Abstract
Four cases of histiocytic sarcoma in domestic ferrets (Mustela putorius furo) are described in the present study. Tumor samples obtained from the abdominal viscera, including the spleen, were submitted for histologic examination. Microscopically, poorly demarcated masses contained numerous round- to pleomorphic-shaped cells with coarsely vacuolated and eosinophilic cytoplasm. Bizarre, binucleated tumor cells and multinucleated giant tumor cells with low phagocytic activity were commonly observed. Immunohistochemically, tumor cells in all of the cases were positive for vimentin, human leukocyte antigen-DR, ionized calcium-binding adapter molecule-1, and lysozyme, but some of them lacked cluster of differentiation (CD)163 or CD208 expression. The survival time after surgical resection was 9 days to 5 months. Histiocytic sarcoma in the ferret is a rare, but highly aggressive, tumor commonly found in the spleen.
The domestic ferret (Mustela putorius furo) has been extensively used for many purposes in the medical research field (e.g., pharmacology, toxicology, and virology) over the past decade. Genetically modified ferrets are also used as models of human disease research. 25 Likewise, it has become a popular companion animal worldwide. Hence, neoplastic diseases in ferrets have been increasingly studied. Endocrine tumors (i.e., pancreatic islet cell tumor and adrenal cortical carcinoma) and lymphomas are common in this species.2,3,7,15,16,23,24
Histiocytic sarcoma is a fatal, aggressive malignant hematopoietic tumor of histiocytes. This tumor has been well described in animals other than humans, especially in dogs and cats.1,8,9,11,19,20 Moreover, cases in horses, 22 cattle,4,17 a rabbit, 14 mice,21,26 and a gerbil 5 have also been reported. In a previous study, 18 the occurrence of histiocytic sarcoma in ferrets was <0.1% (only 1 histiocytic sarcoma case in 945 tumor cases). In our study, histological and immunohistochemical features of histiocytic sarcoma in 4 domestic ferrets are described.
Four tumor samples were obtained from 3 surgical biopsy cases and 1 autopsy case, which were submitted to the Department of Veterinary Pathology, the University of Tokyo, Japan, for histologic examination between 2010 and 2015 (Table 1). The samples, including spleen, stomach, adrenal gland, liver, epigastric and mesenteric lymph nodes, and free-moving abdominal mass were fixed in 10% neutral buffered formalin solution and subsequently processed for paraffin embedding. The sections (2–4-µm thick) were stained with hematoxylin and eosin. To evaluate the mitotic index, the number of mitosis per 10 high power fields (HPFs, 400×) was counted under a light microscope.
Four cases of histiocytic sarcoma in the domestic ferret (Mustela putorius furo).*
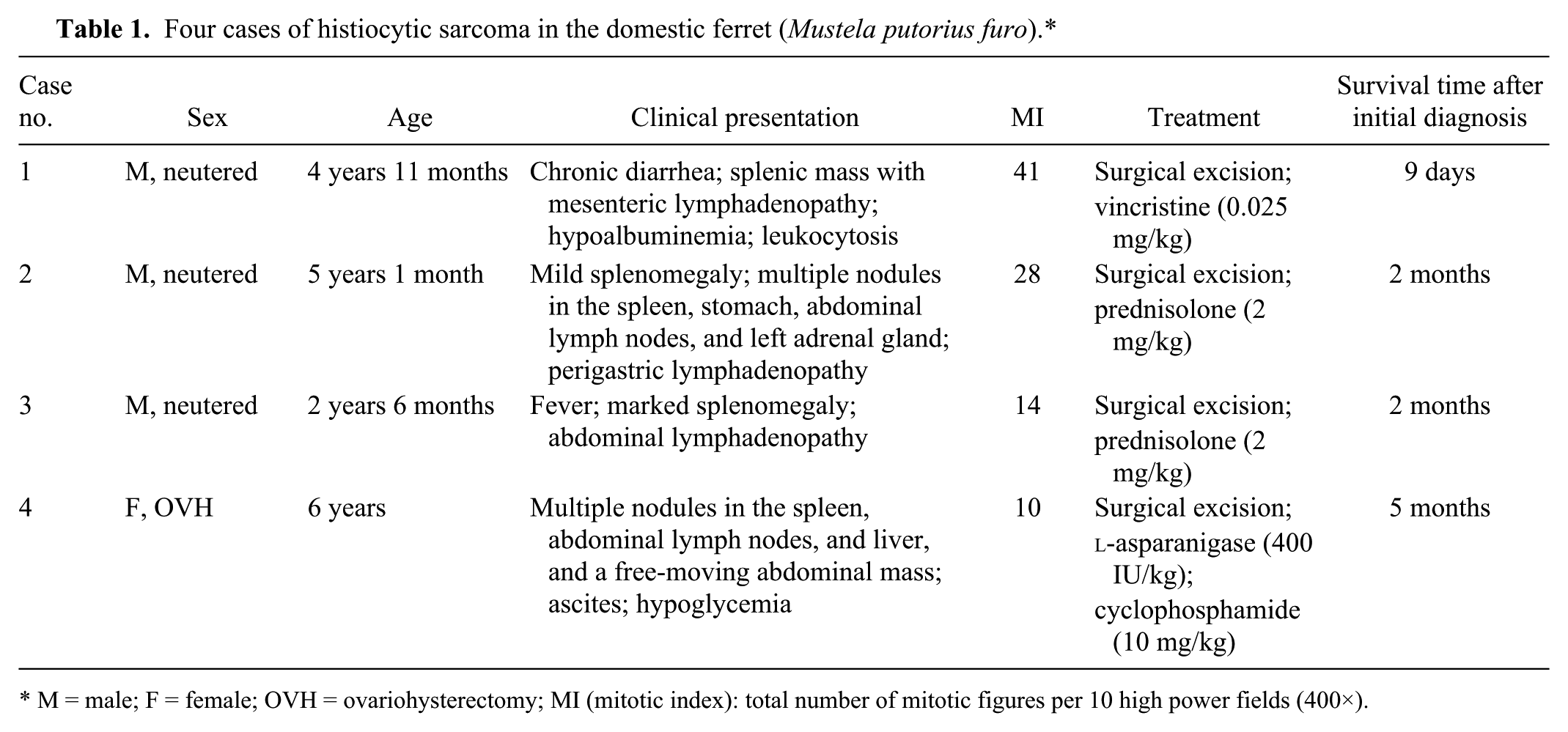
M = male; F = female; OVH = ovariohysterectomy; MI (mitotic index): total number of mitotic figures per 10 high power fields (400×).
In order to identify the cellular origin of tumor cells, immunohistochemical examination was performed. Nonspecific reactions were blocked with 10% hydrogen peroxide (H2O2) in methanol and subsequent 8% skimmed milk in Tris-buffered saline (TBS) prior to applying primary antibodies (Table 2). Tissue sections were then incubated with horseradish peroxidase (HRP)–labeled polymer reagent. a As for the detection of cluster of differentiation (CD) 208, biotinylated secondary antibody (1:400, anti-rat immunoglobulin G (Heavy + Light chains) antibody) b and then streptavidin–HRP reagent c (1:300) were applied. In order to visualize the immunoreactions, DAB (3-3′-diaminobenzidine) solution containing 0.03% H2O2 was employed. All slides were counterstained with Mayer hematoxylin. d Semi-quantitative scoring (– = no positive tumor cells; + = 1–25% positive tumor cells; ++ = 25–50% positive tumor cells; +++ = 50–75% positive tumor cells; ++++ = >75% positive tumor cells) was employed to assess the immunoreactivity of each marker. In addition, normal ferret tissues, including spleen, liver, and lymph node, were also used as positive controls.
Antibodies used for immunohistochemistry.*
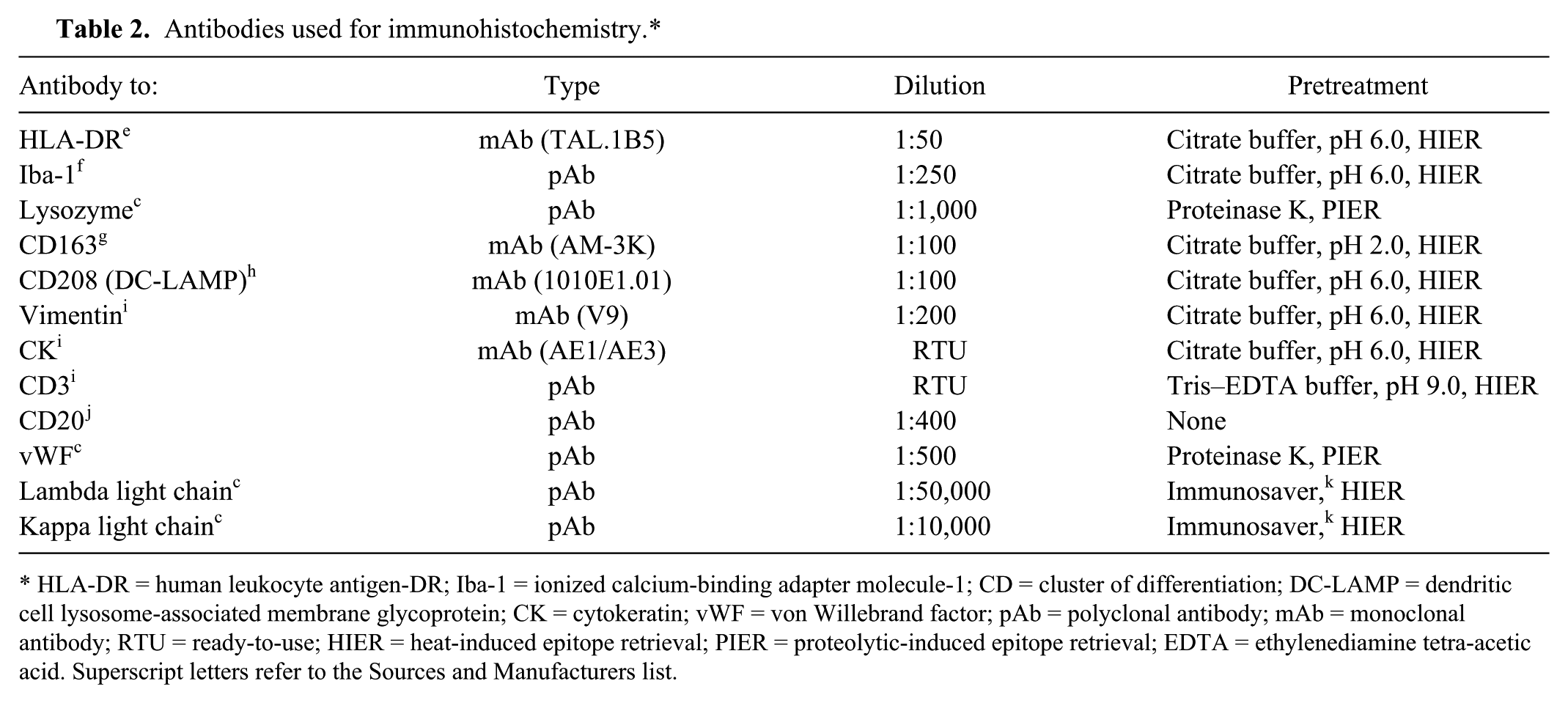
HLA-DR = human leukocyte antigen-DR; Iba-1 = ionized calcium-binding adapter molecule-1; CD = cluster of differentiation; DC-LAMP = dendritic cell lysosome-associated membrane glycoprotein; CK = cytokeratin; vWF = von Willebrand factor; pAb = polyclonal antibody; mAb = monoclonal antibody; RTU = ready-to-use; HIER = heat-induced epitope retrieval; PIER = proteolytic-induced epitope retrieval; EDTA = ethylenediamine tetra-acetic acid. Superscript letters refer to the Sources and Manufacturers list.
Macroscopically, major lesions were found in the abdominal cavity in all of the ferret cases, which included splenomegaly and white to gray, noncircumscribed masses scattered throughout the splenic parenchyma (Fig. 1). Marked splenic necrosis was found in cases 2 and 3. Furthermore, solitary or multiple masses in the regional lymph nodes (cases 2–4; Fig. 2), left adrenal gland (case 2), stomach (case 2), and liver (case 4) were also noted. Free-moving abdominal mass with ascites was present in case 4.
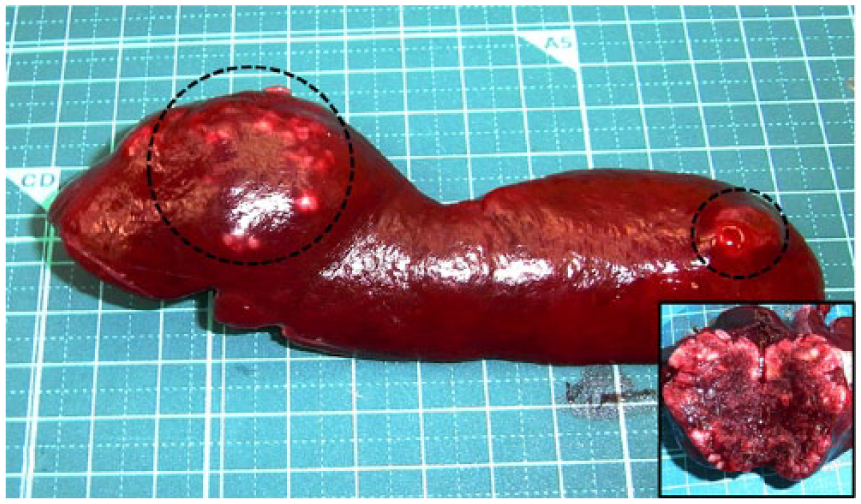
Ferret (Mustela putorius furo; case 2); spleen. Splenomegaly with poorly demarcated nodules (dotted circles). The cut surface of the nodule is whitish to red, indicating hemorrhage and necrosis (inset).
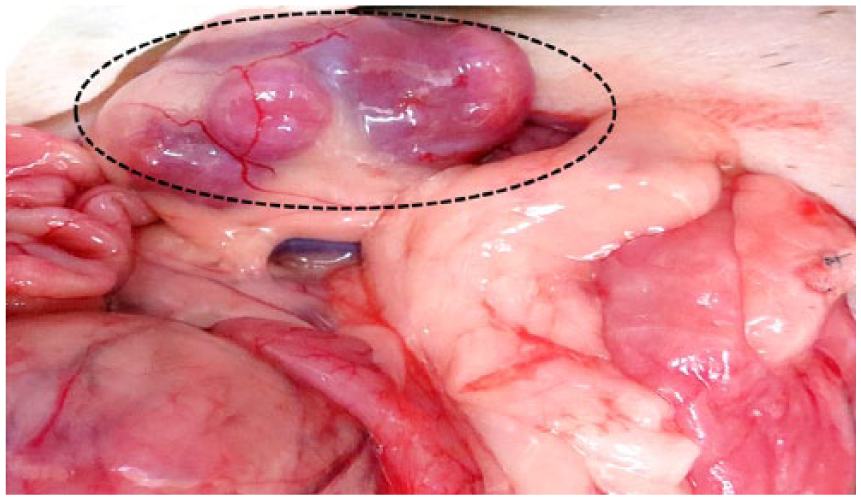
Ferret (Mustela putorius furo; case 3); abdominal viscera. Mesenteric lymphadenopathy (dotted circle).
Microscopic findings of the spleen were common. Poorly demarcated and nonencapsulated tumor masses comprised numerous round to pleomorphic cells of various sizes, often arranged in a loose sheet pattern (Fig. 3). Tumor cells had abundant pale eosinophilic cytoplasm, some of which was coarsely vacuolated, and contained round to ovoid nuclei with fine to coarsely stippled chromatin patterns. One to 3 prominent nucleoli were observed. Atypical mitotic figures with highly variable mitotic index were also noted (Table 1). In addition, interspersing of bizarre, binucleated tumor cells and multinucleated giant tumor cells was commonly found among mononuclear tumor cells. A few tumor cells engulfed red blood cells and granulocytes (Fig. 3). Marked necrosis and moderate lymphocytic infiltration were also noted. Likewise, tumor cells in other organs (i.e., stomach, adrenal gland, liver, and free-moving abdominal mass) were morphologically identical to those of the spleen (Fig. 4). Tumor cell invasion into blood vessels was occasionally seen in cases 2–4. Furthermore, metastases to the sentinel lymph nodes, including perigastric and mesenteric nodes, were found in cases 2–4 (Fig. 5).
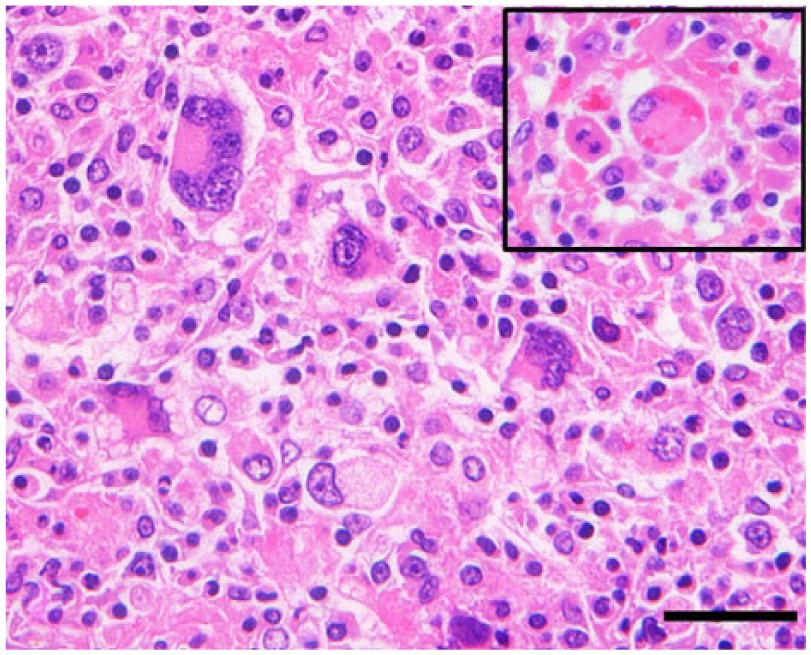
Histiocytic sarcoma; ferret (Mustela putorius furo; case 2); splenic mass. Neoplastic histiocytes are round to polygonal-shaped and characterized by eosinophilic cytoplasm and cytoplasmic vacuolation. Many multinucleated giant tumor cells are also observed. Few tumor cells show erythrophagocytosis (inset). Hematoxylin and eosin. Bar = 20 µm (including inset).
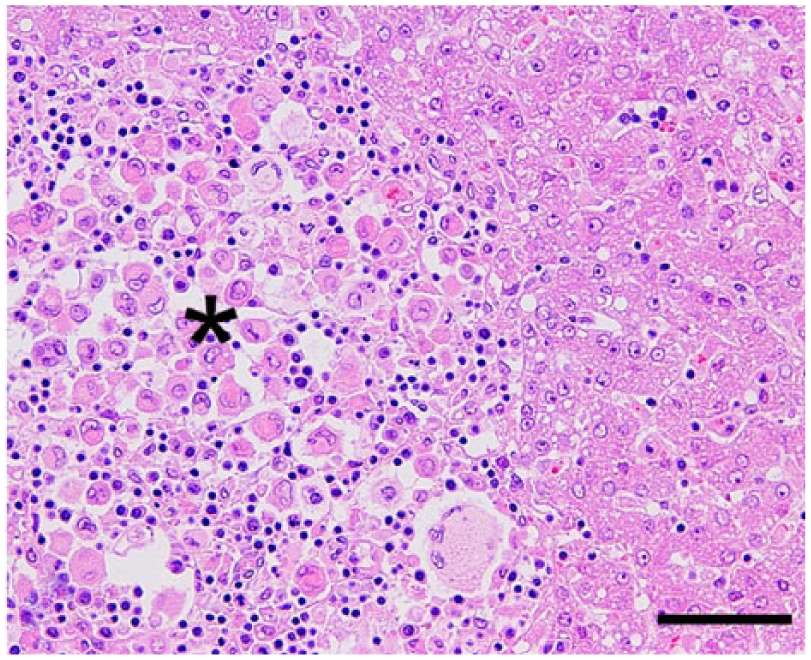
Histiocytic sarcoma; ferret (Mustela putorius furo; case 4); liver. Neoplastic histiocytes admixed with lymphocytes in the expanded hepatic sinusoid (asterisk). Many multinucleated giant tumor cells are also observed. Hematoxylin and eosin. Bar = 50 µm.
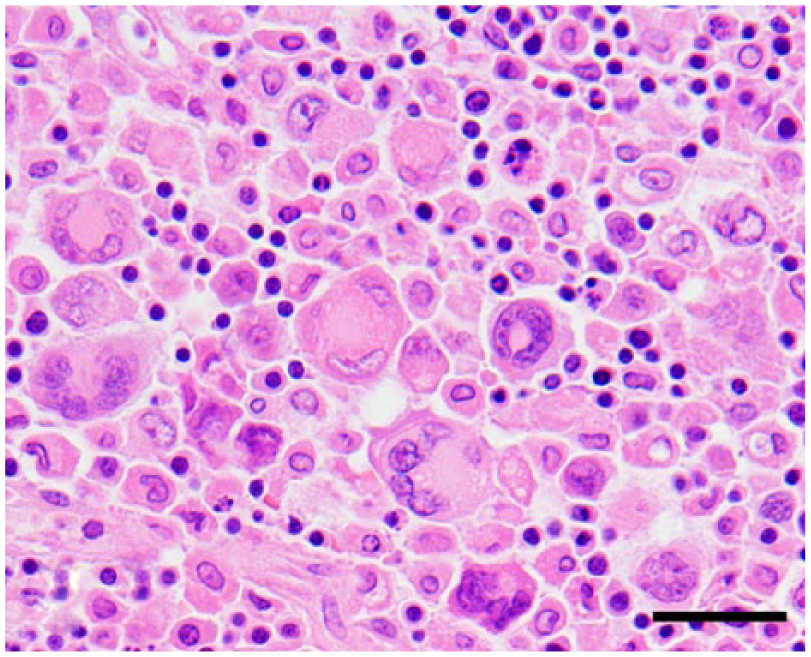
Histiocytic sarcoma; ferret (Mustela putorius furo; case 4); perigastric lymph node. Morphological characteristics of neoplastic histiocytes are similar to those in the liver. Hematoxylin and eosin. Bar = 20 µm.
Immunohistochemical examination revealed that the cytoplasm of the tumor cells was strongly immunopositive for vimentin and human leukocyte antigen–DR (HLA-DR; all cases). Cytoplasmic expression of ionized calcium-binding adapter molecule–1 (Iba-1) and lysozyme was also observed in almost all tumor cells (all cases). Positive granular cytoplasmic and membrane immunoreactivities to CD163 were variably distributed in cases 1, 3, and 4. In addition, cases 3 and 4 had a patchy dot-like cytoplasmic immunoreactivity to CD208 (Fig. 6; Table 3). Tumor cells were negative for cytokeratin, CD3, CD20, lambda and kappa light chains, and von Willebrand factor in all of the cases. Likewise, the expression of histiocytic markers in tissue resident macrophages was identical to that of the present tumor cells (Fig. 6, insets).
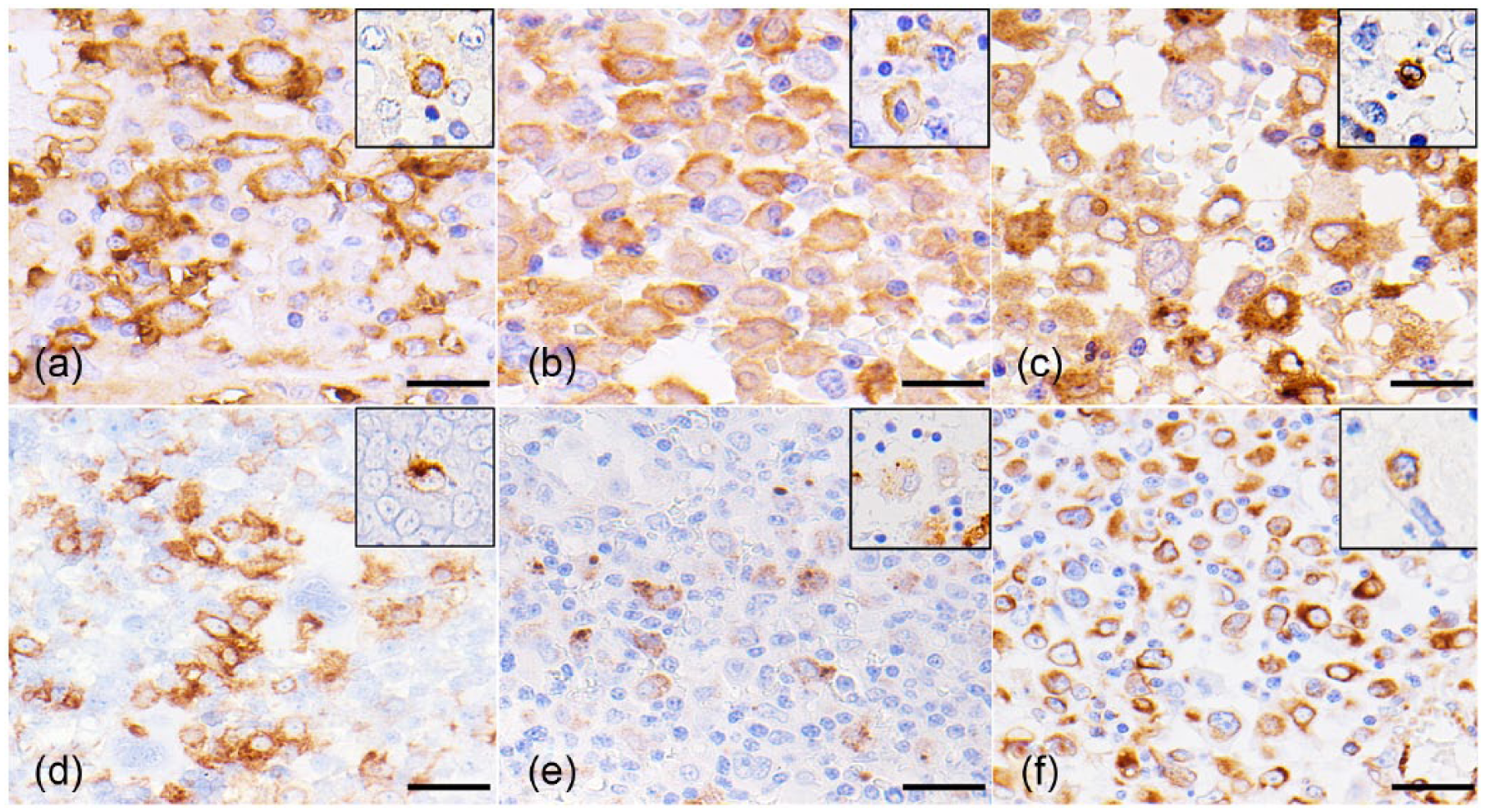
Histiocytic sarcoma; ferret (Mustela putorius furo; case 3); spleen. Tumor cells are positive for HLA-DR (
Results of immunohistochemistry.*
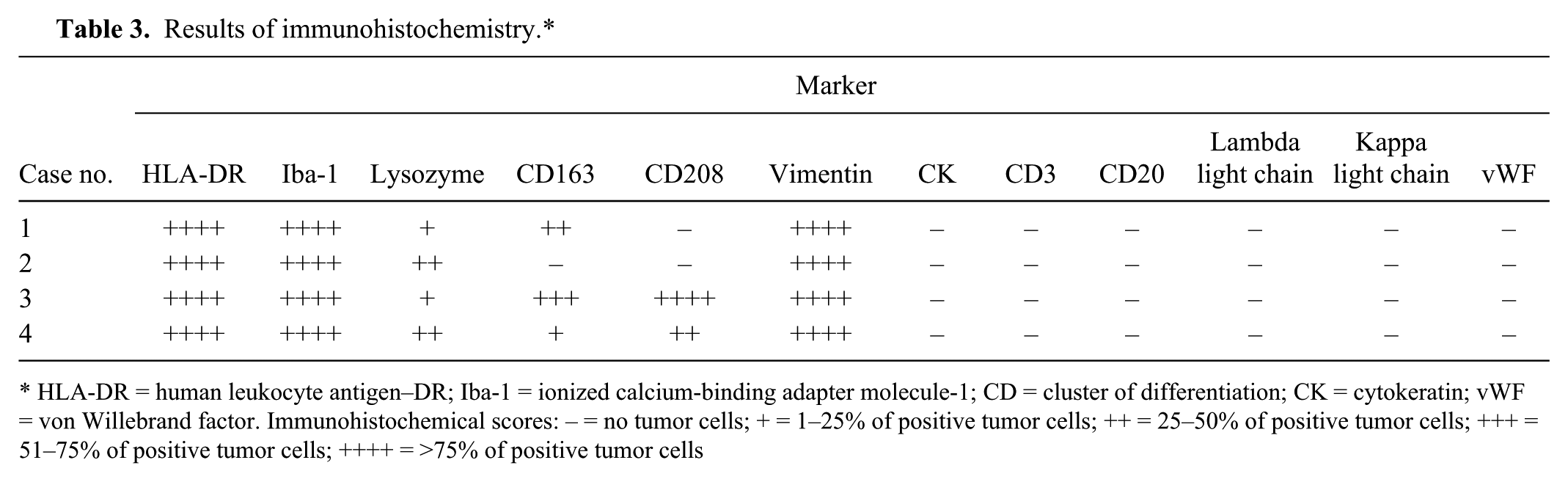
HLA-DR = human leukocyte antigen–DR; Iba-1 = ionized calcium-binding adapter molecule-1; CD = cluster of differentiation; CK = cytokeratin; vWF = von Willebrand factor. Immunohistochemical scores: – = no tumor cells; + = 1–25% of positive tumor cells; ++ = 25–50% of positive tumor cells; +++ = 51–75% of positive tumor cells; ++++ = >75% of positive tumor cells
According to histological and immunohistochemical results, tumor cells in the present cases possessed characteristics of histiocytic differentiation. Therefore, a diagnosis of histiocytic sarcoma was made.
Histiocytic proliferative disorders have been well documented in domestic animals, particularly in dogs. Canine histiocytic sarcoma is generally categorized into 3 types (canine cutaneous histiocytoma, canine reactive histiocytosis, and histiocytic sarcoma), according to clinical and pathological features. 9 Histiocytic sarcoma in dogs, which is recognized as a malignant form of histiocytic tumors, can be subgrouped into localized and disseminated forms based on the number of primary organs and/or the evidence of distant metastases.1,9,19 Histiocytic sarcoma in ferrets can be speculatively categorized into localized (cases 1 and 3) and disseminated (cases 2 and 4) forms, as well.
As mentioned previously, histiocytic tumor in ferrets was first described in the integumentary system. 18 However, in 3 of the 4 present cases, the tumor lesions were confined to the abdominal cavity, especially to the spleen (cases 1, 3, and 4), and only 1 case (case 2) exhibited extra-abdominal lesions (adrenal gland).
Splenomegaly observed in middle-aged or older ferrets is generally associated with extramedullary hematopoiesis. 10 Moreover, such splenic tumors as lymphoma and hemangiosarcoma are also mentioned as causes of splenic enlargement in this species. As cytopathological and histological characteristics of histiocytic sarcoma and other undifferentiated large cell tumors, such as lymphoma, metastatic malignant melanoma, and undifferentiated carcinoma, are often similar, further immunohistochemical examinations are recommended in order to confirm the diagnosis.
Tumor cells of all of the ferrets examined in our study exhibited immunoreactivity to histiocytic markers, such as HLA-DR, Iba-1, and lysozyme. However, the lack of CD163 or CD208 expression was found in cases 1 and 2 (Table 3). In general, CD163 or a monocyte and/or macrophage-restricted membrane protein is expressed exclusively in the monocyte and/or macrophage lineage, particularly in the M2 macrophage phenotype.12,13 This marker is also used for confirming the diagnosis of histiocytic sarcoma in human and canine cases. Most of the canine histiocytic sarcoma cases originate from the dendritic cell lineage, whereas some dominantly exhibit the monocyte and/or macrophage phenotype and are called hemophagocytic histiocytic sarcomas.19,20 Histological features of hemophagocytic histiocytic sarcoma include a marked erythrophagocytosis by neoplastic histiocytes interspersing among the areas of extramedullary hematopoiesis. Tumor cells in 3 of the present ferret cases (cases 1, 3, and 4) had immunoreactivity to CD163, which supports the monocyte and/or macrophage phenotype, though very few tumor cells showed erythrophagocytosis.
CD208 or dendritic cell lysosome-associated membrane glycoprotein (DC-LAMP) is generally expressed in human dendritic cells and is closely associated with the maturation and activation of the cells. 6 In our study, tumor cells in cases 3 and 4 were positive for CD208, suggesting that the tumor cells originated from dendritic cells. Moreover, neoplastic histiocytes of ferrets are also positive for other macrophage markers, like in dogs. 19 Conversely, tumor cells in case 2 were negative for CD163 and CD208 reported for humans.6,12 This indicates that the expression levels of these 2 markers may associate with cell differentiation and maturation of the neoplastic histiocytes. Because the survival time of the ferrets with histiocytic sarcoma after treatment was very short (9 days to 5 months; Table 1), prognosis of histiocytic sarcoma in this species is severe and aggressive based on post-diagnosis longevity.
Footnotes
Authors’ contributions
A Thongtharb contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. K Uchida and JK Chambers contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and critically revised the manuscript. Y Miwa contributed to acquisition and interpretation of data and critically revised the manuscript. Y Murata contributed to interpretation of data and critically revised the manuscript. H Nakayama contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
EnVision+ System-HRP labeled polymer reagent, Dako North America Inc., Carpinteria, CA.
b.
Vector Laboratories Inc., Burlingame, CA.
c.
Dako Denmark A/S, Glostrup, Denmark.
d.
Muto Pure Chemicals, Tokyo, Japan.
e.
Santa Cruz Biotechnology Inc., Santa Cruz, CA.
f.
Wako Pure Chemical Industries, Osaka, Japan.
g.
TransGenic, Kobe, Japan.
h.
Dendritics, Lyon, France.
i.
Dako Japan, Tokyo, Japan.
j.
Thermo Scientific, Life Technologies, Grand Island, NY.
k.
Nisshin EM Corp., Tokyo, Japan.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by JSPS KAKENHI Grant (2629157 and 26660236).
