Abstract
Microsporidia are obligate intracellular, spore-forming fungi. A wide range of vertebrate and invertebrate hosts can be infected; however, cases of infected turtles or tortoises have not yet been described. This is the first description of 4 cases in tortoises (Testudo spp), which showed general signs of illness as well as intestinal and respiratory signs until death occurred or they were euthanized. Granulomatous and necrotizing inflammation was visible in the lung and liver. Fungal organisms were present in heart blood, lung, liver, and intestine. Because of their morphology, staining properties (eg, positive in periodic acid–Schiff and silver reaction), and electron microscopic appearance, they were classified as microsporidia.
Keywords
Microsporidia are an ancient and diverse group of obligate intracellular organisms and are considered atypical fungi without mitochondria. They are most closely related to zygomycetes. 7,14 More than 170 genera and approximately 1300 species are known. 2,9,13,15 They are single-celled eukaryotes (protists) that have the smallest genome of any known eukaryote. 2,15 Although nowadays they are considered as fungi, they are formerly mentioned as parasites in the literature. 7,11,13,15 The host cell cytoplasm is the site of reproduction and maturation, which is quite unusual. 13 Spores are equipped with a tubular extrusion apparatus (polar tubule/tube, polar filament) for injecting spore contents into the host cell. 3,7,15 A unique feature is the emptying of the entire spore contents into the host cell instead of just specific material as occurs in other protists. 13
Microsporidia may be diagnosed by electron or light microscopy, polymerase chain reaction (PCR), cell culture, immunohistochemistry, and serologic methods. 3,7,14 Histologically, the detection of microsporidia can be difficult because these organisms are quite small and can occur without eliciting inflammatory reactions.
Spores of microsporidia have a round to ovoid shape, are refractive, and measure approximately 1 to 1.5 × 2 to 3 µm. Some species, like Heterosporis anguillarum, can measure up to 4 × 7 µm. 9 When stained with hematoxylin and eosin (HE), they appear weakly basophilic. Spores are often located in small intracytoplasmic groups inside histiocytes or free in areas of necrosis. 4,9,15
In modified trichrome stains (eg, Weber stain), the spore wall is of pinkish to red color and the interior appears transparent, whereas the background structures stain green or blue. When stained with periodic acid–Schiff (PAS), spores can show a PAS-positive granule at the anterior end of the spore. 2,4,9 With Giemsa stain, microsporidia appear light to dark blue, sometimes with a darkly stained nucleus. 2,15 With Gram stain, mature microsporidial spores are dark blue to violet (gram positive) and immature spores stain red. Overall, they must be considered as gram variable. 3,4,10 Microsporidia can be silver stained such as by the Grocott methenamine silver (GMS) or Warthin-Starry (WS) stains. 2 Microsporidia are all or partially acid-fast such as with Ziehl-Neelsen or Kinyoun acid-fast stains. 2,10 Heterosporis anguillarum differ in some aspects, for they have a larger size and are negative with Grocott’s methenamine silver stain and PAS reactions. 10
Ultrastructurally, microsporidia possess an electron-lucent endospore (chitinous layer) and an electron-dense exospore and contain 1 to 2 nuclei as well as a variable amount of polar tubule coils (often 6), depending on the species and developmental stage. Some spores contain a posterior vacuole. 2,9,10
Polymerase chain reaction is a sensitive and specific method to diagnose microsporidiosis, and identification of the microsporidia on the subspecies level can be achieved. 7
Microsporidia parasitize a wide variety of invertebrate and vertebrate hosts, including humans. Overall, they have a low pathogenicity, often induce latent or only mildly symptomatic infections in mammals, and are regarded as opportunistic pathogens. 5,11,15 In veterinary medicine, Encephalitozoon cuniculi is the most relevant microsporidia species. 2,15 An overview of microsporidia of veterinary interest is given in published books and reviews. 5,11,14
Microsporidiosis is rarely reported in reptiles and amphibians. 9,11 Spontaneous cases have occurred in tuataras, snakes, skinks, bearded dragons, water dragons, and geckos. 4,9 –11,14 Cases in chelonians (turtles or tortoises) have not yet been described. 4,9 Lizards, skinks, and bearded dragons infected with Encephalitozoon spp or morphologically similar microsporidia have been reported. 4,9 Clinical signs were mostly unspecific and included anorexia, weight loss, diarrhea, and renal failure. 4,9,14 In bearded dragons, necrosis or granulomatous inflammation in different organs (eg, stomach, intestine, liver) as well as systemic disease was described. 4,9 In frogs, toads, and salamanders, infections with microsporidia (among them Microsporidium spp and Pleistophora spp) have been associated with inflammation of the musculature, digestive tract, and liver. 11,14
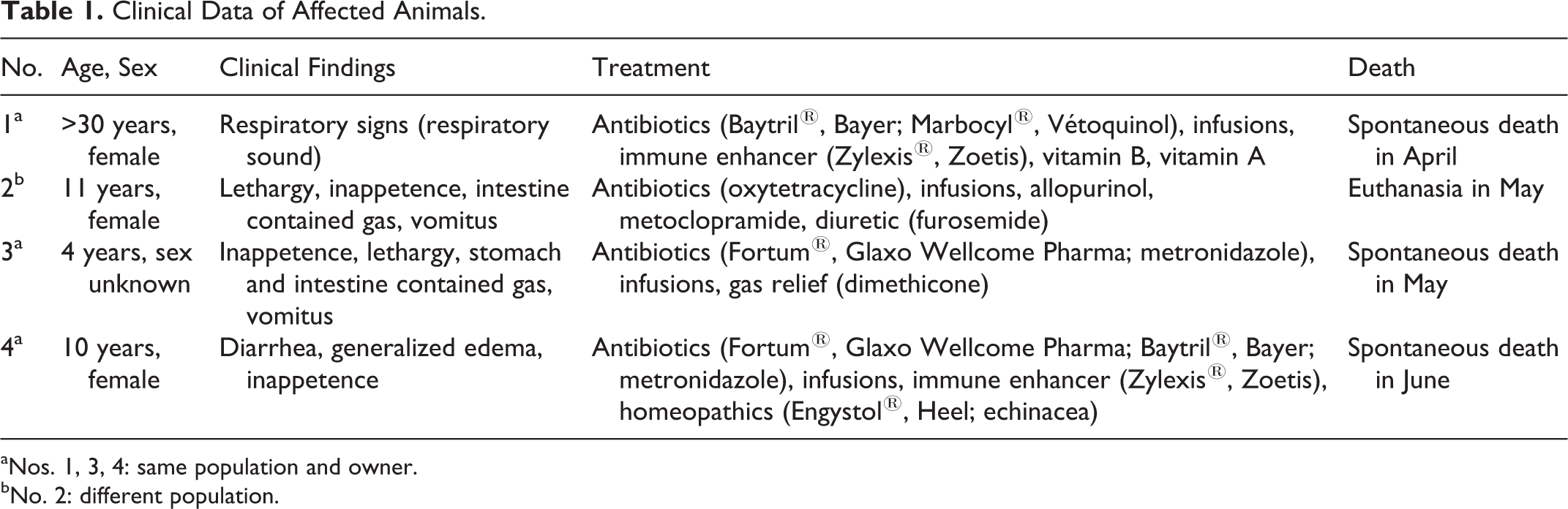
This article describes 4 cases of microsporidiosis in tortoises (Testudo hermanni boettgeri). The animals were sent to our institute for necropsy within a 13-month period by 2 different veterinarians. Three of the supposedly unrelated animals (Nos. 1, 3, 4) lived together in a population of about 200 animals. They were kept outside, in 28 isolated coldframes (Chelonium®; Samenkiste, Karlsruhe, Germany), with permanent access to large meadows and additional feeding of wild herbs, special tortoise feed (Agrobs®; Agrobs GmbH, Degerndorf, Germany), and sepia shell ad libitum. Heating lamps were used when necessary. Sporadic contact with wildlife (eg, raccoons, frogs, colubrids) was possible, but there was no contact with fish. Bacterial infections (Mycoplasma spp, diagnosed by nested PCR), viral infections (Herpesvirus, diagnosed by nested PCR), and parasitic infections (Oxyuris spp, Ascarids, Nyctotherus spp, Trichomonas spp, Cryptosporidia, diagnosed by microscopy of flotated or native samples and carbolfuchsin stained samples) were previously diagnosed in this population in postmortem investigations. Within a 13-month period, about 20 animals (∼10% of the population) showed edema, stertorous breathing, or dyspnea. Some of them also had gas-filled intestines and showed vomiting, diarrhea, inappetence, and lethargy. All animals were treated with antibiotics and some with fenbendazole, immune enhancer, infusions, and vitamins, among others. Exact treatment doses and durations were not known.
The majority of sick animals died after approximately 2 to 3 weeks of illness. Five animals were sent for necropsy.
The single animal of the other population (No. 2) was kept alone. Further information about husbandry or dietary factors was not available. The animal was inappetent and lethargic 2 weeks after hibernation and therefore presented to a veterinarian, where it showed gas-filled intestines without ingesta, apathy, and later, vomiting. The animal was treated with antibiotics, infusions, allopurinol, metoclopramide, and diuretics.
Exact treatment doses and durations were not available. Antiparasitic drugs, like fenbendazole, were not given by the veterinarian. It is unknown if the animal received a fenbendazole treatment by the owner. Animal No. 2 was euthanized due to increasing deterioration after 2 weeks of illness. An overview of clinical data is given in Table 1.
Clinical Data of Affected Animals.
aNos. 1, 3, 4: same population and owner.
bNo. 2: different population.
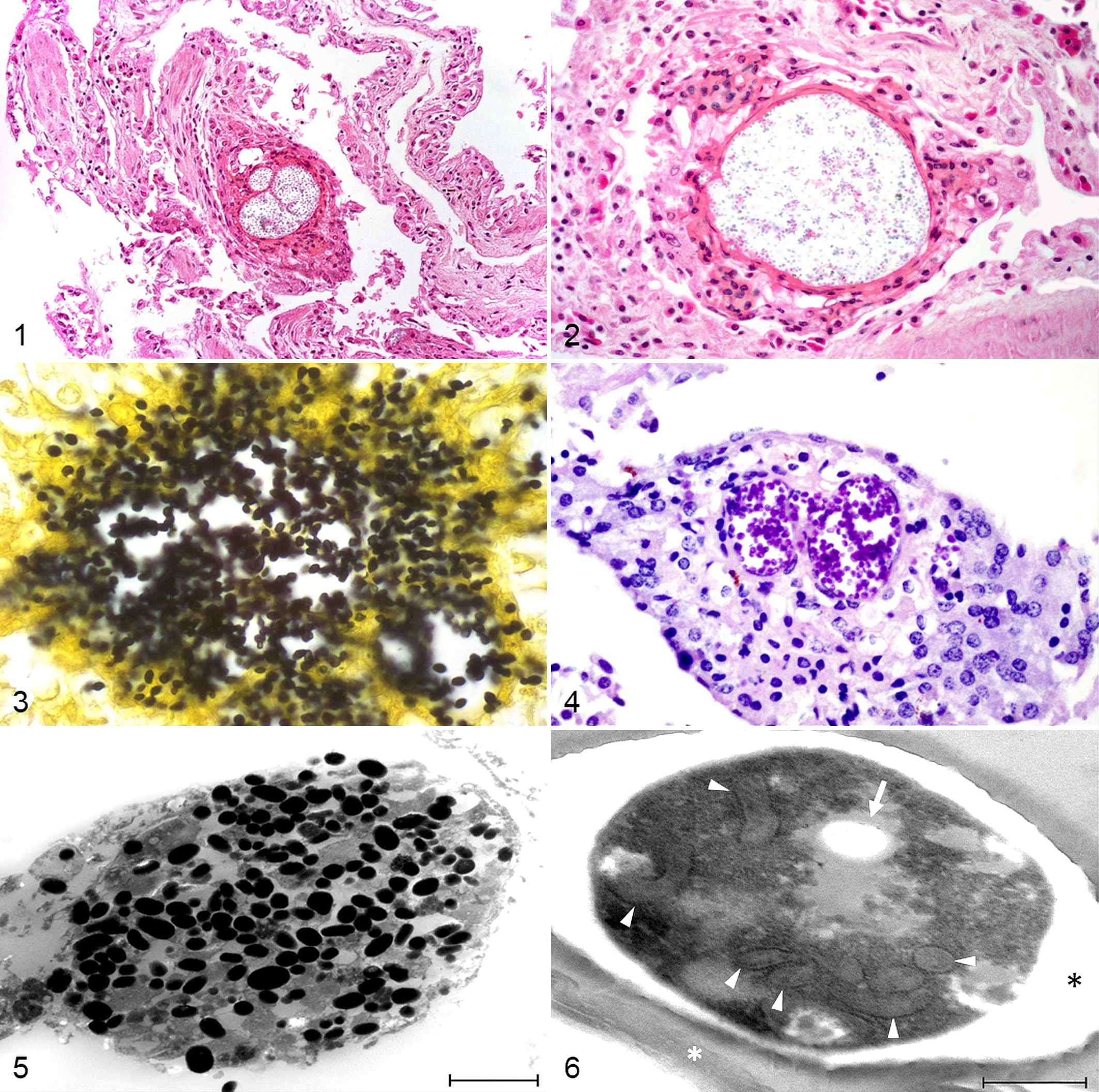
Irrespective of clinical signs, the main findings in all animals were granulomatous and necrotizing hepatitis and/or pneumonia with a variable amount of intralesional spores (Figs. 1, 2) and, in some cases, also intralesional rod-shaped bacteria. In general, inflammation was more severe, and there was a greater amount of spores in the liver than in the lung. Animal No. 1 also had a necrotizing enteritis with pseudomembrane formation, few intralesional spores, and bacteria. In the heart blood of animal No. 3, few spores and bacteria were present without necrosis or inflammation. A detailed overview of macroscopic and microscopic findings is given in Table 2.
Pathologic Findings in Affected Animals.a
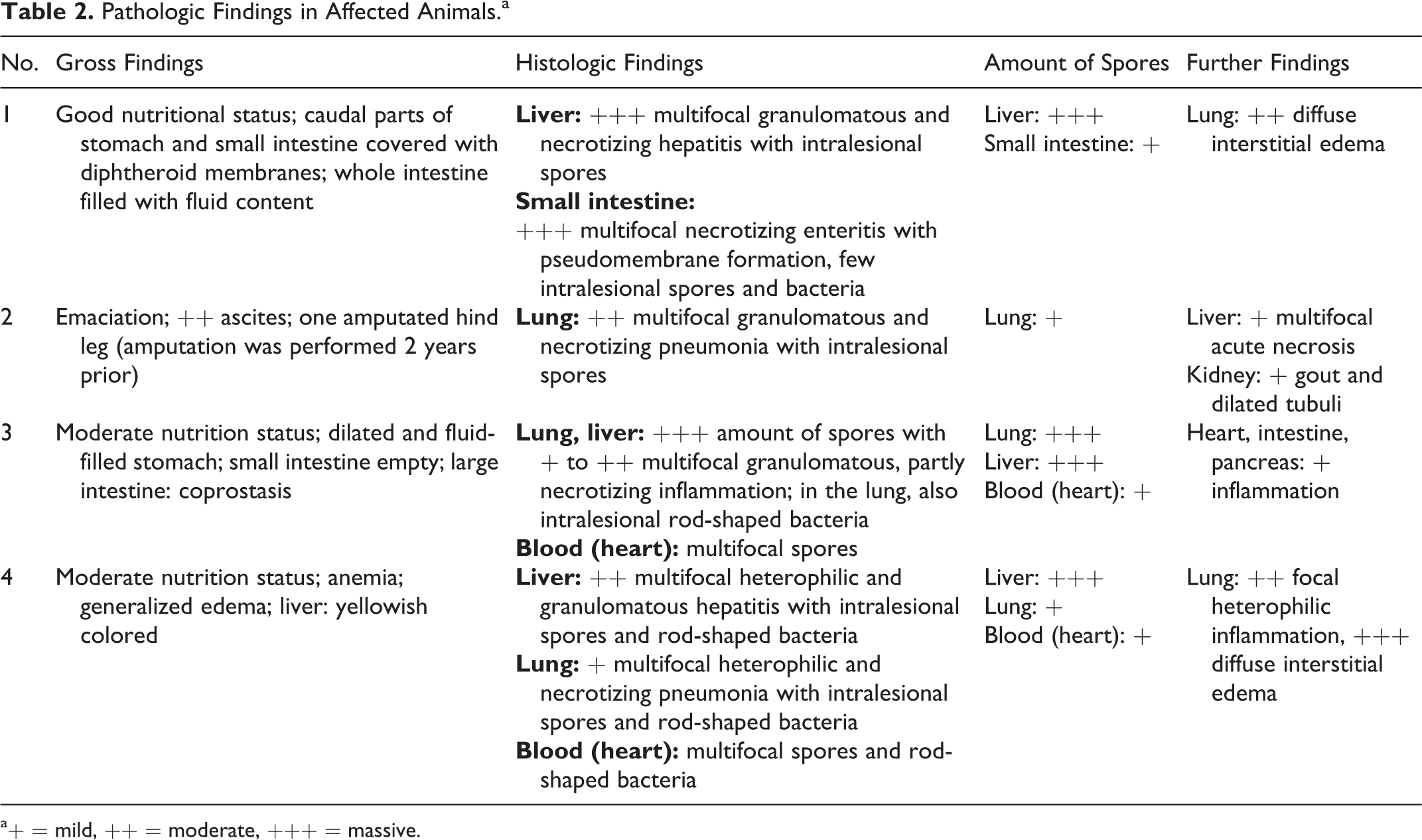
a+ = mild, ++ = moderate, +++ = massive.
Accumulations of spores were often surrounded by a fine fibrous capsule. Spores were pale basophilic in HE stain and measured 2.5 to 2.7 µm × 1.5 to 1.6 µm. They often contained a transparent vacuole. Special stains revealed that spores were positive in silver reaction (Grocott’s methenamine silver stain; Fig. 3). They were dark purple in PAS reaction and contained a transparent vacuole and a nucleus (Fig. 4). They stained mainly reddish, partly green in Weber’s modified trichrome stain, and dark blue to purple in Gram stain (gram positive). In Ziehl-Neelsen stain, they appeared blue and therefore not acid-fast. Under polarized light, spores were not refractive.
Electron microscopy was also performed in selected samples of the lung and liver. Large vacuoles containing numerous spores were visible in the parenchyma of different organs (Fig. 5). They were nearly exclusively located extracellularly and in areas of inflammation, often surrounded by connective tissue (encapsulated). Spores had an electron-dense outer sheath and an electron-lucent inner sheath. They contained an electron-lucent vacuole, most had 8 polar tubule coils in 2 rows, and they measured approximately 3 × 2 µm (Fig. 6).
In aerobic cultures of selected organs, different bacteria species, mainly Enterobacteriaceae and Streptococcus spp, but also yeasts and Candida spp, were cultured. Detailed results are presented in Table 3.
Results of Further Examinations.a
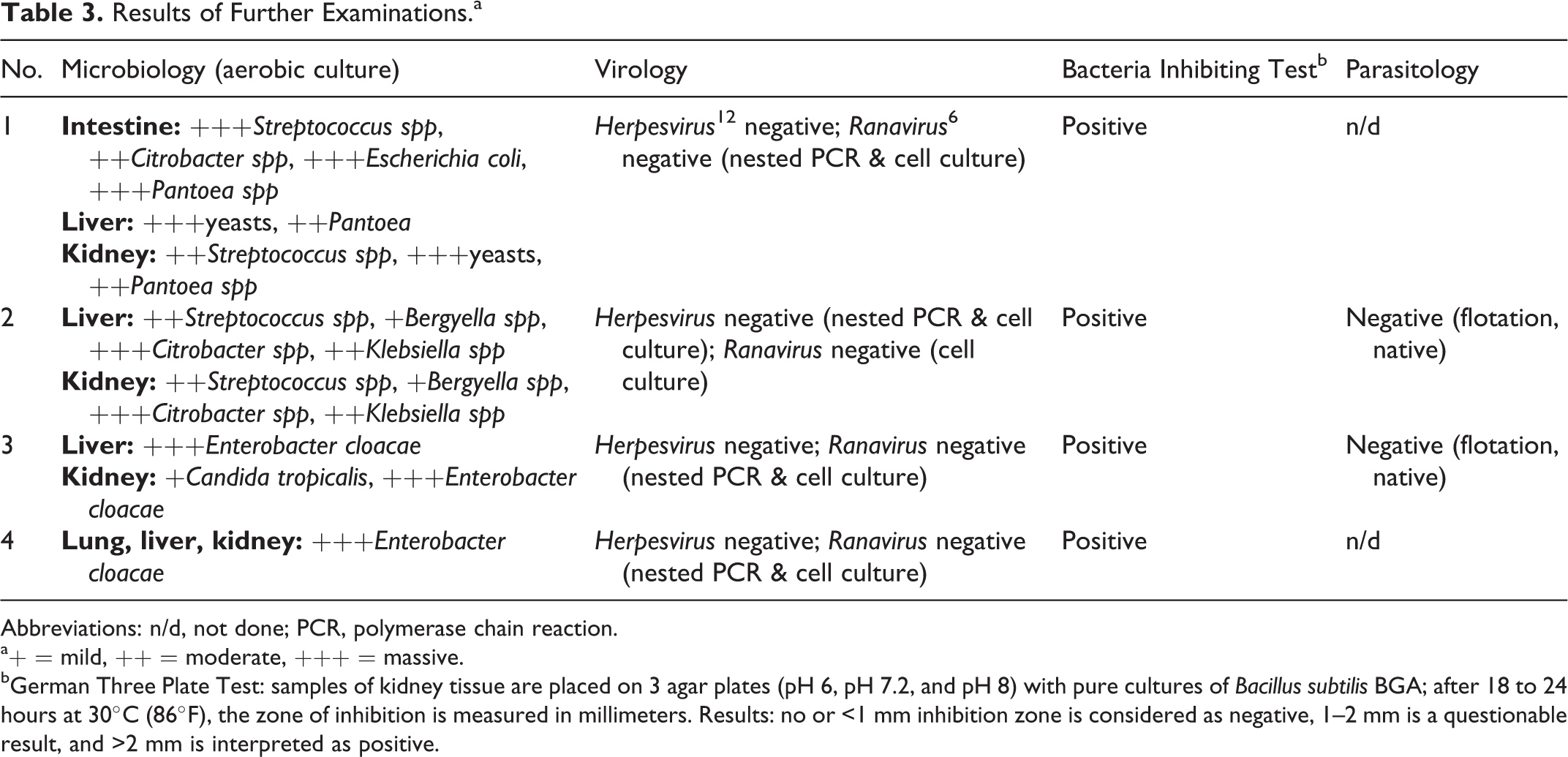
Abbreviations: n/d, not done; PCR, polymerase chain reaction.
a+ = mild, ++ = moderate, +++ = massive.
bGerman Three Plate Test: samples of kidney tissue are placed on 3 agar plates (pH 6, pH 7.2, and pH 8) with pure cultures of Bacillus subtilis BGA; after 18 to 24 hours at 30°C (86°F), the zone of inhibition is measured in millimeters. Results: no or <1 mm inhibition zone is considered as negative, 1–2 mm is a questionable result, and >2 mm is interpreted as positive.
Microsporidia can parasitize almost all animal groups and humans. They have been described in a wide range of species including reptiles such as snakes, bearded dragons, and geckos. 4,9 –11 To the authors’ knowledge, cases in tortoises and turtles have not previously been reported.
This report describes 4 cases that occurred within a 13-month period. Thus, microsporidiosis may be underestimated and more frequent than suspected. It can cause severe problems in populations, as seen in this case where 3 animals with microsporidiosis died within a short timeframe.
Lung, liver, and small intestine were primary sites of inflammation. Therefore, oral ingestion or inhalation of spores may constitute the route of infection. In another 2 cases, not covered by this report, intraluminal spores were found in the stomach without corresponding inflammatory reactions (Eydner and Blahak, 2014, unpublished data), which supports the assumption of oral ingestion. Infection of the lung is most likely acquired by spore inhalation. Since spores have been detected in heart blood, hematogenous spread is the most probable distribution route. A primary infection site (eg, intestines) from which spores could have been distributed to the lungs was evident in only 1 animal. Therefore, inhalation of spores is more likely than hematogenous distribution from another infected organ to the lungs. Although certain bacterial, viral, and parasitic infections were previously verified in 1 population (Nos. 1, 3, 4), these were nonexistent in the examined animals. Therefore, a promotion of the microsporidia infection due to an underlying disease seems unlikely in the present cases. Nevertheless, it is very probable that 3 animals also had a bacterial infection and presumably developed septicemia before death because in different organs, bacteria were visible histologically in HE stain and an equivocal bacterial spectrum was cultured. Necrosis or inflammation directly related to bacteria was not present. Therefore, and also due to the age of the lesions (chronic granulomatous inflammation), the bacterial infection might be secondary to the microsporidiosis, not a predisposing factor for it, but might at least be the cause of death. The extent of postmortem bacterial overgrowth cannot be assessed. The isolated bacterial spectrum was heterogenic and comprised typical species for chelonians (eg, Streptococcus spp and different Enterobacteriaceae) but no obligatory pathogenic bacteria.
Clinical signs were recognized by the animals’ owners shortly after hibernation, and all animals in this report died in spring or early summer (April to June). The 4 that were sent to necropsy showed chronic inflammatory processes associated with microsporidia; the fifth necropsied animal died of Herpesvirus-induced pneumonia. Therefore, the animals with microsporidiosis could have acquired the infection before, during, or shortly after hibernation as a consequence of immunosuppression. Hibernation is supposed to decrease functions of the innate and adaptive immune systems and therefore could have promoted the infection. 1
Treatment with fenbendazole, which causes immunosuppression by damaging the gastrointestinal and hematopoietic systems, can also be a cause for overwhelming systemic bacterial and/or fungal infections in tortoises. 8 Three of the herein described 4 animals had been treated with fenbendazole previously; therefore, this could have promoted the microsporidial infection as well.
Microsporidiosis is generally treated with fenbendazole, which can cause the above-mentioned problems. 11 Alternatively, a treatment with albendazole can be considered. However, this is described as causing toxicosis in birds and therefore may also cause adverse effects in tortoises. 8 To prevent microsporidiosis, optimal husbandry, diet, and environment must be provided; other diseases shall be treated appropriately and sources of infections shall be eliminated. If a preventive treatment is to be given, this should happen before hibernation because the immune system is working better than after hibernation.
On postmortem histology, microsporidiosis is relatively easy to diagnose if it causes severe alterations, like in these cases. With silver stains, melanin pigments in melano-macrophages, which frequently occur in reptiles, might be confused with microsporidia. However, in HE stain, they can be clearly distinguished due to their morphology and size.
The herein described microsporidia could not be identified to the species level. The number of filament coils and their location (in 2 rows) point to the fact that the spores do not belong to Encephalitozoon spp. Spores were located within a vacuole, but it did not seem to be a parasitophorous vacuole, which supports the hypothesis that these microsporidia are not Encephalitozoon spp, which are surrounded by a parasitophorous vacuole. 2,13 Spores were not acid-fast in Ziehl-Neelsen stain and not refractive under polarized light, which contrasts with reports in the literature and could be a hint to a new species of microsporidia. 2,10 Further examinations, especially molecular biological investigations on the genome level (PCR), are planned from deparaffinized tissue.
Footnotes
Acknowledgements
We would like to thank Katharina Moser-Gomer, Sandra Wolf, Kerstin Rohn, and Deborah Basso for excellent technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
