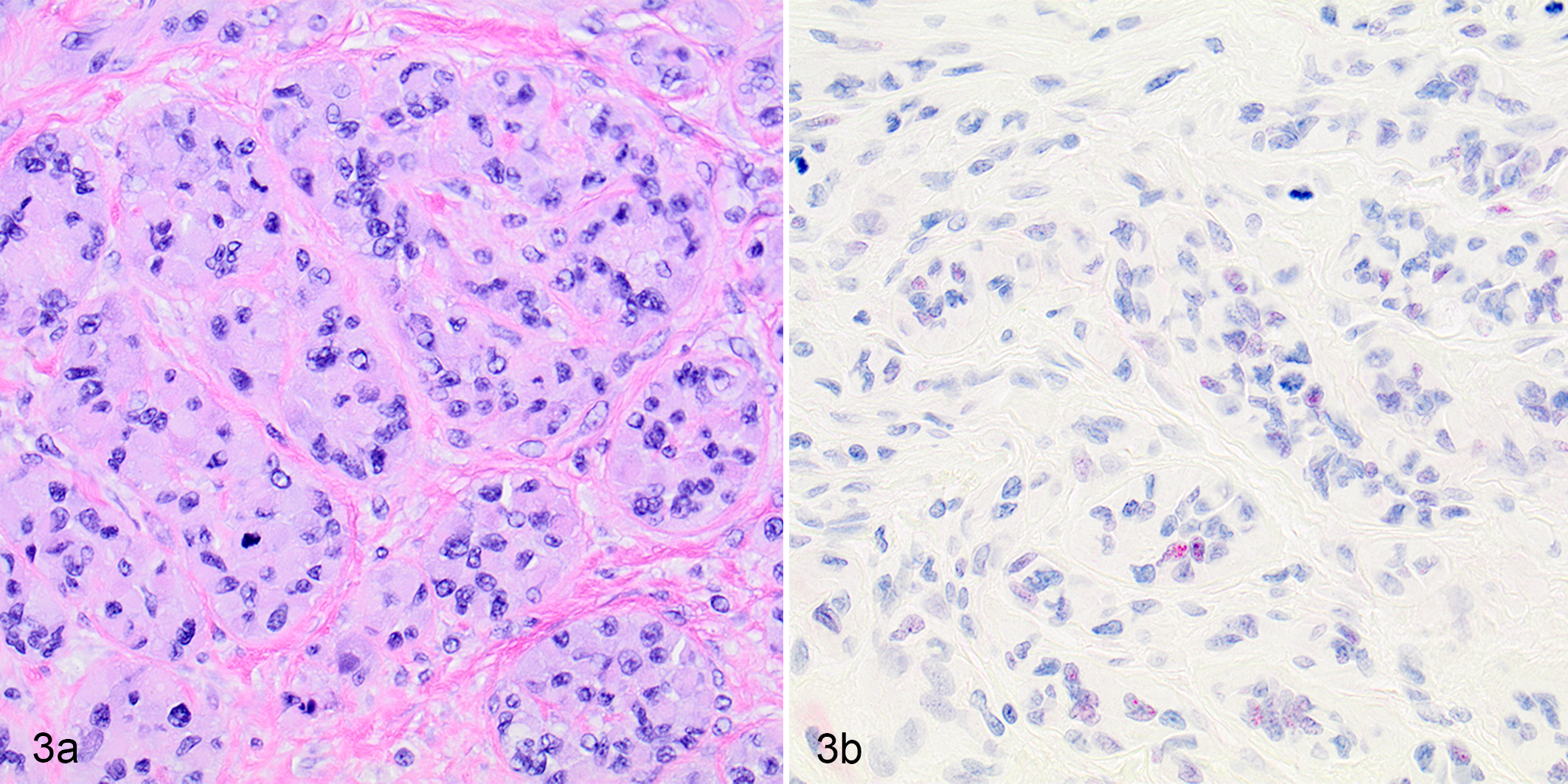
Sclerosing rhabdomyosarcoma, soft tissues adjacent to the stifle, dog, case 6.
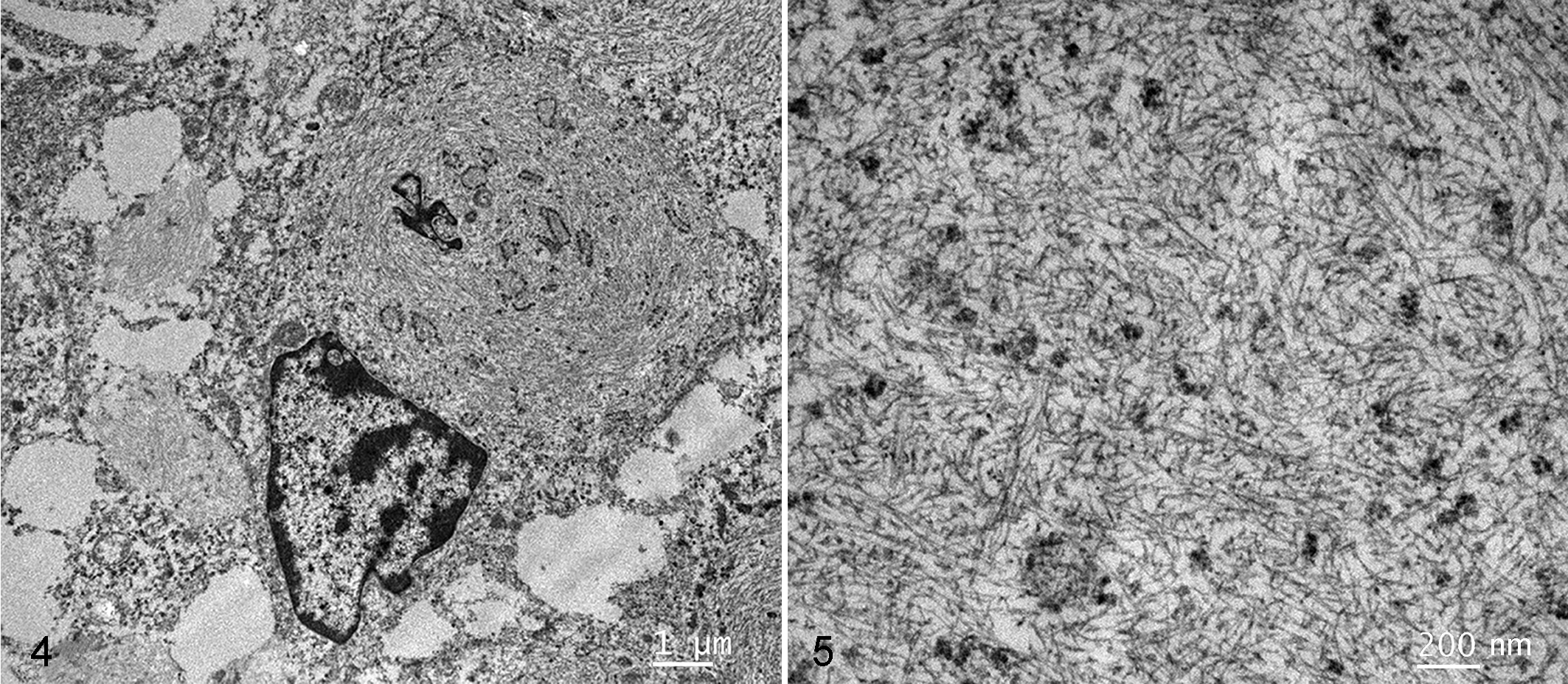
Sclerosing rhabdomyosarcoma, soft tissues adjacent to the stifle, dog, case 6. The cytoplasm of a round rhabdomyoblastic cell contains abundant poorly developed myofilaments that are haphazardly arranged and occasionally associated with electron-dense linear bands and plaques. Transmission electron microscopy.
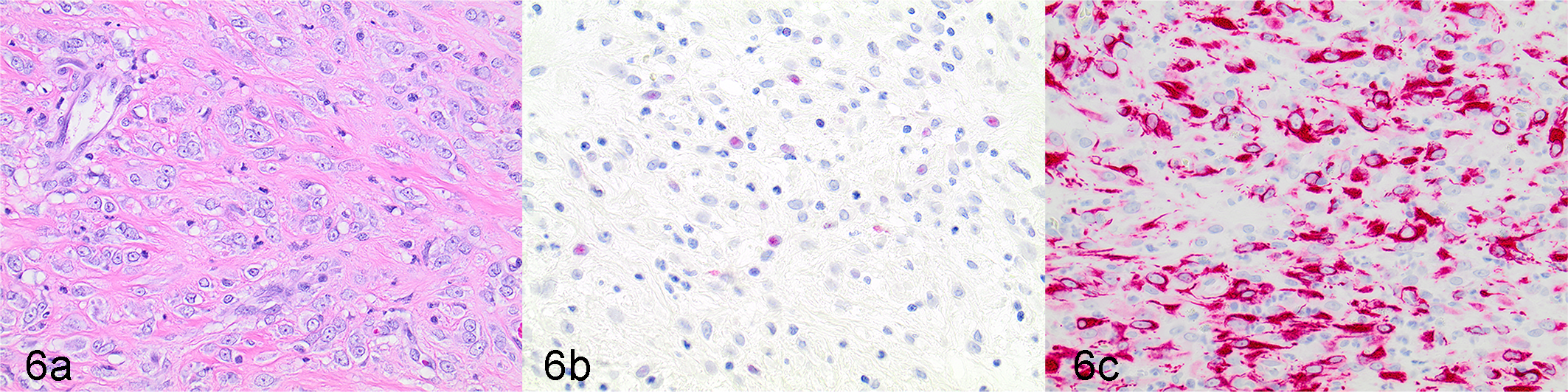
Sclerosing rhabdomyosarcoma, urinary bladder, dog, case 10. (a) Individualized polygonal neoplastic cells are embedded within a dense collagenous stroma. Hematoxylin and eosin. (b) Rare neoplastic cells demonstrate weak nuclear immunolabeling for MyoD1. (c) Neoplastic cells demonstrate diffuse, strong, cytoplasmic immunolabeling for desmin.
Diagnostic Summary
Thirteen of 16 tumors were given a final diagnosis of RMS. Two of the 3 tumors not diagnosed as RMS did not display any histologic, IHC, or TEM (where applicable) features of RMS, and 1 tumor did not display conclusive TEM findings of RMS. The final diagnoses after histopathology, IHC and TEM (where available) for all tumors in this study are indicated in Table 2. In summary, 13 of the 14 MyoD1-positive tumors were diagnosed as RMS. Three of these RMS were desmin-negative. One tumor was MyoD1-positive but not diagnosed as RMS (case 15) based on inconclusive results on TEM evaluation. The degree of cytoplasmic labeling greatly outweighed the nuclear reactivity, suggesting the nuclear labeling in this case was nonspecific. All RMS tumors were positive for MyoD1. All tumors positive for myogenin were diagnosed as RMS.
Histologic Features of 13 Confirmed RMS Cases
Embryonal Subclass
General histopathologic features were conserved across all 7 cases of embryonal RMS, which included rhabdomyoblastic, myotubular and spindle-cell variants. These histopathologic features were characterized by varying populations of spindle-shaped to round to polygonal neoplastic cells that were frequently invasive into surrounding tissues.
The rhabomyoblastic variant of embryonal RMS was diagnosed in 3 cases. These cases had a predominance of neoplastic rhabdomyoblasts that were round to irregularly round and had abundant, very brightly eosinophilic, granular to almost short fibrillar cytoplasmic contents, and large euchromatic nuclei. In many cases, these rhabdomyoblasts were often arranged in sheets of moderate to high cellular density with more pleomorphic bi- and multinucleate cellular forms occasionally interspersed. Less frequently, primarily in regions of lower cellular density, these neoplastic rhabdomyoblasts appeared unipolar with cytoplasmic tails (“tadpole” cells) or had moderate amounts of homogeneously eosinophilic elongated cytoplasm with multiple nuclei approximately arranged in a row (“strap” cells). Rarely, these strap-like cells appeared as more well-differentiated myotubes with poorly defined cytoplasmic cross-striations.
The myotubular variant of embryonal RMS was diagnosed in 2 cases because they were predominated by these multinucleated forms of neoplastic cells. In these cases, most neoplastic cells either had elongated eosinophilic cytoplasm with multiple, large, vesicular nuclei aligned in a row (“strap” cells), or were irregularly globoid with abundant eosinophilic cytoplasm, multiple haphazardly arranged nuclei, and variably distinct unipolar cytoplasmic elongations (“racquet” cells).
In both the rhabdomyoblastic and myotubular variants of embryonal RMS, those regions containing the variant-defining rhabdomyoblastic or myotubular cell morphologies were always admixed with a minority population of more primitive neoplastic myogenic cells with scant to indistinct pale basophilic cytoplasm and uniformly round densely stained nuclei.
A single case in this study was consistent with the spindle-cell variant of embryonal RMS, characterized by a pure population of polygonal to spindle-shaped cells with scant to moderate amounts of brightly eosinophilic, fibrillar cytoplasm, and indistinct cell borders. These cells were arranged in interlacing streams and bundles and embedded within a collagenous stroma.
Alveolar Subclass
One case (case 1) was diagnosed as an alveolar RMS (solid variant). It was characterized by dense sheets of small, primitive round cells with scant cytoplasm and hyperchromatic nuclei, consistent with a prior report. 3
Mixed Morphology
Three tumors displayed mixed histological morphologies. Specifically, case 4 was consistent with the embryonal subclass of RMS but the variant could not be definitively categorized as the tumor did not display a predominance of any one cellular morphology. Instead, this case consisted of roughly equal populations of large, round to polygonal rhabdomyoblastic cells with abundant eosinophilic cytoplasm, multinucleated myotubular cells, and short streaming bundles of spindle cells, depending on the region of the mass evaluated. These varying morphologies were all embedded within moderate amounts of mucinous matrix (rhabdomyoblasts or myotubular cells) or collagenous stroma (spindle cells).
Case 5 was characterized by a heterogeneous population of neoplastic cells which displayed morphological features equally consistent with both the embryonal and alveolar subclasses of RMS. Case 5 had tumor regions composed of neoplastic cells with classic alveolar RMS morphology characterized by lobules of loosely clustered “alveolar-like” packets of neoplastic cells supported on branching fibrous septa. However, these areas of classic alveolar RMS morphology transitioned into equally sized regions of inconspicuous packeting (solid-type variant of alveolar RMS) or areas that were consistent with embryonal RMS, with a predominance of large, multinucleated myotube-like cells (“strap” cells) or large, multinucleated, irregularly round to oval cells with a unipolar cytoplasmic elongation or tail (“racquet” cells), admixed with fewer polygonal rhabdomyoblasts.
Last, case 3 was histologically most consistent with pleomorphic RMS, characterized by a predominance of densely packed sheets of undifferentiated polygonal to spindle-shaped cells with scant, indistinct cytoplasm, large, round to oval nuclei with coarsely clumped to open vesicular chromatin, marked anisokaryosis, and a very high mitotic count. However, this tumor also had smaller subregions of neoplastic cells with embryonal-rhabdomyoblastic and embryonal-myotubular morphologies, as described above.
Sclerosing RMS
The 2 cases were diagnosed as sclerosing RMS and had slightly differing morphologies. One sclerosing RMS occurred in the urinary bladder (case 10) and was characterized by focal segmental submucosal regions of small neoplastic cells with scant cytoplasm and hyperchromatic nuclei, which were subtended by a large, infiltrative mass composed of poorly differentiated polygonal to sometimes unipolar rhabdomyoblastic cells with scant to indistinct, pale basophilic cytoplasm and large, round euchromatic nuclei with a single prominent nucleolus. These rhabdomyoblastic cells were embedded—either individually, in small clusters or packets, or poorly formed short linear aggregates—in abundant amounts of a dense collagenous stroma. The other sclerosing RMS (case 6) was also composed of clusters and packets or rhabdomyoblastic cells embedded in an abundant and very dense collagenous stroma. However, neoplastic rhabdomyoblasts in this case were more often arranged in packets reminiscent of classic alveolar architecture and demonstrated an increased degree of differentiation, appearing larger and more polygonal with increased amophophilic, homogenous to granular cytoplasm (Fig. 3a).
Clinical Data
The median age at diagnosis for the 13 cases of RMS was 7 years (range, 1–13 years). Seven of 9 female dogs were spayed females, and 2 were intact females. Two of 4 male dogs were castrated males, and 2 were intact males. No breed prevalence was observed: 3 cases were mixed-breed, with the remainder represented by individual distinct breeds. Primary tumor locations were variable, with subcutaneous tissue being the most common (
Eleven dogs underwent surgical tumor resection. One dog had a biopsy performed for a urinary bladder lesion noted incidentally during an abdominal exploratory surgery for a possible gastrointestinal foreign body. Four cases were diagnosed at necropsy. Five dogs with RMS had metastatic disease; 2 of these cases were diagnosed at time of necropsy. Of the cases of metastatic disease not diagnosed at necropsy, 1 had pulmonary metastasis, 1 had local recurrence of tumor and pulmonary metastasis, and 1 had both pulmonary and nodal metastases (pulmonary metastases were presumptively diagnosed based on radiographic appearance of pulmonary nodules; lymph node metastasis was cytologically confirmed). One case of RMS had recurrence of disease without metastasis (recurrence was presumptively diagnosed based on tumor growth at the site of previous RMS masses). Three dogs died of RMS-related causes.
Three dogs died of causes unrelated to RMS. The cause of death was unknown in 2 dogs. The survival time of dogs with RMS was available for 5 dogs and ranged from 47 to 1480 days (Table 1). Four dogs with RMS were lost to follow-up; 3 of these were reportedly doing well at the last follow-up, at 339, 586, and 872 days after diagnosis; 1 dog was lost to follow-up immediately after diagnosis.
Discussion
This study demonstrated myogenin and MyoD1 to be markers that can assist in the diagnosis of canine RMS in conjunction with histopathologic characteristics and desmin immunoreactivity. Accurate diagnosis of canine RMS has been challenging and often requires a multimodal approach that includes histopathologic evaluation and IHC labeling. Myogenin and MyoD1 immunoreactivity, as shown in this study, can be used as part of an IHC panel for diagnosing RMS, and can be especially helpful as confirmatory markers in more complex cases, for example, tumors with histological features consistent with RMS but are desmin-negative. TEM is considered the gold-standard diagnostic for human and canine RMS; however, TEM is costly and not a practical diagnostic tool in veterinary medicine. 3 In addition to histological assessment, immunohistochemical evaluation has been used to facilitate the diagnosis of canine RMS, and the basic requirements of diagnosis are the presence of at least one skeletal muscle marker and the absence of smooth muscle markers. 3 Commonly used IHC indicators include positive labeling with desmin, vimentin, and myoglobin, and negative labeling for α-smooth muscle actin. 21 However, these markers can lack sensitivity and specificity. For example, in undifferentiated RMS lesions, myoglobin and desmin labeling may be absent, as expression of these markers occurs later in myogenic differentiation. 2,30
Myogenin and MyoD1 are transcriptional regulatory proteins that stimulate the early stages of myogenesis and are reportedly sensitive and specific IHC markers for human RMS. 4,19,30 Retrospective studies evaluating the utility of myogenin and MyoD1 in human RMS reported positive labeling with myogenin in all RMS samples. 4,15 One study reported lack of myogenin immunoreactivity in some sarcomas including peripheral nerve sheath tumor, leiomyosarcoma, and myofibrosarcoma. 4 In that study, positive MyoD1 labeling was also noted in all RMS samples, but the high prevalence of cytoplasmic and nonspecific background labeling complicated pathological interpretation. 4 A retrospective study of 956 cases of human RMS reported a sensitivity of 97% and specificity of 90% for myogenin, and a sensitivity of 97% and specificity of 91% for MyoD1, in diagnosing RMS. 19 In our study, the sensitivity and specificity was not determined because of the small sample size and because few non-RMS tumors were evaluated. However, all RMS were positive for MyoD1, suggesting MyoD1 may be sensitive in dogs, and all tumors positive for myogenin were RMS.
Myogenin and MyoD1 were useful for diagnosing canine RMS in this study in conjunction with histopathologic characteristics and desmin immunoreactivity. Of the 13 cases diagnosed as RMS in this study, all 13 were MyoD1-positive and 5 were myogenin-positive. One of these tumors (6) was a soft tissue mass in a location typical for STS (soft tissue adjacent to the stifle) and was diagnosed as an anaplastic sarcoma due to absence of desmin immunoreactivity. It however displayed 1+ MyoD1 positive nuclear labeling, which led to a diagnosis of RMS, confirmed via TEM (Figs. 3–5). This case, as well as cases 7 and 14, which were also negative for desmin, highlights the important observation that not all cases of RMS are desmin-positive, and emphasizes the utility of additional markers, such as MyoD1 and myogenin, as part of the toolkit for diagnosing RMS. However, there are still challenges to diagnosing RMS, as evidenced by case 15. This case exhibited Myo-D1 positivity but was desmin-negative, with the majority of tissues not morphologically consistent with RMS except for a metastatic diaphragmatic lesion. TEM evaluation of this case was inconclusive. Case 15 highlights the potential challenges in RMS diagnosis even with the use of advanced diagnostics including histopathology, IHC for multiple markers, and TEM evaluation.
Overall, 14 cases were labeled for MyoD1, although 1 of these cases (15) was not diagnosed as RMS. The 2 cases that were negative for MyoD1 and desmin were not diagnosed as RMS. The location of positive MyoD1 labeling was either exclusively nuclear or a combination of nuclear and cytoplasmic. Five cases stained positively for myogenin, all of which displayed nuclear labeling. Even though the labeling of MyoD1 should theoretically be exclusively nuclear, reports have shown the occurrence of cytoplasmic labeling as well. 19 Although it has been proposed that any nuclear labeling of MyoD1 should be considered as positive, regardless of the presence of concurrent cytoplasmic labeling, case 15 suggests that the degree of cytoplasmic labeling relative to nuclear reactivity might need to be taken into consideration. 19 Potentially, the MyoD1 immunoreactivity in case 15 was a false positive. Similar to this study, tumors in a large human RMS retrospective study exhibited intense and well-defined myogenin labeling that was almost exclusively confined to the nucleus. 19 Interestingly, authors of that case series also observed that MyoD1 labeling can be weak if sections are not immunolabeled within a few days or if they are stored at room temperature. 19 This could potentially account for the variability in the MyoD1 labeling observed in this study.
Desmin IHC is commonly used to aid RMS diagnosis, but it lacks specificity and is not 100% sensitive. One study reported a sensitivity of 99% and specificity of 71% for desmin in human RMS. 19 Desmin labeling can be observed in other human tumors such as alveolar soft part sarcomas. 19 In dogs, desmin positivity has been observed in tumors besides RMS, including leiomyosarcomas, leiomyomas, and malignant fibrous histiocytomas. 17,20 Of the 6 tumors in the current study that exhibited negative desmin immunoreactivity, 3 of them were given a final diagnosis of RMS, highlighting the importance of not relying solely on desmin immunoreactivity to diagnose RMS. This highlights the inconsistent presence of desmin immunoreactivity in canine RMS, especially in poorly differentiated or primitive tumors. 2,3 Myogenin and MyoD1 can be used as additional IHC markers to diagnose RMS, especially in more primitive lesions, desmin-negative RMS lesions, or lesions that exhibit desmin positivity but are not RMS. However, myogenin and MyoD1 IHC immunoreactivity can be observed in other tumor types, such as canine liposarcoma and nerve sheath tumors, emphasizing the importance of combining histopathology and IHC, and ideally a panel of IHC evaluations including myogenin and MyoD1 to diagnose RMS. 16
The median age of dogs with RMS was 7.2 years. Two dogs under 2 years old were affected: one had a subcutaneous RMS in the thigh, and the other had metastatic RMS arising in the urinary bladder. This differs from the observation that canine RMS most often affects dogs under 2 years of age, with 63% of non-laryngeal RMS occurring in dogs under 2 years old. 3 However, the low case numbers and varied RMS locations in this study can influence the degree to which the median age of affected dogs was representative. Anatomic tumor locations for RMS in this study included previously reported locations, such as bladder, larynx, heart, and orbit, but also other locations typical of STS (soft tissues of the thigh, elbow, stifle, hip, thoracic wall, and lip). 3,29 Metastasis was documented in over a third of the cases. The survival data for 5 dogs included a wide range of 47 to 1480 days, and the low number of dogs precluded statistical analysis.
Accurate classification of RMS subtypes in humans is important due to the different prognoses for each subtype and hence different treatment approaches. Alveolar RMS carries a poorer prognosis compared to embryonal RMS; thus, more aggressive treatments are used for alveolar RMS. 19,32 Current veterinary literature lacks reports detailing the clinical outcomes of canine RMS, especially when compared to other STSs. There are also no data associating RMS histologic subtypes with different prognoses in dogs. Canine RMS potentially can be classified into subtypes similar to human RMS, as was done in this study based on histopathologic characteristics. While acknowledging the very limited data available in this study, it is interesting to note that the dog with the alveolar subtype had the shortest survival (47 days) and dogs with the longest survivals (1268 and 1480 days) had the embryonal subtype. With only 13 confirmed RMS in this study, 2 were of the sclerosing subtype, which is a rare subtype in humans. 33 In human RMS, myogenin and MyoD1 have also been used to distinguish between the various RMS subtypes. 4,19 Alveolar RMS reportedly has strong and consistent myogenin labeling compared to embryonal RMS, which has more variable positivity and intensity. 4,9,15 In this study, MyoD1 labeling was more consistently present compared to myogenin labeling. However, due to the presence of only one alveolar subtype in this study, distinguishing potential differences in myogenin and MyoD1 labeling between embryonal and alveolar subtypes could not be made. Further studies will be required to determine whether differences in labeling patterns of myogenin and MyoD1 are associated with RMS subtypes in dogs.
Potential limitations of this study include the use of samples that were stored for a period of time before myogenin and MyoD1 immunolabeling. MyoD1 labeling can be weaker if not performed within a few days of the paraffin section being cut, or with storage of the paraffin sections at room temperature. 19 Future studies using freshly prepared samples are warranted. Another limitation of this study is the small number of cases included. Due to the low prevalence of RMS in dogs, a large multi-institutional study will be needed to acquire larger sample sizes for future studies. This study also did not systematically evaluate MyoD1 and myogenin immunolabeling in other types of canine sarcomas for comparison with canine RMS. The study is retrospective in nature, which limits the availability of historical and follow-up data. Due to limited resources, TEM was not performed for all cases in this study; instead, select cases that would most benefit from TEM evaluation were submitted.
In conclusion, this study demonstrated that myogenin and MyoD1 are effective IHC markers to aid in the diagnosis of canine RMS. However, these results also highlight that myogenin and MyoD1 IHC is not to be relied upon as a sole method for diagnosing canine RMS. Instead, these antibodies should be included with desmin as part of a diagnostic IHC panel that complements standard histological evaluation of HE-stained sections. Importantly, not all RMS cases are desmin-positive, and RMS can be very challenging to diagnose even with a combination approach of histopathology, IHC and TEM. RMS can clinically present as similar to STS and may be misdiagnosed as such without the proposed IHC. Moving forward, continued improvement in distinguishing RMS cases from other canine STS will provide the necessary foundation to further characterize and determine the prognostic significance of the subtypes of canine RMS. Ultimately, this pathological information will allow for better characterization of the biological behavior and clinical outcomes of RMS, enabling veterinarians to provide more accurate prognostic information and treatment recommendations to owners, while also providing the groundwork to facilitate future comparative investigations of canine RMS.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_0300985820988146 - Evaluation of Myogenin and MyoD1 as Immunohistochemical Markers of Canine Rhabdomyosarcoma
Supplemental Material, sj-pdf-1-vet-10.1177_0300985820988146 for Evaluation of Myogenin and MyoD1 as Immunohistochemical Markers of Canine Rhabdomyosarcoma by Joanne L. Tuohy, Brittney J. Byer, Suzanne Royer, Charles Keller, Margaret A. Nagai-Singer, Daniel P. Regan and Bernard Seguin in Veterinary Pathology
Footnotes
Acknowledgements
The authors thank Dr Brendan Podell for his assistance with developing the MyoD1 and myogenin IHC staining assay.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: DPR is supported by National Institutes of Health (NIH) award numbers: K01OD022982 and L30 TR002126.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
