Abstract
Calcification of large arteries has been sporadically reported in horses. The pathogenesis is still unknown, but recent studies in humans suggest that this is a regulated biomineralizing process. This study surveyed the prevalence, distribution, and severity of vascular calcification in Thoroughbred and Standardbred racehorses. Histopathologic, ultrastructural imaging, and energy dispersive X-ray elemental analyses were used to examine the lesions. Calcification of the tunica media, predominantly the pulmonary artery, was found in 82% of horses (83/101). Young adult horses (mean [SD] age in years, 4.44 ± 2.17) of both breeds and sexes were similarly affected. Lesions appeared as white-to-yellowish, hard, and gritty plaques of variable size. On microscopic examination, elastic fibers within the tunica media were thinned, fragmented, and calcified, and surrounded by dense collagen matrix. Elemental analysis showed distinct peaks for calcium and phosphorus, consistent with hydroxyapatite mineral. The frequent occurrence of calcification in the tunica media of large pulmonary arteries of young racing horses indicates the need to investigate its pathogenesis and potential clinical implications.
Introduction
Arterial calcification involves calcium mineral deposition along one or more layers of the arterial vessel wall. 15, 20 It may occur in the tunica media of the artery (known as Mönckeberg's sclerosis) and/or in the intima, where it is always associated with atherosclerosis in humans. 28 The pathogenesis of medial artery calcification in humans is still poorly understood, but it is hypothesized to result from a regulated biomineralization process that resembles embryonic osteogenesis, rather than as a passive response to degeneration. 6, 10 Results of recent studies demonstrate that various proteins (i.e., alkaline phosphatase, osteocalcin, and collagen II) normally involved in bone mineralization are also present in calcified areas of the arterial wall. 28, 33 Furthermore, vascular smooth-muscle cells may ultimately acquire potential biomineralizing capabilities by undergoing osteogenic and/or chondrogenic differentiation. 28 The structural and morphologic changes that occur with vascular calcification affect the elastic compliance of the arterial wall and cause vascular stiffness. 23 Arterial stiffness is a significant independent factor associated with organ dysfunction and increased risk for cardiovascular morbidity and mortality. 27, 34
Calcification of large arterial vessels, such as aorta, pulmonary artery, pulmonary artery branches, and carotid arteries, has been reported in horses. 9, 17, 25 In an abattoir survey, Cranley 9 observed calcification of the pulmonary artery in nearly 27% of the Thoroughbred horses versus less than 6% in ponies and cobs. In humans, medial artery calcification usually occurs as a degenerative process that becomes more severe with age, whereas it has been reported in horses at a relatively early age (<5 years) and particularly in those with a racing background. 17 Nakamura et al. 25 found histologic abnormalities of the carotid artery in 30 of 64 Thoroughbred horses (2–4 years old) and in 3 of 13 Thoroughbred foals. The histopathologic description of such lesions included fragmentation of elastic fibers; mucoid matrix deposition; and calcification with extensive fibrosis of the tunica media. Osseous and collagenous tissue and lacuna that contain chondroid matrix had also been identified within the wall of affected arteries. 9
We are investigating the potential rheologic and clinical consequences of medial artery calcification of the major arteries in horses of racing age. Although this condition might be a background change unrelated to cause of death or racing performance, there is a possibility that such lesions are a degenerative disease that might predispose to occasional acute failure of the arterial wall in racing horses. The objectives of this study were to survey the prevalence, distribution, and severity of medial artery calcification in the major arteries of racehorses at necropsy and to compare the histopathologic and ultrastructural features with those described for calcified arteries in humans.
Materials and Methods
Animals and tissue collection
Tissues were obtained at a necropsy of 108 racing horses that died or were euthanatized between February 2006 and May 2007. Samples came from racing horses examined as part of the Death Registry Program of the Ontario Racing Commission to determine or confirm the cause of death of any registered racing horse that died or was euthanatized within 14 days of training or racing. There were 76 males and 32 females: 51 Thoroughbred and 57 Standardbred, with a mean age of 3.84 and 5.01 years, respectively (range, 1–12 years, including both breeds). Carcasses were transported, within 12 hours of death, to the Animal Health Laboratory (AHL), University of Guelph, where necropsies were conducted within 24 hour of carcasses being delivered to the AHL. A necropsy examination was conducted on a problem-oriented basis, with detailed gross and histologic examination of all organ systems. During the necropsy examination, the heart and lungs were removed en bloc from the thoracic cavity to maintain anatomic integrity. The aortic trunk (n = 108), pulmonary arterial trunk (n = 106), and each pulmonary artery branch (∼10-cm long) (n = 101 × 2) were identified and dissected. At least 2 ring-tissue sections from each vessel were collected from the same location. In addition, tissue segments from other vessels (OV), including coronary artery (n = 18), carotid arterial trunk and branches (n = 26), and femoral and mesenteric arterial trunk (n = 6) were collected. Tissue segments from all vessels were fixed in 10% buffered formalin and embedded in paraffin for routine and special staining histologic analysis. For scanning electron microscopic (SEM) and transmission electron microscopic (TEM) analyses, multiple small tissue specimens were cut from areas surrounding and within the calcified areas and were fixed in 3% glutaraldehyde/paraformaldehyde and 0.1 M cacodylate buffer (pH 7.4).
Histology
Formalin-fixed tissues were routinely processed (without decalcification), embedded in paraffin, and cut into sections of 5-μm thickness. Tissue sections from all the cases were stained with HE for routine histologic examination and detection of calcium deposition. Selected sections were stained with Verhoeff's–van Gieson stain for elastin detection, elastin-trichrome for elastin and collagen localization, periodic acid–Schiff and Alcian blue for mucopolysaccharide detection, and von Kossa's stain to confirm calcium mineral deposition.
Electron microscopy
For SEM, fixed tissues were rinsed with 0.1 M cacodylate buffer (pH 7.4) and postfixed in 1% osmium tetroxide (OsO4), dehydrated through graded ethanol, and critical-point dried with carbon dioxide. Dried samples were snap frozen in liquid nitrogen for further sectioning, then mounted on aluminum pins by using double-sided tape (Canemco, Ltd., Lakefield, QB, Canada). After this, samples were sputter coated with 20 nm of gold/palladium in the Emitech K550X sputter coater (Emitech, Ltd., Ashford, UK). Samples were viewed by using the Hitachi S-570 scanning electron microscope (Hitachi High Technologies, Tokyo, Japan) at 10 kV, and images were digitally captured by using Quartz PCI image acquisition software (Quartz PCI, Vancouver, BC, Canada).
For TEM, the tissue sections were postfixed with 2% w/v OsO4 and 2% w/v uranyl acetate, and dehydrated through a graded ethanol series. Then, the tissues were embedded in LR white resin, cured for 1 hour at 60°C, and ultrathin sections were cut and collected on copper grids. Ultrastructural examination was performed by using a Philips CM10 electron microscope at an accelerating voltage of 80 kV. Images were collected by using an SIS Morada CCD camera with SIS Item software. For energy-dispersive X-ray elemental microanalysis, nonstained resin-embedded tissue sections were examined on an EDAX Sapphire energy-disperse X-ray microanalysis system attached to a Phillips CM10 electron microscope with an accelerating voltage as above.
Statistical analysis
Simple summaries for binary outcomes were constructed by using Proc FREQ in SAS 9.1.3. Proportions and exact 95% confidence intervals (CI) were computed for each binary outcome variable. Because the data were non-normally distributed, to test for differences in age, sex, and breed, an exact Mann Whitney-Wilcoxon test was performed by using Proc NPAR1WAY. Means and standard errors are shown.
Logistic regression models were used to examine the relation between pulmonary artery branch (PAB) and pulmonary artery (PA), aorta, and OV, when adjusting for sex and breed; all 2-way interactions and 3-way interactions were included in the model. Any terms not significant at alpha = 0.10 were removed, except that PAB, PA, and OV were retained regardless. Exact P values were computed, along with conditional odds ratio (OR) estimates and exact 95% Sterne (shorter) CIs on the OR. Proc LOGISTIC was used to fit the models, but special routines were used to compute the Sterne CIs. 8
Results
Fifty six of the horses had a limb fracture, 11 had other fractures (i.e., pelvis), and 53 horses had other conditions, such as pulmonary hemorrhage, sudden death, and internal bleeding, among others. Histologic examination of HE-stained slides confirmed the presence of calcium deposition in major arterial walls of 83 of 101 of the horses (82%). The mean (SEM) age in years of Thoroughbred horses with calcification was 3.84 ± 0.30 years, and this was significantly (P = .006) younger than in affected Standardbreds (5.01 ± 0.29 years). The mean age of males (4.09 ± 0.31 years) was not significantly (P = .45) different from the age of females (4.61 ± 0.27 years). The mean (SD) age in years of affected horses (4.44 ± 2.17 years) was not significantly different from that for nonaffected horses (4.33 ± 2.11 years). There were no statistically significant differences with regard to vascular calcification and sex (P > .25), age (P = .77) or breed (P > .25). The pulmonary artery branches were the most commonly (83/101) and most severely affected arteries, but other vessels, such as the aortic trunk (17/108), pulmonary trunk (33/106), and carotid trunk (4/26), were also affected. The prevalence and severity of the lesions according to the vessels are shown in Table 1.
Frequency of calcification of the major arteries in Thoroughbred and Standardbred horses that died during or within 14 days of racing.
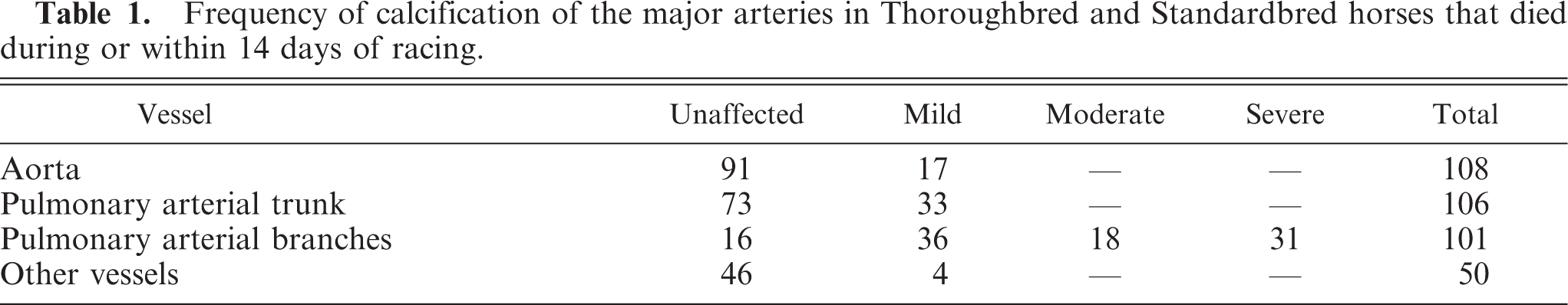
Macroscopic description
Macroscopic and palpable areas of calcification of the wall of the major pulmonary artery branches were identified in approximately a third of the affected horses. In most horses, lesions were restricted to the pulmonary branches, but 5 horses also had similar macroscopic lesions in the pulmonary artery trunk and the aortic trunk. The most obvious lesions recorded were located immediately after the pulmonary artery bifurcation, either bilaterally or unilaterally. In some cases, the lesions were also present distal to the bifurcation, several centimeters deeper into the lung parenchyma. The lesions appeared white to yellowish in color, hard to the touch, gritty, brittle, and their size ranged from a small-circumscribed plaques (0.5 cm) to multiple extensive patches that, in many cases, involved the whole circumference of the arterial wall (Fig. 1). The endothelial surface of the artery at the site of the lesion was irregular, thickened, and fractured, and some sharp spicules protruded into the lumen of the vessel.
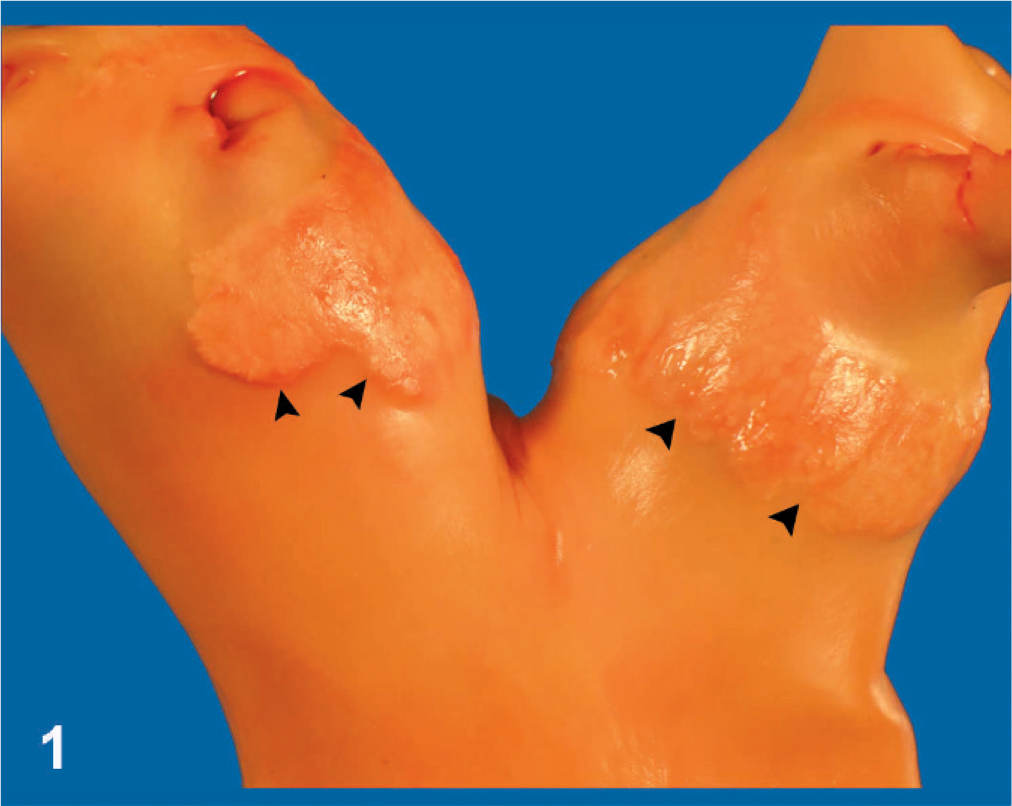
Pulmonary artery trunk and main branches; 2-year-old Thoroughbred horse. Severe calcification of the main branches showing thickening and deformation of the tunica intima (arrowheads).
Histology
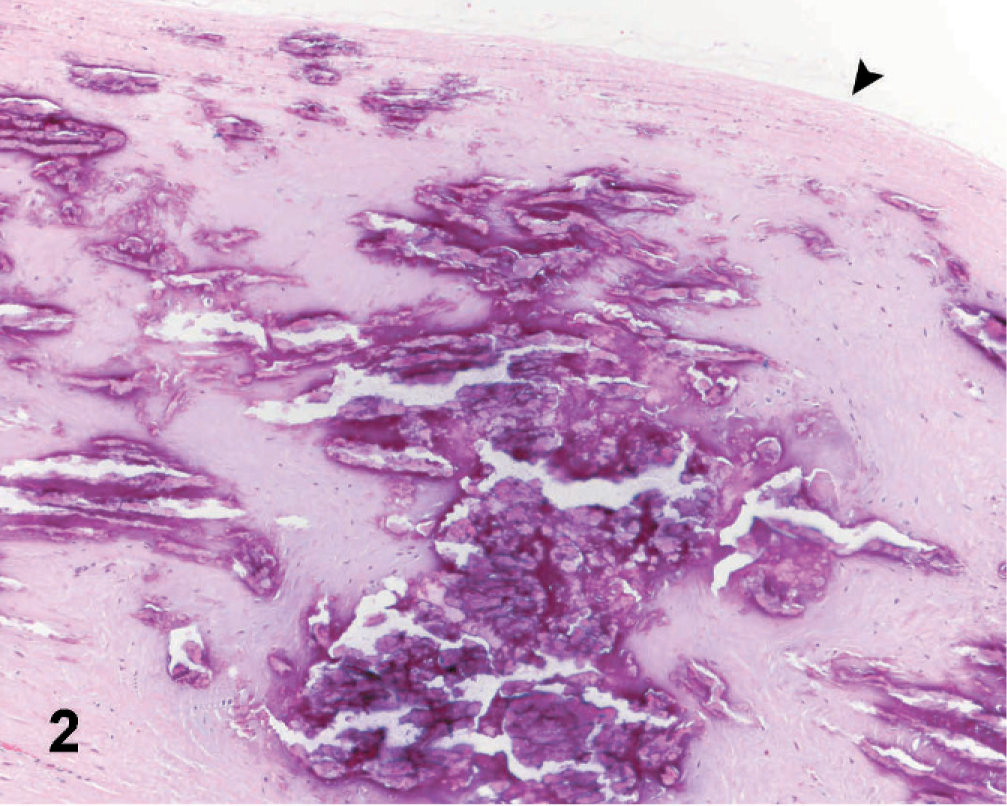
Microscopic lesions within pulmonary arteries ranged from mild or early-to-severe or advanced degrees of calcification. They were located mainly within the tunica media, but the intimal and/or adventitial layers were also affected in more severe or advanced cases. There was marked thickening of the vessels at the site of the lesion. Calcified areas encountered in the adventitial layer were only mild and focal. In some cases, calcification was associated with the vasa vasorum of the aorta, pulmonary artery, and carotid trunk. Calcium mineral deposition appeared dark blue to purple with HE staining (Fig. 2) and brown to black with von Kossa's staining. Areas of calcification varied in size from a few-millimeters-long segments of calcified elastic fibers to extensive and multiple areas of mineralized lacunae. The mineralized substance appeared as a basophilic, compacted, acellular, and amorphous material, with irregular borders, and these large areas of calcification were surrounded by an extensive collagen lattice network that, in some cases, invaded the mineralized areas. Collagen deposition was characterized by disorganized bundles of collagen fibers arranged in all directions within the areas of calcification and occupied most of the lesion. Conversely, elastic fibers were scattered, usually discontinuous and difficult to identify within the fibrocalcified areas. The identifiable elastic fibers were thin and fragmented, and segments of variable sizes were calcified (Fig. 3).
Affected pulmonary artery branch; Horse. Extensive areas of calcification (dark purple) within the tunica media of the arterial wall. Arrow indicates the endothelium. HE (10×).
Affected pulmonary artery branch; Horse. Fragmented, thinned, and calcified elastic fibers (arrowheads), surrounded by a disorganized lattice network of collagen fibers bundles (light blue). Elastin-trichrome (100×).
Large round-to-oval cells, with clear cytoplasm and large nuclei and occasional multinucleation, were present at the periphery of the calcified zones. Those cells could be found individually but more commonly were as clusters of a few or multiple cells (Fig. 4). Such cells resemble the chondrocyte/osteoblast-like cells observed in the periphery of calcified areas of human arteries.
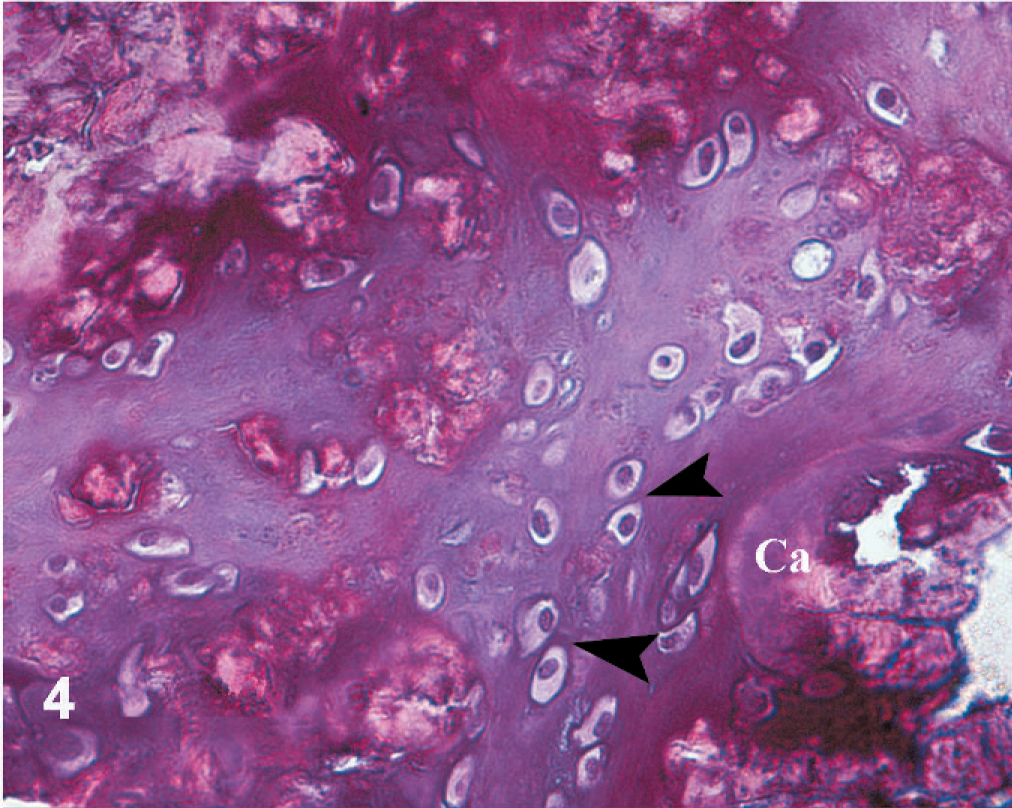
Affected pulmonary artery branch; Horse. Large round-to-oval cells with clear cytoplasm and large nuclei (arrowheads), occasional multinucleation, seen at the periphery of the calcified zones. Ca = calcified area. HE (40×).
Cases were grouped by histologic severity in HE-stained sections. Mild cases were those with small and circumscribed areas of calcification that were almost always associated with the elastic fibers. Moderate and severe lesions had similar but more extensive histologic changes that affected larger circumference and/or thickness of the vessel wall involved.
Transmission and scanning electron microscopy
Ultrastructural analysis showed a marked increased proportion of collagen fibers in-between the affected elastic fibers and surrounding the calcified areas (Fig. 5). Collagen fibers, identified as tightly pack fibrils, with prominent characteristic transverse cross-banding pattern, were arranged as bundles running in all direction in the surroundings of the affected areas.
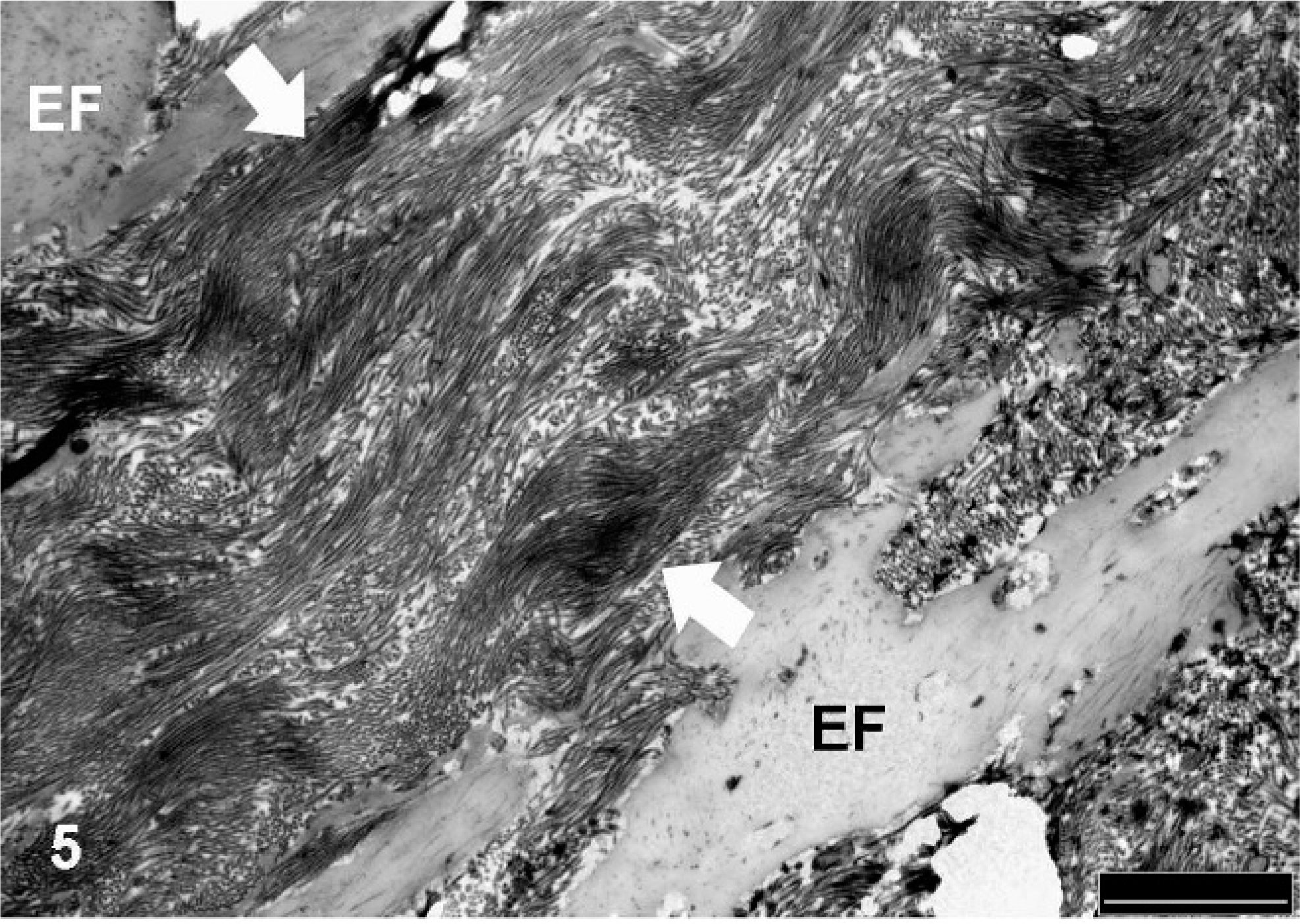
Affected pulmonary artery branch; Horse. Ultrastructural appearance of a calcified artery, showing disorganized bundles of collagen fibers (between arrows) in the periphery of the elastic fibers (EF). TEM. Bar = 2 μm.
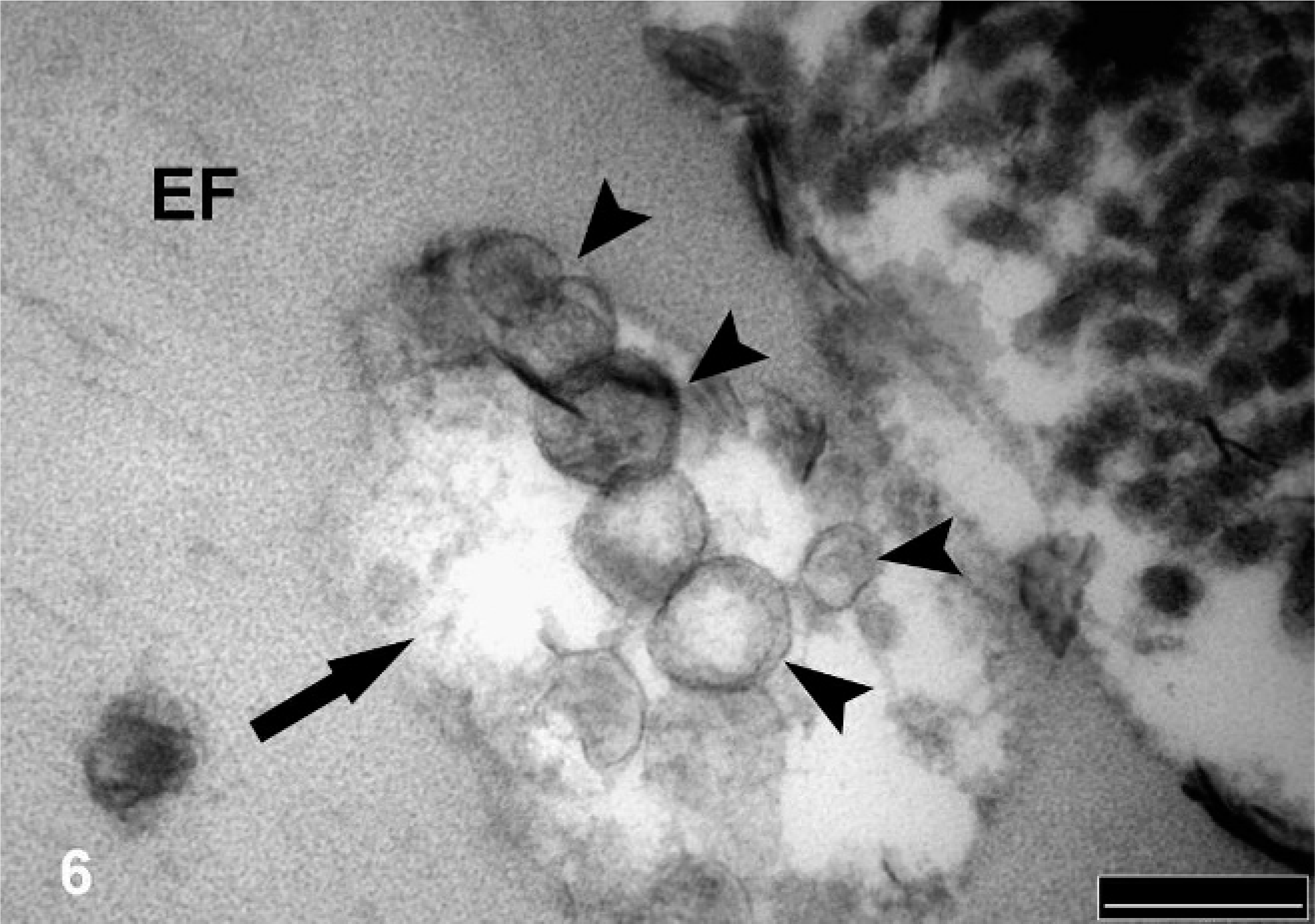
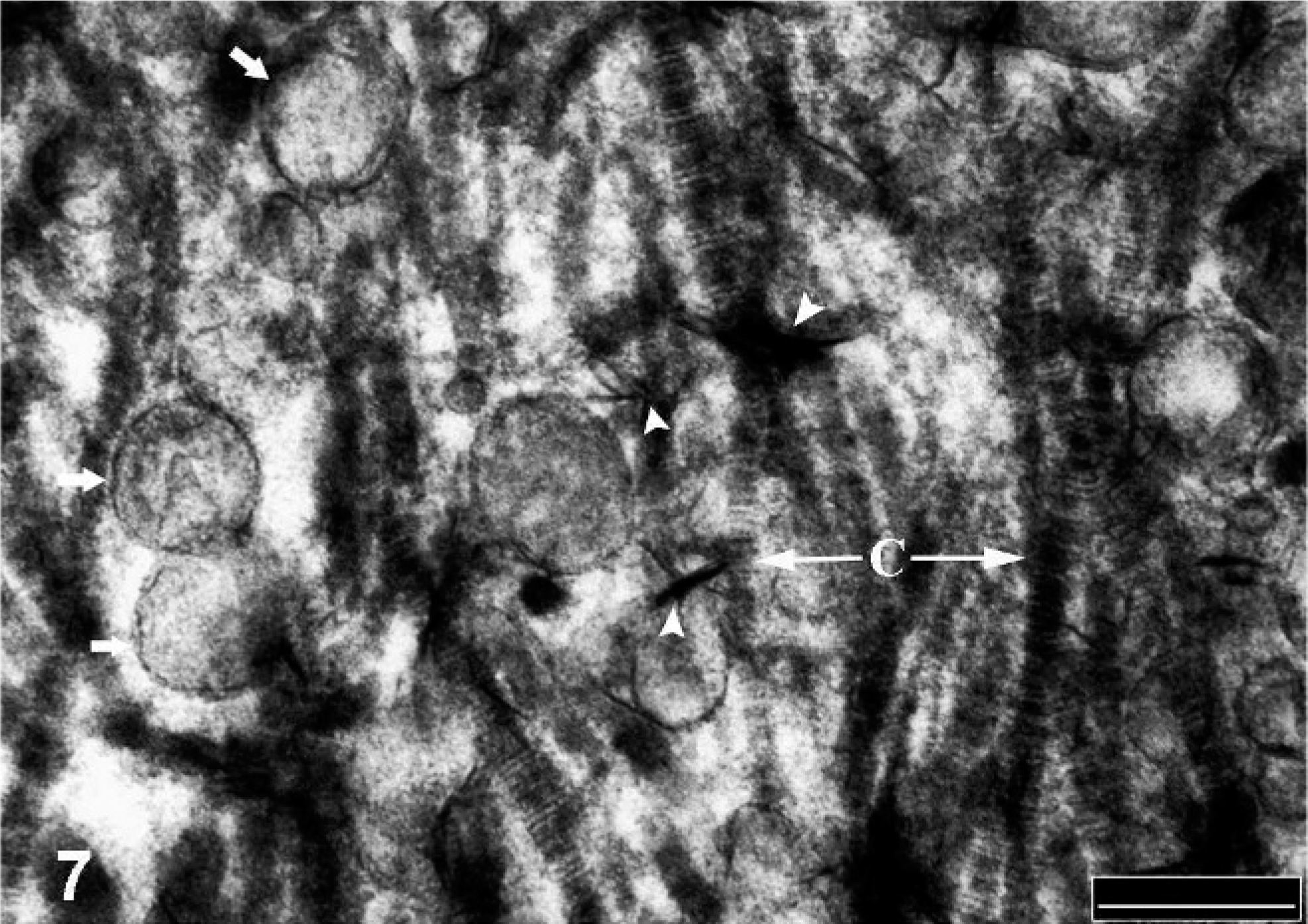
Elastic fibers in some locations had disrupted normal architecture with cavities within the fibers (Fig. 6). Multiple vacuoles were observed within the cavities in the elastin matrix, as well as in the periphery of the fiber. Some fibers had small, round, or linear areas of calcium deposition, and more affected fibers showed extensive areas of calcification along the fiber. Microspicules or needle-like structures of crystalline mineral were scattered in-between the collagen and elastic fibers in the extracellular space (Fig. 7). Multiple vesicular structures were observed in-between the collagen fibers in the extracellular space, but occasional intracellular vesicles were also noted, some of which were calcified (Fig. 7). The chondrocyte/osteoblast-like cells were rich in rough endoplasmic reticulum, and bundles of collagen fibers were observed in the periphery of the cell (Fig. 8).
Affected pulmonary artery branch; Horse. Elastic fiber (EF), showing cavities (arrow) and disruption of its normal architecture. Vacuoles (arrowheads) are present within the cavities in the elastin matrix as well as in the periphery of the fiber (not shown). TEM. Bar = 200 nm.
Affected pulmonary artery branch; Horse. Microspicules or needle-like (arrowhead) and multiple vesicular structures (small arrows) scattered in between collagen fibers (C) in the extracellular space. TEM. Bar = 200 nm.
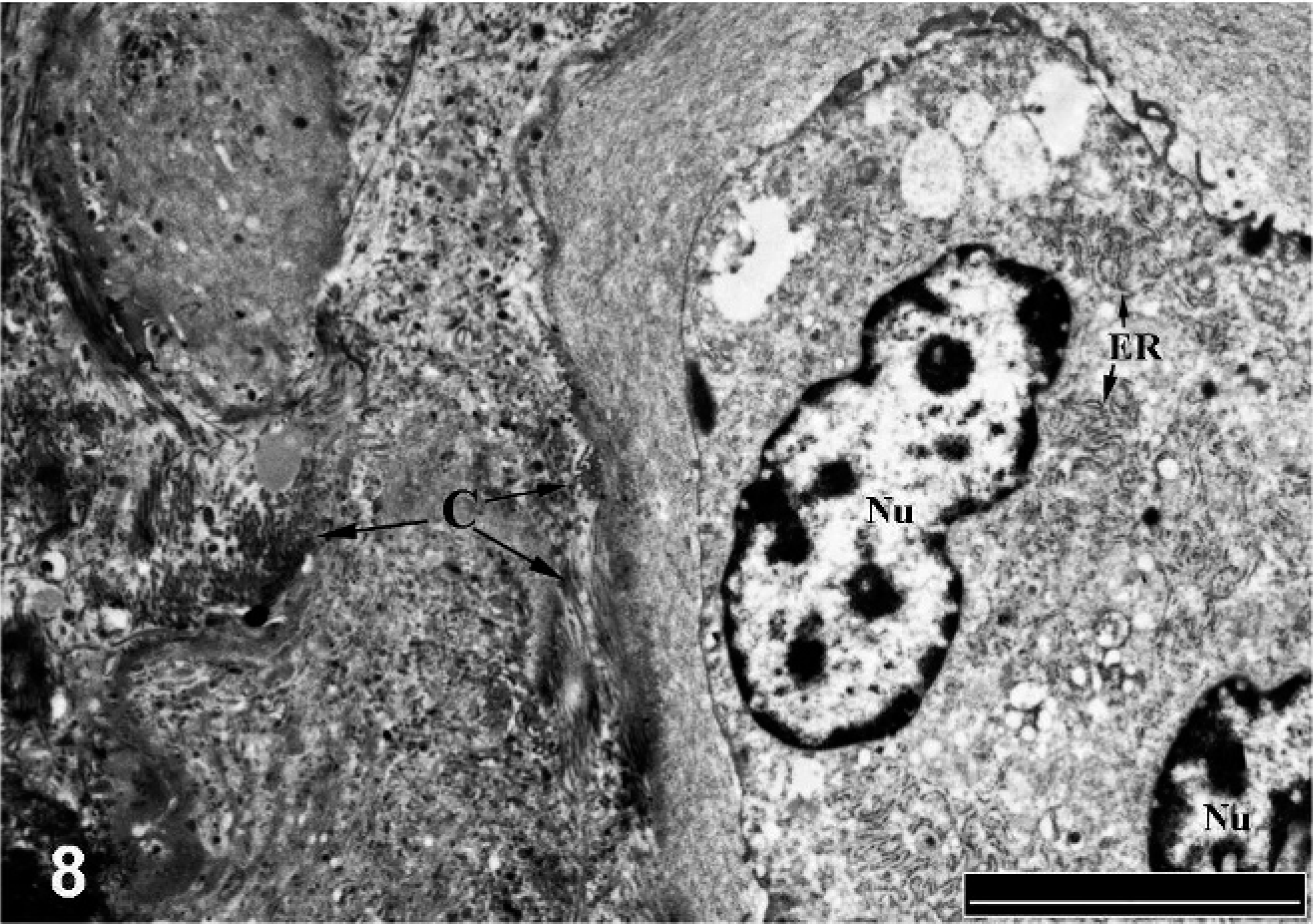
Affected pulmonary artery branch; Horse. Binucleated chondrocyte/osteoblast like-cell with clear cytoplasm and rich in rough endoplasmic reticulum (ER). A dense mesh of collagen fibrils (C) is arranged in the periphery of the cell. Nu = nucleus. TEM. Bar = 5 μm.
SEM images taken from cross sections of the affected vessels showed “mineralized blocks” of different sizes mainly scattered along the tunica media (Fig. 9) and, in some cases, extending to the tunica intima of the vessel, as demonstrated on light microscopy. Mineralized elastic fibers appeared as thick blocks that lost their normal wavy morphology and were surrounded by a large number of thin fibers, which was confirmed as collagen by using special staining techniques. This dense mesh of collagen fibers also surrounded the mineralized blocks, as observed with the mineralized elastic fibers.

Affected pulmonary artery branch; Horse. Large mineral deposits scattered within the tunica media of a severely calcified vessel. Arrowhead indicates the endothelium. SEM. (500×).
The X-ray microanalysis of affected arterial sections obtained from several horses showed that electron-dense calcified deposits contained high levels of calcium and phosphorus. The ratio between Ca : P was approximately 1 : 6, which is indicative of an apatitic mineral substance (Fig. 10).
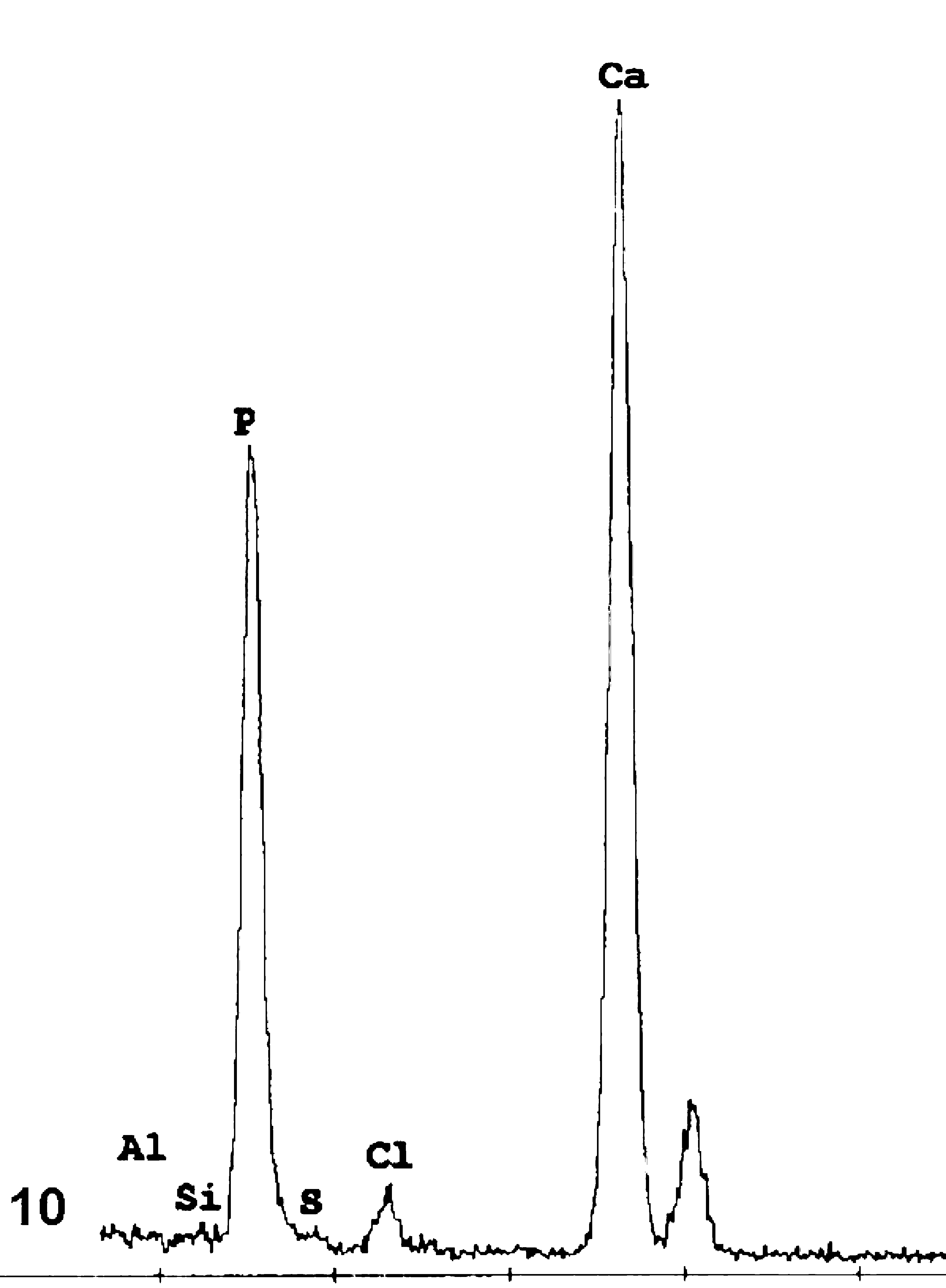
Energy dispersive X-ray elemental analysis of calcified areas, showing distinct peaks for calcium and phosphorus, characteristic of an apatitic material.
Discussion
The light microscopic and ultrastructural features of the medial artery calcification and fibrosis observed in the equine pulmonary arteries were similar to calcification of the tunica media of large arteries in humans. 5, 24 The morphologic similarities suggest a similar pathogenesis, but the initiating causes are likely different for 2 reasons. First, medial artery calcification occurred at a relatively early age in horses, whereas, in humans, it mainly occurs in older individuals. 12 Second, the anatomic distribution of lesions is different; in humans, medial artery calcification can be observed in the aorta and in coronary, carotid, renal, and iliac arteries, 2 whereas, in horses, the pulmonary arteries are the most obviously affected.
Calcification of large arteries has been described in horses, cattle, and cats, 9, 16, 22 but the pathogenesis and functional significance has not been determined. Medial artery calcification of larger arteries was previously reported in 27 to 48% of Thoroughbred horses, 9, 25 but no published reports of medial artery calcification in Standardbred horses were found. By comparison, in our study, Imaizumi et al. 17 observed degeneration and sclerosis of the tunica media of the aorta and pulmonary artery of Thoroughbred horses between 2 to 5 years of age. They noted that the severity of the lesions was associated with horses that had raced rather than with increasing age. 17 Our findings of pulmonary medial artery calcification in 82% of Thoroughbred and Standardbred horses that were obtained as casualties or unexplained deaths within 14 days of racing (i.e., in active training) supports this interpretation. However, it is not clear whether calcification was found more often in Ontario racehorses because the condition is more prevalent or because it is more prevalent with racing casualties or because horses in our study were subjected to a different necropsy protocol.
If medial artery calcification is indeed more common in racing horses, as suggested by this and other studies, 9, 17, 25 then the pathogenesis and functional implications should be examined further. Evidently, many horses have the medial artery calcification, but it is plausible that the condition might sometimes be involved with or associated with adverse conditions or outcomes. Four histologic patterns of arterial calcification in human patients have been categorized, namely atherosclerotic (fibrotic), medial artery calcification (Mönckeberg's sclerosis), cardiac valve, and vascular calciphylaxis. 15, 31 Although the topographic and age differences of medical calcification in horses are not equivalent, the microscopic pattern of lesions observed most resemble Mönckeberg's sclerosis, most commonly observed in elderly patients and in patients with diabetes and renal disease. 13, 28
The pulmonary arterial branches were most commonly and most severely affected in horses in our study, in agreement with previous reports, 9 , 17 but the reasons for this distribution are unknown. This distribution suggests that large elastic vessels are most susceptible in horses. The muscular nonelastic vessels, such as the mesenteric, carotid, or femoral arteries, were minimally mostly unaffected in horses with severely calcified pulmonary arteries. By comparison, elastic and muscular vessels of different diameters, such as the aorta, renal, and coronary artery, can all exhibit medial artery calcification in human patients. 10 Payo et al. 26 surveyed vascular calcification of the spleen and pulmonary arteries in elderly people and observed medial artery calcification only in the splenic artery. These microscopic features of medial artery calcification in horses are consistent with an active biomineralizing process, as described for humans. 20, 31 Chondrocyte/osteocyte-like cells similar to those described in lesions in humans 11, 19 were consistently arranged in the periphery of the calcified areas in all moderately and severely affected pulmonary arteries in horses. Transmission electron microscopic images showed that such cells are probably responsible for collagen synthesis in the lesions, because they were rich in rough endoplasmic reticulum and were surrounded by a large number of collagen fibers. During normal ossification, osteoblasts produce a matrix of predominantly type I collagen that is subsequently mineralized by the deposition of hydroxyapatite onto the collagen and matrix lattice. There is some controversy with regard to the origin of these cells in humans, but vascular smooth-muscle cells, resident pericytes, and circulating stem cells have been suggested as potential candidates. 33 Vascular smooth-muscle cells can undergo phenotypic transformation under the influence of several stimuli, such as oxidative stress, bone morphogenetic proteins, and increased phosphate levels. 18, 20 Results of recent studies showed that some proteins normally associated with chondrocytes and osteoclasts cells, such as bone morphogenetic proteins (BMP-2 and BMP-4), Sox 9, and Runx 2, among others, are also present in calcified human vessels. 7, 11 Further, the elemental microanalysis of the mineral deposition showed the same fingerprint for calcium/phosphorus complex found in calcified arteries in humans. The ratio between calcium/phosphorus was highly similar to hydroxyapatite, which is the main mineral component of bone. 7
Currently, there is little known regarding the cause, predisposing factors, and molecular or cellular components of arterial calcification and its potential clinical repercussions. Although vascular calcification has been reported in humans since the 18th century, 32 it has recently been “rediscovered,” 29 and a great deal of attention has been directed to understanding the pathogenetic mechanism(s) involved. Mounting evidence suggests that vascular calcification is a dynamic phenomenon that results from the interaction between promoters (phosphates, BMP) and inhibitors (MGP, osteopontin) of calcification. 1 Nonatherosclerotic arterial calcification is common in humans 12 and is associated with degradation and remodeling of elastic fibers. Degenerative changes observed in the elastic fibers in areas of calcification were consistent with those described in humans 3 and horses. 9, 17 Large arteries have a higher proportion of smooth-muscle fibers and collagenous matrix composition. 30 Also, cyclic stretching of elastic fibers can cause structural changes and elastin degradation products that promote myofibroblastic and osteogenic differentiation of fibroblasts in vitro. 30 Calcification and remodeling of large elastic arteries reduces vascular compliance and causes stiffening of the vessel wall. 20 Collagen, is an important structural protein of the vessel wall, but it is 100 to 1000 times stiffer than elastic fibers, so a proportional increase in collagen would increase vessel-wall stiffness. 14 Severe collagen matrix deposition observed in the tunica media and intima of the calcified pulmonary arteries in horses is predicted to diminish elasticity and resilience in the pulmonary artery. The elevated pulmonary arterial pressures that occur during racing performance in horses (>90 mm Hg), in conjunction with calcification and fibrosis of large arteries, may result in vascular failure as reported in humans. 4, 21 An understanding of the pathogenesis and functional significance of this condition in horses might also contribute to the understanding of arterial calcification in other conditions of humans.
Footnotes
Acknowledgements
We thank Robert Harris and Sandy Smith for their assistance in tissue processing and analysis of TEM and SEM, respectively. We also thank Dave Hilchie and Mandy Mulder for help with special histologic tissue staining. We thank the pathologists and staff, especially Bruce Cornfield, Heidi Chambers, Rick Roth, and Drs. B. McEwen, M. Hazlett, G. Tompson, A. Van Dreumel, P. Lusis, and M. Stalker of the Animal Health Laboratory at the University of Guelph for some necropsies and tissue collection. This project was kindly funded by the Ontario Racing Commission.
