Abstract
Gross and histopathologic evaluation of skeletal muscle was performed in 229 equids (217 horses, 8 ponies, 3 donkeys, and 1 mule) 1 year of age or older undergoing postmortem examination at Oregon State University in a 2.5-year period. Animals were evaluated for grossly evident muscle lesions, and muscle samples were fixed in formalin, processed routinely, and stained with hematoxylin and eosin (HE) and periodic acid-Schiff (PAS) for glycogen. Muscle lesions were detected in 149 animals (65%). Chronic myopathic change (excessive fiber size variation and internal nuclei) was evaluated in horses without polysaccharide storage myopathy and was the most common finding (36 animals; 15.7%). Chronic myopathic change was more common in older animals. Generalized muscle atrophy was present in 30 animals (13.1%). Myonecrosis was attributed to endotoxic injury (11 animals; 4.8%), bone fracture (8 animals; 3.5%), bacterial infection (5 animals; 2.2%), muscle rupture (3 animals; 1.3%), selenium deficiency (2 animals; 0.9%), and exertional rhabdomyolysis (1 horse; 0.4%); cause was not determined in 9 animals (3.9%). Intramyofiber protozoa were detected in 19 horses and ponies (8.3%). Denervation atrophy was detected in 14 animals (6.1%). Neoplasia involving muscle occurred in 3 animals (1.3%), injection site reactions were detected in 4 animals (1.7%), and focal lymphocytic infiltrates were found in 6 animals (2.6%). Other findings were ring fibers (2 horses; 0.9%), fiber splitting (2 horses; 0.9%), and fat infiltration (1 horse; 0.4%). Skeletal muscle lesions are common in equids examined at postmortem. Transverse sections stained with HE and PAS are invaluable when evaluating equine muscle.
Introduction
Disorders affecting skeletal muscle and peripheral nerves are common in horses. 9,17,28 Myopathic disorders can adversely affect athletic ability. In a previous study, underlying myopathy was identified in 18% of horses evaluated for poor performance. 28 Severe degenerative myopathy in equids can be fatal. 17,19,25,31,32,35 Causes of muscular disorders in adult horses include nutritional myopathy, 5,17,31 infectious disease, 10,17,32,35,38 immune-mediated disorders, 17,25,26 toxic myopathy, 19,36 inherited myopathies, 5,29 endocrinopathy, 2 pasture-associated rhabdomyolysis, 15,44,45 malignant hyperthermia, 1 and exertional rhabdomyolysis. 4,6,23,39 Disorders affecting the peripheral nerves resulting in denervation atrophy of muscle also occur. 8,13,14,34,41
Most survey studies of equine neuromuscular disease are of clinical cases. 9,17 A slaughterhouse study of equine muscle reported only on the incidence of intramuscular protozoa. 18 The prevalence of polysaccharide storage myopathy in the horses and ponies in the population in the current study has been previously reported, 40 and findings of abnormal polysaccharide storage, increase in fiber size variation, and increase in internal nuclei associated with this myopathy are not included in this current study. All other findings in muscle from horses with polysaccharide storage myopathy are included in this current study. Two horses from the present study's population were included in a report of 3 horses with suspect endotoxin-related myonecrosis. 43 The purpose of the current study was to determine the prevalence and nature of skeletal muscle lesions in horses, donkeys, and mules presented for postmortem examination.
Materials and methods
Examination of muscle and of medical records was performed in 229 equids (217 horses, 8 ponies, 3 donkeys, and 1 mule) 1 year of age or older undergoing postmortem examination at the Veterinary Diagnostic Laboratory at Oregon State University (Corvallis, OR) over a 2.5-year period. Breeds examined were Quarter horse (68), Arabian (34), Paint (27), Thoroughbred (22), Appaloosa (16), Warmblood (14), Morgan (11), draft (7), Tennessee Walking Horse (6), Pony of the Americas (4), other horse (12), and other pony (4). Ages ranged from 1 to 30 years.
Examination of muscle included subjective evaluation of overall muscle mass, histologic examination of muscle with grossly apparent lesions, and histologic examination of a semimembranosus or semitendinosus muscle sample obtained from all cases. Bacterial culture was performed when indicated. Postmortem examination was performed by one of 4 board-certified veterinary pathologists, and histologic evaluation of muscle samples from all cases was performed by the author. Muscle samples were placed on portions of wooden tongue depressors, fixed in formalin, processed routinely to provide transverse and longitudinal sections, and stained with hematoxylin and eosin (HE) and periodic acid-Schiff (PAS) for glycogen as previously described. 40 Hematoxylin and eosin-stained sections were evaluated for necrotic myofibers, inflammation, fibrosis, and fat infiltration. Periodic acid-Schiff-stained transverse sections were evaluated for ring fibers. Periodic acid-Schiff-stained transverse sections with amylase digestion were evaluated for overall fiber diameter, angular atrophy, and for chronic myopathic change, specifically increase in fiber size variation and presence of internal nuclei. Fiber size variation was determined by subjective evaluation. Internal nuclei were defined as myonuclei present 1 nuclear diameter or more from the sarcolemma.
The degree of fiber size variation and number of internal nuclei were scored from 0 (normal) to 3+ (severe) to determine a chronic myopathic score as previously described. 40 Specifically, fiber size variation was subjectively judged to be absent (0), mild (1+), moderate (2+), or severe (3+). Internal nuclei were judged to be absent (0), present but with an average of less than 1 per 10X microscopic field (1+), present with an average of 1–2 per 10X microscopic field or occurring as 2 or more within myofibers (2+), and present with an average of 3 or more per 10X microscopic field (3+). Horses and ponies with evidence of polysaccharide storage myopathy were excluded from this portion of the study. The age of horses with a combined chronic myopathic change score of 2 or greater was compared with the age of horses with chronic myopathic change scores of 0–1 using the Student's t-test assuming unequal variance.
Results
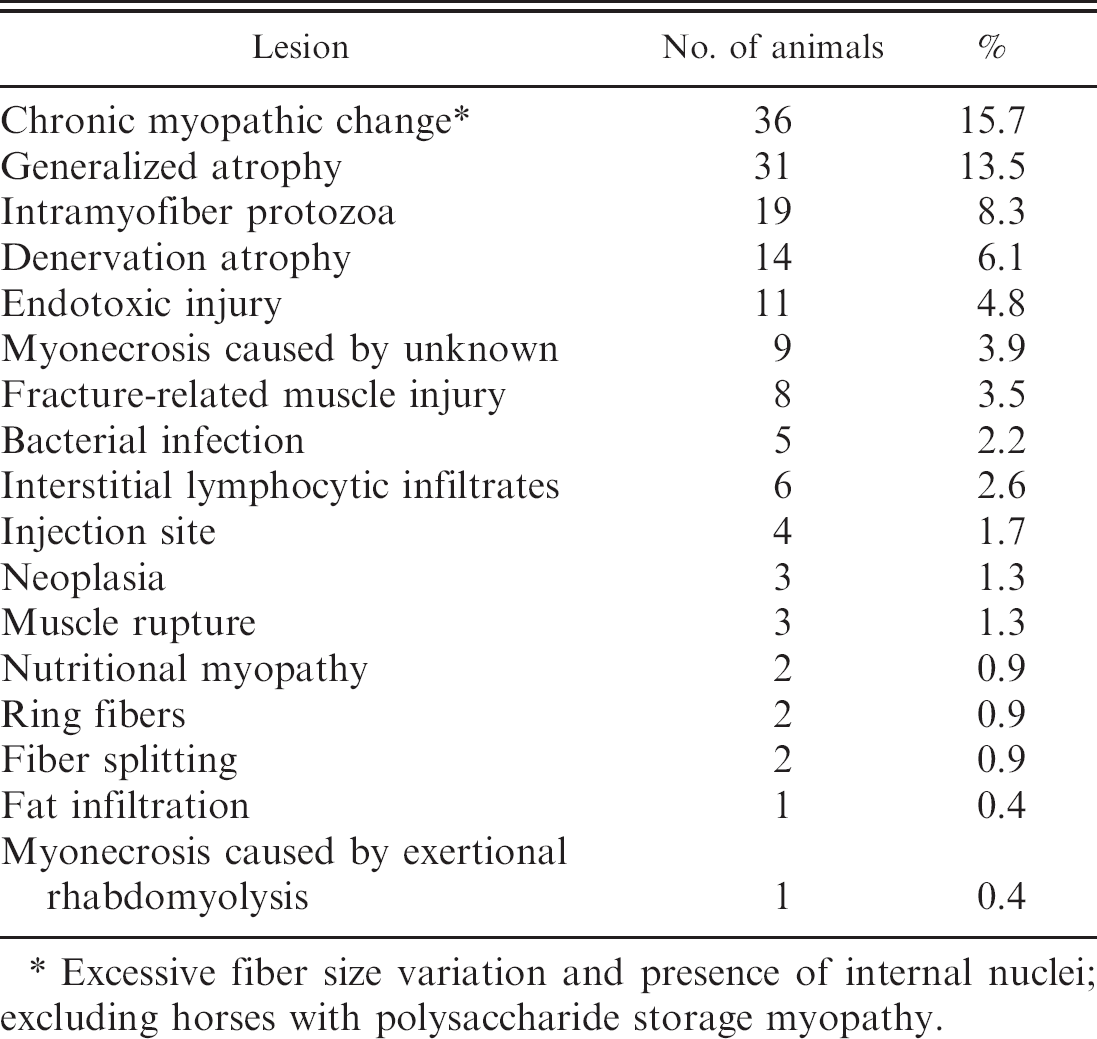
Muscle lesions of one or more type were detected in 149 of 229 animals (65%). Findings are summarized in Table 1. Normal semitendinosus and semimembranosus muscle (Fig. 1) exhibited 3 myofiber diameters consistent with type 1 fibers (smallest diameter, comprising approximately 10–20% of fibers), type 2A fibers (intermediate diameter, comprising approximately 30–40% of fibers), and type 2B fibers (largest diameter, comprising approximately 40–50% of fibers). 3,27 Myonuclei in normal muscle were located at the periphery of the fibers (Fig. 1) as has been previously reported. 3 Histologic evidence of increased fiber size variation or internal nuclei indicative of chronic myopathic change (Fig. 2) was the most common finding in the current study and occurred in 2 donkeys and the mule, as well as in 32 horses and 1 pony. Chronic myopathic change was associated with a wide variety of disorders, most of which were not determined to be primary neuromuscular diseases or to be related to the cause of death. The degree of chronic myopathic change in this study, in which chronic myopathic change associated with polysaccharide storage myopathy was not included, did not correlate with any specific diagnosis. Chronic myopathic change was difficult to detect with HE stain, but fiber size variation and presence of internal nuclei were readily detected in PAS-stained transverse sections with amylase digestion, counterstained with Harris modified hematoxylin. Other chronic myopathic changes detected were fat infiltration, fiber splitting, and cytoarchitectural alterations (ring fibers). Fat infiltration was rare, occurring in only 1 horse. A small number of myofibers with peripheral rims of maloriented myofilaments, characteristic of ring fibers (Fig. 3), was detected in 2 horses, 1 with polysaccharide storage myopathy and 1 with botulism. Ring fibers were best detected in PAS-stained sections. Fiber splitting, caused by focal ingrowth of the sarcolemma to create the appearance of more than 1 myofiber within a single basal lamina, was detected in 2 horses. Internal nuclei (3+) were the only muscle change detected in a horse with hyperkalemic periodic paralysis. The horse with botulism also had 1+ fiber size variation and 2+ internal nuclei, changes considered to be unrelated to the acute toxicosis. Mean age of equids with chronic myopathic change (15.1 years) was significantly greater than mean age of horses without chronic myopathic change (10.6 years; P = 0.003). However, 9 horses from 20 to 28 years of age had no evidence of chronic myopathic change.
Pathologic findings in skeletal muscle in 229 equids examined at necropsy.
Excessive fiber size variation and presence of internal nuclei; excluding horses with polysaccharide storage myopathy.

Normal equine semitendinosus/semimembranosus muscle from a 12-year-old Thoroughbred mare with volvulus of the large colon. Three fiber diameters are evident, consistent with type 1 fibers (smallest), type 2A fibers (intermediate), and type 2B fibers (largest). Periodic acid-Schiff stain with amylase digestion. Bar = 40 μm.

Chronic myopathic change in equine semitendinosus/semimembranosus muscle from an 18-year-old Arabian gelding with small intestine ischemic necrosis. There is a mild increase in fiber size variation, characterized by an increased range of fiber diameters, and many fibers contain 1–3 internal nuclei. Periodic acid-Schiff stain with amylase digestion. Bar = 40 μm.
Three myofibers with a peripheral rim of maloriented myofibrils characteristic of ring fibers (arrows) in semitendinosus/ semimembranosus muscle from a 14-year-old Quarter horse gelding with polysaccharide storage myopathy. Periodic acid-Schiff stain for glycogen. Bar = 40 μm.
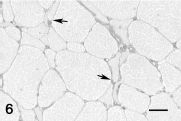
Generalized muscle atrophy was the second most common finding and occurred in 29 horses and 2 donkeys. The most common cause of generalized muscle atrophy was neoplasia (i.e., lymphoma, disseminated melanoma, disseminated carcinoma, and pituitary adenoma [6 horses]). Other causes of muscle atrophy were chronic gastrointestinal disease caused by endoparasites (4 horses), other gastrointestinal disorders (5 horses and 1 donkey), chronic lameness (5 horses), chronic bacterial infection (4 horses), spinal cord disease (2 horses), severe chronic renal disease (1 horse), severe chronic hepatic disease (1 horse), starvation (1 donkey), and atrophy caused by unknown (1 horse). Histologic findings in horses with generalized muscle atrophy were either overall decrease in myofiber diameter (Fig. 4) or increased fiber size variation. Angular atrophy of fibers was uncommon in horses with generalized muscle atrophy.
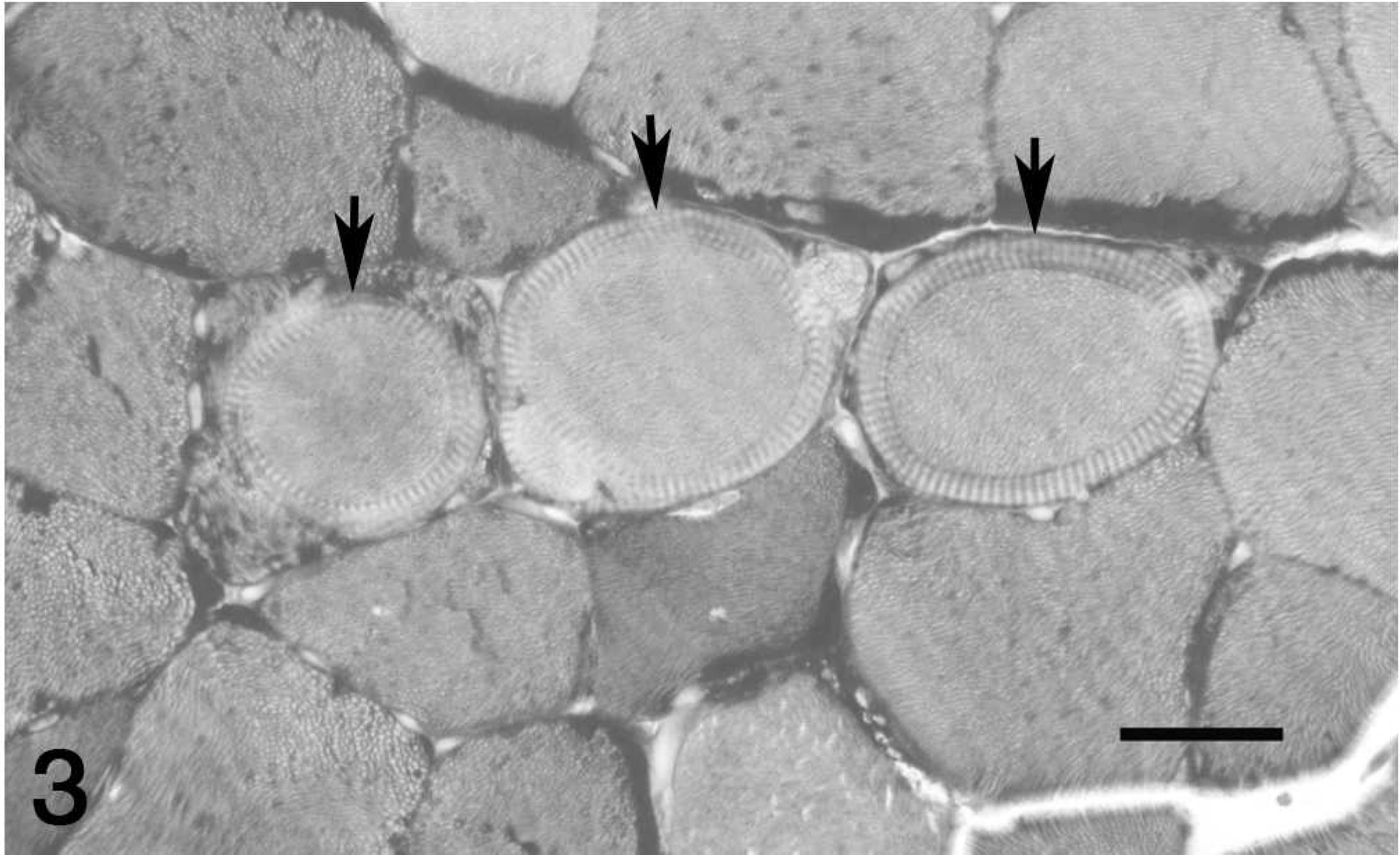
The presence of intramyofiber protozoal cysts consistent with Sarcocystis spp. was the third most common lesion, detected in 18 horses and 1 pony (Fig. 5). Protozoa were not detected in the 3 donkeys or in the mule. Affected myofibers were often enlarged, but protozoa were not associated with inflammation or myofiber necrosis in any case. Intramyofiber protozoa in the current study were considered an incidental finding in all cases.
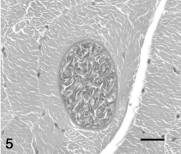
Denervation atrophy was the fourth most common finding, detected in 13 horses and 1 pony. Denervation atrophy was characterized by grossly evident asymmetric atrophy of laryngeal muscle (2 horses) or gluteal muscle (1 horse with equine protozoal myeloencephalitis), and by small group angular atrophy in microscopic sections (Fig. 6). In most cases, microscopic evidence of denervation atrophy was a localized lesion.
Active degenerative muscle lesions were detected in 33 horses and 1 donkey (14.8%). The most common degenerative lesion detected was scattered single-cell necrosis suggestive of endotoxic injury (11 horses; Fig. 7). 43 The finding was most often detected in semimembranosus or semitendinosus samples. Localized necrosis associated with bone fracture was present in 8 animals. Muscle rupture was present in 2 horses with acquired diaphragmatic hernias and 1 postpartum mare with a ruptured prepubic tendon. Myonecrosis associated with selenium deficiency myopathy was found in 1 horse and 1 donkey, and in 1 horse with renal failure secondary to exertional rhabdomyolysis. The cause of myonecrosis was not determined in 9 horses, but muscle damage was not considered to be related to the cause of death or euthanasia.
Bacterial myositis included infection due to Streptococcus zooepidemicus, Actinobacillus equuli, Fusobacterium spp., and Corynebacterium pseudotuberculosis, and occurred in 5 horses. Locally extensive zones of myofiber regeneration (characterized by small diameter, cytoplasmic basophilia, and large euchromatic nuclei), dissecting granulation tissue, or dissecting fibrosis consistent with damage caused by prior intramuscular injection were detected in 4 horses (Fig. 8). Interstitial and perivascular lymphocytic infiltrates were detected in 6 horses (Fig. 9). Inflammatory infiltrates surrounded normal myofibers as well as necrotic myofibers. Neoplasms involving muscle were melanoma (2 aged gray horses) and gastric squamous cell carcinoma with metastasis to the diaphragm (1 horse).
Neuromuscular disease was the cause of death or euthanasia in 14 horses and 1 donkey (6.7%). Three horses were euthanized as a result of peripheral neuropathy causing stringhalt, 3 horses with muscle rupture died or were euthanized, 2 horses with bacterial myositis were euthanized as a result of the infection, 1 horse and 1 donkey with nutritional myopathy died, 2 horses with peripheral neuropathy (1 with laryngeal paralysis and 1 with radial paralysis) were euthanized as a result of peripheral nerve disease, and 1 horse each died as a result of renal failure secondary to exertional rhabdomyolysis, hyperkalemic periodic paralysis, and botulism.
Discussion
Results of the current study indicate that muscle lesions are common in horses examined at necropsy. Chronic myopathic change was the most common lesion detected. Chronic myopathic change is a nonspecific finding associated with a variety of neuromuscular disorders. 12 In the horse, chronic myopathic change has been reported in association with equine motor neuron disease, 41,42 malignant hyperthermia, 1 equine polysaccharide storage myopathy, 16,40,42 recurrent exertional rhabdomyolysis, 6,39 and myotonic dystrophy-like myopathy. 21,33 It is possible that chronic myopathic change is also associated with increasing age in equids. Increased fiber size variation caused by angular atrophy of type 2 fibers occurs in dogs 7 years of age or older. 7 Excessive fiber size variation in the aged equids in the current study was not often associated with angular atrophy of fibers. Chronic myopathic change was more common in older animals, but the fact that 9 animals 20 years or older lacked chronic myopathic change suggests that age is not the only factor leading to this change in equine muscle. Three of the older horses with chronic myopathic change were diagnosed with pituitary adenoma. Excessive fiber size variation, as well as overall muscle atrophy, is associated with pars intermedia dysfunction resulting in type 2 selective myofiber atrophy. 2 It is possible that chronic myopathic change and generalized muscle atrophy detected in other older equids in the current study could have been the result of pituitary dysfunction, as the pituitary was not examined in all cases. Ring fibers are common in horses with myotonic dystrophy-like myopathy 21,33 but are also reported in horses with recurrent exertional rhabdomyolysis, 6 in horses with malignant hyperthermia, 1 and in horses with immune-mediated myositis. 26 Ring fibers in the current study were uncommon but were found in a horse with polysaccharide storage myopathy and in a horse with botulism, indicating that they are likely to be a nonspecific myopathic change in horses. Detection of chronic myopathic change requires sectioning to obtain transverse sections 12,40,42 and stains that emphasize fiber margins, nuclei and, in the case of ring fibers, sarcomeric arrangement. Many chronic myopathic changes are not readily detectable on routinely processed muscle sections stained with HE, but a combination of HE and PAS stain for glycogen provides appropriate sections for analysis. Morphometric analysis of fiber diameter and determination of atrophy and hypertrophy factors provide objective data related to fiber size variation 3,7,12 but are not practical tools for routine veterinary diagnostic pathology. Subjective evaluation, however, can provide valuable information.
Mild to severe degenerative myopathy was a common finding in the current study and was attributed to a variety of causes. Eleven horses with clinical histories and pathologic findings characteristic or suggestive of endotoxemia or septicemia were considered to have myodegeneration characteristic of endotoxic injury. Myodegeneration associated with endotoxemia/systemic inflammatory response syndrome is recognized in people, 37 pigs, 20 and sheep. 22 Evidence for a similar phenomenon has recently been reported in the horse. Characteristic histologic findings are scattered, single myofibers or small groups of myofibers undergoing lysis. 43 Endotoxic muscle injury should be suspected when scattered, acutely necrotic myofibers are detected in horses with confirmed or suspected endotoxemic/systemic inflammatory response syndrome in the absence of other factors that could predispose to skeletal muscle injury. Selenium deficiency myopathy, confirmed by liver selenium analysis, was the cause of myonecrosis and death in a 6-year-old horse and a 16-year-old donkey. A prior study of horses in the Pacific Northwest documented the occurrence of severe selenium deficiency myopathy causing trismus or dysphagia in horses from 6 months to 22 years of age. 31 Myofiber necrosis also occurs in horses with polysaccharide storage myopathy 16,42 and in horses with equine motor neuron disease. 41,42 Myofiber necrosis with no identifiable cause was detected histologically in 9 cases in the current study.

Overall reduction in myofiber diameter in semimembranosus/semitendinosus muscle from a 1-year-old Paint filly with cachexia caused by severe intestinal parasitism. Periodic acid-Schiff stain with amylase digestion. Bar = 40 μm.
An intramyofiber protozoal cyst consistent with Sarcocystis spp. in semitendinosus/semimembranosus muscle from a 19-year-old Arabian gelding with traumatic cervical spinal cord injury. Hematoxylin and eosin stain. Bar = 40 μm.
Severe angular atrophy of a small number of contiguous myofibers (arrows) characteristic of small group atrophy indicative of denervation atrophy in semitendinosus/semimembranosus muscle from a 22-year-old Standardbred mare with volvulus of the large colon. Periodic acid-Schiff stain with amylase digestion. Bar = 40 μm.
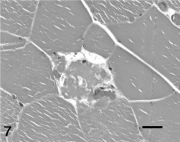
Single myofiber lysis consistent with endotoxic injury in semitendinosus/semimembranosus muscle from a 1-year-old Paint colt with gastric rupture. Hematoxylin and eosin stain. Bar = 40 μm.

Locally extensive dissecting granulation tissue consistent with injury caused by intramuscular injection in semitendinosus/semimembranosus muscle from an adult (age not specified) Thoroughbred mare with a rectal tear. Hematoxylin and eosin stain. Bar = 20 μm.
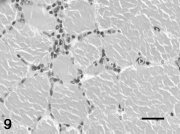
Interstitial lymphocytic infiltrates surrounding intact myofibers in semitendinosus/semimembranosus muscle from an 8-year-old Warmblood mare with volvulus of the large colon. Hematoxylin and eosin stain. Bar = 40 μm.
Denervation atrophy was diagnosed histologically, based on finding severe angular atrophy occurring in small contiguous groups (small group atrophy). Denervation atrophy is characterized by type 1 and type 2 myofiber atrophy. 12 Fiber typing was not performed in the current study, but small group atrophy is characteristic of denervation atrophy. 12,42 Denervation atrophy was not diagnosed in any of the horses with pituitary adenomas, and therefore it is not likely that the selective type 2 myofiber atrophy that occurs in horses with pituitary dysfunction 2 was mistaken for denervation atrophy. Interestingly, 2 of the cases with histopathologic evidence of denervation atrophy also had severe concurrent chronic hepatic disease. Laryngeal paralysis suggestive of peripheral neuropathy has been reported in ponies with severe chronic hepatic disease. 30 Further studies on the possible relationship between chronic hepatic disease and peripheral neuropathy in the horse are needed. Peripheral neuropathy leading to stringhalt was identified as a cause of denervation atrophy in 3 horses. In most cases, denervation atrophy was a localized lesion that was not related to clinical disease.
Other findings considered to be incidental were also common in the current study, including intramyofiber protozoa, fibrosis secondary to intramuscular injection, and focal to multifocal interstitial lymphocytic infiltrates. Protozoal cysts have been previously reported as an incidental finding in horses examined at slaughter. 18 The Sarcocystis spp. reported to occur in equine muscle are S. bertrami, S. equicanis, and S. fayeri. 24 Prior slaughterhouse studies within the United States indicate regional differences in prevalence of infection in horses, from 0.5% to 21%. 18 Sarcocystis fayeri is the only Sarcocystis spp. reported in equine muscle in the United States. 11 Speciation was not attempted in the current study. Although Sarcocystis spp. infection has been considered to be the cause of clinically apparent disease in horses, 17,38 the results of this and prior slaughterhouse studies indicate that careful evaluation for other causes of muscle disease are warranted when protozoa are detected in muscle samples from horses with suspect neuromuscular disease.
Semimembranosus and semitendinosus muscle samples proved to be very useful when screening for muscle lesions in horses. These muscles are common sites for intramuscular injection in the horse, and therefore it is not surprising that this type of injury was detected in 4 cases in the current study. Injury to intramuscular nerves following intramuscular injection might also explain the evidence of localized denervation atrophy detected as an apparently incidental finding in 8 horses in this study, but this association is purely speculative.
Interstitial and perivascular lymphocytic infiltrates, with or without associated myofiber injury, were detected in 6 horses and were considered an incidental finding. Similar infiltrates were described associated with a variety of neuromuscular disorders in a study of equine muscle biopsy samples. 26 Foci of interstitial lymphocytic infiltrates were also detected in normal canine muscle. 7 It is possible that interstitial infiltrates are associated with protozoal infection of equine muscle, but in the current study, only 2 cases with focal interstitial lymphocytic infiltrates also had detectable intramyofiber sarcocysts. The cause of these foci of interstitial and perivascular lymphocytic inflammation in equine muscle, especially those associated with intact myofibers, remains unknown.
Suspect immune-mediated disease resulting in interstitial and perivascular lymphocytic myositis occurs in horses, but is uncommon. 26 A diagnosis of myositis should not be made based on a small number of interstitial and perivascular lymphocytic infiltrates in equine muscle samples.
Results of the current study indicate that lesions within skeletal muscle are a common finding in horses undergoing postmortem examination. This is especially true given the high prevalence of abnormal polysaccharide storage characteristic of polysaccharide storage myopathy (101 horses and ponies in the present population). 40 Protozoal cysts, interstitial inflammation, and fibrosis are readily detected on HE-stained sections. Periodic acid-Schiff stain with amylase digestion on transverse sections will facilitate evaluation of fiber size variation, internal nuclei, small group atrophy, and ring fibers, as well as allow for detection of abnormal polysaccharide storage. Findings in the current study indicate that gross evaluation of muscle and sampling of muscle for histopathologic evaluation, and utilizing PAS stain for glycogen as well as HE stain, are important procedures when performing equine postmortem examinations.
Acknowledgements
The author thanks Dr. Jerry Heidel, Dr. Christiane Löhr, Dr. Rob Bildfell, Mehmet Kupel, and Jermey Becker for collection of samples, and Patricia DeVaughn for assistance with medical record data collection. This study was funded by a donation from Tom and Mindy Wolfe.
