Abstract
A neuronal storage disease affecting 5 captive Humboldt penguins is described. One bird died after 3 days of lethargy and anorexia. The 4 remaining birds died after a slowly progressing course of disease with signs that included lethargy, weakness, and neurologic dysfunction. Neurologic signs included dysphagia and ataxia. Gross lesions in the first animal to die consisted of hepatosplenomegaly indicative of avian malaria, which was confirmed histologically. The 4 remaining animals were mildly to moderately emaciated. Moderate to marked vacuolation of the neuronal perikarya was observed in Purkinje cells, neurons of the brainstem nuclei, and motorneurons of the spinal cord in all birds. By electron microscopy the vacuoles represented multilayered concentric lamellar structures. These findings were indicative of sphingolipidosis. All animals had been prophylactically treated for avian malaria, aspergillosis, and possible bacterial infections with chloroquine, itraconazole, and enrofloxacin. circumstantial evidence implicates chloroquine therapy as the possible cause of the storage disease.
Lysosomal storage diseases may be inherited or acquired and are due to enzymatic insufficiency. These diseases are classified based on the exact type of enzyme defect, biochemical composition of the accumulated material, or morphology. The substrate may accumulate primarily in neurons, or it may be more generalized. 8, 19 Any form of neuronal storage disease is uncommon in birds, but a hereditary mucopolysaccharidosis type III and an inherited gangliosidosis have been documented in emus. 2, 3, 17 In addition, a cerebral lipidosis was reported in 2 captive Hawaiian geese, but it was unclear whether this condition was hereditary or acquired. 20 Acquired lysosomal storage diseases generally are caused by the ingestion of alkaloid-containing plants or can be induced by the administration of certain cationic amphiphilic drugs, although there have been no reports of acquired lysosomal storage diseases in birds, with the possible exception of the above-mentioned case in the Hawaiian geese.
The 5 Humboldt penguins in this study were adult females (age range: 4–11 years) that were acquired by the zoo in 2001 from 2 different breeding programs. Three animals were received from one zoological garden (bird Nos. 1, 2, and 3); these birds had identical parents but were of different ages. Two birds received from another institution also were the offspring of one breeding pair and also were of different ages (bird Nos. 4 and 5). In addition, the dam of the 3 siblings and the sire of the 2 siblings had the same parents (Fig. 1). The birds were fed various fish species, including capelin and smelt, and were kept in an outdoor exhibit. The penguins were treated prophylactically with approximately 25 mg/kg chloroquine (100 mg per bird) once per day beginning June 1 for the entire mosquito season (June until the end of October or until they died) to prevent avian malaria. When the animals began exhibiting clinical signs, itroconazole (12 mg/kg twice per day) and enrofloxacin (16 mg/kg once per day) also were adminstered orally. The first bird (No. 5) died after 3 days of lethargy and anorexia on September 1. The second bird (No. 1) at the same time began exhibiting progressive weakness which continued for 4 weeks. In this bird neurologic signs began as slight ataxia and progressed to a marked ataxia. The animal had difficulty standing and supported itself using its wings. Nevertheless, it fell over with increasing frequency and had marked problems righting itself after the falls. It had difficulty holding its head up and grabbing with its beak any fish that was offered. Shortly before death the penguin was incapable of prehending food, although swallowing appeared to be functional when the fish was put deep into the throat. At about the time of death of the second penguin, the remaining 3 animals began a similar course of the disease and died 3 (bird Nos. 2 and 4) to 5 (bird No. 3) weeks later. Evaluation of blood smears of all animals failed to demonstrate intraerythocytic inclusions indicative of Plasmodium sp infection.
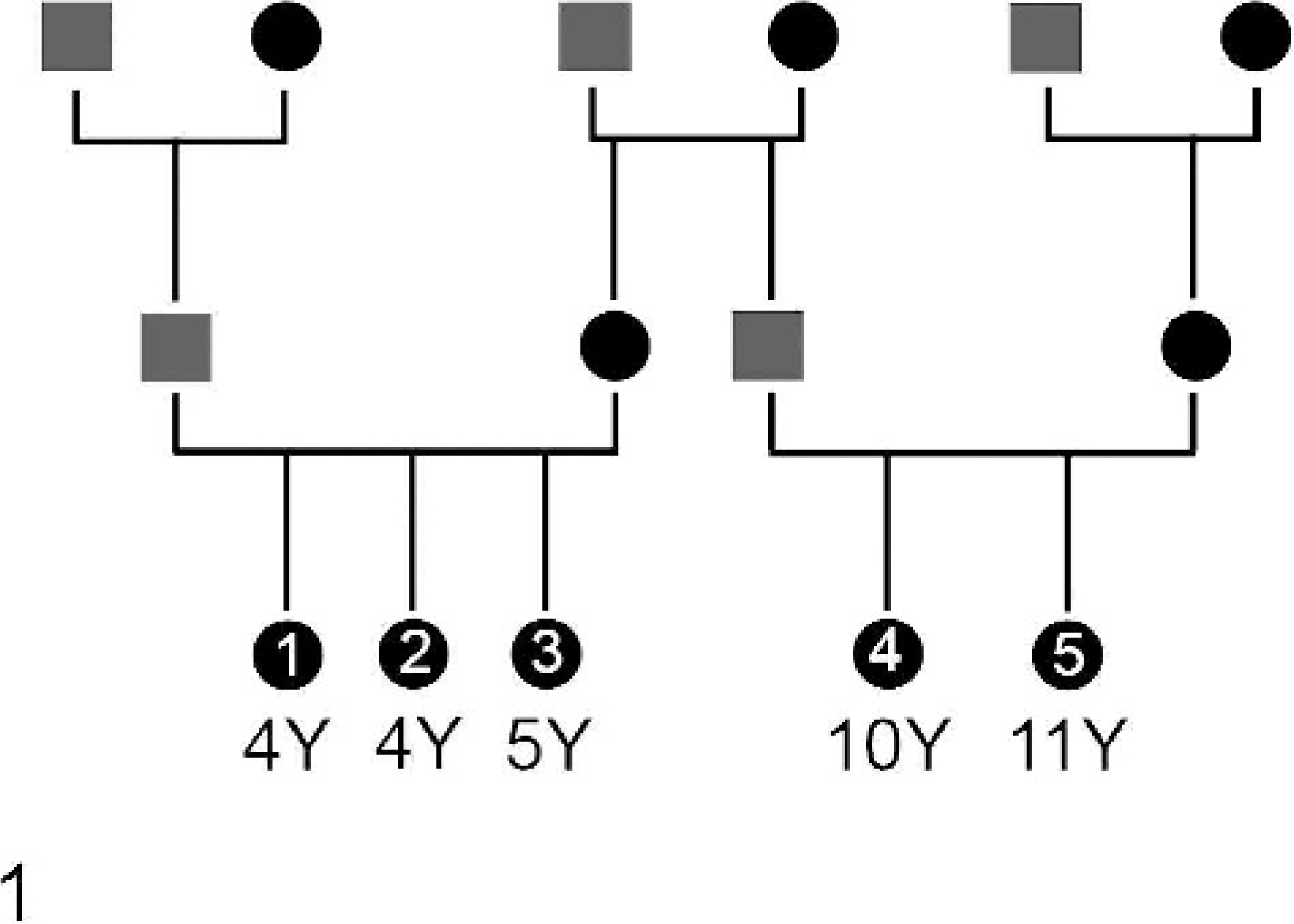
Pedigree and ages (in years) of the 5 penguins of the study. The bird numbers (Nos. 1–5) are indicated.
All animals were submitted for necropsy to the Minnesota Veterinary Diagnostic Laboratory 1 to 3 days postmortem. The first animal exhibited splenomegaly and hepatomegaly with a mildly friable liver. Serosal petechiae and approximately 10 ml of clear, watery, slightly reddish fluid were present in the cardioabdominal cavity. The intestinal content was slightly blood tinged. The lungs were markedly edematous. The remaining 4 penguins were moderately to markedly emaciated. In 2 of the penguins (bird Nos. 1 and 5) there were both white to yellow plaques and nodules approximately 2 to 5 mm in diameter in the serosal lining of the cardioabdominal cavity, as well as air sacs consistent with mild fungal airsacculitis and serositis.
Brain, spinal cord, spinal ganglia (only in bird No. 3), eyes, liver, kidney, spleen, femoral bone marrow, proventriculus, intestine, pancreas, skeletal muscle, heart, adrenal gland, thyroid gland, ovaries, and lungs were collected for histologic evaluation. In the spleen of the first penguin to die (bird No. 5) there were numerous macrophages containing protozoal organisms and black granular pigment. Similar organisms were seen in the liver and bone marrow, suggesting a diagnosis of avian malaria, which was considered to be the cause of death. Nonneurological lesions in the remaining penguins included a mild myofibrillar degeneration and necrosis in pectoral and axial skeletal muscles, characterized by rare hyaline degeneration of few individual myofibers.
All penguins exhibited similar histologic neuronal changes in the absence of other significant histologic findings of the nervous system. The perikarya of numerous neurons of the spinal ganglia, spinal cord (motorneurons), brainstem nuclei, and mesencephalon contained aggregates of numerous fine, clear vacuoles that were commonly evenly distributed (Fig. 2). The Nissl substance in affected neurons was either pushed to the periphery or missing. Occasionally, fine clear vacuoles distended the axon hillock and the proximal initial axon segment. A few swollen axons with a granular appearance were present in white matter tracts of the cerebellum, brainstem, and spinal cord (spheroids). The vacuoles in smaller neurons and small clusters of vacuoles in large neurons (e.g., brainstem ganglia and spinal motorneurons) were luxol fast-blue positive. The vacuoles stained only faintly with periodic acid schiff (PAS) and did not exhibit autofluorescence. The magnitude of neuronal vacuolar change appeared to be proportionate to the duration of clinical signs. Accordingly, vacuolar changes were most pronounced in the last animal to die (bird No. 3).
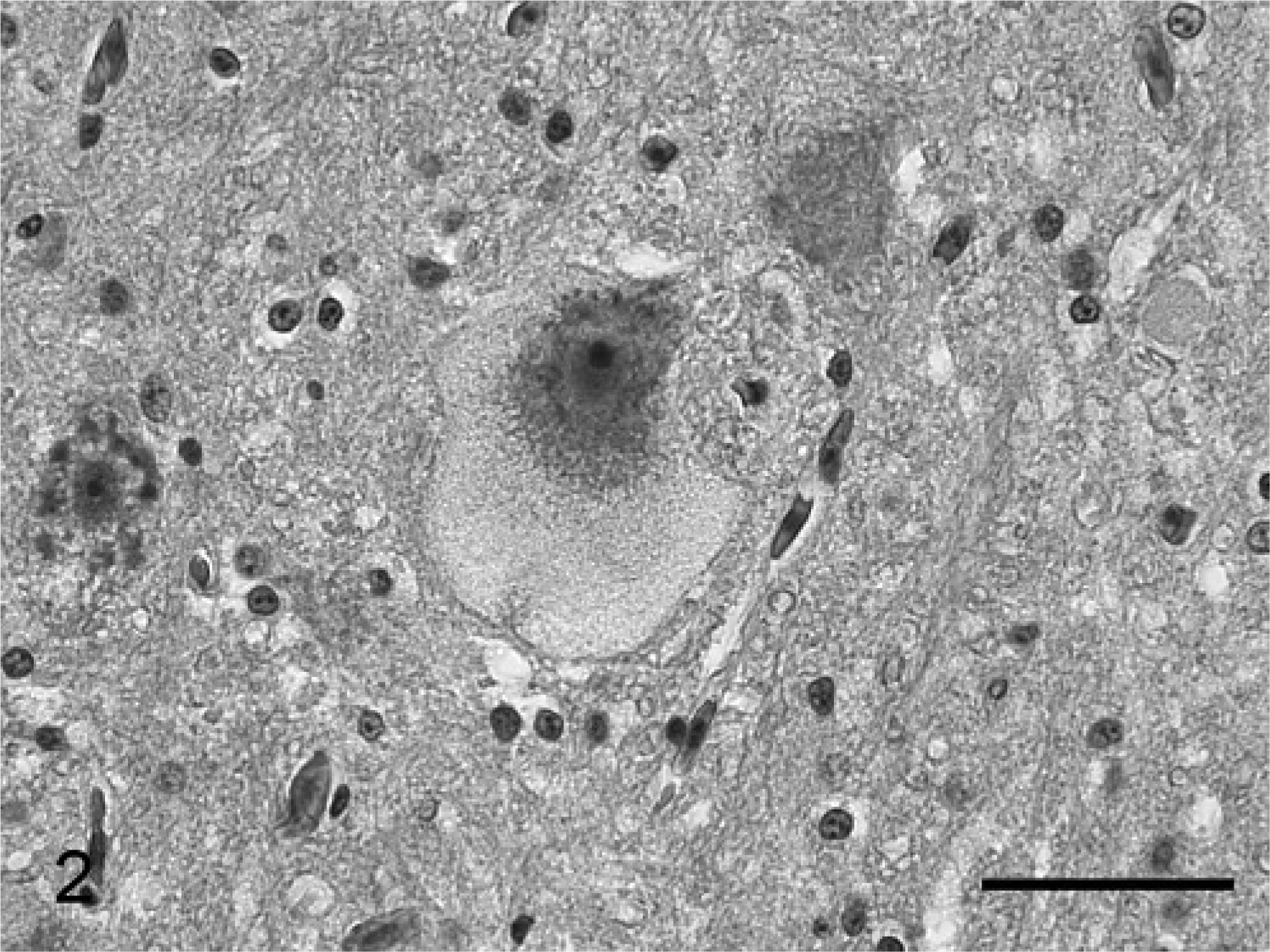
Brainstem; bird No. 3. Histologic section of a brainstem neuron with vacuolated neuronal perikaryon. HE stain. Bar = 100 μm.
Transmission electron microscopy (TEM) of the brainstem and spinal cord of 2 animals (bird Nos. 2 and 4) and the spinal cord and a spinal ganglion of 1 animal (bird No. 5) was performed using the formalin-fixed samples in order to characterize the neuronal vacuolar change. Tissues were postfixed in 3% glutaraldehyde, stained with osmium tetroxide, and embedded in Medcast resin. The ultrathin sections were examined using a Philips 300 transmission electron microscope as previously described. 15 TEM revealed that the perikarya of neurons were distended by fairly uniform vacuoles that contained whorled layers of alternating dense and pale material (multilayered concentric lamellar bodies; Fig. 3). Some lamellar bodies were partially enclosed by membraneous material, whereas other lamellar bodies did not have evidence of membraneous enclosures. In addition, occasional neurons contained vacuoles with loosely and haphazardly arranged membranous material. Also, few myelin bodies characterized by regular concentric arrangement of double-layered membranes were detected in the cytoplasm of neurons. Occasional axonal spheroids were identified in the spinal cord and cerebellum. These were characterized by segmental distention with accumulation of residual bodies. Multilayered concentric lamellar bodies were not detected in glial cells or endothelial cells in the examined specimen; neither were these structures present in hepatocytes or Kupffer cells of 2 animals (bird Nos. 2 and 3) nor the renal tubular epithelial cells of 1 animal (bird No. 2).
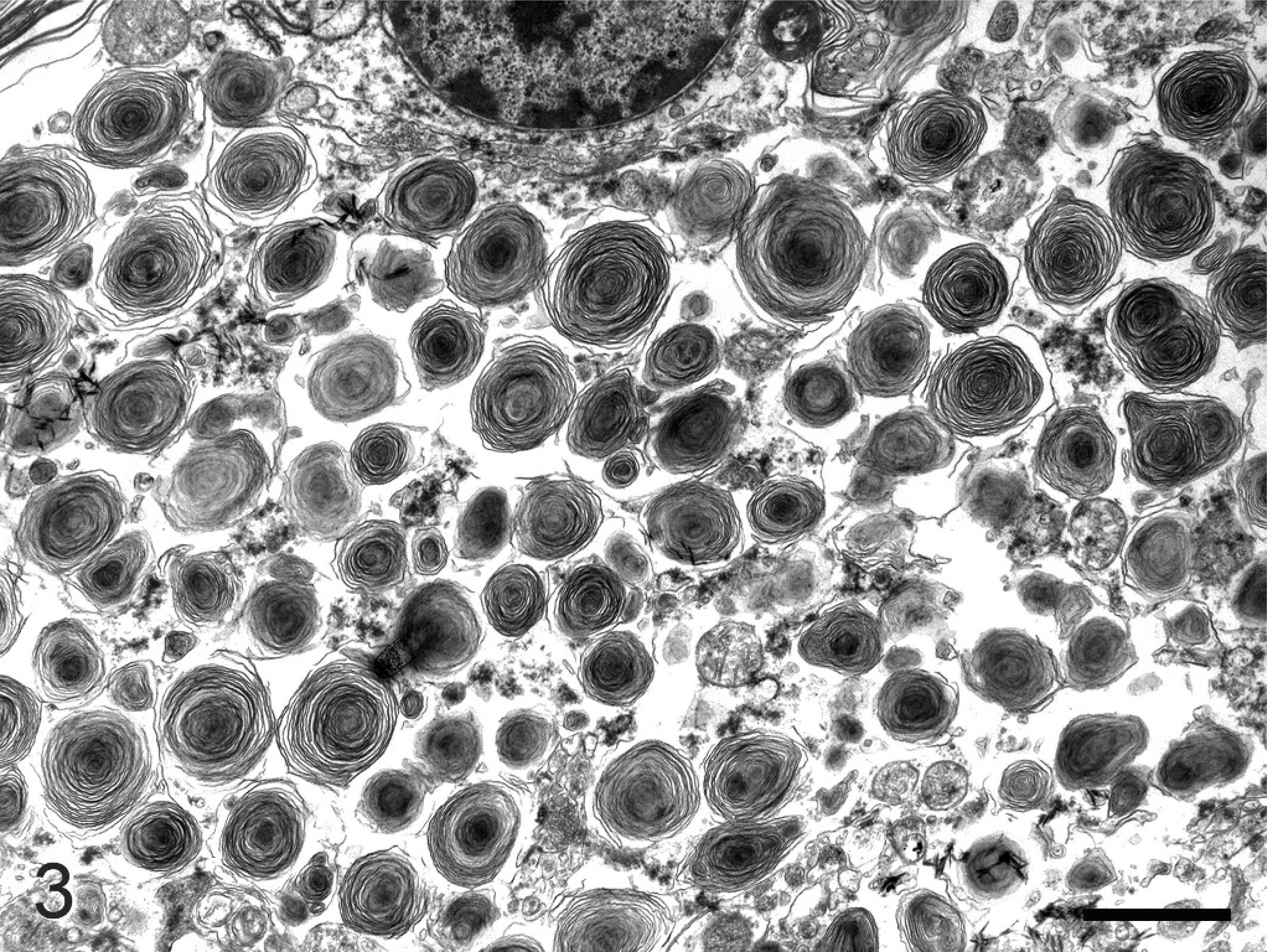
Spinal cord; bird No. 2. Electron micrograph of a spinal cord motorneuron with distended perikaryon due to the presence of numerous multilayered concentric lamellar structures. Bar = 300 nm.
The clinically slowly progressive course of the disease and the pathologic findings were consistent with a neuronal storage disease. The ultrastructural characteristics of the large majority of the vacuoles suggest that the stored material may be sphingolipid, and this material resembled material stored in cases of gangliosidosis. 8, 19 Based on the presence of membraneous enclosures, some of the vacuoles resembled secondary lysosomes, whereas other vacuoles may have been extralysosomal (e.g., residing free in the cytoplasm). The histologically detected accumulation of granular material in axons may be the result of an impaired axonal transport in neurons with massive somatic storage.
Sphingolipidoses are a group of lysosomal storage diseases that include gangliosidoses and sphingomyelinosis. 19 Unfortunately, unfixed brain tissues of the diseased animals and control animals were not available for biochemical analysis or analysis of the enzyme activities, which would have more precisely characterized the identity of the accumulated lipids. Although an inherited storage disease may be suspected since the animals were at least indirectly related to each other, the simultaneous disease expression in a population of animals of variable age is most consistent with an acquired disorder.
Chloroquine emerges as the probable cause of the neuronal storage disease based upon its ability to cause similar structural changes in neurons and the history in the present cases. Penguins are highly susceptible to aspergillosis and avian malaria, this being a particular problem in zoo collections. Therefore, in the Northern hemisphere penguins are treated prophylactically during the mosquito season (summer and fall months) when kept outdoors. Treatment protocols vary between the institutions (zoos and private collections) but occasionally include the application of chloroquine. Side effects of chronic chloroquine administration have been described in humans, in whom the immune modulatory effect of the drug is used to treat autoimmune diseases such as rheumatoid arthritis and lupus erthematosus. 14 These side effects include impaired neuromuscular function. 1, 11, 13, 14, 18 The pathophysiologic basis for these side effects is poorly characterized, although chloroquine has been shown to cause a neuronal storage disease (gangliosidosis) in miniature pigs and rats in experimental settings. 4, 9, 10 Chloroquine is a weak base and is known as a lysosomotropic amine. It may cause storage disease by increasing the intralysosomal pH and disintegrating the lysosomal membrane. 5– 7, 12, 14, 16
Chloroquine has been used previously in various institutions and in various species of penguins, including Humboldt penguins, without any reported problems. However, the cumulative dose used in the current cases (approximately 25 mg/kg per day for 3–4 months) appears to be higher than recommended. An informal inquiry at 8 other facilities that house penguins outdoors showed that prevention protocols for malaria are quite variable, although several institutions follow the guidelines of the Penguin Taxon Advisory Group (TAG): a combination of 125 mg sulfadiazine and 4 mg pyrimethamine per animal once every day or every 2 or 3 days for the duration of the mosquito season. The other surveyed institutions that used chloroquine administered the drug at lower doses, intermittently, or for shorter periods of time. Intoxication with another unknown lysosomotropic toxin was also considered to be a possible cause of the neuronal storage disease. Penguins are obligate fish eaters. The fish that were fed to the animals were acquired from a reliable source, and the same fish were fed to other fish-eating birds that did not develop any clinical signs. An inspection of the outdoor exhibit and holding area of the birds performed by three of the authors (AA, AW and MW) did not show evidence of toxic plants or toxic substances within the reach of the animals.
The present case raises concern that long-term administration of high doses of chloroquine may cause fatal neuronal storage disease in penguins. Further investigation is warranted to determine whether lower dosages result in lesser degrees of neuronal vacuolar change, as well as the degree to which these lesser changes may be functionally significant. Until then, chloroquine should be used judiciously, and animals should be closely monitored for side effects. It would also be desirable to standardize malaria prophylaxis protocols for penguins.
Footnotes
Acknowledgements
We thank Evelyn Handley (transmission electron microscopy section of the Department of Veterinary Biosciences; Ohio State University), Dr. Alexander DeLahunta (Department of Biomedical Sciences, Cornell University), H. L. Shivaprasad (California Veterinary Diagnostic Laboratory System, University of California, Davis), and Dr. Joseph Alroy (Department of Pathology, Tufts University Schools of Medicine and Veterinary Medicine and Tufts New-England Medical Center Boston) for their technical assistance and comments.
