Abstract
Subdural histiocytic sarcomas from 15 dogs (mean age: 7.8 years) were histopathologically examined. Among the 15 dogs, there was a marked breed predominance (toward Pembroke Welsh Corgi dogs, 47%), but no gender predilection. Focal solitary subdural masses were detected in the cerebrum (12 cases) and spinal cord (1 case), whereas diffuse infiltrative lesions were observed in the cerebral leptomeninges in 2 cases. All neoplastic lesions had common histological features characterized by the proliferation of pleomorphic histiocytic cells combined with various inflammatory reactions. Multinucleated giant cells, phagocytosis, and atypical mitotic figures in the neoplastic cells were commonly observed. Most of the pleomorphic neoplastic cells in the present cases were immunopositive for monocytic, histiocytic, or both markers, such as human leukocyte antigen (HLA)-DR, ionized calcium-binding adaptor molecule 1 (Iba1), cluster of differentiation (CD)163, and CD204, except for the neoplastic cells in 2 focal and 2 diffuse histiocytic sarcomas. The findings suggest that differences in cell origin, molecular expression, or both patterns are responsible for the distribution patterns of canine subdural histiocytic sarcomas.
The World Health Organization classification for brain tumors in domestic animals includes malignant histiocytosis, 6 and the involvement of systemic central nervous system (CNS) lesions is associated with the disorder. However, there have been several case reports of canine histiocytic proliferative disorders confined to the intracranial or vertebral canals, without any cutaneous or other visceral organ involvement. 1,3,12,14,16 The pathological features of the intracranial disorder have not yet been definitively evaluated, and the cellular origin of such tumors has not been well elucidated. In addition, histiocytic sarcomas within the subdural region have been described to show 2 distinct distribution patterns, focal mass lesions that affect the leptomeninges and CNS, 1,3,13,14,16 and diffuse leptomeningeal lesions. 12,13,15 However, the reason why these different patterns exist has not been clarified. The authors of the current study, therefore, hypothesized that the differences in tumor distribution patterns could reflect different cellular lineages or marker expression patterns. Thus, the expressions of selected monocytic, histiocytic, or both markers that are characteristic of canine subdural histiocytic sarcomas were examined in 15 dogs.
Canine histiocytic sarcomas that were confined to the subdural region were collected from 15 dogs (i.e., from 3 postmortem and 12 surgical cases that were encountered during the last 5 years [2005–2009]). The breed, sex, age, major neurological signs, tumor location, and distribution pattern are shown in Table 1. Among the 15 dogs were 7 Pembroke Welsh Corgis (47%), 2 Bernese Mountain Dogs (13%), 1 Wire Fox Terrier (7%), 1 Rottweiler (7%), 1 Newfoundland (7%), 1 Norfolk Terrier (7%), 1 Chihuahua (7%), and 1 Miniature Schnauzer (7%). No gender predilection was detected. The mean age of the dogs was 7.8 years (5 years 10 months to 11 years). The majority of the dogs had a history of neurological signs, including seizures, postural reaction deficits, and paresis. Focal solitary subdural masses were found in the cerebrum (12 cases) and spinal cord (1 case), whereas diffuse infiltrative lesions were observed in the cerebral leptomeninges in 2 cases. Clinicopathological evaluation of the cases revealed no involvement of other visceral organs.
Tissue lesion samples were fixed in 10% phosphate buffered formalin solution before being embedded in paraffin. The paraffin sections (2–4 mm thick) were then stained with hematoxylin and eosin. Histological classification of the canine histiocytic sarcomas was performed according to whether the tumor was distributed focally or diffusely. Immunohistochemistry was performed using the dextran polymer method. a The primary antibodies used and their positive reactions in canine tissues are listed in Table 2. After being deparaffinized, the sections were autoclaved at 120°C for 5 min for antigen retrieval. The tissue sections were then treated with 1% hydrogen peroxide in methanol at room temperature for 30 min and incubated with 8% skimmed milk in Tris buffered saline (TBS) at 37°C for 40 min to block nonspecific reactions before being incubated with the primary antibodies at 4°C overnight. After being washed 3 times in TBS, the sections were then incubated with the polymer reagent a at 37°C for 40 min. Finally, the reaction products were visualized with 0.05% 3-3′-diaminobenzidine and 0.03% hydrogen peroxide in Tris-HCl buffer, and the sections were counterstained with Mayer hematoxylin. The mean percentage of strongly immunopositive neoplastic cells in 5 high-power (400×) fields was then recorded. A chi-square test was performed to determine the statistical significance of differences between each marker expression and tumor distribution patterns by using a scale of 1–3 for immunoreactivity (1 = 1–10% positive cells, 2 = 11–50% positive cells, 3 = 51–100% positive cells). The level of P < 0.05 was considered to be significant. Data analysis was performed by means of a computer-based statistical package. g
Canine subdural histiocytic sarcomas examined in the present study. *
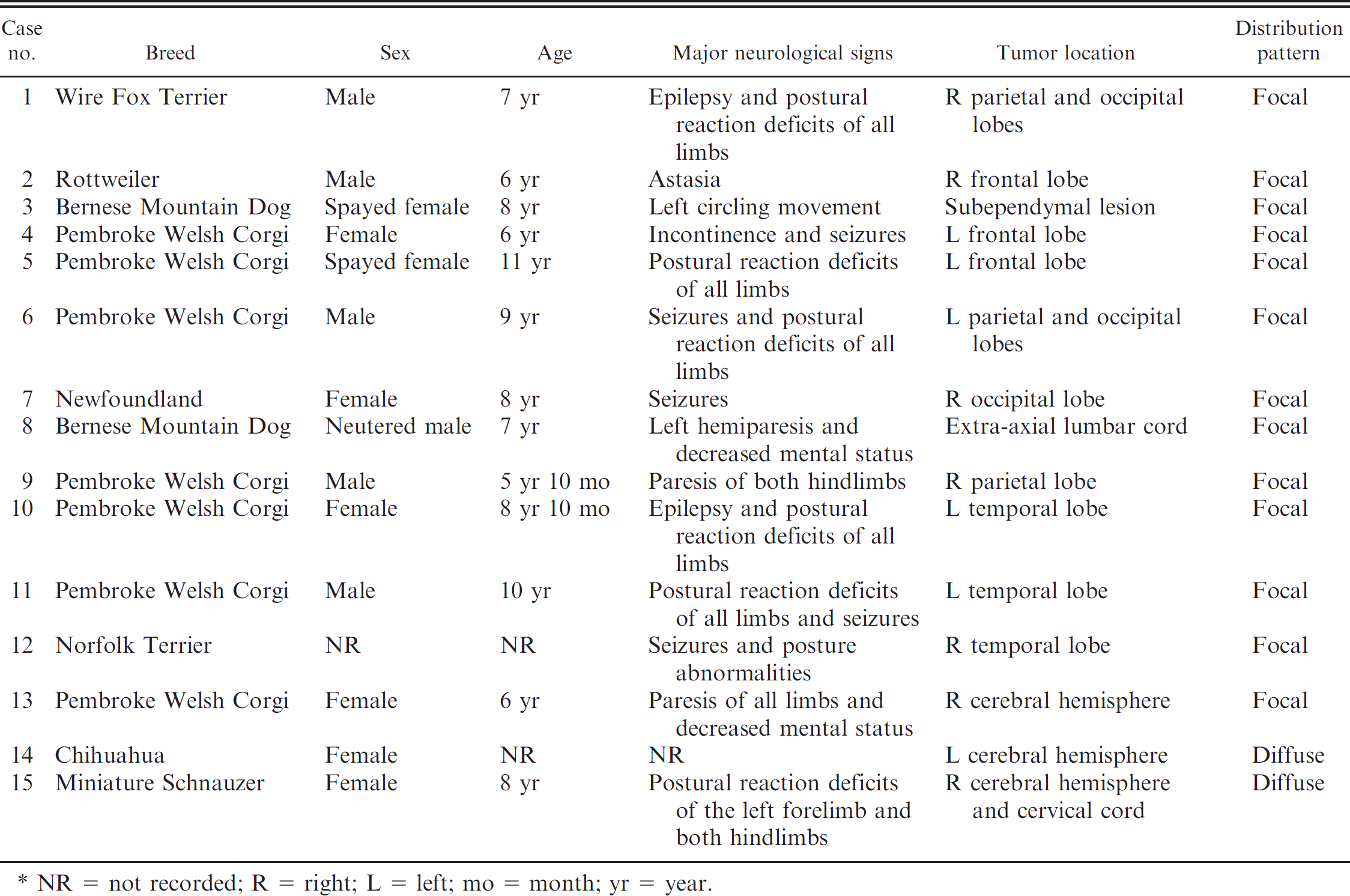
NR = not recorded; R = right; L = left; mo = month; yr = year.
Primary antibodies used in the present study. *
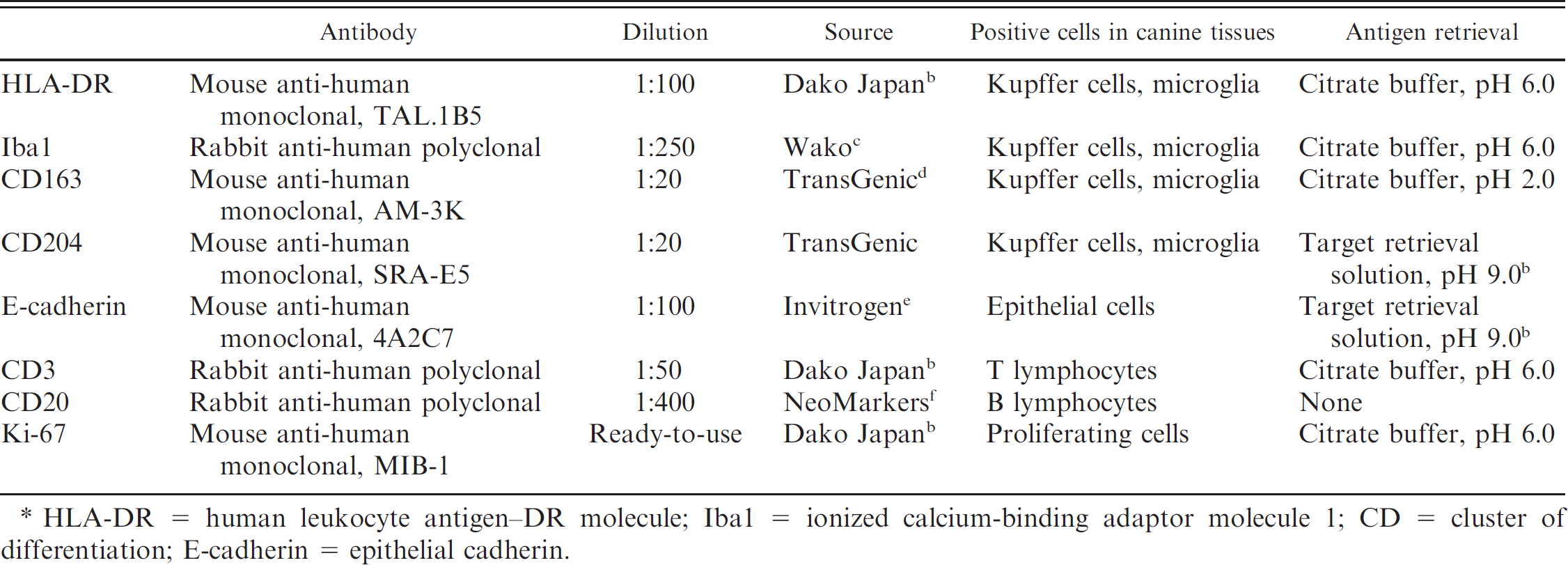
HLA-DR = human leukocyte antigen–DR molecule; Iba1 = ionized calcium-binding adaptor molecule 1; CD = cluster of differentiation; E-cadherin = epithelial cadherin.
In each focal histiocytic sarcoma (Fig. 1), the mass lesion demonstrated pleomorphic histiocytic cell proliferation and various degrees of small lymphocyte infiltration. In all cases, the neoplastic cells had invaded the leptomeninges and the brain or spinal parenchyma. Most neoplastic cells contained abundant eosinophilic cytoplasm with fairly well delineated cell margins, eccentric nuclei of various sizes, and many mitotic figures. Multinucleated giant cells were often scattered throughout the lesion, and phagocytic figures were occasionally observed. The pleomorphic histiocytic cells frequently accumulated around small arterioles. The cells exhibited the characteristic morphology of foamy macrophages, with degenerating myelin sheaths or lipid granules in their cytoplasm. Moderate numbers of small reactive lymphocytes were admixed within the neoplastic cell population.
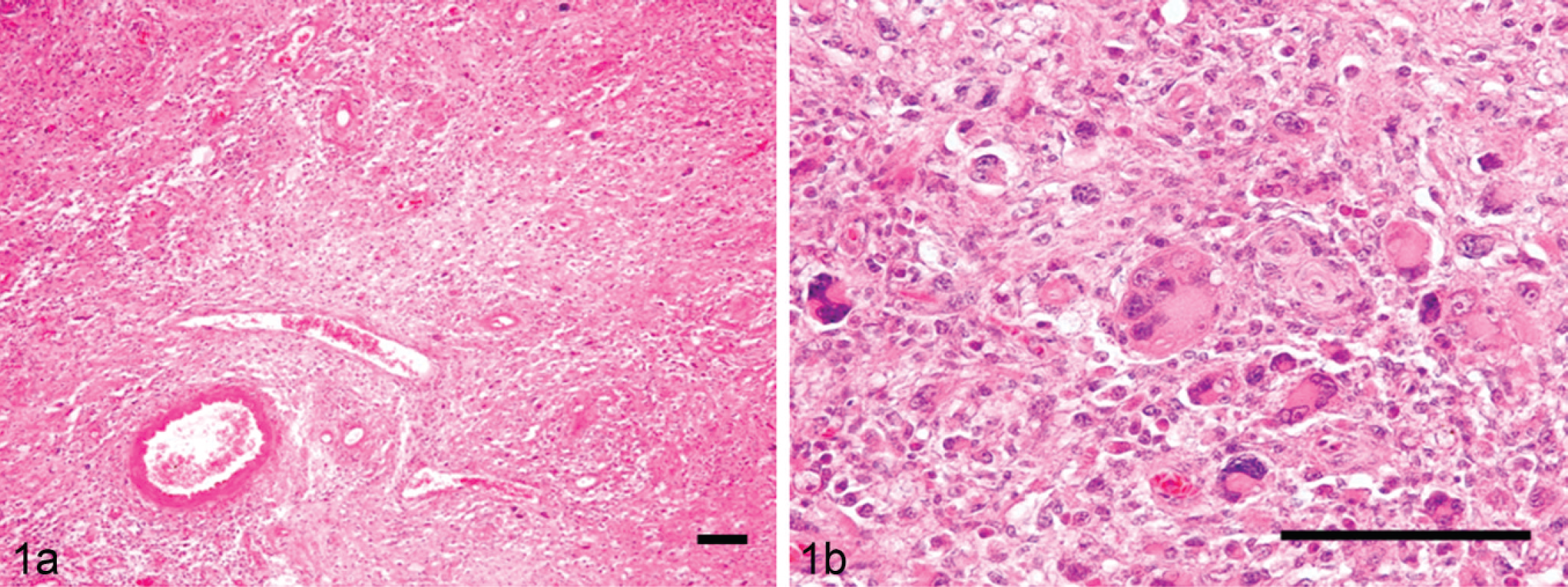
Focal histiocytic sarcoma; dog 13.

Diffuse histiocytic sarcoma; dog 15.
In the diffuse histiocytic sarcomas (Fig. 2), the lesions were characterized by marked infiltration of pleomorphic histiocytic cells admixed with small lymphocytes. These histiocytic cells were distributed diffusely within the leptomeningeal and ventricular areas of the brain, spinal cord, or both. The dura mater, arachnoidal space, and leptomeninges had been infiltrated by large numbers of pleomorphic histiocytic cells. A large number of the cells were seen infiltrating the brain parenchyma from the cerebral surface to the cortex. Most of the cells exhibited morphological variations, such as a clear to eosinophilic cytoplasm. Some were bi- or polynucleated giant cells with abundant eosinophilic cytoplasm, and phagocytic cells and cells with mitotic figures were frequently observed.
The results of immunohistochemistry for the human leukocyte antigen (HLA)-DR alpha-chain, ionized calcium-binding adaptor molecule 1 (Iba1), cluster of differentiation (CD)163, CD204, epithelial cadherin (E-cadherin), and Ki-67 are summarized in Table 3. In the normal arachnoidal tissue from a 5-year-old male Beagle dog without any neurological lesions, HLA-DR–, Iba1-, CD163-, CD204-, and E-cadherin–positive cells were observed (Fig. 3). In the canine subdural histiocytic sarcomas, the immunoreactivity for HLA-DR varied from case to case; however, there tended to be more Iba1-positive cells than HLA-DR–positive cells (Figs. 4a, 4b, 5a, 5b). Very large numbers of CD163- and/or CD204-positive cells were observed in 11 focal histiocytic sarcomas, while most neoplastic cells were immunonegative in both diffuse histiocytic sarcomas (Figs. 4c, 4d, 5c, 5d). No pleomorphic neoplastic cells were positive for E-cadherin. The number of neoplastic cells with Ki-67–positive nuclei varied (7.7–43.5%) in the focal histiocytic sarcomas but was low (8.9–10.5%) in the diffuse histiocytic sarcomas. Many CD3-positive T cells and CD20-positive B cells had infiltrated into the lesions. Significant differences were found between the focal and diffuse histiocytic sarcomas for CD163 (P = 0.012) and CD204 (P = 0.010) expression. On the basis of tumor location, histological characteristics, and marker expression, 13 dogs were diagnosed with focal histiocytic sarcomas in the leptomeninges, and 2 dogs were diagnosed with widely disseminated diffuse histiocytic sarcomas.
Immunohistochemistry results of canine subdural histiocytic sarcomas in the present study. *
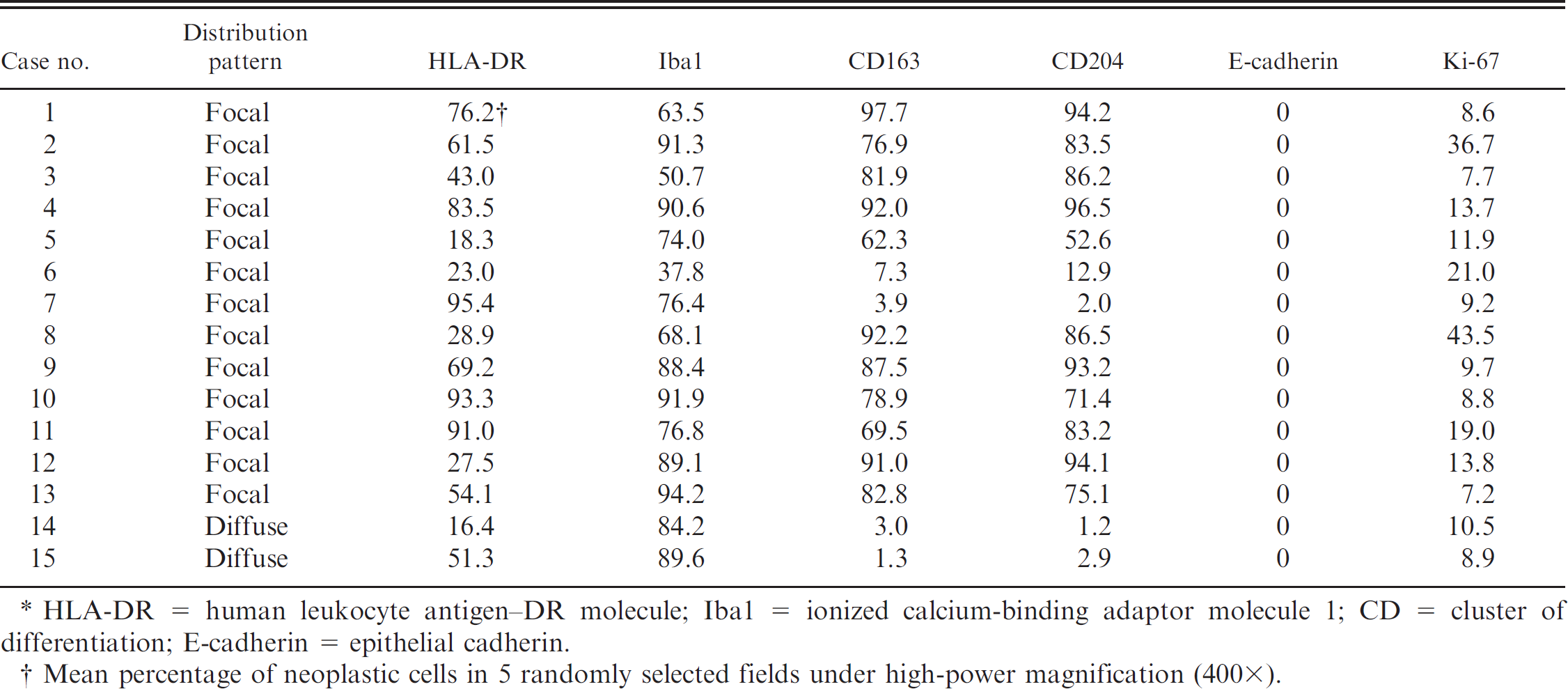
HLA-DR = human leukocyte antigen–DR molecule; Iba1 = ionized calcium-binding adaptor molecule 1; CD = cluster of differentiation; E-cadherin = epithelial cadherin.
Mean percentage of neoplastic cells in 5 randomly selected fields under high-power magnification (400×).
Canine histiocytic proliferative disorders have been classified into 3 major categories: canine cutaneous histiocytoma, which is mostly found in young dogs; canine reactive histiocytosis, which includes the cutaneous and systemic types; and the histiocytic sarcoma complex, which includes localized and disseminated histiocytic sarcoma (Moore PF: 2004, The histiocytic disease complex. In: Proceedings of the American College of Veterinary Internal Medicine annual meeting, pp. 437–438. Minneapolis, MN, June 9–12, 2004). The histiocytic sarcoma complex was more commonly observed in Bernese Mountain Dogs, Rottweilers, and retrievers (Golden, Labrador, and Flat-Coated), 1 whereas subdural histiocytic sarcomas were most frequently detected in middle-aged Pembroke Welsh Corgis (47%) in the present study. The difference in breed predominance between subdural histiocytic sarcomas and other canine histiocytic sarcomas might indicate that they possess different etiological properties.
Canine monocytic, histiocytic, or both lineages of cells express CD1, CD11b, CD11c, CD14, CD68, and major histocompatibility complex (MHC) class II antigens, which are considered to be reliable cell markers. 1 Because only formalin-fixed tissues were obtained in the present study, antibodies against HLA-DR (MHC class II), Iba1, CD163, and CD204 were utilized to identify the canine monocytic and histiocytic lineages. It was shown in the present study that Iba1, which mediates calcium signals in the monocytic and histiocytic cell lineages, 4,5 can also be used as a marker for identifying monocytic and histiocytic cells in dogs. Cluster of differentiation 163 is a membrane protein belonging to the endocytic scavenger receptors for hemoglobin–haptoglobin complex. 2,8,11 Cluster of differentiation 204 is also a scavenger receptor for a wide range of ligands. 8,10 The expression of these molecules is known to be associated with the anti-inflammatory M2 macrophage population, 7–9 which includes hepatic Kupffer cells and microglial cells. 2,7
Histological feature and cell phenotype between the localized and disseminated forms of canine visceral histiocytic sarcoma are not different. 1 Although they are known to arise from myeloid dendritic cells, which display CD1, CD11c, MHC class II, and intercellular adhesion molecule 1 (ICAM-1) phenotype, such dendritic lineage–specific markers were not available on formalin-fixed tissues. The present study revealed that the focal and diffuse histiocytic sarcoma of this study could not be differentiated by histological features alone. Immunohistochemically, the neoplastic cells in the majority (11/13) of canine focal histiocytic sarcomas had high levels of CD163- and CD204-positive cells. On the contrary, the neoplastic cells in the diffuse subdural histiocytic sarcomas (2/2) and some of the focal type histiocytic sarcomas (2/13) had low levels of CD163- and CD204-positive cells. These results suggest that marker expression is not definitively associated with cellular origin of tumor cells. Although these findings suggest that the different lesion distribution patterns of subdural histiocytic sarcomas do not depend on their marker expression patterns to identify the cellular origin of canine subdural histiocytic sarcomas, further case reports and more immunohistochemical information on frozen sections is needed.

Normal arachnoidal tissue; 5-year-old Beagle dog. The following positive cells were detected:

Focal histiocytic sarcoma; dog 13. Neoplastic cells immunopositive for:
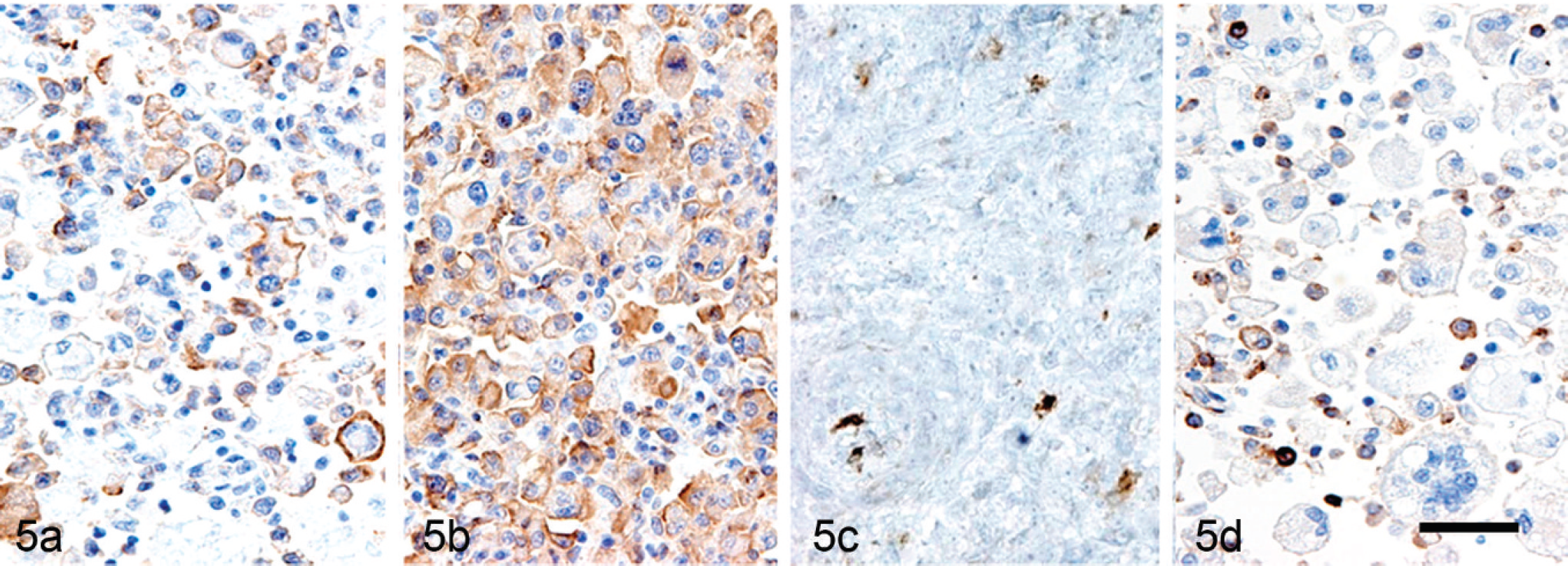
Diffuse histiocytic sarcoma; dog 15. Neoplastic cells immunopositive for:
Footnotes
a.
Envision, Dako Japan Inc., Tokyo, Japan.
b.
Dako Japan Inc., Tokyo, Japan.
c.
Wako Pure Chemical Industries Ltd., Osaka, Japan.
d.
TransGenic Inc., Kumamoto, Japan.
e.
Invitrogen Corp., Carlsbad, CA.
f.
NeoMarkers Inc., Fremont, CA.
g.
SPSS version 15.0, SPSS Inc., Chicago, IL.
