Abstract
Here we characterize 6 cases (4 autopsies and 2 biopsies) of histiocytic sarcoma in the CNS of cats. All affected cats had chronic, progressive clinical signs. Three autopsied cats were euthanized because of a poor prognosis, and one died. The clinical outcome for the biopsy cases remains unknown. Tumors occurred in the brain (4 cases), spinal cord (1 case), and brain and spinal cord (1 case). Neoplasms were restricted to the CNS in 3 cases. Reported gross changes in the 4 autopsy cases consisted of neuroparenchymal swelling with or without tissue pallor or gray discoloration (2 cases) and a yellow or dark-gray mass (2 cases). Histologically, pleomorphic, round-to-elongate neoplastic cells with typical histiocytic morphology effaced the neuroparenchyma and leptomeninges. Multinucleate neoplastic cells were observed in all cases. The mitotic count was 1–24 in 2.37 mm2 (10 FN22 40× fields). Neoplastic cells in all cases had positive immunolabeling for Iba1; immunolabeling was negative for E-cadherin, CD3, CD79, and MUM1, confirming their histiocytic origin.
In domesticated animals, proliferative histiocytic diseases are reported most frequently in dogs and cats. 11 In dogs, reactive and neoplastic histiocytic disorders consist of cutaneous and systemic reactive histiocytosis, cutaneous histiocytoma, histiocytic sarcoma, and dendritic cell leukemia. 11 In cats, histiocytic diseases are diagnosed less often and include pulmonary Langerhans cell histiocytosis, feline progressive histiocytosis, and histiocytic sarcoma. 11
Histiocytic sarcoma is a malignant neoplasm that originates from interstitial dendritic cells and less often from macrophages.5,6,11 The anatomic distribution and biologic behavior of these neoplasms in dogs and cats is similar. 11 Histiocytic sarcomas can be localized (when affecting a single tissue) or disseminated (when affecting multiple tissues),5,11 and primarily affect lymph nodes, spleen, liver, lungs, bone marrow, articular and periarticular tissues, skin, and rarely other tissues.4–6,11 Histiocytic sarcoma of the CNS is relatively common and well described in dogs but is not well characterized in cats.8–10,16 Here we describe the neuroanatomic distribution, neuropathology, and diagnostic immunohistochemistry (IHC) of 6 histiocytic sarcomas in cats with CNS involvement.
Cases of histiocytic sarcoma in cats were searched in the archives of the Athens Veterinary Diagnostic Laboratory (AVDL; College of Veterinary Medicine, University of Georgia, Athens, GA, USA) between 2000 and 2022 and the New York State Animal Health Diagnostic Center (AHDC; College of Veterinary Medicine, Cornell University, Ithaca, NY, USA) between 2007 and 2022 using the keywords “feline” and “histiocytic sarcoma”. Submission forms and autopsy or biopsy reports were reviewed for cases in which tumors affected the CNS (brain and/or spinal cord [SC]) with or without extraneural involvement. Archived slides were retrieved, and H&E-stained tissue sections were reviewed to characterize the morphology and distribution of the tumors within the affected tissues. In the CNS, tumors affecting the brain or the SC were characterized as intraparenchymal (when affecting the gray or white matter and/or perivascular spaces) or extraparenchymal (meningeal). CNS tumors were subjected to IHC for Iba1 (histiocytic immunomarker), E-cadherin (Langerhans cell immunomarker), CD3 (T-lymphocyte immunomarker), CD79 (B-lymphocyte immunomarker), and MUM1 (plasma cell immunomarker) for diagnostic confirmation (Table 1).
Details of immunohistochemical stains performed on sections of CNS histiocytic sarcomas in cats.
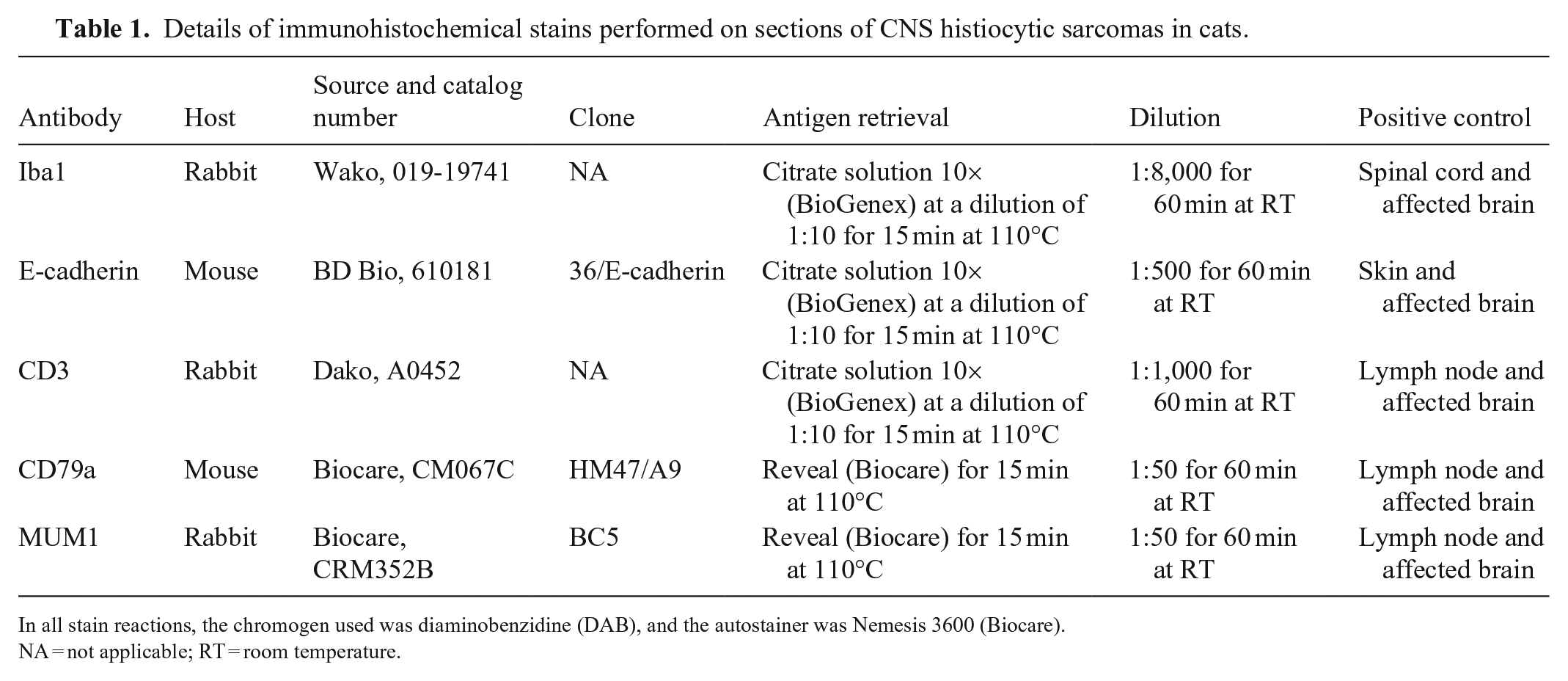
In all stain reactions, the chromogen used was diaminobenzidine (DAB), and the autostainer was Nemesis 3600 (Biocare).
NA = not applicable; RT = room temperature.
The initial search for feline cases with a routine diagnosis of histiocytic sarcoma retrieved 28 submissions (15 biopsies and 13 autopsies) at the AVDL and 33 submissions (28 biopsies and 5 autopsies) at the AHDC. CNS involvement was reported in 7 cases from both institutions; 1 AVDL case was excluded from our study because histologic and IHC evaluation was consistent with a B-cell lymphoma. After diagnostic confirmation, 4 AVDL autopsy cases (cases 1–4, comprising 14% of the retrieved AVDL feline histiocytic sarcomas) and 2 AHDC biopsy cases (cases 5 and 6, comprising 6% of the retrieved AHDC feline histiocytic sarcomas) met our inclusion criteria (Table 2). None of the other histiocytic sarcomas had CNS involvement. Brain was available for evaluation in 5 cases; SC was examined only in cases 2 and 5.
Signalment, clinical signs, outcome, and gross changes of histiocytic sarcoma with CNS involvement in 6 cats.
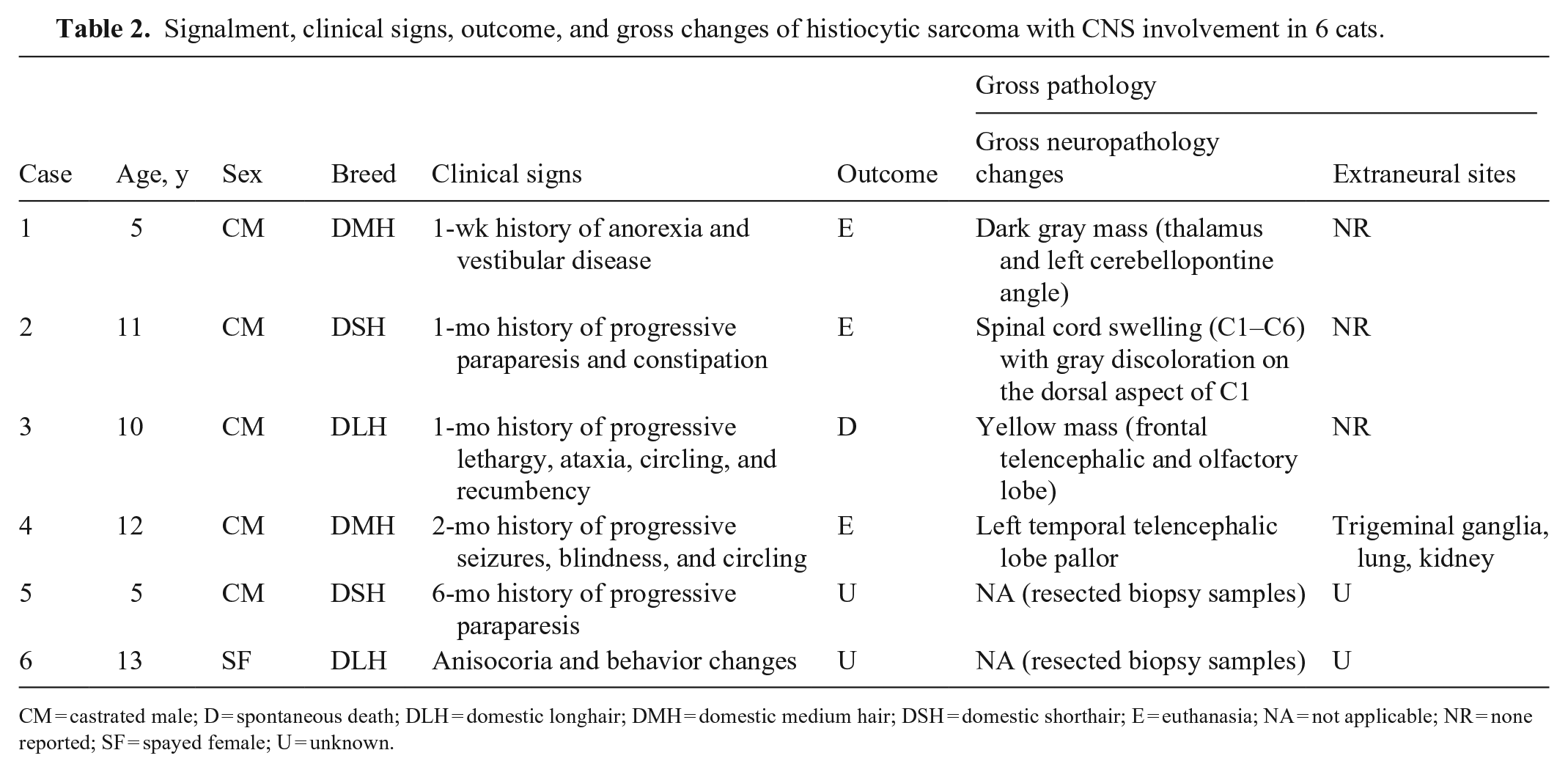
CM = castrated male; D = spontaneous death; DLH = domestic longhair; DMH = domestic medium hair; DSH = domestic shorthair; E = euthanasia; NA = not applicable; NR = none reported; SF = spayed female; U = unknown.
The average age of affected cats was 9.3 y. Feline immunodeficiency virus (FIV) or Feline leukemia virus (FeLV) status was not reported for any case. Clinical signs were variable and mainly chronic and progressive. Magnetic resonance imaging (MRI) data were available for case 2, and consisted of contrast-enhancing, hyperintense meningeal areas extending from the caudal fossa to the cervical SC. Three cats were euthanized because of a poor prognosis, and 1 cat died spontaneously; the clinical outcome remains unknown for the 2 AHDC biopsy cases.
Tumors occurred in the brain in 4 cases (cases 1, 3, 4, 6), SC in case 5, and brain and SC in case 2. Reported gross changes in the 4 autopsy cases consisted of neuroparenchymal swelling with or without tissue pallor or gray discoloration in 2 cases, and a yellow or dark-gray mass in 2 cases (Fig. 1). Tumors were reported to be restricted to the CNS in cases 1–3.
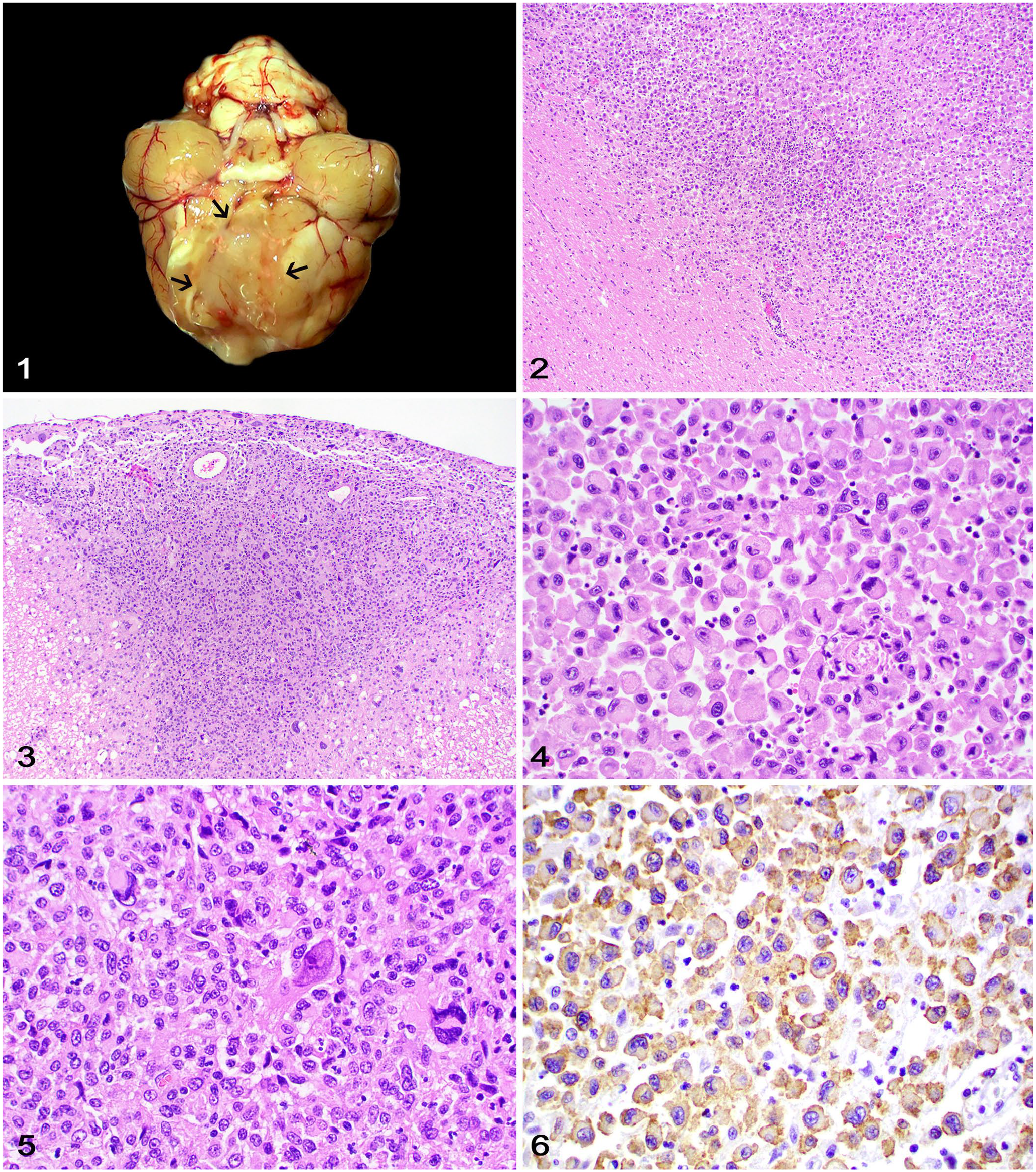
Histiocytic sarcoma with central nervous system involvement in cats.
Histologically (Table 3), all tumors were intraparenchymal and partially effaced the gray and white matter (Fig. 2). In cases 2 and 4, neoplastic cells were also extraparenchymal and expanded the leptomeninges (Fig. 3). Neoplastic cells were arranged in sheets or clusters supported by scant remaining neuroparenchyma or leptomeningeal tissue. Neoplastic cells had moderate-to-marked pleomorphism and typical histiocytic cell morphology characterized by round-to-polygonal-to-elongate shape, distinct cell margins, and a moderate-to-large amounts of variably vacuolated, eosinophilic cytoplasm (Figs. 4, 5). Nuclei were round-to-indented and had finely stippled-to-coarse chromatin with 1–2 large nucleoli and occasional round, 6–10-µm diameter, eosinophilic cytoplasmic inclusions. Multinucleate neoplastic cells were observed in all cases. The mitotic count was 1–24 in 2.37 mm2 (10 FN22 40× fields). Neoplastic cells in all cases had positive cytoplasmic immunolabeling for Iba1 (Fig. 6), negative membranous immunolabeling for E-cadherin, CD3, and CD79, and negative nuclear immunolabeling for MUM1, confirming their histiocytic origin. 2 A similar amount of perivascular and less often intratumoral CD3+ T lymphocytes and CD79+ B lymphocytes were distributed throughout the neoplasms. Scattered MUM1+ plasma cells were rarely observed.
Neurohistopathology and diagnostic confirmation of histiocytic sarcoma with CNS involvement in 6 cats.
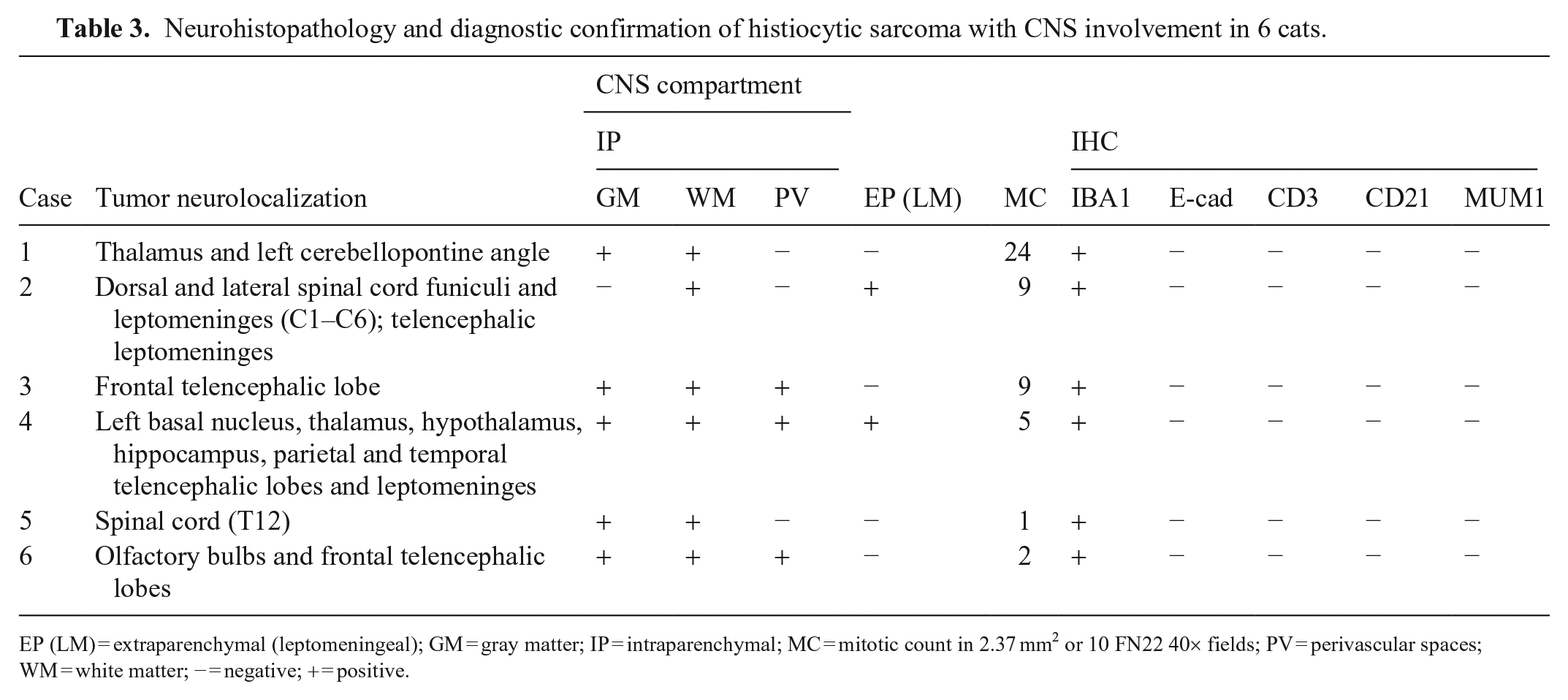
EP (LM) = extraparenchymal (leptomeningeal); GM = gray matter; IP = intraparenchymal; MC = mitotic count in 2.37 mm2 or 10 FN22 40× fields; PV = perivascular spaces; WM = white matter; − = negative; + = positive.
Histiocytic sarcomas involving the CNS of cats are reported only rarely in the veterinary literature and were rarely diagnosed in our searched database (10% of all histiocytic sarcomas diagnosed at both institutions). Histiocytic sarcomas can be localized or disseminated, 11 and can affect a wide range of tissues. Based on detailed examination of the medical history, autopsy results, and histologic examination of neural and extraneural tissues, cases 1–3 were diagnosed as localized (primary) CNS histiocytic sarcomas affecting the brain (2 cases) and the brain and SC (1 case), which has been reported only rarely in cats.8,16,17 Based on these criteria, CNS histiocytic sarcomas with tumors at extraneural sites cannot be confirmed as primary CNS histiocytic sarcomas.10,14 A similar diagnostic approach has been described for histiocytic sarcomas of the CNS of dogs. 9
The reported tumor neurolocalization for primary CNS histiocytic sarcomas in cats includes the fornix, 8 cervical and thoracic SC and adjacent nerves, 16 and brain (neurolocalization not specified). 17 Case 4 had a telencephalic tumor that also affected the trigeminal nerve, lung, and kidney, which is consistent with a disseminated histiocytic sarcoma involving the CNS. 11 Cases 5 and 6 were diagnosed based on histologic evaluation of biopsy samples and thus the presence of tumors in other tissues could not be assessed. No neuroanatomic predisposition was found in the CNS of dogs affected by histiocytic sarcoma. 9 However, too few cases have been reported to account for possible predisposing areas to be affected by histiocytic sarcomas in the brain or SC of dogs and cats. Given that only one autopsy case in our study had the SC examined, it is possible that spinal tumors may have been present and missed in the other autopsied cats.
As with many CNS tumors, computed tomography (CT) or MRI can be useful for the clinical diagnosis and neurolocalization of brain and/or SC lesions in affected patients.8,9,16 In dogs, the MRI appearance of extraparenchymal CNS histiocytic sarcoma closely resembles that of meningioma, making clinical differentiation between these 2 neoplasms difficult. MRI was performed in only one of our cases, and was successful in determining the neurolocalization of the tumor.
CNS histiocytic sarcomas are typically characterized by white-to-yellow, single-or-multiple masses that can occur throughout the brain or SC.8,9,16 In cats, gross differential diagnoses for CNS histiocytic sarcomas include granulomatous lesions, lymphoma, and meningioma.12,15 However, meningiomas tend to occur as a focal mass with characteristic firm consistency, which can be differentiated grossly from a histiocytic sarcoma. 12 Histologically, neoplastic cells with histiocytic morphology (round-to-polygonal-to-elongate, eosinophilic-or-vacuolated cytoplasm) associated with scattered multinucleate neoplastic cells allowed for a presumptive diagnosis of histiocytic sarcoma in our cases.
In cats, the main differential diagnosis for histiocytic sarcoma is Langerhans cell histiocytosis, a proliferative disorder of Langerhans cells that leads to nodules in the lungs and occasionally other tissues.2,3 Although histiocytic sarcoma and Langerhans cell histiocytosis can share similar morphologic features, diagnostic confirmation for the latter relies on the immunolabeling of histiocytic cells for E-cadherin,2,11 which was consistently negative in our cases. In addition, feline progressive histiocytosis, a cutaneous dendritic cell neoplasm that can spread to internal organs in its terminal stage, could be considered in situations in which cutaneous nodules are present, which was not the case in our patients. 1 Neither Langerhans cell histiocytosis nor feline progressive histiocytosis have been described in the CNS.1,2
Histiocytic sarcoma should also be differentiated from other round-cell neoplasms, such as lymphoma and plasma cell tumor, which were ruled out by the lack of immunolabeling for lymphocytic and plasma cell immunomarkers. One of the previously diagnosed histiocytic sarcomas in our case series was confirmed to be a B-cell lymphoma after widespread membranous immunolabeling for CD79 was observed throughout the neoplasm.
Histiocytic sarcoma arises from interstitial dendritic cells and less often from macrophages.5,6,11 Iba1 is a reliable immunomarker for the diagnosis of histiocytic lineage in dogs and cats, but it does not discriminate between dendritic versus monocyte or macrophage cell lineages.7,8,13 The determination of the cell of origin of histiocytic sarcomas may be challenging because it relies on a combination of immunomarkers that are not always available to be used on formalin-fixed, paraffin-embedded tissues. For example, specific interstitial cell immunomarkers (such as CD1 and CD11c) and macrophage and/or monocyte immunomarkers (such as CD11b) can be used only on acetone-fixed, snap-frozen tissue sections. 11 Immunomarkers such as CD14, MHC-2, MAC387, and lysozyme can be used on formalin-fixed tissues but are not able to differentiate between interstitial dendritic cell and macrophage or monocyte lineages. 11 Our previous attempts to characterize the cell of origin of CNS histiocytic sarcomas in dogs and cats have revealed inconsistent results given the lack of specificity of available immunomarkers and the low number of cases assessed (Rissi DR, pers. comm., 2022 Oct 3). A prospective study examining a larger number of histiocytic disorders is necessary to completely characterize these tumors using a wide range of immunomarkers or molecular techniques, which goes beyond the scope of our study.
Footnotes
Acknowledgements
We thank Dr. James Stanton (Department of Pathology, College of Veterinary Medicine, University of Georgia, Athens, GA, USA) for contributing case 4.
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors declared that they received no financial support for their research and/or authorship of this article.
