Abstract
Necropsy was performed on 2 smallmouth bass (Micropterus dolomieu) that died 8 days after experimental gill inoculation with Actinonaias pectorosa glochidia. The salient feature at necropsy was ragged gills containing multiple petechial hemorrhages and numerous white, iridescent dust-like particles easily confirmed as glochidia in squash preparations of the gill filaments. Microscopically, the gills contained multiple rounded glochidia encysted at the tips or along the length of the filaments. Parasitized filaments were thickened, blunted, and often fused. Lamellae were extensively fused and obliterated, with proliferation of the epithelial cells giving the filament a smooth outline. Sometimes glochidia were associated with necrosis and/or hemorrhage. These fish most likely died due to asphyxia associated with the severe branchial lesions caused by the glochidia.
The Unionoida, commonly known as freshwater mussels, naiads, or unionids, is a diverse order of bivalved mollusks whose larvae are parasitic, generally on fish. 4 Three families within the order, the Unionidae, Margaritiferidae, and Hyriidae, have glochidium-type parasitic larvae. 4 Glochidia are small, bivalved larvae that invade and encyst in the gills and fins of fishes and then metamorphize into to a free-living juvenile with adult internal organs. Juvenile mussels then excyst, drop off the host, and settle to the bottom to develop into mature mussels. Although hosts of most mussels are unknown, glochidia of some mussels are species specific, whereas others can use a wide variety of fish as hosts. 3, 5, 8
Freshwater mussels play an important role in an aquatic ecosystem, and their presence or absence provides clues about water quality and environmental health. Typically, infection with glochidia is subclinical, but if gill infection is severe enough, asphyxiation can occur. Mortality has been reported in both natural and experimental glochidial infections in salmonids. 1, 7, 9, 10 Because glochidial infections are rarely described in the literature, the purpose of this report is to heighten awareness of glochidia in fish and to describe the lesions associated with infection with glochidia of Actinonaias pectorosa in smallmouth bass (Micropterus dolomieu).
Two 7-cm long smallmouth bass died 8 days after inoculation with an unknown quantity of glochidia of the freshwater mussel A. pectorosa. To prepare the inoculum, gravid female mussels, collected from streams in Virginia, USA, were placed in crystalizing dishes half filled with water and the marsupia (gills containing glochidia) punctured with a scalpel and perfused with water from a pipette to expel glochidia into the dish. Glochidia from several females were mixed and a few drops collected in a pipette and placed onto the host fish's gills.
At necropsy, the fish were covered by white slime and had petechial hemorrhages in the skin and reddening of the skin at the base of the tail. The fins were ragged and livers pale. However, the most striking changes were in the gills. The gills were ragged and contained multiple petechial hemorrhages, as well as numerous white, iridescent dust-like particles.
Squash preparations of gill filaments contained numerous thin-shelled organisms compatible with glochidia (Figs. 1, 2). The organisms were composed of 2 nearly symmetrical, rounded (referred to as subelliptical in the glochidia literature 6 ) valves, 242 to 247 μm (246 ± 2.5 μm, n = 4) in length and 247 to 258 μm in height (251 ± 5.32 μm, n = 4), joined by a hinge (the dorsal margin) that was apparent when organisms were seen on edge.
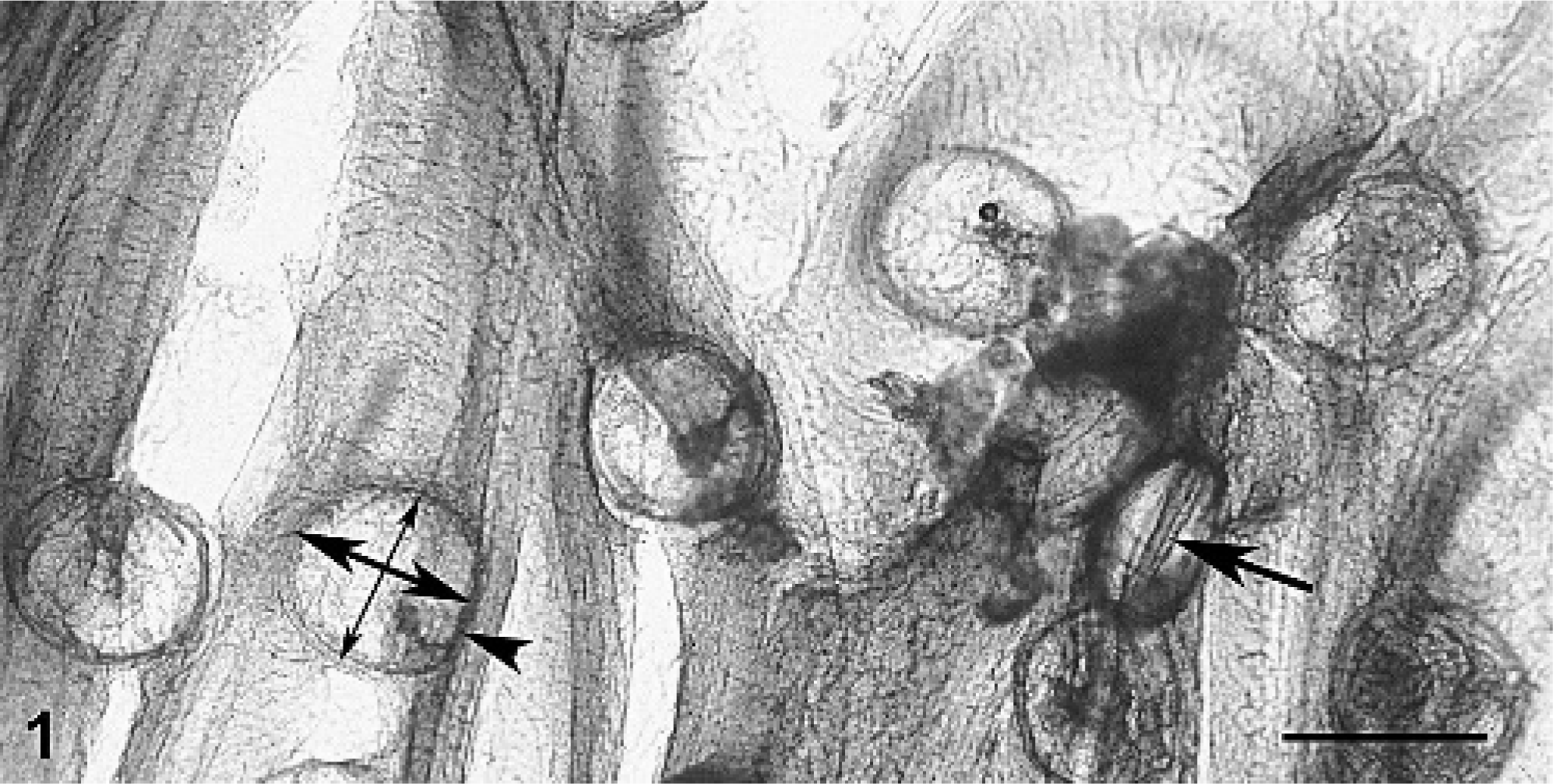
Gill; smallmouth bass. Squash preparation of gill filaments parasitized by glochidia of Actinonaias pectorosa. Note numerous glochidia that are roughly round when viewed sagitally. A single organism is seen on edge revealing its hinged bivalve nature (single-headed arrow). One organism is used to demonstrate the dorsal margin (arrowhead), which is the hinged margin, height (dorsal to ventral, thicker double-headed arrow), and length (anterior to posterior, thinner double-headed arrow). Bar = 250 μm.
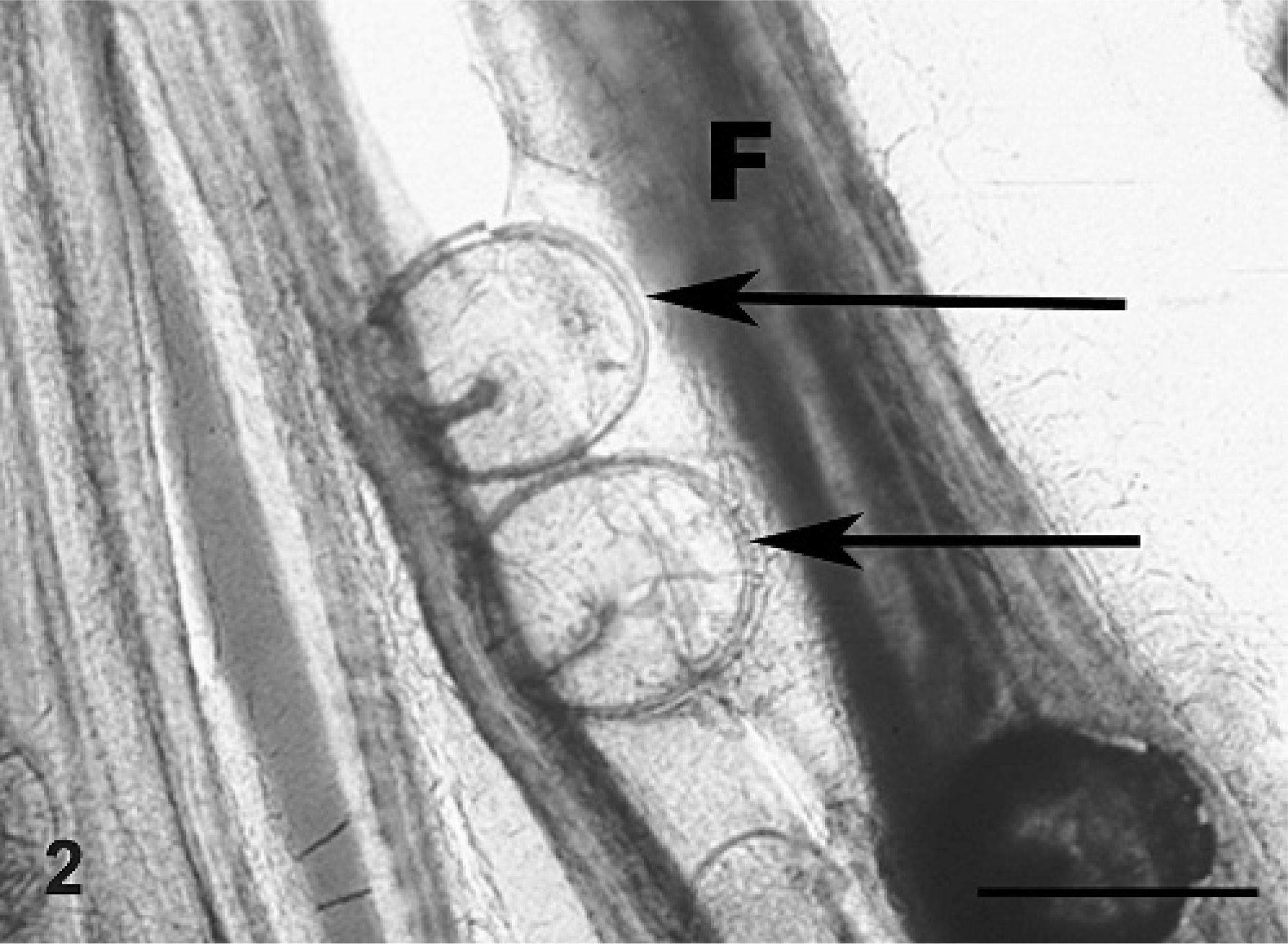
Gill; smallmouth bass. Squash preparation of gill filament. Note attachment of 2 glochidia along the length of a filament (F). The arrows point to the ventral margin of the glochidia where they have grasped the filament for attachment. Bar = 250 μm.
Tissues were fixed in 10% buffered formalin and embedded in paraffin, and 3-μm sections were stained with hematoxylin and eosin. Microscopically, gills contained multiple glochidia encysted at the tips or along the length of filaments (Figs. 3, 4). Parasitized filaments were thickened, blunted, and often fused (Fig. 5). Lamellae were extensively fused and obliterated, with proliferation of the epithelial cells giving parasitized filaments a smooth outline (Figs. 3–5).
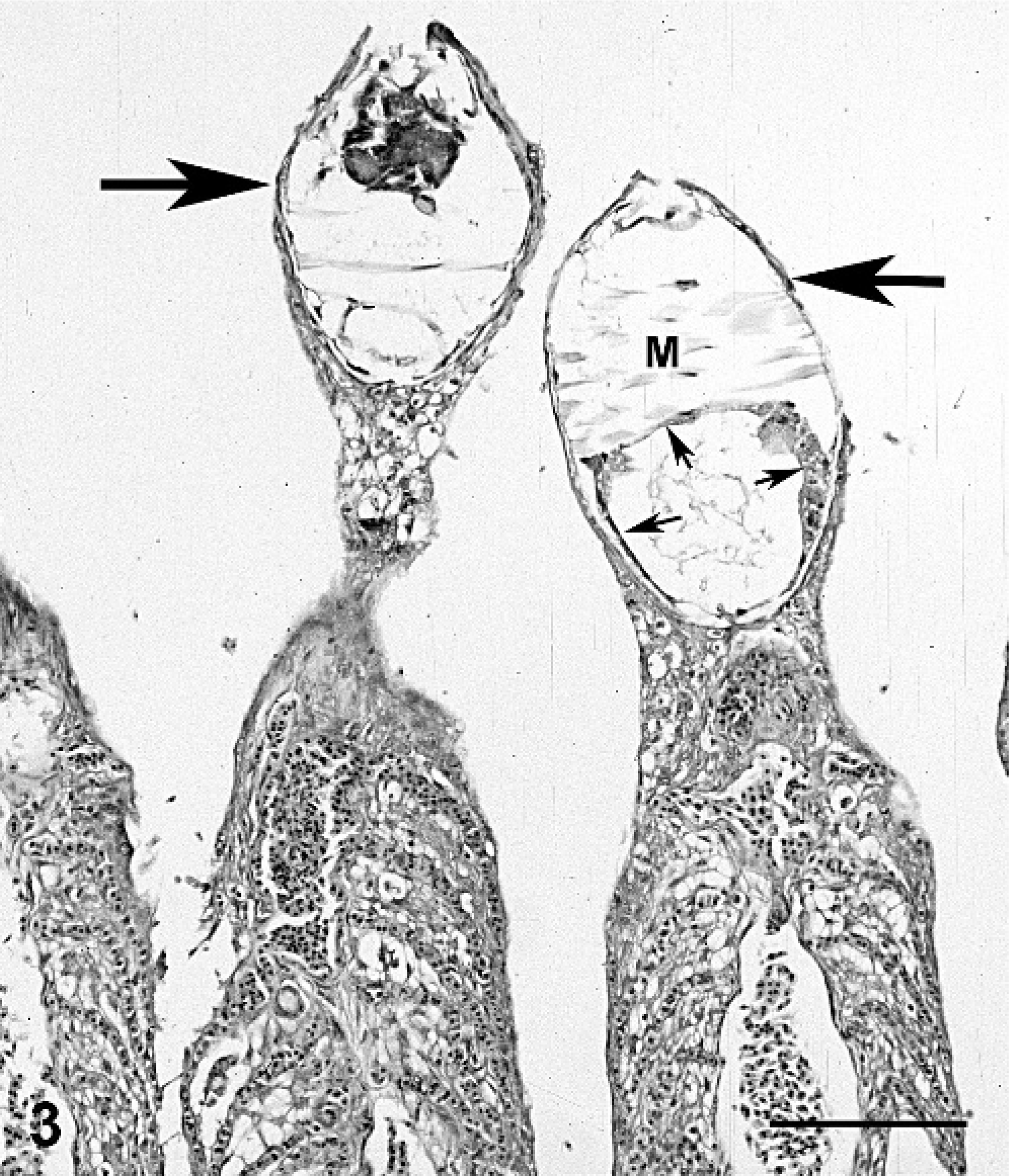
Gill; smallmouth bass. Two gill filaments have a glochidium of Actinonaias pectorosa at the tip that is encysted by a single layer of host cells (arrows). Internal structures seen are adductor muscle (M) and mantle (small arrows). Filaments are smooth in outline due to lamellar fusing and epithelial hyperplasia. HE stain. Bar = 130 μm.
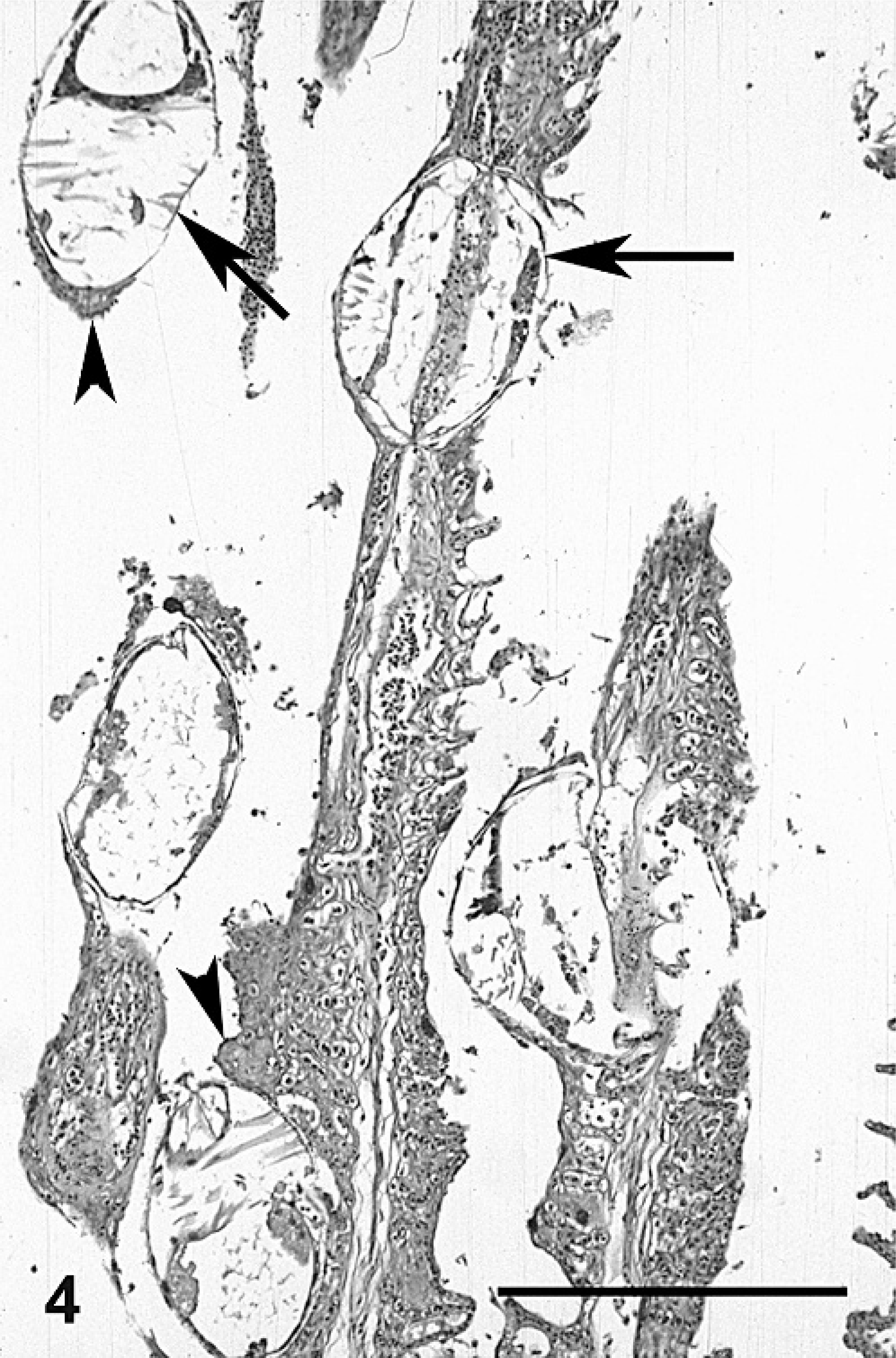
Gill; smallmouth bass. Five encysted glochidia of Actinonaias pectorosa along the gill filaments that have extensively fused lamellae. Glochidia are encysted by single (arrows) and multiple (arrowhead) layers of epithelium. HE stain. Bar = 250 μm.
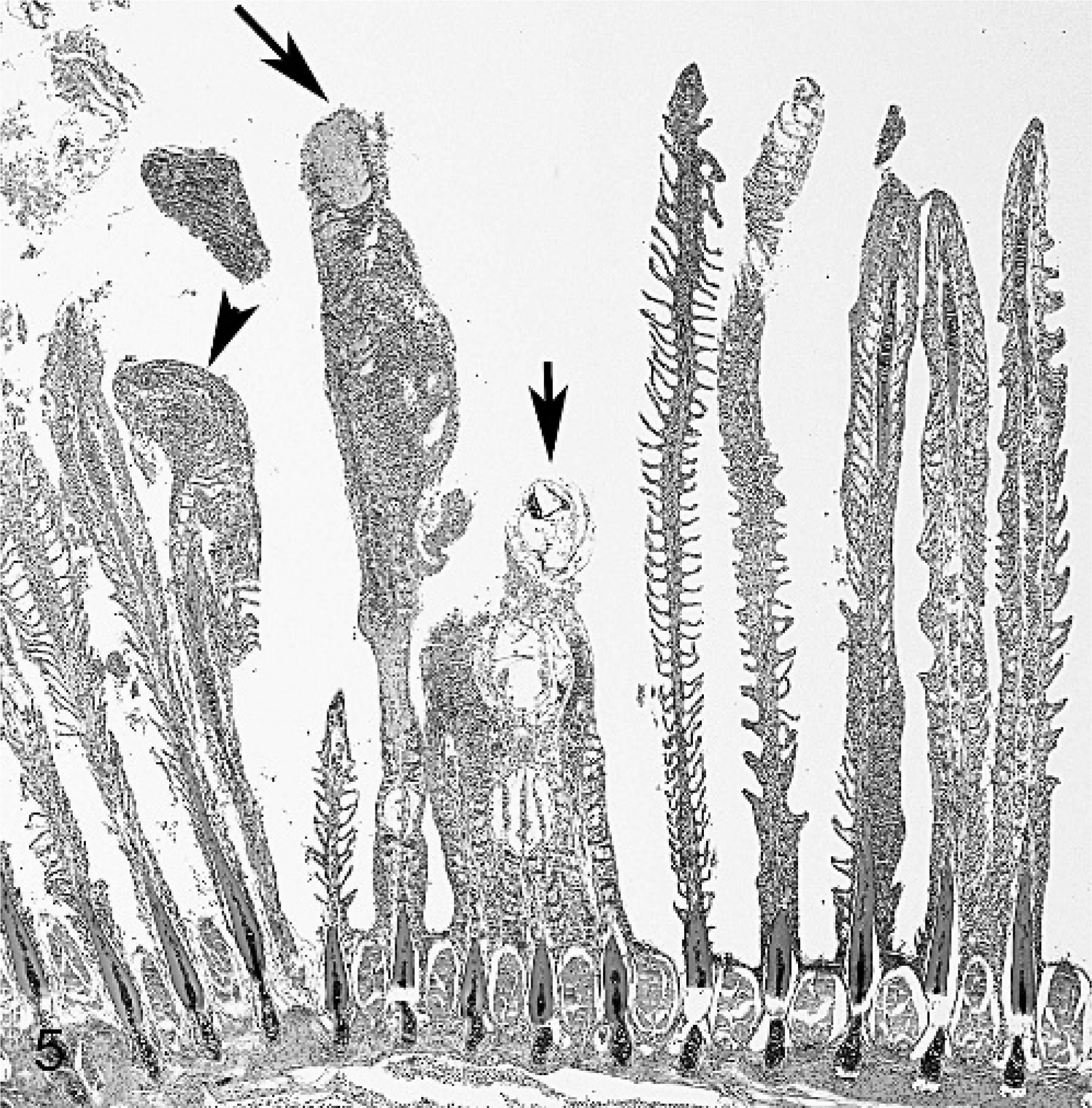
Gill; smallmouth bass. Centrally, 4 filaments are fused encysting 3 glochidia of Actinonaias pectorosa (shorter arrow). Many filaments have extensive fusing of lamellae, resulting in a smooth filament outline and epithelial hyperplasia that is sometimes extensive (arrowhead). A filament with multiple areas of necrosis (longer arrow) is shown at higher magnification in Fig. 6 . HE stain.
The encysted glochidia were roughly round in outline and sometimes surrounded by a wall composed of host epithelial cells. When located at the tips of filaments, a single layer of squamous epithelium surrounded the glochidia (Fig. 3), but when located along the sides of filaments, the cyst wall was a single to multiple layer of epithelium (Figs. 4, 6). The glochidia had a minimal internal structure that consisted of muscle type tissue (the adductor muscle) and rings of cuboidal cells that composed the mantle (Fig. 3). Sometimes glochidia were associated with lamellar necrosis and/or hemorrhage (Fig. 6).
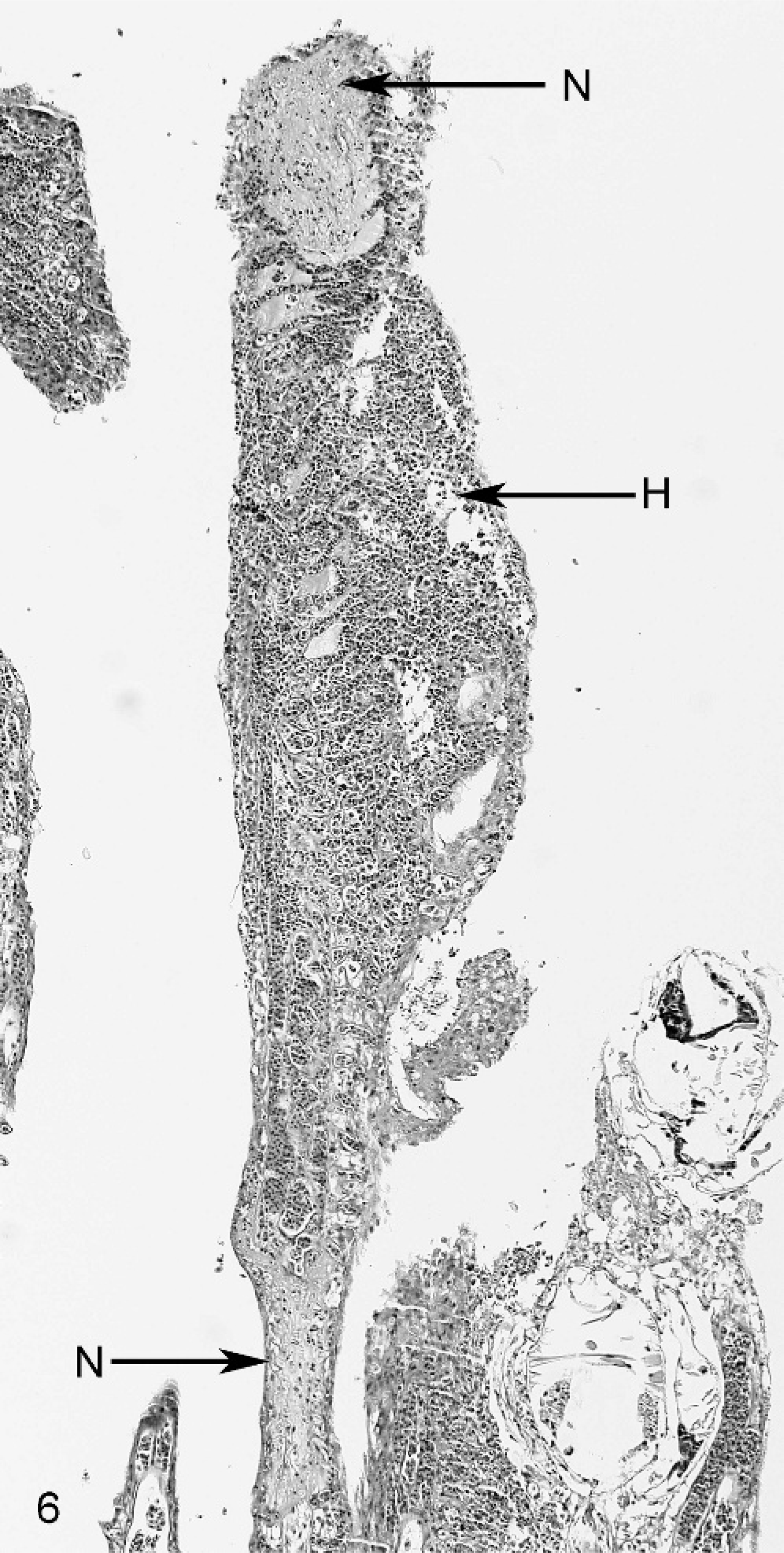
Gill; smallmouth bass. The filament at the longer arrow in Fig. 5 , in addition to having fused lamellae and epithelial hyperplasia, has multiple foci of necrosis (N) and hemorrhage (H). HE stain.
Microscopic lesions in other tissues included foci of epidermal hyperplasia on fins and subcutaneous edema and mild inflammation at the base of the tail. The tail lesion contained a few gram-negative bacilli, but bacteria were not seen in visceral organs. Multiple bacteria were cultured and included Aeromonas hydrophila from the skin, liver, and gill of both fish; Plesiomonas shigelloides from the skin of 1 fish; Pseudomonas stutzeri from liver and gill of 1 fish; and Pseudomonas putida from the skin of both and liver and gill of 1.
The iridescent dust-like particles seen grossly in the gills of these fish were glochidia, and their identity was easily confirmed by squash preparations of the gill filaments. In fact, these organisms were much easier to identify in squash preparations than in histologic sections. Glochidia of various species vary in size and may be rounded (as A. pectorosa), rectangular, or triangular. 3 There are problems with identification to the species level using light microscopy, and scanning electron microscopy may be needed to identify such features as protuberances to differentiate species within a genus. 11
Because branchial lesions in these fish were severe and contained numerous encysted glochidia, asphyxia was most likely the immediate cause of death. Although bacteria were not seen in the viscera, the presence of petechial hemorrhages suggests that sepsis may have been a complicating factor. However, it is uncertain if any of the isolated bacteria are significant. Secondary fungal infections are relatively common in salmonids infected, either experimentally or naturally, with glochidia of Margaritifera margaritifera, 7, 9 so invading or exiting parasites may provide a portal of entry for secondary invaders, such as bacteria and fungi.
The histologic changes in the gills of these fish were similar to those described for infection of salmonids with glochidia of M. margaritifera. 7 Microscopic findings in salmonids infected with M. margaritifera glochidia suggest that once glochidia attach to the gill by biting or grasping, they are encysted by epithelial proliferation and lamellar fusion. 2, 9 The microscopic appearance of the gill filaments and glochidia cysts in this report would support a similar mechanism of cyst formation for the glochidia of A. pectorosa. Necrosis and hemorrhage in parasitized filaments as seen in these fish have not been described for other glochidia, and their cause is uncertain. It may have been the result of vascular damage caused by the sheer number of parasitizing glochidia. The glochidia had not metamorphized to the juvenile form, so this was not a manifestation of excystment, which is due to the juvenile mussel breaking the wall with its foot. 7
Susceptibility to a particular glochidium may vary with host fish. For example, when 6 species of salmonids were infected with M. margaritifera glochidia, a range of susceptiblity was observed. Chinook salmon were the most susceptible, with mortality occurring even at low doses, whereas coho salmon were relatively resistant. 7 Length of infection and lesions also may vary depending on host susceptibility. 7 In highly susceptible chinook, parasites were retained for 12 weeks through complete metamorphosis to the juvenile form, but histologically there was little hyperplasia in the gills. On the other hand, relatively resistant coho sloughed glochidia in about 5 days and infection was associated with a severe hyperplastic response in the gills. Similar differences in encapsulation and gill tissue reactions and length of infection have been seen with Lampsilis radiata siliquoidea infections in walleye (Stizostedion vitreum vitreum), a suitable host, versus common carp (Cyprinus carpio), a resistant host. 12
Although most natural infections of glochidia go undetected, this report demonstrates the potential for lethal glochidia infections. Introduction of the zebra mussel, an aggressive, nonglochidia-forming species, demonstrates the potential for introduction of glochidia-producing mussels that might out compete and replace native mussel species. Susceptibility of native fishes to exotic mussel glochidia could vary substantially from native mussel species, and the potential for exotic glochidia to cause lethal disease in certain species needs to be recognized.
