Abstract
A 1.5-year-old American quarter horse gelding (case 1) and an 11-month-old American quarter horse filly (case 2) were presented for acute onset pelvic lameness and lethargy. Case 1 had nasal discharge, while case 2 developed rapid muscle atrophy. Both horses had elevated serum creatine kinase activity. The horses showed similar polyphasic histiocytic and lymphoplasmacytic myositis with necrosis, mineralization, and regeneration. Additionally, case 1 had Streptococcus equi subsp. equi-induced suppurative retropharyngeal lymphadenitis with renal purpura hemorrhagica and myoglobinuric nephropathy. A focal pulmonary abscess caused by Actinobacillus equuli was found in case 2. Genetic testing revealed case 1 as heterozygous and case 2 as homozygous for the E321G MYH1 variant, supporting the diagnosis of myosin heavy-chain myopathy, with concomitant bacterial disease as potential triggers.
Myosin heavy-chain myopathy (MYHM) is an autosomal codominant muscle disease with variable penetrance that affects American quarter horses (AQH) and related breeds, such as Appaloosa and American paint horse.5,16,17,20 It is associated with a mutation in the myosin heavy chain 1 (MYH1) gene, 5 and it can manifest into 3 clinical syndromes: immune-mediated myositis (IMM), nonexertional rhabdomyolysis (non-ER), and systemic calcinosis.14,16 Affected animals are usually younger than 8 years old or older than 17 years old,5,10,18 and have a concomitant trigger, such as vaccination or infectious respiratory disease, in approximately 50% of the cases.7,10,14,20 However, cases where a trigger was not identified are also described. 1 We report 2 cases of MYHM in quarter horses following concurrent bacterial disease.
Case 1 was a 1.5-year-old AQH gelding that developed lethargy, hind limb lameness, and bilateral cool, pitting swelling distal to the tibiotarsal joint 11 days prior to presentation to the Texas A&M Veterinary Medical Teaching Hospital. Four days after the clinical signs first presented, the horse had a large amount of yellow nasal discharge. The gelding mildly improved after treatment but 4 days later developed red-tinged urine. Endoscopy revealed bilateral abscessation of the retropharyngeal lymph nodes, suggestive of streptococcal lymphadenitis (strangles). Serum creatine kinase (CK) and aspartate aminotransferase (AST) activities were increased (60,291 U/L and 8825 U/L, respectively). The calcium-phosphorus product was 79.86. Other findings included neutrophilia and azotemia, with increased creatinine (5.34 mg/dL) and increased blood urea nitrogen (41 mg/dL). Despite aggressive fluid therapy, the patient progressed into renal failure, and euthanasia was elected.
At necropsy, case 1 had locally extensive, well-demarcated, pale, and dry areas in the skeletal muscle (Fig. 1a), with multifocal areas of pinpoint, white, gritty deposits consistent with mineralization. The muscles primarily affected were in the hind limbs. Mineralization was noted in the left m. rectus femoris and in the left m. vastus medialis. Histologically, the skeletal muscles had multifocal to coalescent myofiber degeneration and necrosis with mineralization, satellite cell hyperplasia, regeneration, and lymphoplasmacytic and histiocytic myositis (Fig. 1b, c). The blood vessels in the muscle were unremarkable.
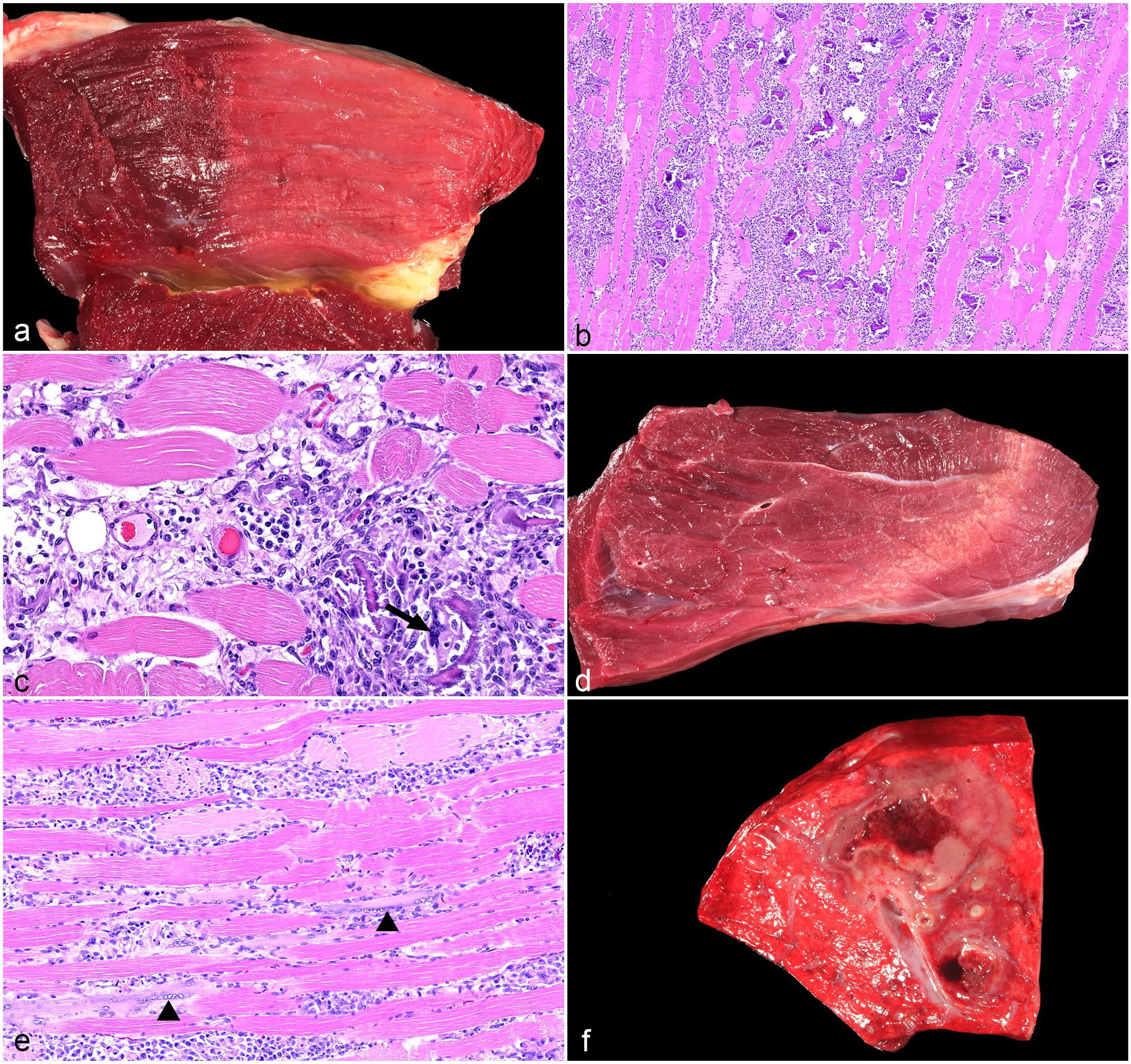
Pathological findings of myosin heavy-chain myopathy in 2 quarter horses. (a-c) Case 1, 1.5-year-old gelding, skeletal muscle. (a) The skeletal muscle has pale areas of muscle necrosis. (b) The skeletal muscle is fragmented and necrotic with mineralization. The perimysium and endomysium are infiltrated by numerous inflammatory cells. Hematoxylin and eosin (HE). (c) Myositis. Lymphocytes and plasma cells surround blood vessels, and macrophages are centered around necrotic myofibers. Rarely, multinucleated giant cells phagocytize minerals (arrow). Note the absence of vascular lesions in the skeletal muscle. HE. (d-f) Case 2, 11-month-old filly, skeletal muscle and lung. (d) Skeletal muscle. Affected muscles have multifocal pale areas with pinpoint mineralization. (e) Skeletal muscle. The myofibers are multifocally swollen, hypereosinophilic, and fragmented (muscle necrosis) with frequent rowing of satellite cells, indicating regeneration (arrowhead). HE. (f) Lung. Cut section of a pulmonary abscess, which cultured Actinobacillus equuli.
The retropharyngeal lymph nodes were bilaterally enlarged and filled with pus. Bacterial culture of the lymph node yielded a 1+ growth of Streptococcus equi subsp. equi, confirming the clinical diagnosis of strangles. Additionally, the kidneys of the gelding were surrounded by edema and diffusely dark red with numerous pinpoint pale foci in the cortex and cut surface (Supplemental Figure S1a). Histologically, the kidneys had acute, necrotizing glomerulonephritis with vascular fibrinoid necrosis, tubular necrosis, intratubular hemorrhage, and myoglobin casts (Supplemental Figure S1b and 1c). Intratubular myoglobin was confirmed by immunohistochemistry (Supplemental Table S1; inset Supplemental Fig. S1c). The renal lesions were consistent with purpura hemorrhagica associated with strangles and pigmentary nephropathy secondary to myositis and muscle degeneration.
Case 2 was an 11-month-old AQH filly that had a 5-day clinical course of lethargy and rapidly progressive bilateral symmetrical muscle atrophy affecting the dorsum and hind limb muscles (Supplemental Fig. S1d). The rectal temperature was 102.3 ºF. Thoracic auscultation revealed bilaterally increased bronchovesicular signs, being more prominent on the left side. Ultrasound showed numerous broad and narrow-based comet tails in the left mid-thorax to ventral field. The right side had a few narrow-based comet tails. These findings were suggestive of pneumonia. The filly had increased serum CK (49,275 U/L), AST (14,450 U/L), elevated blood urea nitrogen (34 mg/dL), and hypoalbuminemia (2.3 g/dL). Calcium-phosphorus product was 71.19. Muscle wasting persisted, and euthanasia was elected.
At necropsy, the gross findings in the skeletal muscle were similar to case 1 and were comprised of well-demarcated, pale, and dry areas with multifocal areas of mineralization (Fig. 1d). The hind and forelimb, pectoral, and thoracic muscles were affected bilaterally, particularly the mm. pectoralis ascendens, latissimus dorsi, obliquus externus abdominis, and, to a lesser extent, the mm. biceps femoris, semitendinosus, gluteus, quadriceps femoris, triceps brachii, and deltoideus. The histological findings in the skeletal muscle were similar to those in case 1, consisting of multifocal myofiber degeneration and necrosis with satellite cell hyperplasia, regeneration, mineralization, and lymphoplasmacytic and histiocytic myositis (Fig. 1e). However, muscle mineralization and inflammation were less prominent in case 2 compared to case 1. The blood vessels were unremarkable in all examined organs in case 2.
A 7.5 x 4 x 3 cm abscess filled with brown, opaque, malodorous material was identified in the left cranial lung lobe of case 2 (Fig. 1f). Histologically, numerous gram-negative bacilli were seen within the abscess. Bacterial culture of the pulmonary abscess revealed a 2+ growth of Actinobacillus equuli.
Immunohistochemistry for CD3 (T lymphocytes), CD20 (B lymphocytes), IBA-1 (macrophages), and MUM-1 (plasma cells) were performed on sections of the affected muscle (Supplemental Table S1). In both cases, macrophages and CD3+ T lymphocytes were the main inflammatory population, comprising approximately 80% and 15% of the inflammatory cell population, respectively, while plasma cells and CD20+ B lymphocytes were approximately 10% and 5% of the inflammatory population, respectively. CD3+ T lymphocytes, scattered CD20+ B lymphocytes, and plasma cells multifocally infiltrated the perivascular spaces, endomysium, and perimysium. Histiocytes were concentrated around necrotic myofibers, particularly in case 1, where mineralization was more prominent. Fewer histiocytes were seen scattered in the endomysium, perimysium, and around blood vessels (Fig. 2a–h).
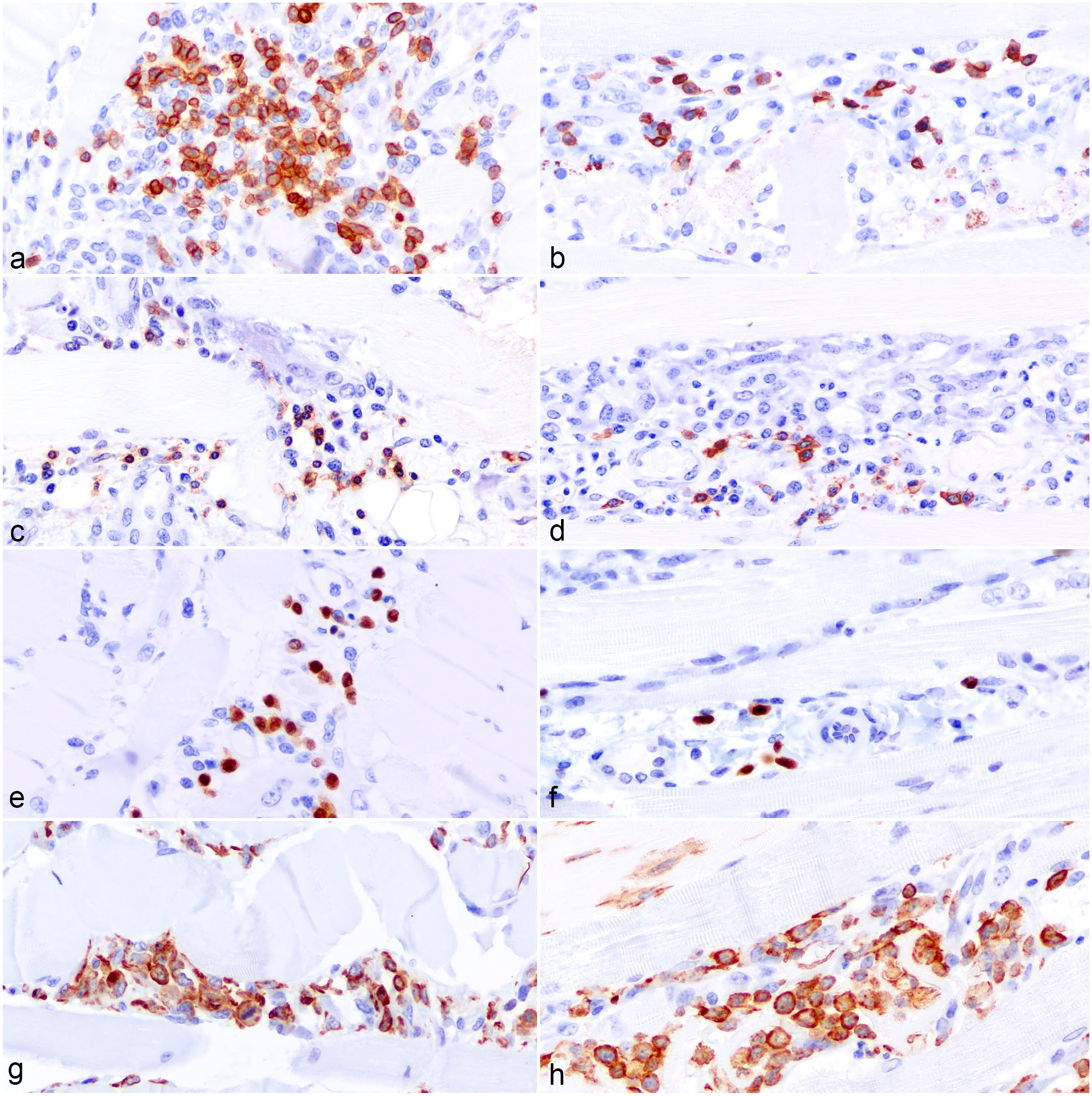
Immunohistochemical findings of myosin heavy-chain myopathy in the skeletal muscle of 2 quarter horses. (a) Case 1, 1.5-year-old gelding. Moderate numbers of CD3+ T lymphocytes surround blood vessels and infiltrate the perimysium and endomysium. Immunohistochemistry (IHC) for CD3. (b) Case 2, 11-month-old filly. Few CD3+ T lymphocytes infiltrate the perimysium and endomysium. IHC for CD3. (c) Case 1. Few CD20+ B lymphocytes infiltrate the perimysium and endomysium. IHC for CD20. (d) Case 2. Few CD20+ B lymphocytes are around blood vessels. IHC for CD20. (e) Case 1. Scattered plasma cells are around blood vessels and in the perimysium and endomysium. IHC for MUM-1. (f) Case 2. Rare plasma cells are seen around blood vessels. IHC for MUM-1. (g) Case 1. Numerous histiocytes infiltrate around fragmented myocytes and areas of mineralization. IHC for IBA-1. (h) Case 2. Numerous histiocytes infiltrate the perimysium and endomysium around areas of muscle necrosis. IHC for IBA-1.
In both cases, plucked hair containing hair bulbs from the tail was submitted for the quarter horse and related breeds disease panel (5-panel plus) at the Veterinary Genetics Laboratory at the University of California, Davis. The genetic testing was performed postmortem in case 1 and antemortem in case 2. Case 1 was heterozygous and case 2 was homozygous for the E321G MYH1 variant. Results for glycogen branching enzyme deficiency, hereditary equine regional dermal asthenia, hyperkalemic periodic paralysis, malignant hyperthermia, and polysaccharide storage myopathy type 1 were negative in both cases. Based on the pathological and genetic results, we concluded that both horses had MYHM, associated with infection by S. equi subsp. equi (strangles) in case 1 and A. equuli in case 2.
Myosin is composed of 2 heavy chains and 4 light chains, occurring in different isoforms. In horses, 3 heavy-chain isoforms are expressed: type 1, 2A, and 2X. 13 MYHM results from a missense mutation affecting the MYH1 that encodes the myosin filaments’ heavy chains in type 2X myofibers. 5 The mutation responsible for MYHM results in a substitution of glutamic acid by glycine (E321G) in highly conserved helix loops of the myosin globular head of subfragment-1. These helix loops are located between the SWITCH1 and SWITCH 2 proteins that act on ATP binding. This substitution results in decreased contact between different domains of the myosin type 2X globular head and significantly decreased protein stability. 5 The mutation does not affect the relaxed conformations of myosin filaments and its ATP consumption. However, the mutated fibers have increased force production and higher sensitivity to calcium, indicating the affected myofibers have a hyper-contractile phenotype, which could contribute to the development of MYHM. 11 Approximately 6% to 14% of quarter horses are heterozygous for the mutation,1,6 mainly in reining, working cow, and halter horses in the American quarter horse population, 6 and reining and barrel racing horses in the Brazilian quarter horse population. 1 The increased prevalence in high-performance animals is likely due to common ancestors derived from limited genetic lines. 6
Type 2X fibers in AQH are located primarily in the mm. psoas major, iliocostalis, longissimus dorsi, and within the hind limbs, particularly mm. gluteus, semimembranosus, semitendinosus, and biceps femoris.5,8,19 MYHM manifests primarily in the gluteal and epaxial muscles, while the semitendinous and semimembranous muscles tend to be unaffected. 3 In case 1, the inflammation was primarily located within the hind limb muscles. In case 2, the epaxial, pectoral, abdominal, and forelimb muscles were most affected. The reason for localized atrophy is still unknown. 3
The MYH1 mutation is associated with 3 distinct clinical presentations: IMM, 5 non-ER, 18 and systemic calcinosis,14,16 described under the umbrella terminology of MYHM. IMM was first described when 37 biopsy cases from primarily quarter horse bloodlines were diagnosed with lymphohistiocytic myositis, predominantly with CD4+ and CD8+ T lymphocytes, affecting the epaxial and gluteal muscles. 10 The affected horses had rapid muscle atrophy and stiffness with high serum CK and AST activities. 10 Non-ER is associated with the same MYH1 mutation, with 67% of non-ER animals homozygous (75%) or heterozygous (25%) for the mutation. 18 The primary sign of non-ER is muscle stiffness, 20 and differs from IMM by the absence of rapid-onset muscle atrophy. 18 However, approximately 35% of affected horses subsequently develop acute muscle atrophy similar to IMM 16 and the mutation increases the risk for muscle atrophy 12-fold. 18 Recently, systemic calcinosis has been described in a 9-year-old AQH gelding homozygous for MYH1 mutation. 14 This is a rare fatal syndrome characterized by disseminated heterotopic mineralization affecting the skeletal muscle, lungs, kidneys, heart, and blood vessels.4,14 –16 It initially presents similar to IMM, and as the calcification progresses, other clinical signs related to tissue calcification may surface. 14 In both case 1 and case 2, mineralization was restricted to the skeletal myofibers and did not involve blood vessels in the muscle or any other body system.
The pathogenesis of IMM is not fully understood. Lymphocytic infiltrates are found in type 2X myofibers. Affected myofibers express major histocompatibility complexes I and II, which are not normally expressed in mature skeletal muscle, suggesting that myofibers can act as antigen-presenting cells. 2 Other mechanisms include loss of self-tolerance and activation of self-reactive T lymphocytes, such as epitope mimicry; release of superantigens; and massive cytokine release, leading to proteolysis and muscle degradation. 3 Up to 47% of cases have a concomitant triggering factor, usually an infectious disease such as S. equi subsp. equi, S. equi subsp. zooepidemicus, C. pseudotuberculosis, Anaplasma phagocytophylum, equine influenza virus, or equine herpesvirus-1. Vaccines against strangles, influenza, and herpesvirus-1 have also been associated with MYHM.7,16,20 Findings include fever and increased CK and AST. 18 Fever was reported only in case 2; however, both horses had elevation of serum CK and AST. Even though elevated serum CK and AST activities have also been reported with Streptococcus equi-associated purpura hemorrhagica, 9 there were no compatible gross and histological lesions of purpura hemorrhagica in the muscle of either cases. Case 1 had concomitant strangles caused by S. equi subsp. equi, while case 2 had a pulmonary abscess caused by A. equuli.
Severe muscle atrophy tends to be worse in homozygous patients, as does a higher recurrence rate of atrophy and lower recovery rate, 20 as observed in case 2. Even though case 2 had muscle necrosis and fragmentation, there was no gross or histological evidence of myoglobinuria. Conversely, case 1 did not show signs of muscle atrophy but presented with myoglobinuria, which is common with MYHM non-ER. 16
Myositis was observed in both cases. The pattern of inflammatory infiltration with scattered CD3+ T lymphocytes, CD20+ B lymphocytes, and plasma cells in the perimysium and endomysium, along with macrophages mostly located around necrotic myofibers, is consistent with previously described cases of IMM.2,3,7 Homozygous horses also have significantly greater lymphohistiocytic muscle infiltration when compared to non-ER horses without the mutation. 18
Case 1 also had histological renal lesions of purpura hemorrhagica characterized by fibrinoid vascular necrosis and glomerulonephritis, which likely contributed to the renal failure. Vascular changes were restricted to the kidney and were not appreciated in the skeletal muscle. Purpura hemorrhagica is usually associated with S. equi subsp. equi infection, although other associated bacterial and viral agents have also been described. 12 It is hypothesized that S. equi infection or vaccination results in high production of S. equi M protein-IgA immune complexes, triggering a type III hypersensitivity reaction that causes vasculitis and complement activation around blood vessels, leading to leukocytoclastic vasculitis. 3 In case 1, the skeletal muscle changes were attributed to MYHM rather than purpura hemorrhagica due to the absence of vascular changes, hemorrhage, and infarction in the skeletal muscle.
This report describes 2 cases of MYHM in AQH, with a clinical presentation of nonexertional rhabdomyolysis (case 1) and acute onset of muscle atrophy (case 2). Lesions were characterized by extensive muscle degeneration, necrosis, and mineralization, with lymphoplasmacytic and histiocytic myositis and concomitant bacterial diseases. To the authors’ knowledge, A. equuli has not been previously associated as a triggering factor of MYHM. Genetic testing for the MYH1 mutation in quarter horse-related breeds is recommended in cases of acute onset muscle atrophy and/or rhabdomyolysis, particularly in those patients that present with fever, other signs of infection, or recent history of vaccination.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231204253 – Supplemental material for Myosin heavy-chain myopathy in 2 American quarter horses
Supplemental material, sj-pdf-1-vet-10.1177_03009858231204253 for Myosin heavy-chain myopathy in 2 American quarter horses by Mayane Faccin, Kirsten A. Landsgaard, Sarai M. Milliron, Alexis H. Jennings, M. Keith Chaffin, Paula R. Giaretta and Raquel R. Rech in Veterinary Pathology
Footnotes
Acknowledgements
We want to acknowledge the Histology Laboratories at Texas A&M University for performing the HE stains and myoglobin immunohistochemistry, the University of Georgia for performing CD3, CD20, MUM-1, and IBA-1 immunohistochemistry, the staff at the Gastrointestinal Laboratory at Texas A&M University for slide scanning, and the necropsy technicians at the Pathology Laboratory at Texas A&M University.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
