Abstract
Rhabdoid meningioma is a rare type of meningeal neoplasm in humans. This study reports the clinical, pathological, and ultrastructural features of 4 cases of canine meningioma with rhabdoid features. The cases were female and 8 to 12 years of age. Biopsies from complete surgical resections were examined for all cases. The whole brain with tumor recurrence was collected at necropsy in 2 dogs. Histologically, the tumors consisted of discohesive sheets of oval-polygonal cells with abundant eosinophilic cytoplasm and occasional paranuclear hyaline-like inclusions. Cells were intensely immunopositive for vimentin, negative for melan A and S100 protein in all cases, and showed variable immunolabeling for cytokeratin in 2 cases. Focal glial fibrillary acidic protein (GFAP)-immunopositive cells were present in 1 case. Ultrastructurally, the rhabdoid cells in case 1 contained prominent cytoplasmic whorls of intermediate filaments, recapitulating the ultrastructural features of rhabdoid meningioma in humans. In cases 2 and 3, the meningioma cells contained interdigitating cell processes folded in a maze-like fashion resembling rhabdoid-like meningioma in humans. In case 4, the voluminous cytoplasm contained many round-to-flattened mitochondria admixed with rough endoplasmic reticulum, indicating a predominant oncocytic differentiation and not the rhabdoid differentiation suggested by light microscopy. Thus, rhabdoid morphology occurs in different types of meningiomas, and ultrastructural findings are essential for a correct diagnosis.
Meningioma is the most common extra-axial brain neoplasm in dogs, making up to 45% of primary canine brain tumors. 34 Affecting mainly middle-aged-to-older animals, spontaneous canine meningioma shares several similarities to the human counterpart and is currently being intensively investigated in the effort to establish the dog as a naturally occurring preclinical model for human meningioma.6,14
Reflecting their complex histogenesis and divergent phenotype, several histologic types of meningiomas are currently described in dogs, including meningotheliomatous, fibroblastic, transitional, psammomatous, angiomatous, microcystic, secretory, atypical, chordoid, clear cell, papillary, granular cell, and anaplastic.12,25,37 Several variants of meningioma displaying unusual focal features, such as papillary meningioma with secretory patterns 15 and a myxoid meningioma with rhabdoid features, 33 were described in dogs. Most of the histologic types of canine meningioma have similar prognosis, except for anaplastic and papillary meningioma that have biologically aggressive behavior.21,22
In humans, rhabdoid meningioma is an uncommon type of meningioma first included in the 2000 World Health Organization (WHO) classification of nervous system tumors and assigned as a WHO grade III malignancy.17,19,20 However, based on the fact that many of the rhabdoid meningiomas lack other histologic features of malignancy, the latest WHO classification of tumors of the central nervous system recommends a case-by-case histologic gradation of these meningiomas, independent of the rhabdoid features.31,39 Histologically, rhabdoid meningioma is characterized by loosely cohesive sheets of cells with distinctive rhabdoid morphology: large, plump cells with abundant eosinophilic cytoplasm, paranuclear hyaline inclusions, eccentric nuclei with open chromatin and prominent nucleoli, and polyphenotypic immunohistochemical profile.20,24,39 The typical ultrastructural feature of the rhabdoid meningioma used for the initial description, and currently the gold standard for defining a “true rhabdoid phenotype” of meningioma, is the presence of whorls of intermediate filaments (identified with vimentin immunopositivity) that often entrap cell organelles.5,24,27,28,39
Although the routine diagnosis of rhabdoid meningioma is based on classical histopathology, ultrastructural and immunohistochemical confirmation of the presence of rhabdoid differentiation was often necessary to differentiate it from several other tumors that exhibit rhabdoid morphology (eg, teratoid rhabdoid tumor in the brain, choroid plexus carcinoma, metastatic carcinoma, rhabdomyosarcoma, or melanoma).11,24 In 2014, a rhabdoid-like meningioma with virtually identical histology to rhabdoid meningioma was described in a human patient. 11 In this case, ultrastructurally, the cytoplasm was replaced by complex interdigitating cell processes rather than the paranuclear aggregates of intermediate filaments that define the “classical” forms of rhabdoid meningioma.
Herein, we present the clinical and pathological aspects of meningioma with rhabdoid morphology in 4 dogs. Describing the gross, histologic, immunohistochemical, and ultrastructural details, we show the key features, challenges, and limits in diagnosing this aggressive form of meningioma.
Material and Methods
Case Selection
The 4 meningioma cases with rhabdoid features were identified in the archive of the Comparative Pathology Shared Resource of the University of Minnesota, USA. From a total of 310 cases of brain tumors examined between 2008 and 2018, 73 cases of meningiomas were retrieved, histologic slides examined, and tumor cases with rhabdoid features were chosen for further analysis. Clinical data, magnetic resonance imaging (MRI) results, and follow-up outcomes were recorded for all 4 subjects included in the study.
The rhabdoid morphology of the meningiomas was defined by the diagnostic criteria reported by Perry and Brat
27
and Perry et al:
28
sheets of loosely cohesive cells with abundant eosinophilic cytoplasm, eccentric nuclei, and hyaline-to-fibrillar paranuclear inclusions, associated with the presence of meningothelial differentiation at the light microscopic, immunohistochemical, or ultrastructural level. Animals with meningiomas showing only focal rhabdoid characteristics (covering less than 50% of tumor surface) (
Gross Examination, Histopathology, and Immunohistochemistry
A complete necropsy was performed for cases 1, 2, and 4. Biopsies obtained by complete surgical resection of the tumor were examined in all 4 dogs. Whole brains with tumor recurrence were collected during necropsy and further examined histologically from cases 2 and 4.
For all 4 cases, samples were fixed in 10% neutral buffered formalin, routinely processed in paraffin wax, sectioned at 4 µm, and stained with hematoxylin and eosin, and periodic acid–Schiff (PAS). Independent of the presence of rhabdoid features, the tumors were graded as grade I/benign, grade II/atypical, and grade III/malignant according to the histologic criteria described in the WHO classification of tumors of the central nervous system of humans. 20 Meningioma grade was based only on the histologic criteria without considering the type. Histologic criteria for assigning a tumor as Grade II/atypical meningioma were an increased mitotic count (4–19 mitoses/10 HPF), presence of brain invasion, or meeting 3 or more of the following cytological features: hypercellularity, presence of small cells with high nuclear-to-cytoplasmic ratio, prominent nucleoli, sheeting (loss of whorling or fascicular histologic patterns) or foci of spontaneous necrosis. Grade III meningioma was diagnosed based on the presence of anaplastic (malignant) cytological features as markedly increased mitoses (≥20 mitoses/10 HPF) and overtly malignant cytologic appearance (resembling high-grade sarcoma, carcinoma, or melanoma). If the above-described histologic criteria of malignancy were absent, the tumor was classified as grade I meningioma.
All cases were immunolabeled for vimentin (Abcam, mouse monoclonal, 1:100), wide-spectrum cytokeratin (CK) (Dako, polyclonal rabbit, 1:4000), glial fibrillary acidic protein (GFAP) (Dako, rabbit polyclonal, 1:1000), melan A (Dako, monoclonal mouse, prediluted antibody), and S100 protein (Dako, rabbit polyclonal, 1:8000). The additional antibodies used in cases 1 and 4 are shown in Supplemental Materials.
Transmission Electron Microscopy (TEM)
For ultrastructural characterization, all 4 cases were evaluated by TEM at the University of Minnesota Veterinary Diagnostic Laboratory (VDL). Areas of interest were localized using the H&E-stained slide. For cases 1, 2, and 4, the tissues were formalin-fixed. For case 3, the samples were obtained from the paraffin-wax block. Six 1-mm 3 pieces of tumor tissue were dissected from each tumor. Samples were postfixed with 2.5% glutaraldehyde in 0.1 M sodium cacodylate buffer followed by a second postfixation with 1% osmium tetroxide in 0.1 M sodium cacodylate buffer. After 3 washes in distilled water, samples were dehydrated using a 25% to 100% ethyl alcohol gradient. Tissues were then infiltrated with 2:1 ethanol: EMbed 812 resin (Electron Microscopy Sciences) for 1 hour and subsequently transferred to a 1:2 ethanol: EMbed 812 resin mixture for 1 hour. Tissues were further infiltrated with 100% resin and embedded and incubated at 58°C for 24 hours to polymerize the resin. Embedded samples were trimmed and sectioned on an ultramicrotome (UC6, Leica Microsystems, Vienna, Austria). Thin sections (60–70 nm) were obtained and collected on 200 mesh copper grids (Electron Microscopy Sciences). Grids were contrasted with 5% uranyl acetate for 20 minutes and Santos’ lead citrate for 6 minutes. These samples were visualized using a transmission electron microscope (JEOL 1400, JEOL LTD, Tokyo, Japan). Images were obtained using an AMT Capture Engine Version 7.00 camera and software (Advanced Microscopy Techniques Corp, Woburn, MA, USA).
Results
Clinical History
All cases were female (1 intact, 3 spayed), with a mean age at the time of diagnosis of 10 years (ranging from 8 to 12), presenting with seizures (cases 1 and 3), acute blindness (case 2), and change in activity/playfulness (case 4) (Table 1). The cases included 2 Labrador Retrievers, 1 Chihuahua, and 1 Norwich Terrier. The seizures in all cases were well controlled with oral anti-epileptic drugs.
Signalment and clinical findings in dogs with rhabdoid meningioma.

Abbreviations: OS, overall survival time (days); FI, female, intact; FS, female, spayed.
Age at surgery (years).
By MRI, all cases had solitary masses involving the olfactory-frontal (cases 1 and 2) or frontal lobes (cases 3 and 4). The meningeal tumor volume ranged from 2.0 to 7.8 cm3 (mean volume 4.7 cm3). The initial diagnosis of rhabdoid meningioma was based on biopsy examination, and all cases underwent surgery for complete tumor resection. Two cases (cases 1 and 4) had immediate postsurgical MRIs showing gross total resection of the meningioma. All cases were discharged from the hospital. However, dog 1 died at home 3 days after surgery. The remaining 3 cases received adjuvant immunotherapy with a series of 6 autologous tumor lysate vaccinations. Dog 2 was euthanized 45 days after surgery due to aspiration pneumonia and, subsequently, multi-organ failure. The other 2 cases were euthanized 348 and 419 days after surgery due to uncontrollable seizure activity.
Gross and Microscopic Findings
During the necropsy, no residual neoplasm was grossly evident for case 1. Although there were cardiac and renal findings suggestive of chronic hypertension, and adenomas were found in the pituitary, left adrenal, and left parathyroid glands, no definitive cause of death was identified on necropsy. Infiltration of meningioma into the cribriform plate of the ethmoid bone was present in case 2 (Fig. 1), and recurrence of the meningioma was observed at postmortem in case 4 (Fig. 2). At necropsy, no other tumors were identified in any extraneural tissues for case 4.
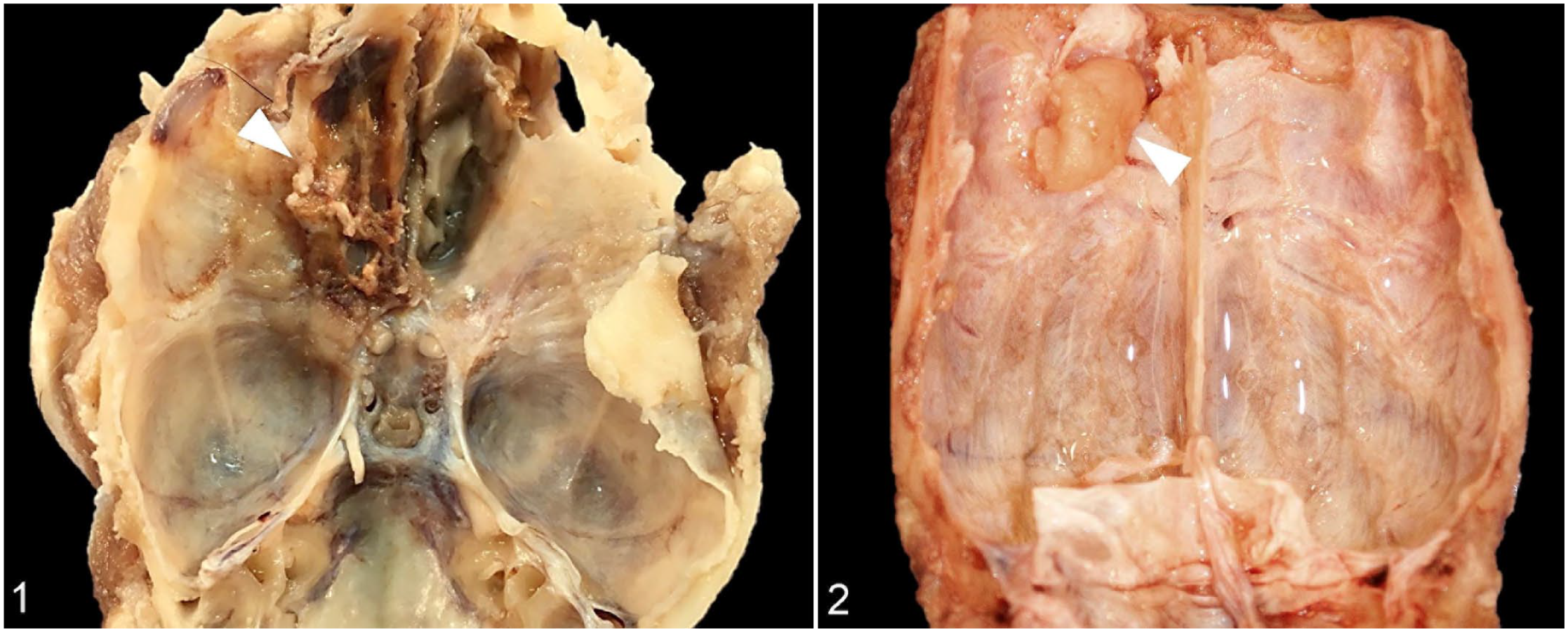
Rhabdoid meningioma, cranial vault, dog.
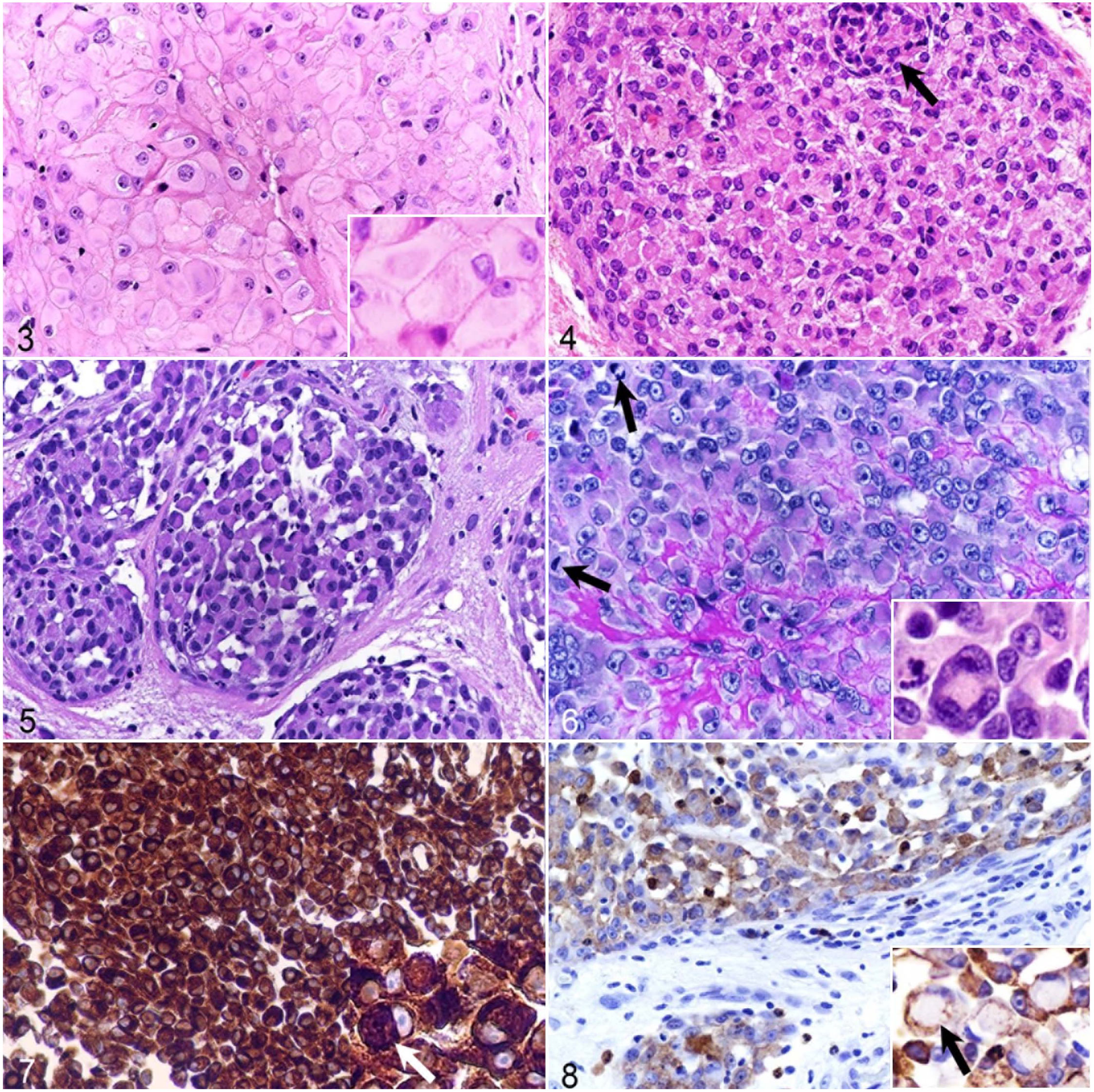
Histologically, the tumors were locally invasive, nonencapsulated, moderately-to-densely cellular and pseudolobulated, and contained sheets or occasionally cords of ovoid to polygonal cells separated by a fine fibrovascular stroma (Figs. 3–6). The areas of meningiomas showing rhabdoid differentiation was approximately 70% for cases 2 and 4, 90% for case 1, and 100% for case 3. In case 1, a focal perivascular pseudopapillary organization was observed. Multifocal to coalescing areas of necrosis affecting less than 5% of the tumor were observed in the first 3 cases. The rhabdoid cells were focally discohesive, had well-defined cell borders, contained abundant eosinophilic-to-glassy cytoplasm, often with eosinophilic, globular, paranuclear inclusions, large oval to reniform, eccentric nuclei with vesicular chromatin, and conspicuous eosinophilic to amphophilic nucleoli. In cases 1 and 4, focal areas of rhabdoid cells with anaplasia and pleomorphism were present. In all cases, nuclear cytoplasmic inclusions (pseudonuclear inclusions) and invasion of the dura mater and the subjacent cortical neuropil were observed. The PAS stain was largely negative in all cases. Except for case 2, all tumors contain giant tumoral cells. The mitotic counts were 0 to 1 for cases 1, 2, and 3, and 7 for case 4.
Cases 2 and 3 were assigned as atypical/grade II meningioma based on the presence of brain invasion, spontaneous necrosis, and prominent nucleoli. Based on the marked anaplasia, with loss of recognizable meningothelial features, cases 1 and 4 were assigned as grade III meningiomas.
In all 4 cases, the cytoplasm was diffusely and intensely immunopositive for vimentin, mainly restricted to the paranuclear inclusions (Fig. 7). For cases 1 and 3, the cytoplasm was also positive for CK (minimal for case 1 and marked for case 4) (Fig. 8). S100 and melan A expression was negative in all cases, while weak, focal immunolabeling for GFAP was present in 1 case (case 4). Key features are summarized in Table 2.
Rhabdoid meningioma, brain, dog.
Immunohistochemical and histopathological findings in dogs with rhabdoid meningioma.

Abbreviations: TEM, Transmission Electron Microscopy; CK, cytokeratin; GFAP, glial fibrillary acidic protein.
Mitoses in 10 HPF (2.37 mm2).
Ultrastructural Findings
Ultrastructurally, for case 1, the rhabdoid cells were round-to-oval, separated by thin collagen bundles or pre-existent neuropil, and delimited by a discontinuous basal lamina. Cells had peripheral nuclei with prominent central nucleoli, and intranuclear cytoplasmic invaginations (nuclear pseudoinclusions) were frequent. There was abundant cytoplasm filled with haphazardly arranged intermediate filaments of 5 to 8 nm in diameter, often forming cytoplasmic whorls and entrapping few organelles including lysosomes, RER, or mitochondria (Figs. 9, 12). Cells were connected by desmosomes and hemidesmosomes and by fine fibrils to the basal membrane (Figs. 10, 11). The intermediate filaments corresponded to the vimentin-immunopositive cytoplasmic inclusions.
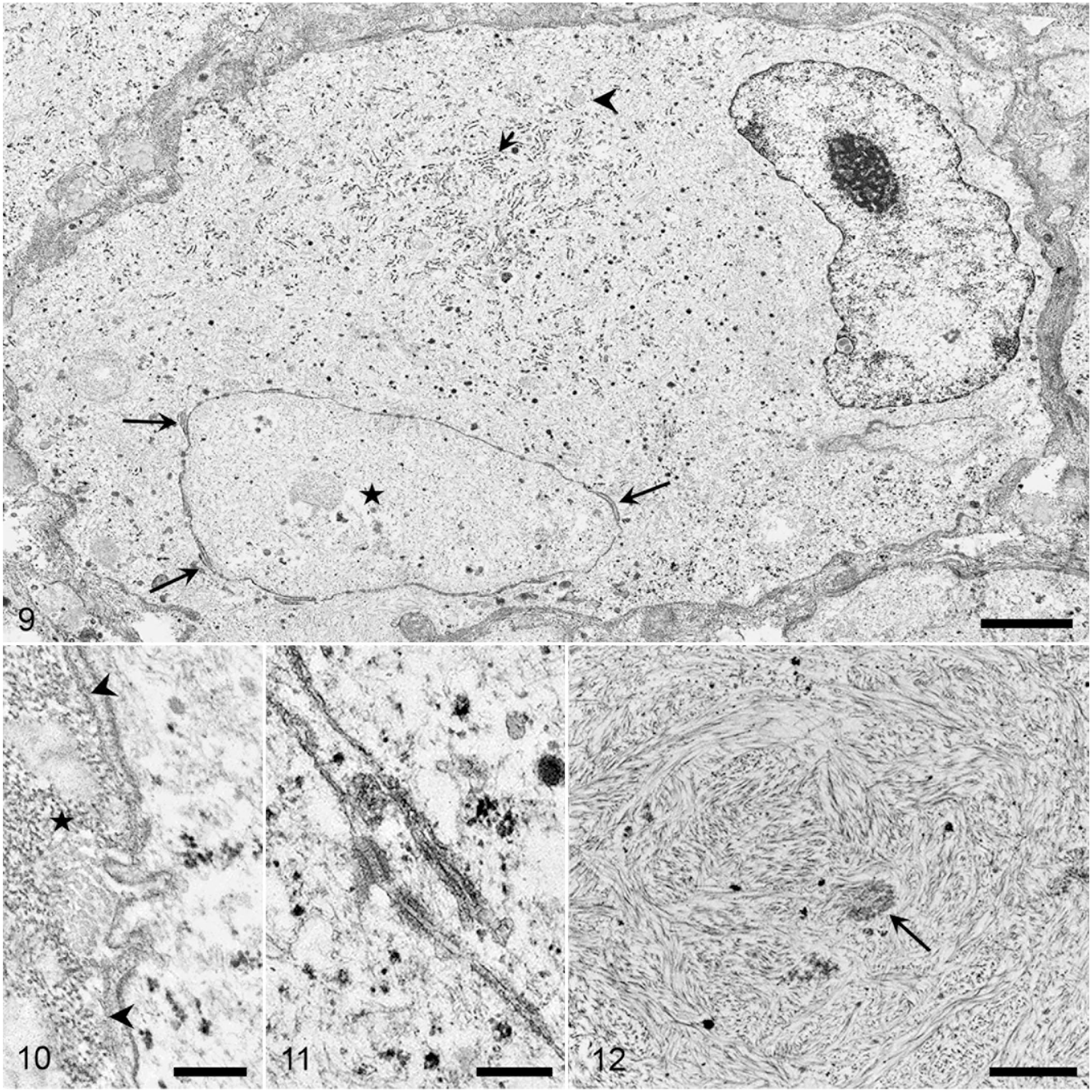
Transmission electron micrograph. Rhabdoid meningioma, brain, dog. Case 1.
In cases 2 and 3, the cell body contained innumerable arrangements of interacting cell processes folded in a maze-like fashion with scattered intercellular junctions (Fig. 13). Cells were usually in close contact but occasionally were separated by few collagen fibers. Within the cytoplasm, there were few organelles, represented mainly by mitochondria.
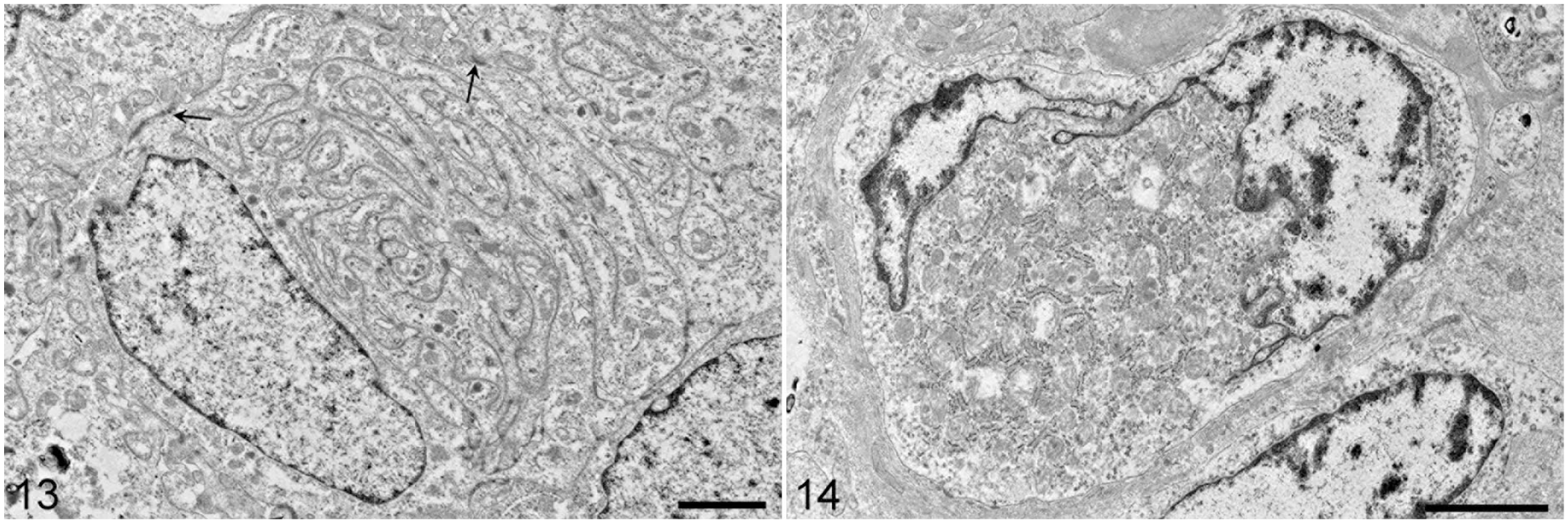
Transmission electron micrograph. Rhabdoid-like meningioma, brain, dog. Case 2. Neoplastic cells have large cytoplasmic processes folded in a maze-like fashion. Between processes, there are numerous intercellular attachments (visible as dark spots). The cell borders have intercellular attachments (arrows). Scale bar = 2 µm.
In case 4, the cytoplasm consisted of many round-to-flattened mitochondria admixed with rough endoplasmic reticulum and Golgi cisternae (Fig. 14). Therefore, tumor cells showed oncocytic differentiation in this case and not the rhabdoid differentiation suggested by light microscopy.
In all cases, the interdigitating cell processes were joined by desmosomal intercellular connections consisting of a perimembranous accumulation of electron-dense material accompanied by intracellular intermediate filaments (Fig. 10). Premelanosomes and actin/myosin-like filaments were absent.
Discussion
This study presents the clinical, pathological, and ultrastructural features of 4 canine meningioma cases with rhabdoid morphology, an uncommon and aggressive form of meningioma in humans. Rhabdoid meningioma is an uncommon variant of malignant (WHO grade III) human meningioma comprising predominantly plump cells with eosinophilic cytoplasm (“rhabdoid cells”) containing eosinophilic paranuclear inclusions and eccentric nuclei with open chromatin and a prominent nucleolus.19,20 In all 4 canine cases presented here, by histopathology, the meningiomas consisted of loosely cohesive sheets of rhabdoid cells with abundant eosinophilic-to-glassy cytoplasm with paranuclear, hyaline-like inclusions. However, the accurate diagnosis of rhabdoid meningioma is often difficult and requires correlation of histopathology, immunohistochemistry, and transmission electron microscopy, with the ultrastructural findings being essential for confirmation of this meningioma type.11,24,28 This peculiar diagnostic feature derives from the fact that the term “rhabdoid” is a general histologic descriptive commonly used for several tumors (eg, Wilms’ tumor, rhabdoid kidney tumor, and atypical teratoid rhabdoid tumors of the brain, carcinoma, sarcoma, gemistocytic astrocytoma, and melanoma), and because rhabdoid meningioma frequently shows histologic features of other histologic types (eg, papillary).28,39
Ultrastructurally, rhabdoid meningiomas are defined by the intracytoplasmic aggregations of whorled intermediate filaments associated with classical features of meningothelial cells such as intercellular desmosomal and gap junctions formed by the interdigitating cell processes.5,2,28,39 In our case series, case 1 closely recapitulated these ultrastructural features of rhabdoid meningiomas in humans, as the majority of the cytoplasm contained whorls of vimentin entrapping few organelles and glycogen. Cases 2 and 3 ultrastructurally resembled rhabdoid-like morphology, as the paranuclear inclusions consisted mainly of interacting interdigitating large cell processes folded in a maze-like fashion and joined by desmosomal cell connections. Rhabdoid-like (or “pseudo-rhabdoid”) meningiomas are rare forms of rhabdoid meningioma that histologically appear rhabdoid but are ultrastructurally characterized by accumulations of complex interacting cell processes rather than intermediate filaments.11,30 For case 4, the cytoplasm of the rhabdoid cells consisted mainly of round to flattened mitochondria admixed with fewer flattened Golgi cisternae and rough endoplasmic reticulum. This ultrastructural pattern of meningeal cells with mitochondria-rich cytoplasm resembles oncocytic meningioma, a rare and poorly known form of meningioma. 40 Oncocytic meningioma has not been previously described in dogs.
The mitotic count in our case series of rhabdoid meningioma was low (for cases 1, 2 and 4. Most human rhabdoid meningiomas have a high mitotic rate and show more anaplasia, 16 although rhabdoid meningiomas occasionally lack other morphological features of malignancy.8,39 As in our case series, focal tumor necrosis is frequently observed in human rhabdoid meningioma.23,28
Immunohistochemically, rhabdoid meningioma shows a polyphenotypic nature, indicating a divergent differentiation. 22 As in our case series, rhabdoid cells typically show strong and diffuse cytoplasmic immunolabeling for vimentin, occasionally coexpressed with focal positivity for CK. Variable immunolabeling for S100 and epithelial membrane antigen (EMA) and weak expression of GFAP and neuronal markers (such as synaptophysin) can also be occasionally observed in human rhabdoid meningioma.13,23,24
There is a spectrum of central nervous system (CNS) tumors from which rhabdoid meningioma should be differentiated,11,24,28 including gemistocytic astrocytoma, metastatic carcinoma, melanoma, rhabdomyosarcoma, and atypical teratoid rhabdoid tumors in the brain. Negative immunolabeling for GFAP and melan A and immunopositivity for vimentin were used in this study to differentiate rhabdoid meningioma from astrocytoma, metastatic carcinoma, and melanoma. The ultrastructural confirmation of meningothelial differentiation and absence of actin myosin-like filaments and pinocytotic vesicles helped distinguish rhabdoid meningioma from metastatic rhabdomyosarcoma and smooth muscle tumors. 9 The distinction of rhabdoid meningioma from atypical teratoid/rhabdoid tumors is particularly challenging. 24 Atypical teratoid/rhabdoid tumors in the brain are characterized by variable admixtures of components indicating neuroectodermal, mesenchymal, and epithelial lines of differentiation,4,18,20 which was not observed in rhabdoid meningioma.
Rhabdoid transformation of meningioma following surgical removal has been described in humans for several histologic types of meningioma, such as meningothelial or fibroblastic, suggesting that rhabdoid morphology could be an acquired, more aggressive phenotype of meningioma.10,16,30 Rhabdoid meningioma may be focally combined with other cytoarchitectural meningioma patterns, such as papillary and epithelioid.3,32
The rhabdoid morphology of human meningiomas is associated with an aggressive clinical behavior highlighted by the early postsurgical recurrence and low overall survival time. However, the overall survival time for the 2 cases in this study treated by surgery and vaccine-mediated immunotherapy (348 and 419 days) was comparable with dogs with grade I meningiomas with high vascular endothelial growth factor (VEGF) expression treated by surgery and hypofractionated radiotherapy (442 days) 29 (tumor-related survival times were not considered in case 1 because there was no definitive cause of death, or in case 2 because it was euthanized due to aspiration pneumonia). In addition to the histologic type, larger meningioma volume could be associated with a poorer prognosis in dogs. 38 This has been observed in our cases as well.
A rhabdoid tumor with an aggressive clinical course and uncertain cellular origin invading the right pyriform lobe of the brain was described by Steele et al 36 in a 1.5-year-old dog. The rhabdoid cells show vimentin-positive paranuclear cytoplasmic inclusions, but the possible cellular origin remained uncertain despite the detailed immunohistochemical and ultrastructural examination. In this case, the absence of typical ultrastructural meningothelial features in the rhabdoid cells, such as interdigitating cell processes and desmosomic intercellular connections, makes the meningeal origin of the neoplasm less likely.
Rhabdoid morphology can be focally present in many types of meningioma. Thus, according to WHO recommendations, the diagnosis of rhabdoid meningioma should be made only in cases of meningothelial tumors showing a predominantly rhabdoid morphology. In cases with focal rhabdoid morphology, the dominant histologic pattern of the tumor should be followed by the mention of “with rhabdoid features.” 20 Although rhabdoid meningioma has not been formerly reported in veterinary medicine, a myxoid meningioma with focal rhabdoid features was described in a dog. 33 In this case, the rhabdoid features were present in approximately 30% of the tumor cell population.
In our case series, the rhabdoid meningiomas were all located in the olfactory and/or frontal lobes. In dogs, most meningiomas are located in the meninges covering the dorsal surface of the frontal cortex, olfactory lobes, ventral surface of the brain, falx cerebri, cerebellar tentorium, and cerebellopontine angle.26,37 This can be explained by the concentration of the arachnoid cap cells near the large cerebral veins, basilar plexus, major sinuses, and around the crista galli and cribriform plate of the ethmoid bone. 13
All 4 dogs in our series of rhabdoid meningioma were female. Although canine meningiomas express progesterone receptors 1 as do their human counterparts, and a large population study assigned a female predisposition, 35 it is generally believed that no sex predilection is apparent in canine meningioma.25,26,37 In humans, rhabdoid meningioma shows a slight female predisposition; 39 however, this is not surprising since meningiomas are particularly common in female patients 7 with an overall female-to-male ratio of 2.2:1.
This case series reports the gross, histologic, immunohistochemical, and ultrastructural characteristics of meningiomas with rhabdoid features in dogs. Rhabdoid morphology comprised different types of meningiomas, the ultrastructural findings being essential for a correct diagnosis.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221100436 – Supplemental material for Meningioma with rhabdoid features: Pathologic findings in dogs
Supplemental material, sj-pdf-1-vet-10.1177_03009858221100436 for Meningioma with rhabdoid features: Pathologic findings in dogs by Alexandru-Flaviu Tabaran, Anibal G. Armien, G. Elizabeth Pluhar and M. Gerard OO’Sullivan in Veterinary Pathology
Footnotes
Acknowledgements
We thank our colleagues Paula Overn and Katalin Kovacs in the Comparative Pathology Shared Resource for excellent technical assistance, and Dean Muldoon in the University of Minnesota Veterinary Diagnostic Laboratory for the outstanding electron microscopy preparation.
Supplemental material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
