Abstract
A 12-year-old Persian cat was examined for a firm swelling of the right tarsal region and enlargement of the corresponding right popliteal lymph node. Cytologic evaluation demonstrated a population of malignant cells consistent with large cell lymphoma. Necropsy revealed a multi-lobulated subcutaneous mass involving the tarsus with some extension into adjacent deep muscular tissue. Histologically, the mass was composed of round cells with eosinophilic cytoplasm and pleomorphic anisokaryotic nuclei. Evidence of articular and nodal infiltration by these cells was observed. Differential diagnoses included synovial sarcoma and histiocytic sarcoma. Neoplastic cells were negative for cytokeratin, CD79a, and CD3 and positive for CD18, vimentin, lysozyme, and alpha-1-antitrypsin, most consistent with a diagnosis of histiocytic sarcoma. This is the first report of a histiocytic sarcoma involving a joint of a cat. The final diagnosis was based on the light microscopic appearance in combination with the immunohistochemical stains.
Malignant histiocytic tumors have been reported in various species with few cases observed in cats. The majority of these tumors in cats were disseminated and involved organs such as skin, liver, spleen, lymph nodes, and bone marrow with anemia and thrombocytopenia as common hematologic findings. 9 Histiocytic sarcoma (HS) as a solitary tumor has been rarely described in cats and reports to date include an intramural intestinal mass, a femoral mass, and a mediastinal and vertebral canal HS that resulted in a secondary myelopathy. 2, 10, 11 A brief description of an HS of the central nervous system of a cat was also included in a review article of intraoperative tumor diagnosis in dogs and cats, but without much clinical detail. 12 To the authors' know ledge, this is the first report of an HS involving the joint of a cat. The definitive diagnosis of the tumor in this case was established by using immunohistochemical stains.
A 12-year-old, 4.7 kg, neutered male Persian cat was presented to the Centre Hospitalier Universitaire Vétérinaire (CHUV) for evaluation of a severely swollen right hind limb and associated 3-week duration lameness. On presentation, the cat was alert and responsive with normal vital parameters. Physical examination findings included severe swelling of the right tarsus extending distally, and an enlarged right popliteal lymph node. A subsequent radiographic evaluation revealed severe soft-tissue swelling of the right tarsus, metatarsus, and pes; with the tarsus being most severely affected. There was a mild, slightly mottled decrease in bone density compatible with non–weight-bearing osteoporosis and/or minimal lysis.
A fine-needle aspiration biopsy of the tarsal swelling was obtained. The smears were air dried and stained with a 2-part Romanowski stain (Wescor Inc., Logan, UT). They were highly cellular and were composed principally of individualized, large round cells measuring 15 to 30 μm in diameter that were often aligned in rows (Fig. 1). These cells were characterized by small amounts of finely granular, dark basophilic cytoplasm, which on occasion contained small vacuoles, variably sized nuclei that were round to indented or cleaved and often multi-lobulated, with a coarsely granular to clumped chromatin containing 1 to several prominent nucleoli of varying size. Binucleation was frequently noted. Rare mitotic figures were present. Other cell types present included small mature lymphocytes and occasional vacuolated macrophages. The background contained several small, round, lightly basophilic structures consistent with lymphoglandular bodies. Occasional small clumps of an eosinophilic amorphous material consistent with cartilage fragments were also observed. Based on cytologic evaluation, a large cell lymphoma involving the joint was suspected. Because of the severity of the disease and the age of the cat, the owners elected euthanasia, and a necropsy was performed.
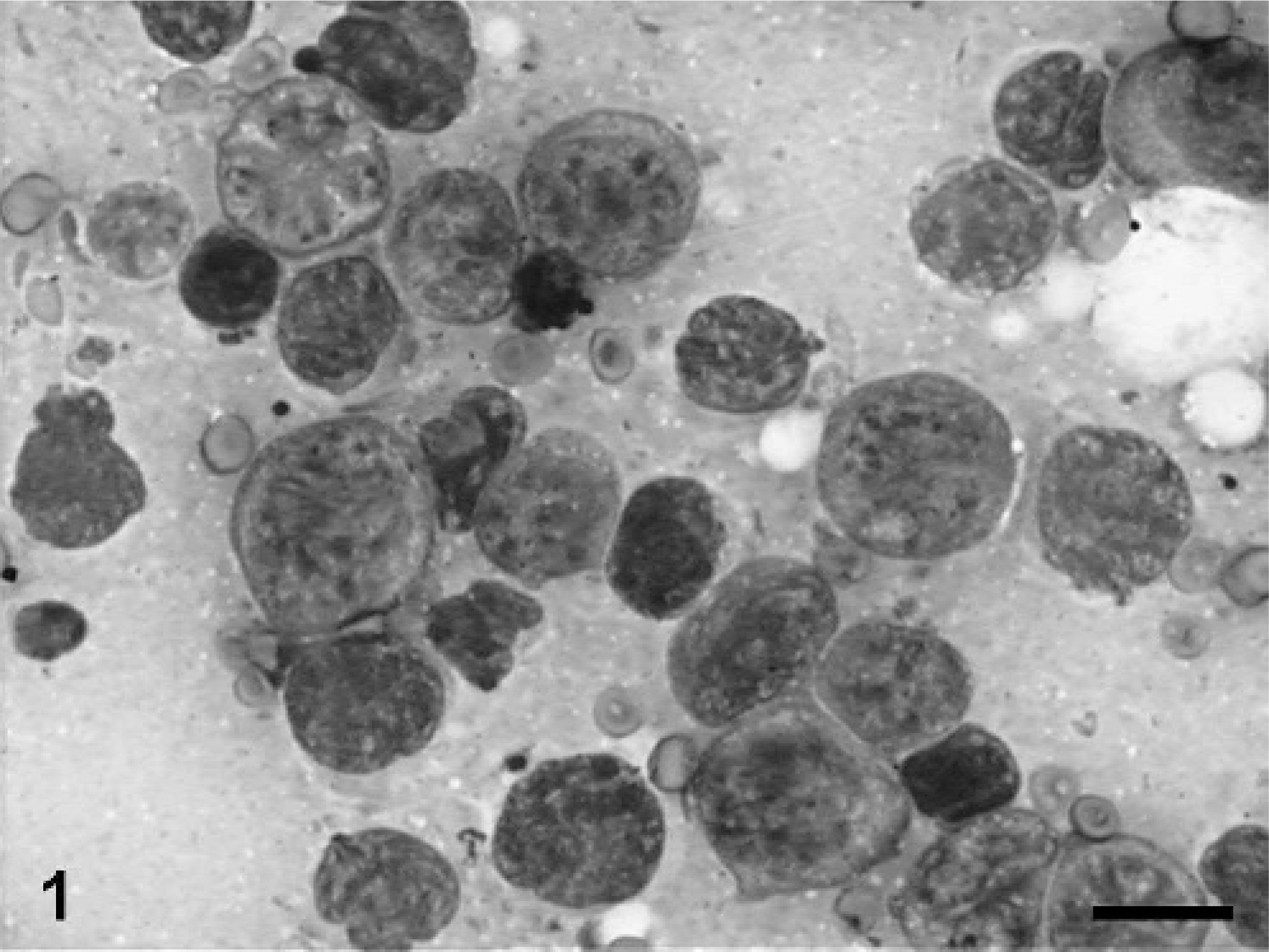
Tarsal mass; cat. Fine needle aspiration showing large round tumor cells with pleomorphic nuclei, prominent nucleoli, and marked anisocytosis and anisokaryosis. Bar = 15 μm.
At necropsy, the right tarsus enlargement corresponded to a firm, subcutaneous, whitish multi-lobulated mass composed of 2 main parts, one lateral (5.5 × 3.0 × 1 cm) and one medial (4.5 × 2.5 × 1 cm) to the articulation, linked caudally by a thin band of the same tissue. A third part was found deeply embedded between the muscles following the proximal third of the tibia. Articular involvement was not detectable grossly. The corresponding popliteal lymph node was slightly enlarged. Other findings included multiple, small hepatic and renal cysts (less than 0.5 cm in diameter).
Histologically, the mass was partially encapsulated and multi-lobular. It compressed surrounding tissues. Neoplastic cells formed densely cellular sheets and sometimes cords delineated by a fine fibrous stroma. Cells were round, approximately 10-μm diameter, with distinct cytoplasmic borders and scant, homogenous, eosinophilic cytoplasm. Nuclei were centrally located, round to oval or sometimes kidney-shaped with moderate pleomorphism and little karyomegaly. Chromatin was finely to coarsely stippled. The nucleolus was not evident. Some binucleated cells and rare multi-nucleated cells were present. Mild anisocytosis and anisokaryosis were observed. Mitoses ranged from 3 to 4 per 40× objective field. There were extensive coalescing foci of necrosis mainly in the center of the mass. A few clusters of well differentiated phagocytes were scattered through the mass. Lymphocytic infiltration was observed mostly at the periphery of the mass. The synovial membrane of the tarsus appeared hyperplastic and formed villi infiltrated by tumor cells (Fig. 2). A 2-mm focus of neoplastic cell infiltration associated with bone necrosis was present in one of the right tarsal bones. Proximal to the right tarsal articulation, the muscles and a nerve sheath were infiltrated by the same neoplastic cells. The popliteal lymph node was multi-focally infiltrated. There was no evidence of neoplastic cells in other tissues including brain, eye, thyroid gland, lung, heart, liver, spleen, small and large intestines, adrenal glands, kidney, and bladder; bone marrow appeared normal.
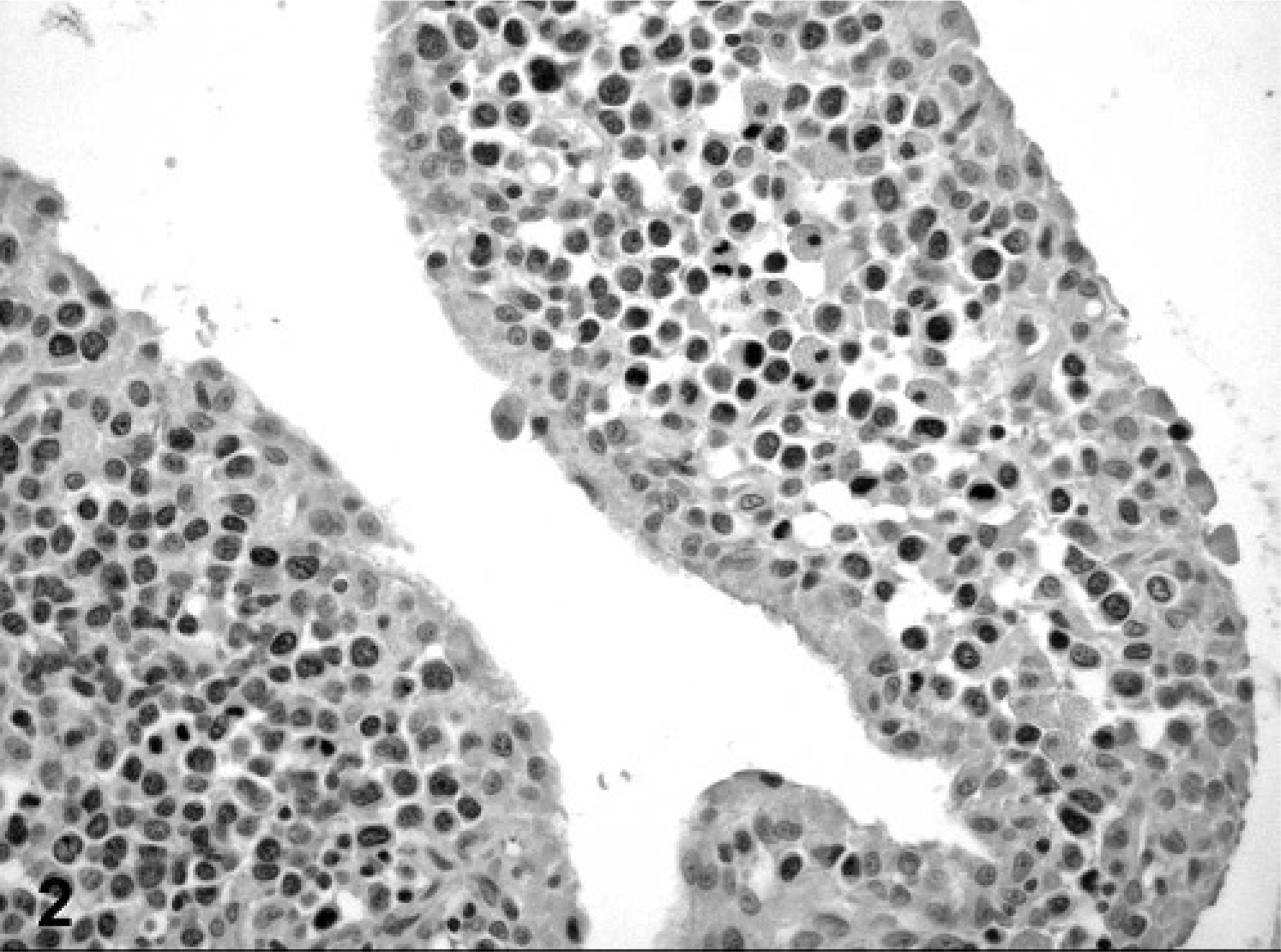
Tarsal mass; cat. Neoplastic cells infiltrating synovial membrane. Immunohistochemistry shows negative reaction for cytokeratin.
In view of the histologic findings, 2 main diagnoses were considered: synovial sarcoma and histiocytic sarcoma, although other round cell tumors such as lymphoma could not be excluded. Representative sections of neoplastic tissue were fixed in 10% neutral-buffered formalin and routinely processed for immunohistochemistry. For immunohistochemical examination, sections of neoplastic tissue were deparaffinized and rehydrated by routine methods. 8 Antigen retrieval was accomplished by incubation of slides in antigen retrieval solution (Dako, Carpinteria, CA) in a steamer (Black & Decker, Hunt Valley, MD) for 20 minutes. Endogenous peroxidase was blocked for 15 minutes with 3% hydrogen peroxide. Nonspecific immunoglobulin binding was blocked by incubation of slides for 10 minutes with a protein-blocking agent (Dako) before application of the primary antibody. The latter was allowed to react for 30 minutes at room temperature. Sections were stained in a Dako autostainer apparatus. The slides were incubated with a mouse monoclonal antivimentin antibody (Dako, dilution of 1°50), a mouse monoclonal antipancytokeratin antibody (MNF116, Dako, protein digestion, dilution of 1°800), a mouse monoclonal anti-CD79a antibody (Dako, dilution of 1°100), a rabbit polyclonal anti-alpha 1-antitrypsin antibody (Dako, dilution of 1°100), a rabbit polyclonal antilysozyme antibody (Dako, 60 minutes retrieval, dilution of 1°1000), a rat monoclonal anti-CD3 antibody (Peter Moore, dilution 1°10), and a mouse monoclonal anti-CD18 antibody (Peter Moore, dilution 1°10). A labeled streptavidin-biotin-immunoperoxidase staining procedure (Dako) was used for immunolabeling. The immunoreaction was visualized with 3,3′-diaminobenzidine substrate (Dako). Sections were counterstained with Mayer's hematoxylin counterstain. Positive immunohistochemical controls included a normal feline lymph node to which the appropriate antisera were added. For negative controls, the primary antibodies were replaced with homologous nonimmune sera. Staining for cytokeratin was negative (Fig. 2) and staining for CD79a (Fig. 3) and CD3 (Fig. 4) revealed scattered B-cells and T-cells throughout the tumor, but the primary neoplastic population was negative for all 3 markers. The neoplasm was diffusely and strongly positive for vimentin, CD18 (Fig. 5) and less intensely positive for lysozyme (Fig. 6) and alpha-1 antitrypsin. Based on the microscopic appearance and the described immunohistochemical staining characteristics, a diagnosis of localized HS was made.
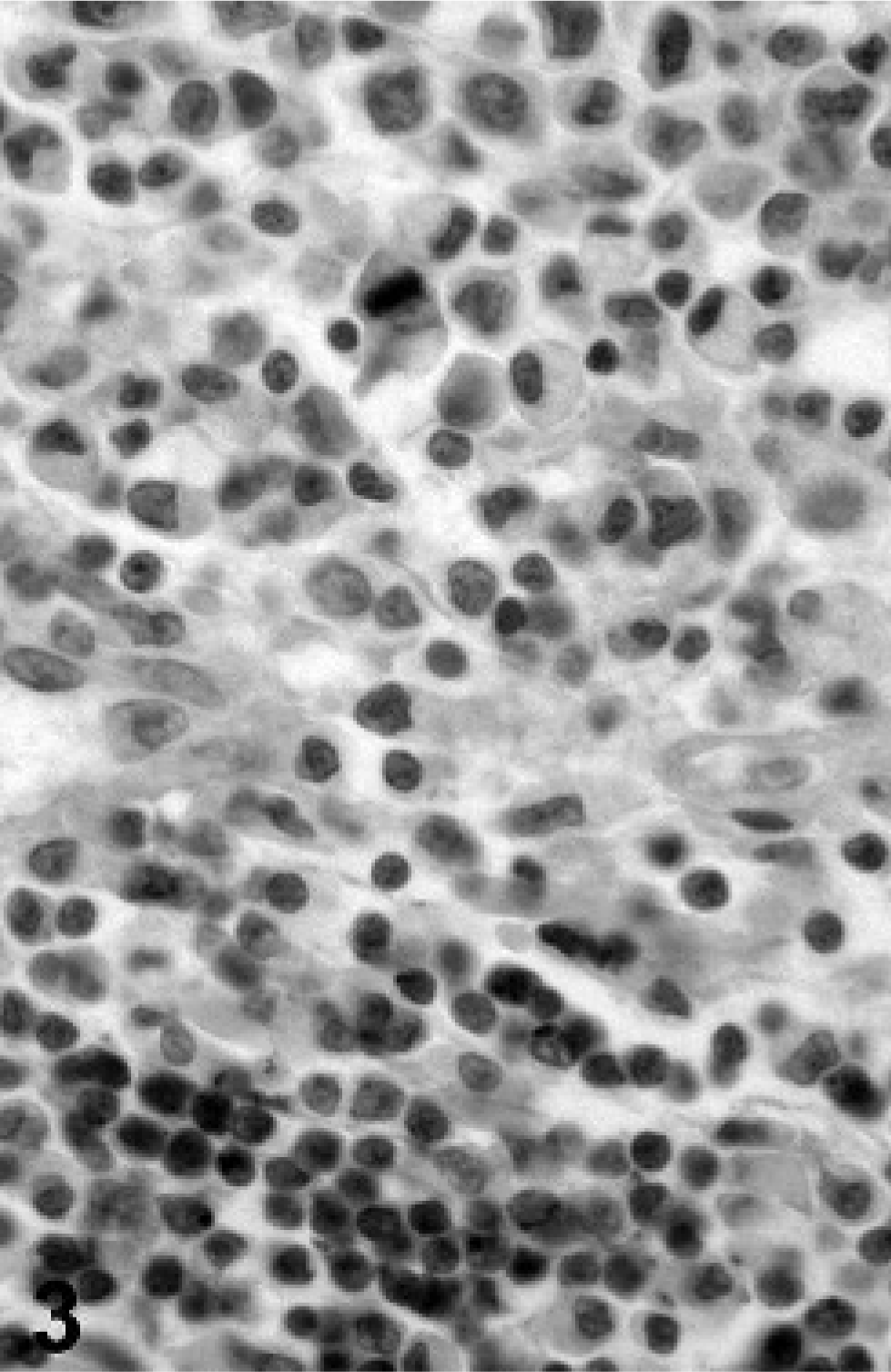
Tarsal mass; cat. Immunohistochemistry shows negative reaction of tumor cells for CD79a. Small lymphocytes act as positive control.
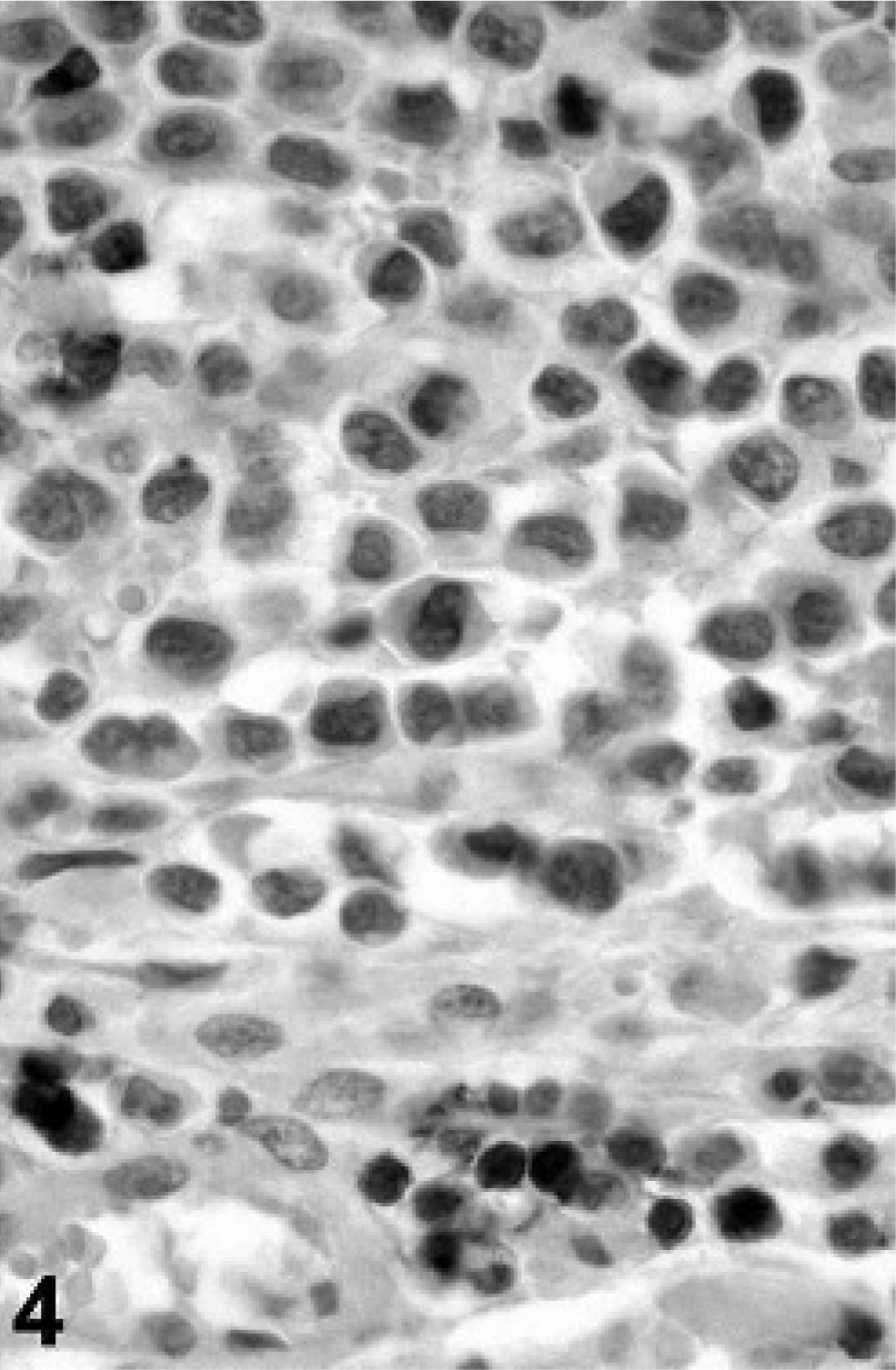
Tarsal mass; cat. Immunohistochemistry shows negative reaction of tumor cells for CD3. Small lymphocytes down the picture act as positive control.
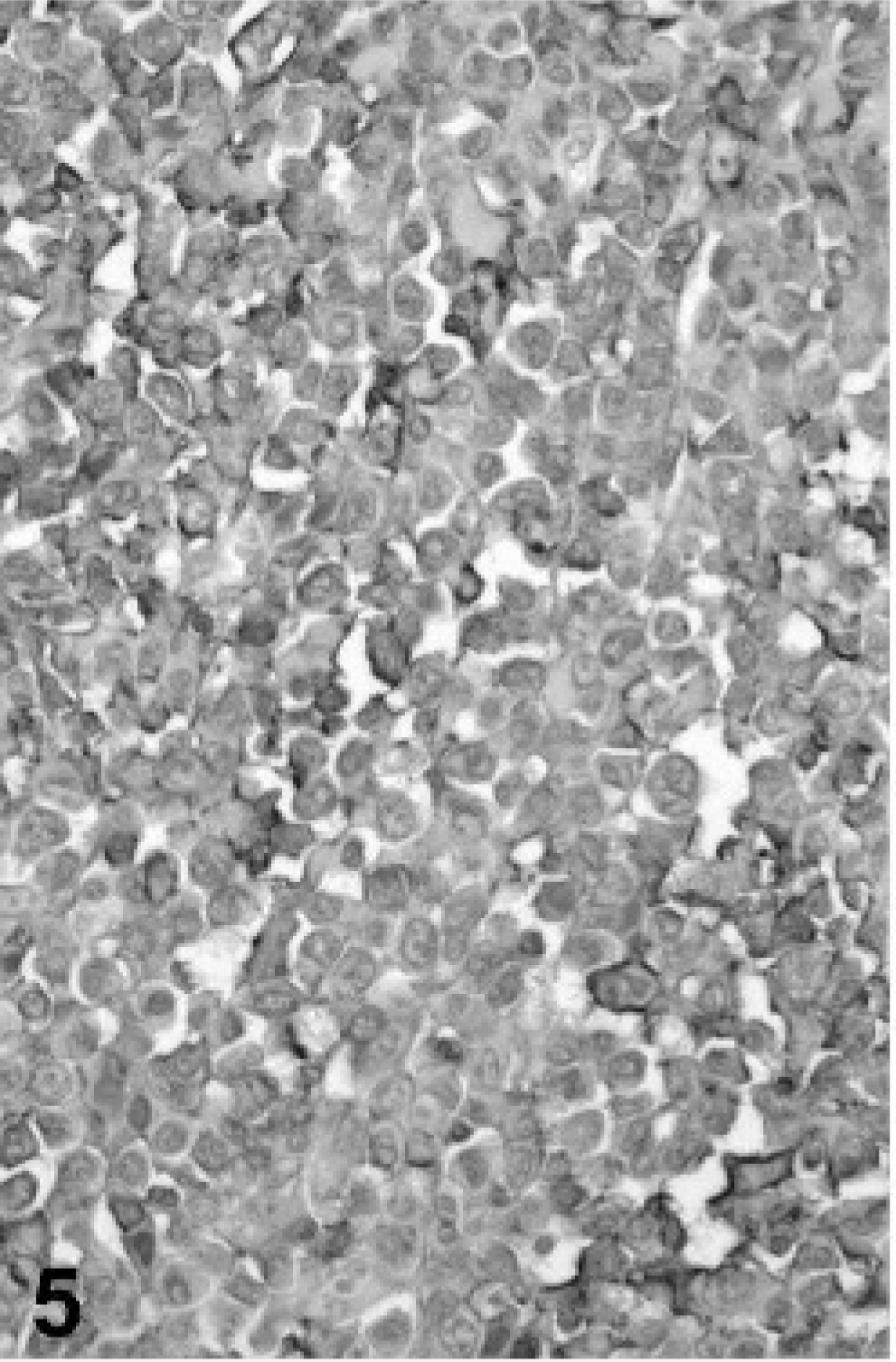
Tarsal mass; cat. Immunohistochemistry shows positive reaction of tumor cells for CD18.
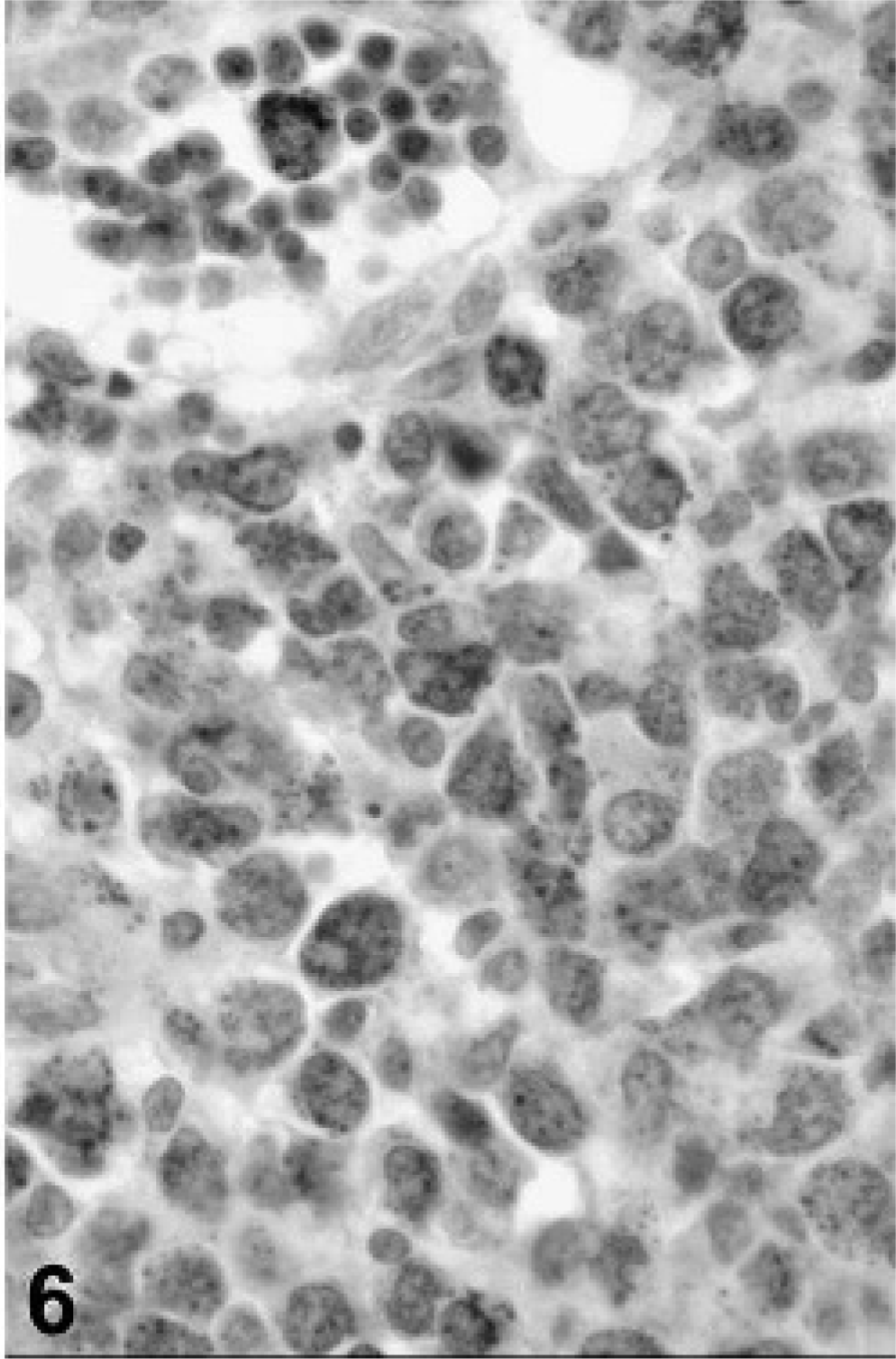
Tarsal mass; cat. Immunohistochemistry shows positive reaction of tumor cells for lysozyme.
The previously reported cases of HS in cats involved the mediastinum, intestine, and the central nervous system. This is to our knowledge the first report of an articular involvement of a localized HS in a cat. Conversely, in the dog most HS originate from the subcutis and underlying tissues of the extremities. 1 A retrospective study of periarticular neoplasms in dogs identified HS as the most common neoplasm in this location. 5
In dogs, localized and disseminated HS are characterized by identical morphologic features, 1 which include moderate to marked cellular pleomorphism, abundant, faintly eosinophilic and finely vacuolated to granular cytoplasm, and round to bean-shaped (pleomorphic) nuclei that usually contain a prominent nucleolus. Phagocytosis and multi-nucleated cells are usually seen, although they are not always prominent features. 2, 4, 6, 9– 13 In this cat, the cytoplasm of the neoplastic cells was scant, there was little evidence of phagocytosis, and multi-nucleated giant cells were rare. Because of the absence of typical morphologic features of HS, immunohistochemistry was used to confirm the diagnosis. In dogs, CD1, CD11b, CD11c, CD14, CD68, and MHC class II are considered the most reliable markers for identifying cells of histiocytic origin. 1 Furthermore, it is possible to distinguish dendritic cells (CD1+, CD11c+) from macrophages (CD11b+, CD14+, CD68+). 1 Most canine histiocytic tumors are of dendritic cell origin, with macrophage origin being an unusual finding. 1 Unfortunately, most of these antibodies are either not available for cat tissues or cannot be applied to formalin-fixed material. If only formalin-fixed tissues are available, CD3, CD79a, and CD18 as well as morphologic features can be used to identify nonlymphoid monocytic neoplasms. 1, 5 In this cat, the neoplastic cells were negative for CD3 (T-cells) and CD79a (B-cells) but were positive for CD18 (monocytic cells), most consistent with a nonlymphoid monocytic neoplasm. Positive CD18 staining also excluded a synovial sarcoma. Furthermore, neoplastic cells were positive for lysozyme and alpha-1 antitrypsin, both well-characterized immunohistochemical markers of mononuclear phagocyte differentiation. 1, 3, 7
Based on the described microscopic features and the positive staining of neoplastic cells for CD18, lysozyme, and alpha-1 antitrypsin and lack of staining with pancytokeratin, CD3, and CD79, the diagnosis of a periarticular HS was made. For future cases, frozen sections of neoplastic tissue should be collected to further characterize the neoplastic cell type (CD1a, CD11c, CD11b, CD11d, and MHC class II) of such lesions.
Footnotes
Acknowledgements
We thank Dr. Pascal Surugue for submission of this clinical case and for providing clinical information. We would also like to thank Dr. Kate Alexander and Dr. Luc Breton for their invaluable aid in the radiographic interpretation of this case.
