Abstract
An 8-y-old, spayed female Bernese Mountain Dog was presented to a referral center for evaluation of right thoracic limb lameness and previously suspected Evans syndrome that had been poorly responsive to immunosuppressive therapy. Based on review of examination findings and laboratory data, Evans syndrome was deemed unlikely and hemophagocytic histiocytic sarcoma (HHS) was strongly suspected. On blood smear evaluation, atypical, histiocytic cells were noted, some of which exhibited siderophagia. Considering that circulating cells are not typically observed in dogs with HHS, additional diagnostic investigation was performed. Autopsy and histopathology revealed that the dog had a mixed form of HS (dendritic-cell origin HS in the lung, and HHS in the spleen, liver, and bone marrow), and immunocytochemical characterization of cultured cells derived from blood suggested that the cells were of dendritic HS origin, rather than HHS origin, as originally suspected. Whole-exome sequencing revealed genetic similarity between cell lines derived from lung tissue and blood, providing additional evidence of the relatedness of these 2 cell populations. Our case highlights the rare entity of mixed HS and typifies the inherent challenges in classifying rare, atypical, circulating neoplastic cells.
An 8-y-old, spayed female Bernese Mountain Dog was presented to a primary care veterinarian for periodic lameness of the right thoracic limb (RTL) and vocalizing with pain on palpation of the right shoulder. On examination, the dog was febrile (40.8°C), lame on the RTL, and resisted manipulation of the neck. Radiographs were performed, and C5-C6 degenerative joint disease, sclerosis in the right bicipital groove, and an alveolar pattern in the right ventral middle lung lobe (interpreted as pneumonia or hemorrhage) were identified by a board-certified radiologist. On abdominal ultrasound, there was a markedly enlarged right medial iliac lymph node with a hypoechoic center, interpreted as a cystic or neoplastic focus. On initial in-clinic CBC evaluation (ProCyte; Idexx), the dog had moderate thrombocytopenia (36 × 109 platelets/L; RI 165–430 × 109 platelets/L) with rare giant forms and evidence of platelet clumping. Over the course of the next 2.5 wk, the dog was treated for presumptive immune-mediated thrombocytopenia with oral prednisolone (0.5 mg/kg q12h) and azathioprine (1.8 mg/kg q24h). The platelet count remained static, and the patient developed icterus, rapid weight loss, and moderate-to-marked, strongly regenerative anemia (Hct 0.22 L/L, RI: 0.39–0.59 L/L; reticulocytes 243 × 109/L, range: <100 × 109/L).
The dog was referred to Wheat Ridge Animal Hospital (Wheat Ridge, CO, USA) for further evaluation of presumed Evans syndrome (combined immune-mediated hemolytic anemia and immune-mediated thrombocytopenia [ITP]). On presentation, the dog was tachycardic (208 bpm) with bounding femoral pulses, moderate icterus, and diffuse muscle wasting. At this time, Evans syndrome seemed unlikely, given that the patient’s thrombocytopenia was moderate, rather than marked, as is typically seen with ITP patients; was likely underestimated (as platelet clumping was present); and was poorly responsive to immunosuppressive therapy. Additionally, there were no indicators of immune-mediated hemolysis evident on the blood smear (agglutination, spherocytosis, ghost erythrocytes). Serum biochemistry (AU480; Beckman Coulter) revealed anomalies that were collectively interpreted as indicative of increased hepatic enzyme activity and cholestasis: alanine aminotransferase 135 U/L (RI: 17–115 U/L), aspartate aminotransferase 69 U/L (RI: 11–46 U/L), alkaline phosphatase 957 U/L (RI: 8–196 U/L), gamma-glutamyl transferase 18 U/L (RI: 0–9 U/L), and total bilirubin 20.5 μmol/L (RI: 0–5.1 μmol/L). Additionally, the patient had slight hypoalbuminemia (26 g/L; RI: 27–39 g/L) and low normal cholesterol (5.2 mmol/L; RI: 3.4–8.9 mmol/L). Fine-needle aspirates were obtained from a markedly enlarged spleen, and a pathologist review of the CBC was ordered for assessment of leukocyte morphology. On this CBC (Advia 2120; Siemens), the platelet estimate was 72 × 109 platelets/L with moderate clumping in the blood smear.
Aspirates of the spleen revealed significant extramedullary erythropoiesis and a moderately to markedly atypical, hemophagocytic, histiocytic cell population. These cells had eccentrically placed oval-to-reniform nuclei, with stippled chromatin, inconspicuous-to-prominent nucleoli, and a moderate amount of heavily vacuolated cytoplasm, often containing erythroid cells, or dark-green pigment (hemosiderin). Some cells had several large nucleoli, and scattered atypical mitotic figures were present. Rare, similarly appearing cells were observed in the feathered edge of the peripheral blood smear (Fig. 1). In light of the clinical history, cytologic findings were most suggestive of histiocytic sarcoma (HS), suspected to be of the hemophagocytic (HHS) type.
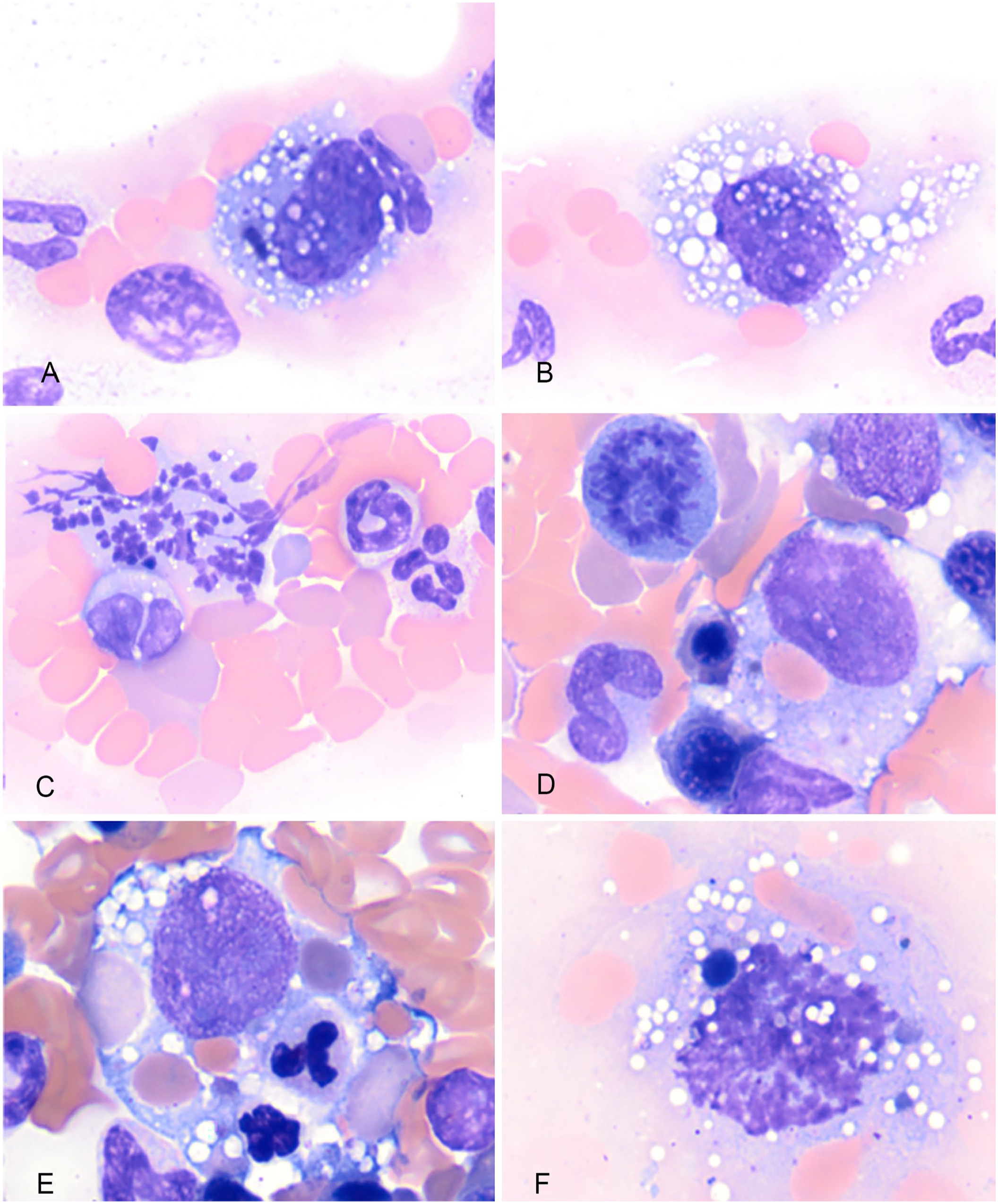
Cytology of histiocytic sarcoma in a Bernese Mountain Dog. Modified Wright–Giemsa stain.
Following rapid clinical decline of their dog, the owners agreed to euthanasia and postmortem examination. On autopsy, the spleen and liver were diffusely enlarged with patchy discoloration. The lung contained many variably sized, white to off-white nodules, which were most prominent in the right middle lung lobe; this was the area that previously contained alveolar infiltrates radiographically. Several enlarged abdominal lymph nodes were noted. There was no evidence of neoplastic tissue affecting the RTL shoulder joint; however, because bone pain has been associated with neoplastic cell infiltration of marrow, a bone marrow aspirate and core biopsy were taken from the right humerus. Postmortem aspirates of spleen, lymph node, and marrow confirmed neoplastic expansion by atypical, histiocytic cells in the sampled areas. Tissues were submitted for histopathology, which revealed disseminated HS, as well as evidence of atypical circulating cells within the blood vessels in several tissues, marked canalicular cholestasis and centrilobular atrophy in the liver, and infarcts in the spleen.
We retrieved no canine cases with circulating HHS cells in a literature search of Google Scholar, PubMed, Scopus, or Web of Science, suggesting that this condition has not been reported in dogs. To further classify the circulating histiocytic cells in this case, we submitted paraffin-embedded tissues and pre-stained blood smears to the Leukocyte Antigen Biology Laboratory (UCDavis; University of California–Davis, Davis, CA, USA) and created a cell tube block (CTB) with peripheral blood enriched for leukocytes. Flow cytometry was not pursued because of the poor cellular viability at the time of shipping, as well as the exceptionally low representation of the rare, remaining cells.
Immunocytochemistry (ICC) and immunohistochemistry (IHC) were performed as described previously17,21 (Table 1). Based on histopathology and IHC, this patient had a mixed form of HS: non-hemophagocytic HS (CD11d–) in the lung, and hemophagocytic HS (CD11d+) in the spleen, liver, and bone marrow (Figs. 2, 3). Initial immunocytochemical staining for CD18, CD204, CD1a, CD11c, and CD11d (UCDavis) was not successful on pre-stained blood smears. The staining protocol for CD11d on previously stained slides was subsequently optimized at Colorado State University (CSU; Fort Collins, CO, USA); however, none of the cells from the original aspirate cytology exhibited positive labeling, even in the cell population in the bone marrow aspirate, which was expected to label positively. This is likely due to the time lapse between collection and staining (~6 mo at the time of the second attempt), as cells on cytology slides may progressively lose reactivity to antibodies over time.2,18 Although CTB creation was successful, there were too few histiocytic cells in the sample for further characterization.
Antibodies used for immunocytochemistry and immunohistochemistry.
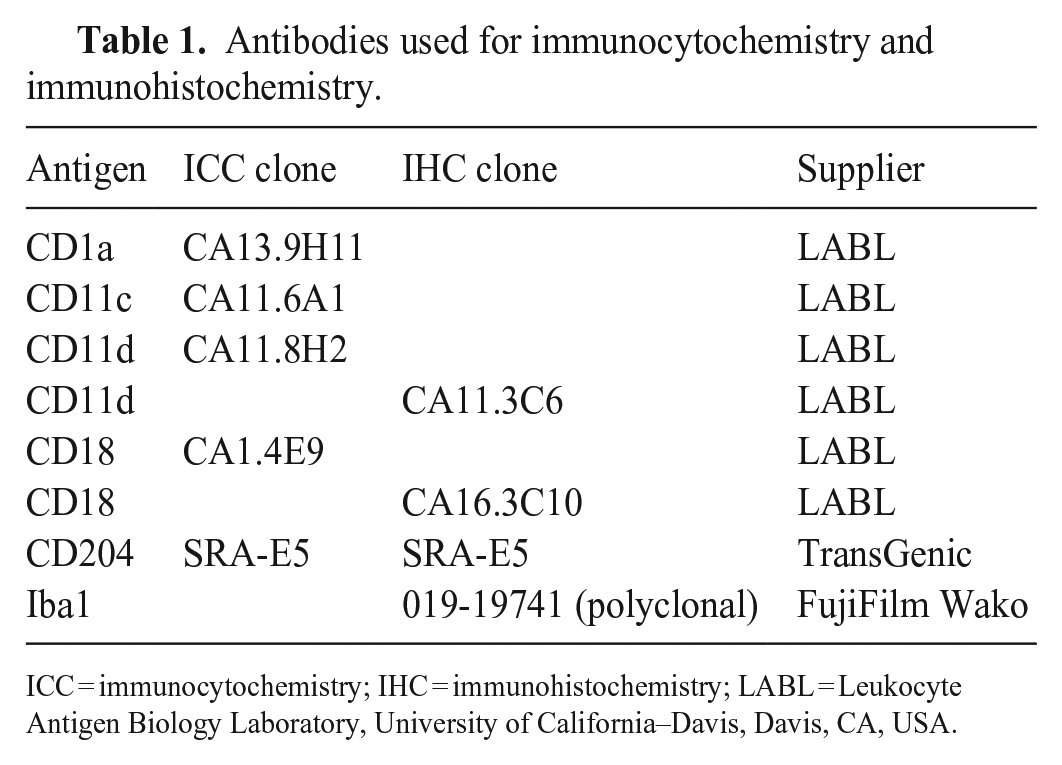
ICC = immunocytochemistry; IHC = immunohistochemistry; LABL = Leukocyte Antigen Biology Laboratory, University of California–Davis, Davis, CA, USA.
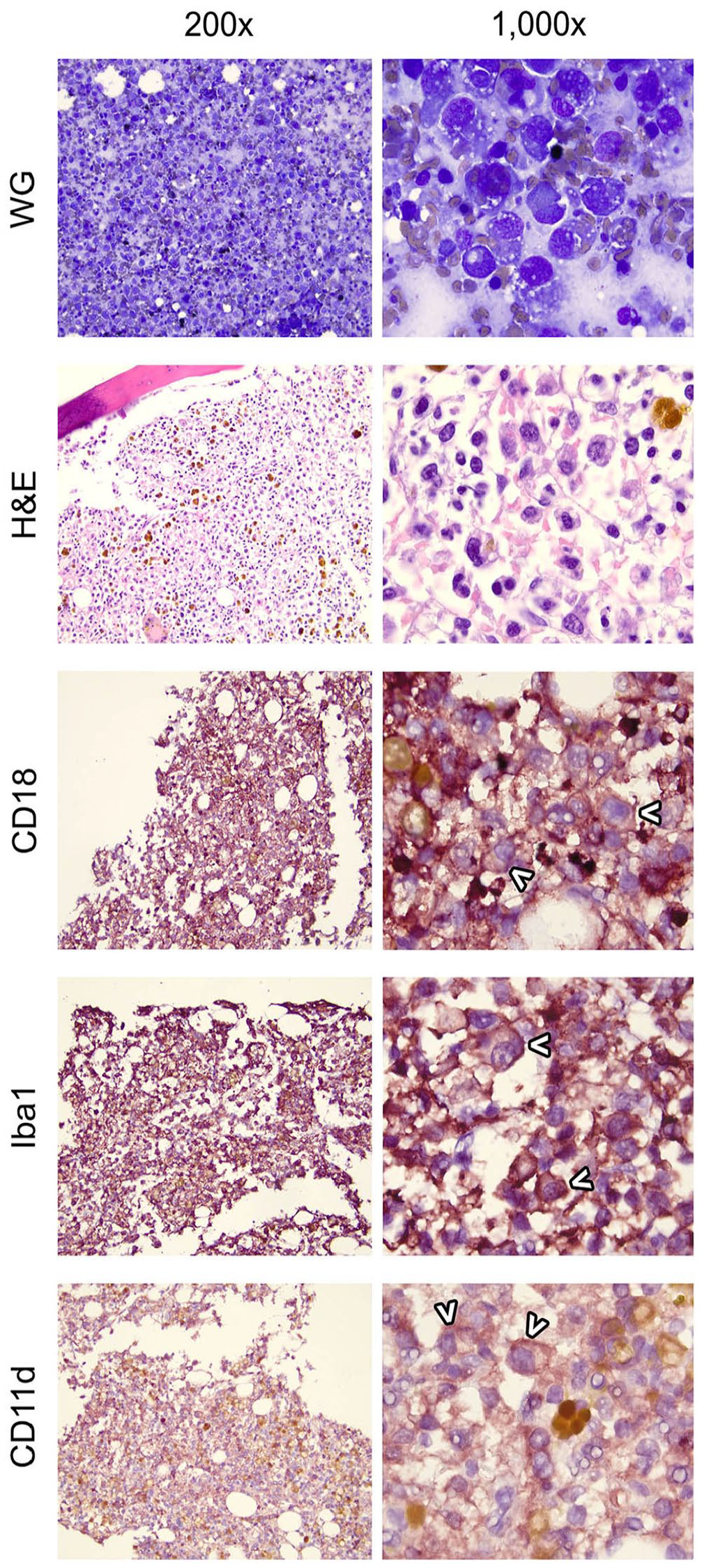
Cytology (WG), histopathology (H&E), and immunohistochemistry of hemophagocytic histiocytic sarcoma in a Bernese Mountain Dog. Cellular aggregation is quite dense in the bone marrow, and most of the cells are atypical, histiocytic cells. White carats denote individualized cells that can be evaluated independently from the thick background. Histiocytic cells have strong, positive, membranous staining of CD18 and positive membranous and cytoplasmic staining of Iba1. This cell population has positive membranous staining of CD11d. WG = modified Wright–Giemsa stain.
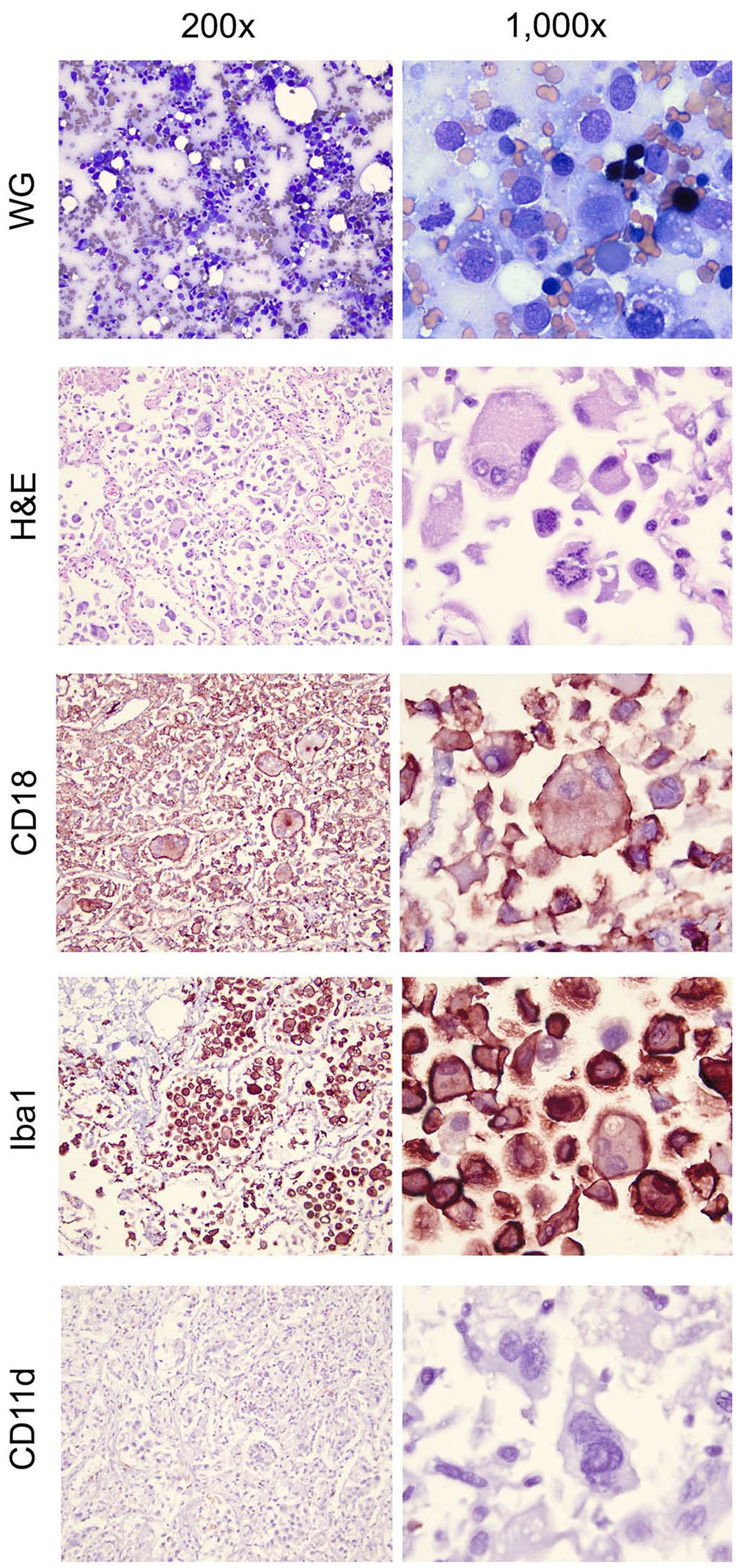
Cytology (WG), histopathology (H&E), and immunohistochemistry of dendritic (APC) histiocytic sarcoma in a Bernese Mountain Dog. Histiocytic cells in the lung have strong, positive, membranous staining of CD18 and positive membranous and cytoplasmic staining of Iba1. This cell population is negative for CD11d staining. WG = modified Wright–Giemsa stain.
We were able to maintain atypical, histiocytic cells from the blood in culture (CSU, Dr. Doug Thamm’s laboratory), and following expansion of these cells, were able to pursue ICC. These cells were strongly CD11c and CD18 positive, variably CD204 and CD1a positive, and did not express CD11d (Fig. 4). Alteration of cell surface markers in histiocytic cells in culture has been documented.3,8 However, another explanation for our ICC results, in a patient with mixed HS, is that the atypical, circulating cells originated from dendritic antigen-presenting cell (APC) HS, and not the patient’s HHS. Although it would have been ideal to definitively characterize the circulating cells directly from blood, as intended, it is worth noting that previously documented circulating HS cells are of dendritic APC HS origin.1,19 This may be because HHS cells tend to lodge in the pulmonary vasculature, rather than stay in circulation; on histologic evaluation, HHS cells are especially visible in septal capillaries (Moore PF, pers. comm., 2023 Mar 22).
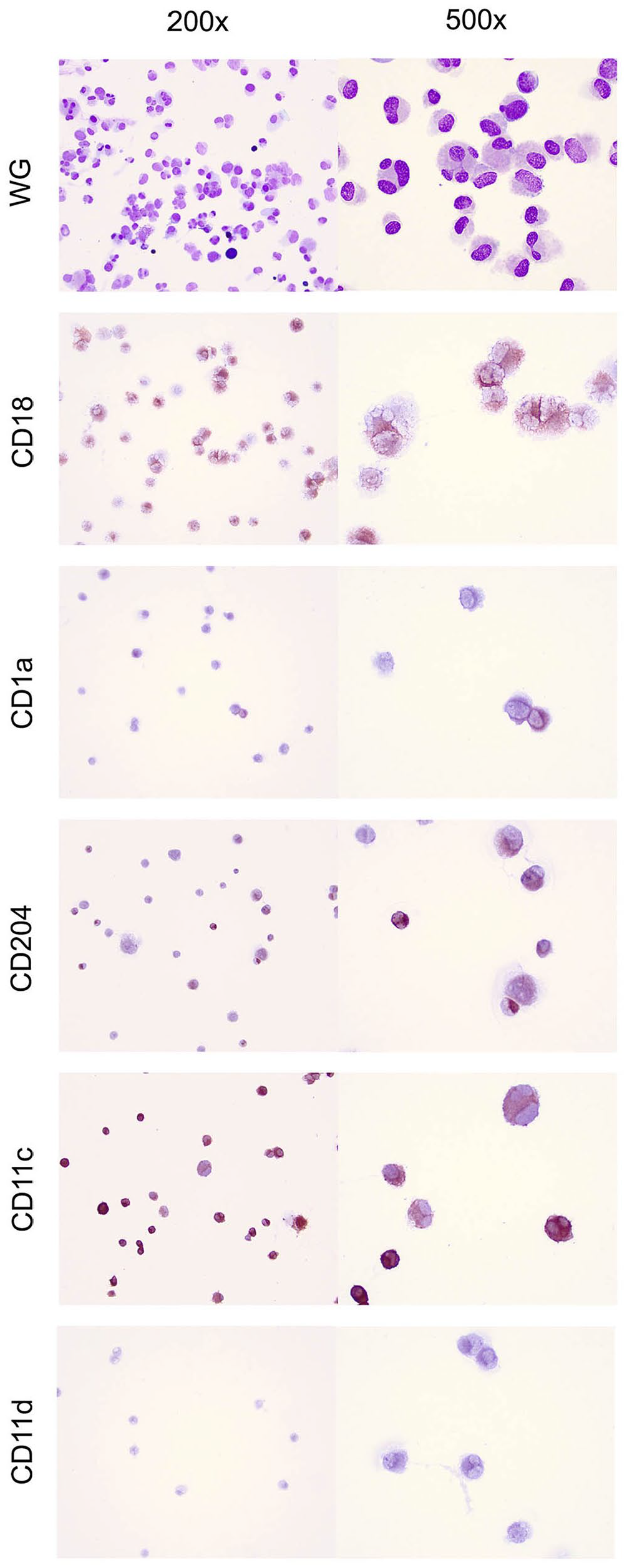
Mixed histiocytic sarcoma in a Bernese Mountain Dog. Cultured cells from blood are positive for CD18 and CD11c, variably positive for CD1a and CD204, and negative for CD11d. Immunocytochemistry. WG = modified Wright–Giemsa stain.
To further characterize the cells isolated in culture and identify candidate driver mutations, whole-exome sequencing was performed. Briefly, DNA was isolated from each cell line and submitted for Illumina library preparation and short-read sequencing to the University of Colorado Anschutz Genomics Core (Aurora, CO, USA). DNA from each sample was sequenced to an average depth of 300× coverage (NovaSeq 6000 sequencer; Illumina) and 150-bp paired-end reads. The acquired raw fastq files were quality-assessed using FastQC (v.0.11.9, https://www.bioinformatics.babraham.ac.uk/projects/fastqc/). Reads that passed QC were trimmed and aligned to the Canis lupus familiaris (Ensembl v.104 CanFam3.1) genome using TrimGalore (v.0.6.6, https://www.bioinformatics.babraham.ac.uk/projects/trim_galore/) and BWA-mem (v.0.7.17, https://github.com/lh3/bwa).11,12 Variants were identified using the GATK best practices pipeline 20 and using the variant caller Mutect2 (v.4.3.0.0, https://github.com/broadinstitute/gatk). A previously acquired panel from 37 clinically normal dogs of various breeds and a panel of germline variants (provided by the Broad Institute, Cambridge, MA, USA) were utilized for variant calling. Variants were de-prioritized if they were identified in the matched normal sample, the panel of normal samples, or in the germline variant call file. Alternatively, variants were prioritized if they had 1) a minimum total read depth >5, 2) occurred at an allelic frequency >0.10, 3) were found in a known cancer gene, and 4) were predicted to have a moderate or high impact via Ensembl Variant Effect Predictor. 14 Variant call files were converted to mutation annotation format (MAF) files using vcf2maf (v.1.6.19, https://github.com/mskcc/vcf2maf) imported to R (v.4.4.1, https://www.R-project.org/) and analyzed using maftools (v.2.14.0, https://github.com/PoisonAlien/maftools). 13 Multiple conserved missense mutations were observed in various genes of interest (Fig. 5; Suppl. Table 1). Of note, both cell populations contain a PTPN11 mutation, which is a mitogen-activated protein kinase signaling proto-oncogene that is commonly altered in both human and canine visceral histiocytic sarcoma 9 ; a CREB1 mutation, which has been documented as a transcription factor of ribonucleotide reductase small subunit M2 in human cancers, and may promote NF-κB activity6,7; and 2 separate mutations in the tumor suppressor gene TP53, which is a commonly mutated gene in both canine and human cancer.4,5,10 The striking genetic similarity in genes of interest strongly implies that the 2 cell lines are related, as suspected, although, again, it would have been ideal to have more straightforward ICC results on the original, circulating cell population, as well.
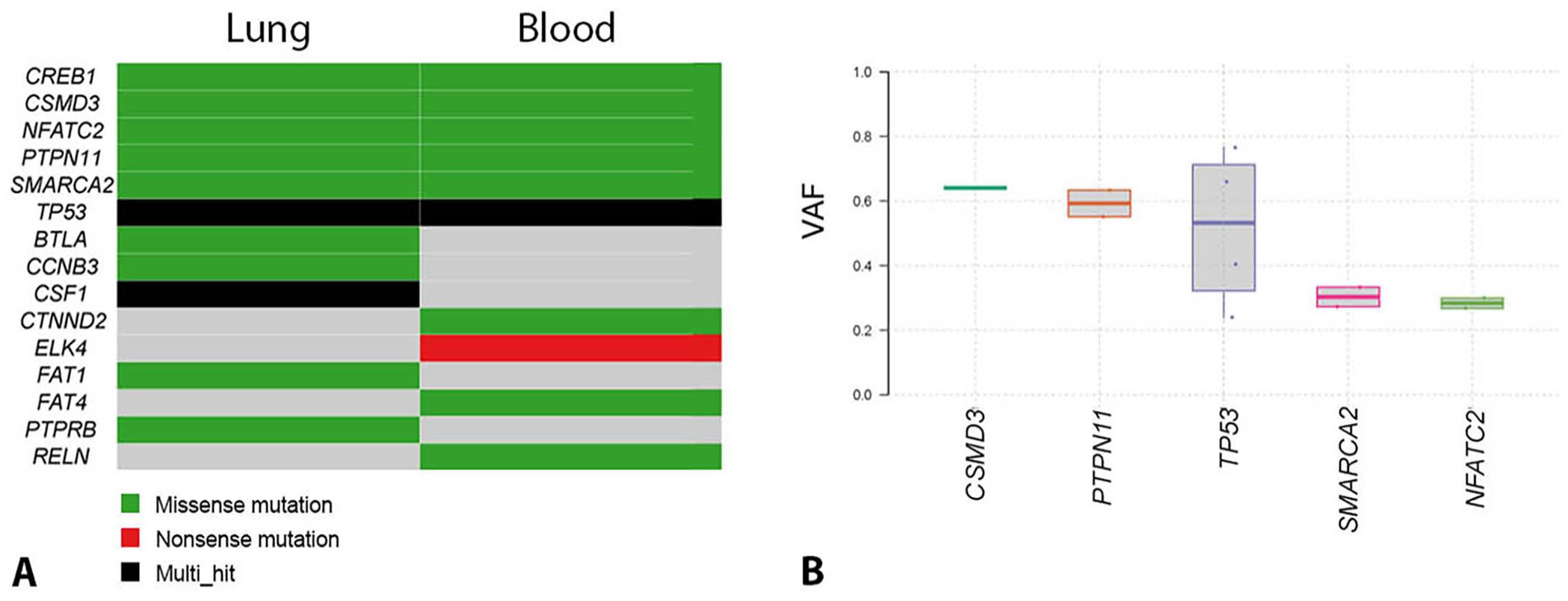
Mixed histiocytic sarcoma in a Bernese Mountain Dog. Whole-exome sequencing result summary.
In most cases, HS cells are derived from interstitial dendritic cells, which are ubiquitous in the body, and can therefore arise in almost any tissue; these cells express CD1a, CD11c, and MHC class II. HHS, a variant of HS, arises from splenic red pulp and/or bone marrow macrophages and expresses CD11d/CD18 rather than CD1a and CD11c/CD18. In rare cases, dogs have been documented to have both HHS and HS of interstitial dendritic cell origin.16,17 Clinically, dogs with HS have hyporexia, weight loss, and lethargy, with other signs dependent on the organ affected, secondary to mass lesion formation. Lesions of HS that arise from interstitial dendritic cells are most often large mass lesions that are white or cream-to-tan on cut surface and may consist of a solitary lesion or multiple masses or nodules within an organ. By contrast, dogs with HHS may have diffuse organomegaly, perhaps an ill-defined mass, and/or bone marrow infiltration. 16 HHS cells tend to have less dramatic cytologic and histologic characteristics of malignancy, such as anisokaryosis and anisocytosis, nuclear pleomorphism, and multinucleation, compared to HS cells. In our case, both the autopsy findings and the microscopic features of cells in tissues are most consistent with mixed HS and HHS.
Dogs with the hemophagocytic form of HS typically have moderate-to-marked, regenerative, hemolytic anemia and thrombocytopenia, hypoalbuminemia, and variable hypocholesterolemia. This may be confused with immune-mediated hemolytic anemia with thrombocytopenia (Evans syndrome), as it was initially in our case. 17 Although these dogs are typically Coombs test (erythrocyte-bound IgG) negative, some dogs may have secondary immune-mediated hemolysis and/or false-positive results on a Coombs test, for example, secondary to neoplasia or recent transfusion. A Coombs test was not performed in our case, and as previously noted, there was no cytologic evidence of immune-mediated hemolysis. Phagocytosis of erythrocytes, leukocytes, and/or other tumor cells (“cellular cannibalism”) may occur in both forms of HS, but this process is more common in the hemophagocytic form of HS.
A retrospective study evaluated blood smears for the presence of macrophage-like (“ML” cells) in canine patients and found this is rare (0.14% of CBC case records evaluated). In cases with circulating ML cells that met inclusion criteria, 46% of patients were ultimately diagnosed with a neoplastic condition, and 5 of 18 had HS. 15 Other causes for circulating ML cells included severe local or systemic inflammatory and/or necrotic disease. The retrospective study and our report highlight the importance of identifying ML cells in blood smears and understanding their potential significance to the patient. Given that cells were most readily identifiable on the feathered edge of the blood smear in our case, it is important that this area of the smear be evaluated carefully in cases of suspected HS, as tumor cells may not be identifiable in the monolayer area typically used for evaluation of individual cell morphology.
The cell lines derived from our patient will provide a valuable resource for further study of canine HS; hence, the additional effort in characterizing these cells serves multiple purposes: for further characterization of the cells in our case, and for future scientific discovery. For practicality, however, the most useful assays for such assessments in a clinical setting are flow cytometry and ICC on freshly prepared samples.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387241312308 – Supplemental material for Mixed histiocytic sarcoma in a Bernese Mountain Dog
Supplemental material, sj-pdf-1-vdi-10.1177_10406387241312308 for Mixed histiocytic sarcoma in a Bernese Mountain Dog by Jillian Hickey, R. Adam Harris, Stacy D. Meola, Samuel Jennings, Peter F. Moore, William Vernau, Kayla Harding, Douglas H. Thamm and Lisa J. Schlein in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Kristy Harmon and Laura Ashton for technical assistance with immunostaining.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors thank Dr. Laura Brandt (Zoetis) for immunostaining funds.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
