Abstract
Cardicola forsteri is a blood fluke that is highly prevalent among cultured southern bluefin tuna (SBT), Thunnus maccoyii, in South Australia. The role of C forsteri in annual SBT mortality outbreaks, which peak 6 to 12 weeks poststocking, is unknown. The objective of this study was to identify lesions unique to cultured SBT that died during a mortality event in 2009 and to determine the significance of C forsteri–associated lesions. Cultured SBT were sampled from 4 pontoons of a single site in Spencer Gulf that experienced a mortality epidemic that spanned 5 to 14 weeks poststocking. Study SBT comprised 7 that died during peak mortality, 27 that did not die, and 10 wild-caught (noncultured) SBT. All cultured SBT had branchitis and myocarditis due to C forsteri, whereas no life stages of C forsteri were histologically identified in any wild-caught SBT. Mortality was associated with the presence of severe branchitis (P < .005), and the odds of severe branchitis were 90 times greater for SBT that died than for SBT that were live caught during peak mortality (95% confidence interval, 5 to 1,684). In SBT that had died, no lesions other than those associated with C forsteri were of sufficient severity or physiologic significance to account for death. Other lesions common among cultured SBT included systemic granulocytic perivascular infiltrate, granulocytic gastric infiltrate, hepatic lipidosis, visceral granulomas, and branchial parasitic infestation. This study shows for the first time that a substantial proportion of poststocking mortality in cultured SBT is strongly associated with severe branchitis caused by C forsteri.
In Australia, individual transferable fishing quotas for southern bluefin tuna (SBT), Thunnus maccoyii, were established in 1984 as a response to a declining wild fishery. Subsequent to quota declines, the SBT aquaculture industry was established in 1991 as a means to improve the value of a restricted catch. The industry now comprises 12 private companies based in Port Lincoln, South Australia, and is presently one of the largest aquaculture sectors in Australia, producing over A$115 million of product annually (2009 data supplied by the Australian Southern Bluefin Tuna Industry Association). Young 15- to 20-kg fish are purse seine captured from December to March, when present along the continental shelf in the Great Australian Bight. Fish are transferred to tow cages and slowly moved to culture sites in waters immediately offshore from Port Lincoln in Spencer Gulf. Fish are grown in pontoons for 3 to 5 months until they reach approximately 30 to 35 kg.
By 2002, consistent patterns of annual mortality epidemics were occurring across several farms. The mortality events peak around 6 to 12 weeks following introduction to grow-out sites, and they appear tightly correlated to the time that fish had been present in the grow-out site, rather than the calendar date. Initially, annual losses were low, reaching 2 to 3% in some farms. By 2008, all farms were affected, and mortalities approached 10% for the worst-affected farms, where some pontoons experienced up to 15% mortality. It is estimated that this level of mortality would correspond to more than A$10 million in annual lost production industry-wide.
In 2008, a diagnostic veterinary investigation by Future Fisheries Veterinary Service (FFVS) and the University of Sydney was conducted for four companies of the SBT industry. SBT dying during peak mortality, 6 to 12 weeks poststocking, were affected with severe branchitis caused by Cardicola forsteri, a sanguinicolid blood fluke. The branchitis appeared of sufficient severity to have a negative physiologic impact. However, despite widespread prevalence of C forsteri infection among cultured Australian SBT, mortality had not been previously attributed to the infection, 3,4 and C forsteri was considered to pose low overall risk to the SBT industry. 14 Because the prevalence of severe C forsteri–associated gill lesions was unknown in fish that had not died during the 2008 mortality spike, its role in SBT mortalities remained uncertain.
The objective of the present study was to describe lesions unique to cultured SBT that died during a mortality event in 2009 and to determine the health significance of C forsteri–associated lesions.
Materials and Methods
Sampling of Fish
All cultured fish evaluated by the present study were obtained from a single culture site managed by one tuna company, Blaslov Fishing Group Pty Ltd, from January 27 to May 7, 2009. Following the 2008 diagnostic investigation of poststocking mortality by FFVS and the University of Sydney, this company elected to fund further investigation in 2009. Since 2004, the tuna company’s stock had been held at three culture sites and was annually affected with mortality epidemics of variable severity, which peaked at 6 to 12 weeks poststocking. In 2009, pontoons were positioned on a virgin lease site, physically remote from other leases in operation (Fig. 1), more than 2.5 km north from the nearest other lease site stocking SBT. The culture site had 6 pontoons, only 4 of which were sampled for this study. Two separate stockings took place, each from a separate fishing trip and tow from the Great Australian Bight. Tow 1, comprising 120 tons of SBT, was fed 5 tons of sardines opportunistically during the tow. Pontoon 1 was populated with 2,495 fish from tow 1 on January 27, 2009. Tow 2, comprising 90 tons of SBT, was fed 12 tons of sardines opportunistically during the tow. On March 6, 2009, a total of 1,771, 1,665, and 1,344 fish from tow 2 were stocked into pontoons 2, 3, and 4, respectively. An additional 780 fish sourced from a tow of another farm were stocked into pontoon 4 on 8 April 2009.
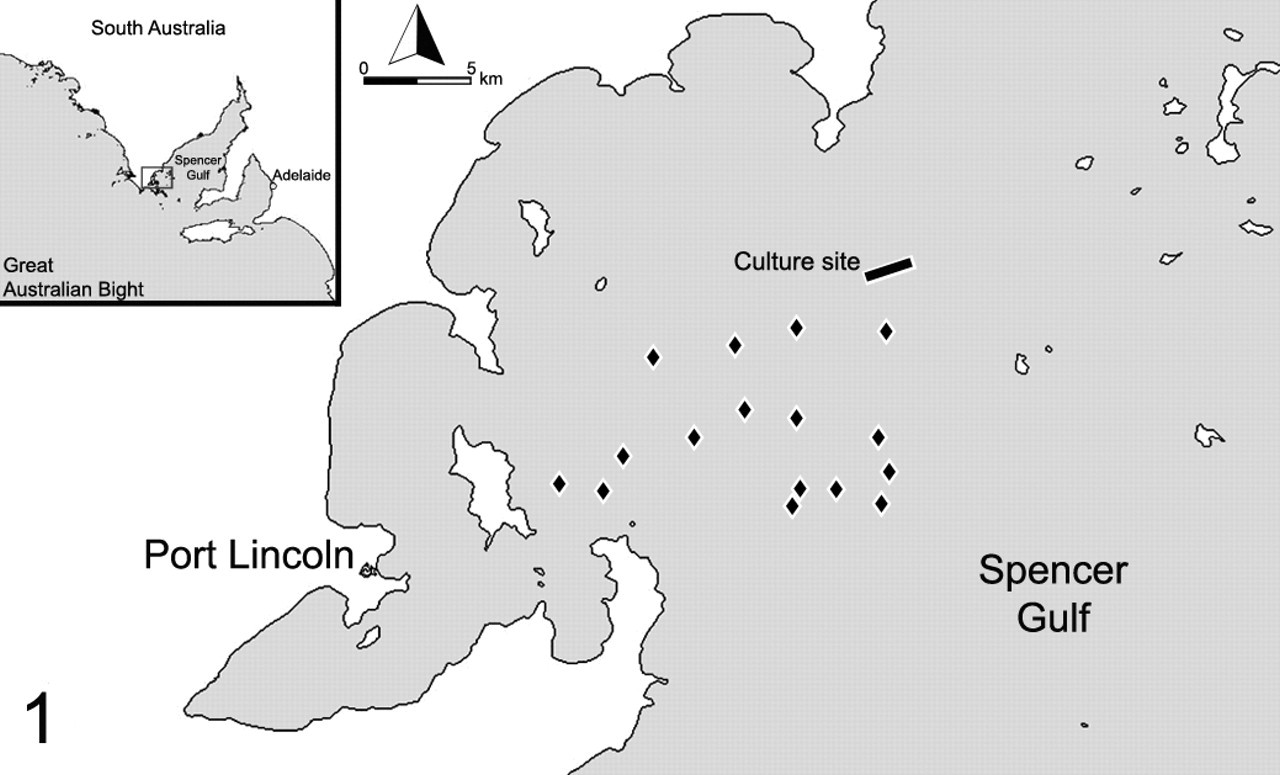
Location of study culture site (rectangle) in Spencer Gulf and approximate locations of neighboring culture sites (diamonds). This site had not been leased before the 2009 season, and it was at least 2.5 km from other culture sites.
A case–control study design was employed to identify histologic lesions that were associated with mortality (Table 1). Cases were defined as any fish that died without premonitory clinical signs and with an absence of obvious external lesions. Cases were sampled from pontoons 2 to 4 during the peak of a mortality epidemic, from May 4 to May 7, 2009. During the study period, each pontoon was inspected daily by divers. Divers involved in collecting mortalities from the pens assessed fish on the basis of the degree of rigor, body temperature, gill color, and degree of mucous to estimate which fish had died most recently. Only the fish that were deemed to be fresh mortalities were sampled, to ensure high-quality material for histologic analysis. Four control groups were sampled: wild-caught fish sampled well outside the grow-out lease area (group A) and fish from each of 2 tows (groups B–D) held in 3 separate grow-out pontoons (pontoons 1–3) for varying durations. Fish of control group A were wild captured with a baited hook and line on March 1, 2009. Wild-caught fish were euthanized through use of a cranial spike. Fish of control group B were sampled as part of commercial net harvest of 140 fish from pontoon 1 on May 5, 2009, approximately 8 weeks after peak mortality had affected its cohort. The commercial harvest method involves the use of a harvest net, which purses up the fish and confines them in shallow water where divers individually catch them. The fish were then euthanized using the Japanese “Ike Jime” method, where a spike is driven through the pineal window to penetrate the brain. A coring cylinder in then used to bore a hole in the head of the fish down to the brain. Following this, a wire is inserted through the hole and down the spinal column to destroy the medulla oblongata and upper spinal cord. Fish of control groups C and D were sampled on May 7, 2009, during the period that peak mortality was affecting each cohort, using baited hook-and-line capture. Once on board, the fish were euthanized through use of a cranial spike. Digital photographs were taken for the comparison of the gross appearance of tissues from sampled fish.
Sampling Groups
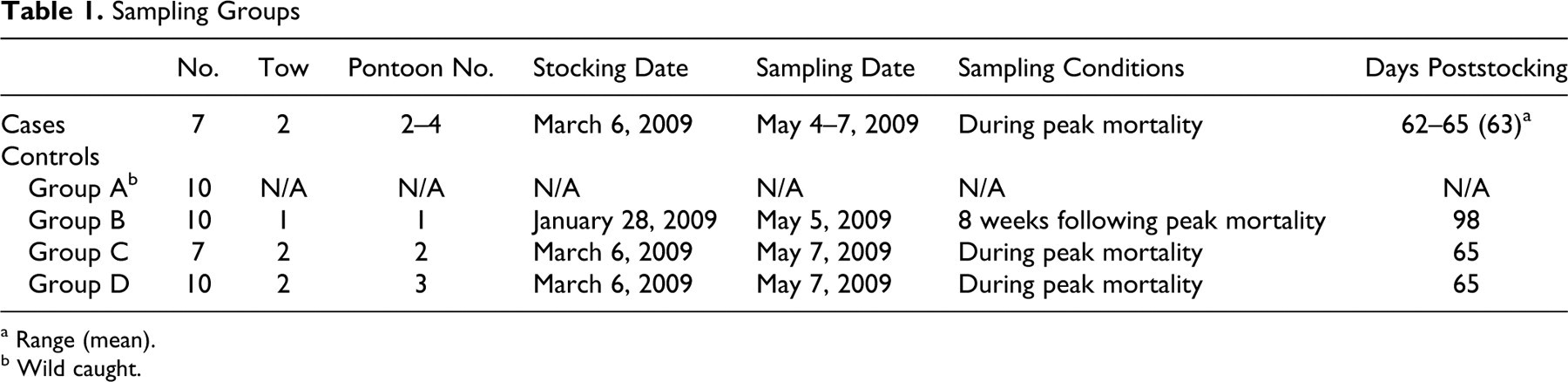
a Range (mean).
b Wild caught.
Pathologic Examination
Case fish were collected by divers, placed on ice, brought to shore, and then immediately necropsied, within 3 to 5 hours of sampling. Control fish were necropsied immediately following euthanasia. For control group A, only cardiac ventricle and gill were collected and immersed in 10% formalin in sea water. For all other fish, gill, atrium, ventricle, stomach, intestine, pyloric cecae, liver, and spleen were collected and immersed in 10% formalin in sea water. The same representative location of tissue was used for all samples. The middle of the outermost (first) gill arch was sampled. The spleen sample was generally taken from the center of the organ. The intestine sections were sampled in the terminal rectum and from the proximal intestine, immediately distal to the pylorus of the stomach.
For histopathology, at least two 1.5-cm-diameter sections of ventricle and atrium were prepared from each heart evaluated. Gill samples were decalcified using 12.5% EDTA before histopathology preparation. Six sections of gill were prepared from the outer (lateral) row of each gill sample. A segment of lamellae 1 cm in width was trimmed and sectioned such that filaments were evaluated from base to tip, on both inner and outer filament surfaces. Care was taken when filament samples were embedded within paraffin wax such that 3 sections represented the outer (efferent) lamellar surface and 3 sections represented the inner (afferent) lamellar surface.
Tissues were processed routinely for histopathology and sectioned at 5 μm. All sections were stained with hematoxylin and eosin (HE). Ziehl Neelsen acid fast, Fite-Faraco acid fast, and gram Twort stains were used where indicated by pathologic findings. Histopathologic examination of all sections was completed by one pathologist (M.M.D.) who had no prior knowledge of the history or clinical findings of study fish.
Statistical Analysis
The gill lesions of fish in the case and control groups C and D were dichotomized based on severity of C forsteri branchitis. Similarly, the heart lesions of fish in the case and control groups C and D were dichotomized based on the presence of acute–subacute C forsteri myocarditis. Fisher exact test and odds ratio were produced to compare differences in the occurrence of severe C forsteri branchitis and acute–subacute C forsteri myocarditis among fish that died and those that did not. Fisher exact test was used to test for associations of the occurrence of moderate to severe granulocytic gastric infiltrate, branchial parasitic infestation, visceral granulomas, visceral didymozoidiaisis, and hepatic lipidosis, with the occurrence of mortality. Pearson chi-square was used to test for the association between severity of systemic granulocytic perivascular infiltrate and the occurrence of mortality. A P value of < .05 and a confidence interval that did not contain the value 1 were considered to be statistically significant.
Results
In sum, 173 mortalities occurred in pontoons 1–4 during the 2009 season. Cumulative mortality for pontoons 1–4 totaled 2.1% and ranged from 0.3 to 3.6% per pontoon (mean, 2.3%). Mortality epidemics occurred in all pontoons from 5 weeks poststocking (Fig. 2) but were most notable in pontoons 2–4. Epidemics peaked between 6 and 9 weeks poststocking and spanned 5 to 10 weeks poststocking in pontoon 1, 6 to 10 weeks poststocking in pontoon 2, 5 to 8 weeks poststocking in pontoon 3, and 5 to 14 weeks poststocking in pontoon 4. The epidemic in pontoon 4 initially subsided at around 10 weeks poststocking but then resumed at 12 weeks poststocking. Mortality was notably lower in pontoon 1 relative to other pontoons of the culture site; however, fish of this pontoon were stocked over a month earlier than other study fish. The fish were also among the first in the industry to be moved into the growing area for the 2009 season. Compared with the other pontoons, pontoon 1 was also harvested around 4 weeks earlier; hence, the grow-out duration was significantly shorter.
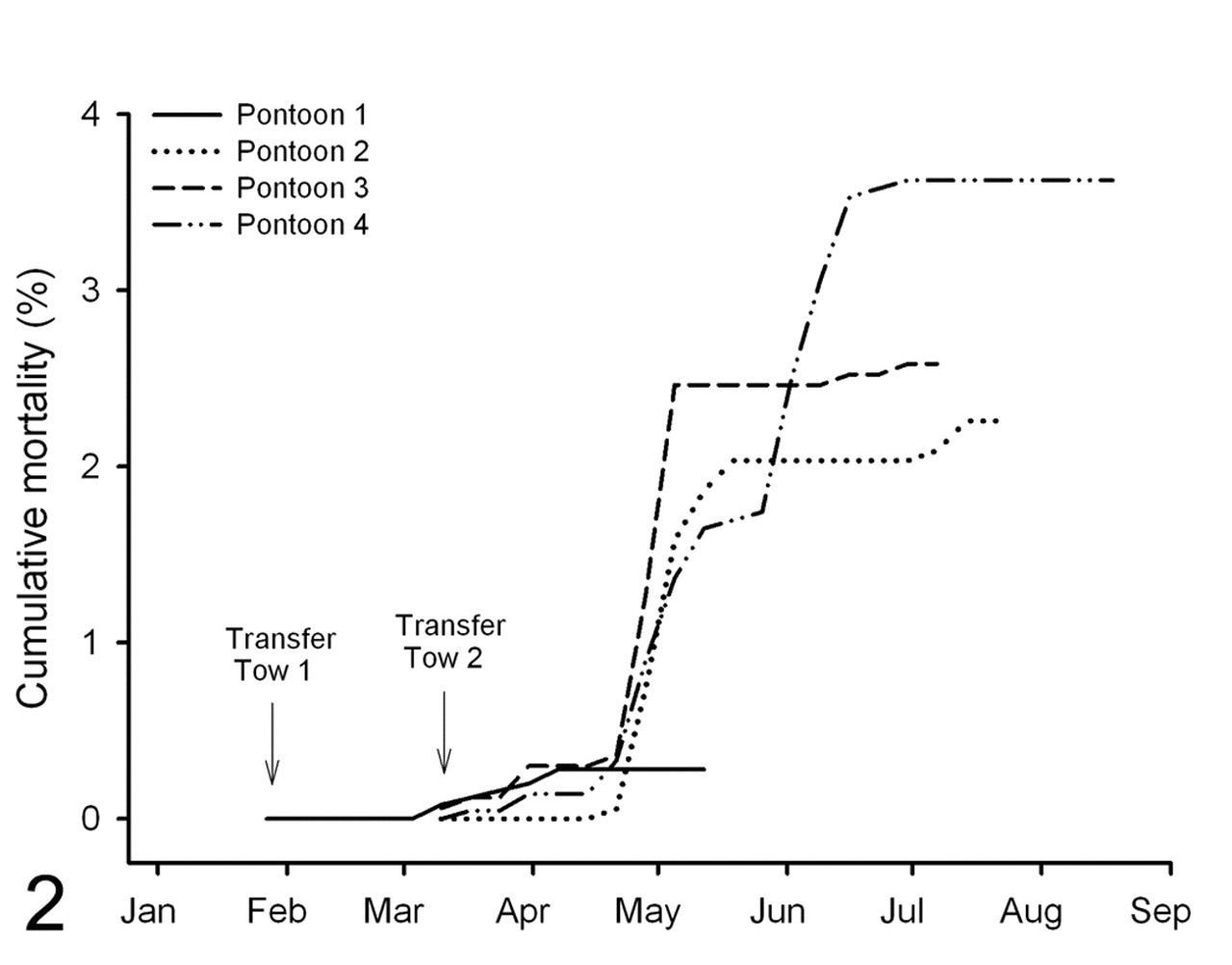
Cumulative mortality per pontoon throughout the 2009 season. Pontoon 1 was populated with fish from tow 1 on January 27, 2009. Pontoons 2, 3, and 4 were populated with fish from tow 2 on March 6, 2009. An additional 780 fish sourced from a tow of another farm were stocked into pontoon 4 on April 8, 2009. Note that lines cease after fish have been harvested from pontoons.
C forsteri–Associated Branchitis
Branchitis associated with C forsteri occurred in all fish, except those of control group A (wild-caught fish). Grossly, gills with severe branchitis were segmentally covered with white-pink opaque mucoid exudate. The gills had a streaked appearance owing to alternating pallor and reddening of filaments (Fig. 3).
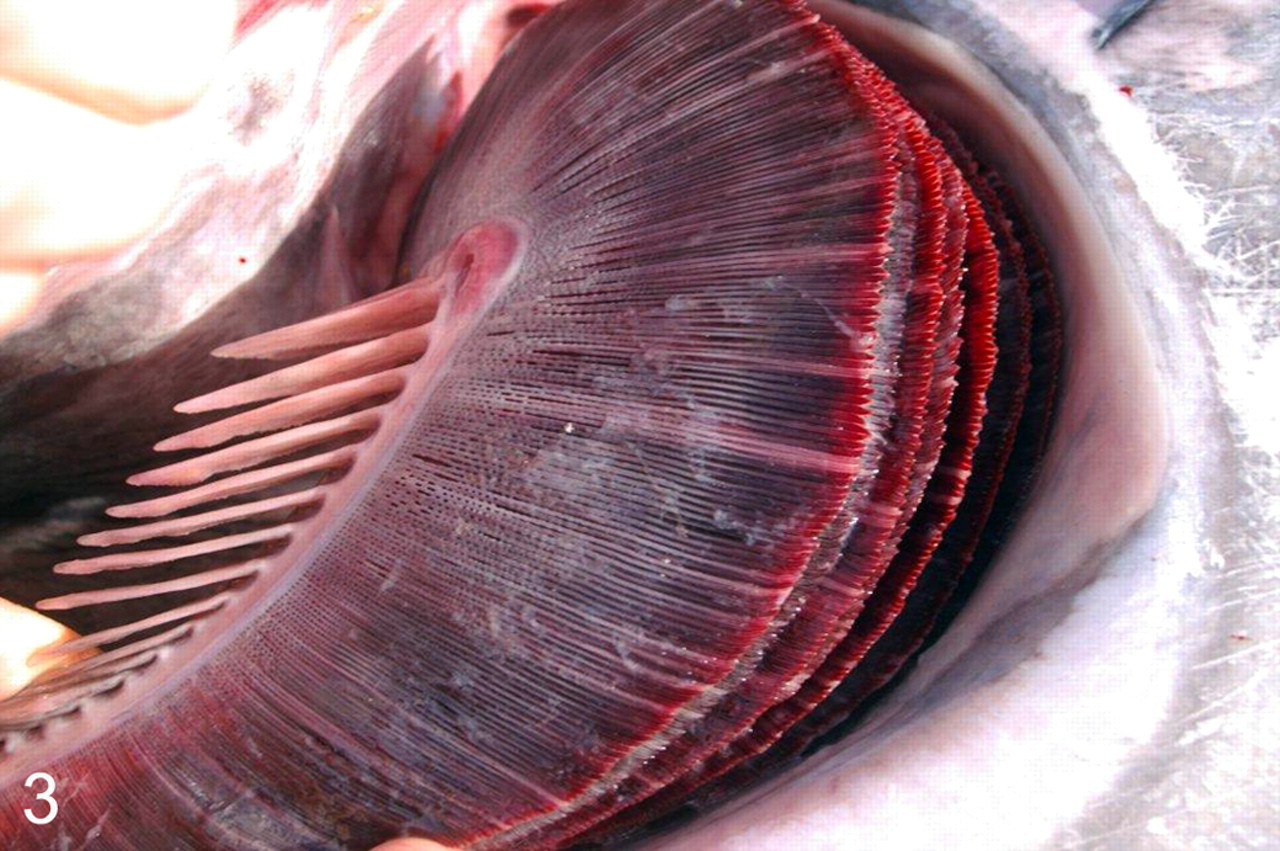
Gill; Southern bluefin tuna, mortality from 2008 investigation. Similar changes occurred in all study fish with severe acute branchitis due to Cardicola forsteri. Filaments are streaked with pallor and segmentally covered in mucoid exudate.
Histologically, in acute branchitis, myriad eggs were localized to lamellar tips (presumptively ruptured from lamellar capillaries) and, to a lesser extent, were within afferent filament arteries (Figs. 4–6). Eggs, consistent with those described by Colquitt et al, 4 were round to oval and 25 to 50 μm in diameter; they had very thin walls and contained well-developed ciliated miracidia. Lamellar tissue adjacent to an egg was necrotic, contained hypertrophic epithelial cells, and was infiltrated with scattered mononuclear leukocytes and granulocytes; its surface was often covered with hemorrhage, protein, and/or necrotic cell debris. The architecture that adjoins opposing lamellae was lost in these areas. The distribution of acute branchitis was multifocal but widespread, involving most of the gill sections evaluated. Small afferent filament arteries frequently contained degenerate eggs that were associated with thrombi and/or mural granulocytic infiltrates.
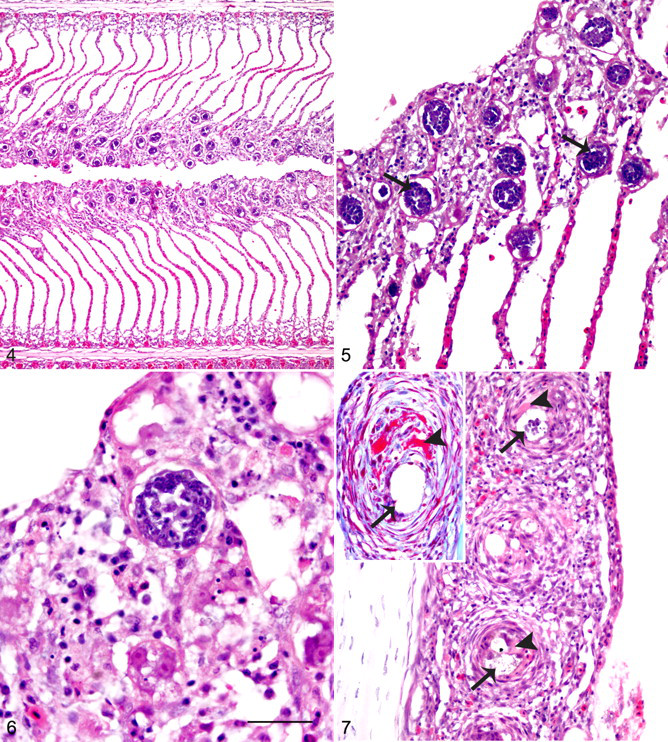
Gill; Southern bluefin tuna, case group. Severe acute branchitis. There is widespread distortion of lamellar architecture, and numerous eggs are within the tips of lamella. HE.
Gill; Southern bluefin tuna, control group B. Moderate chronic branchitis. Granulomas (arrows) are centered on degenerate eggs (inset). HE.
Subacute branchitis was also multifocal, although less widespread than acute branchitis. Subacute branchitis also contained several eggs within lamellae and afferent filament arteries but fewer than in sections with acute branchitis. In addition, subacute branchitis had less severe lamellar necrosis than sections with acute branchitis. In contrast, foci of lamellar fusion due to epithelial hyperplasia and thrombi and eggs within afferent filament arteries (Fig. 7) were more common in subacute relative to acute branchitis.
Chronic branchitis was mild to moderate in every case and comprised small scattered granulomas within filaments (Fig. 8). Eggs were sparsely within afferent filament arteries, and when present, they were often fragmented or degenerate. Eggs were very rare within lamellae. In addition, there was adventitial hyperplasia and smooth muscle hypertrophy and hyperplasia of the tunica media of filament arteries, whereas lamellar necrosis and branchial vasculitis and thrombosis were rare.
C forsteri–Associated Myocarditis
C forsteri–associated myocarditis occurred in all fish, except those of control group A (wild caught), and was mild in every case. Acute myocarditis was slight and consisted of few eggs entrapped within the atrium and the spongiosa layer of the ventricle, with little host response other than minimal villous endothelial hyperplasia and granulocyte infiltration of the endocardium.
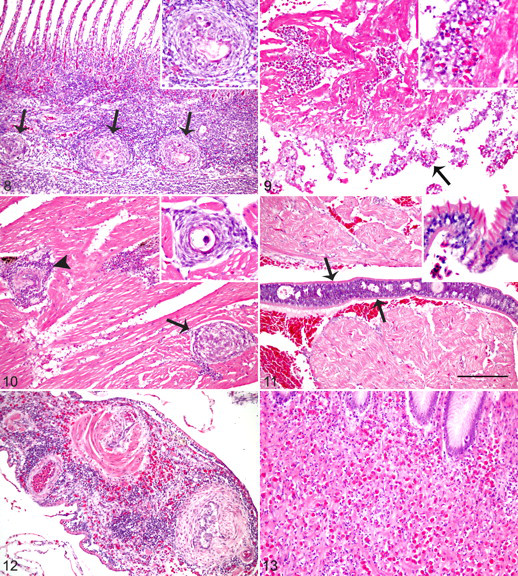
In subacute myocarditis, granulocytes and lesser mononuclear leukocytes expanded the endocardium and epicardium (Fig. 9). The degree of villous endothelial hyperplasia of the endocardium was much more prominent than in acute myocarditis. Subacute myocarditis tended to have more entrapped eggs than acute myocarditis, and eggs were more abundant in the ventricle than the atrium. In 2 fish, epicarditis extended into epicardial ganglia.
Chronic myocarditis comprised large numbers of granulomas scattered throughout the spongiosa layer of the myocardium, often centered on degenerate eggs (Fig. 10). Villous endothelial hyperplasia and endocardial and epicardial leukocyte infiltrate were mild or absent.
Histologically evident adult flukes were within atria of 2 fish from groups C and D (Fig. 11). Adult flukes, consistent with those described by Cribb et al, 5 were dorsoventrally compressed and 100 to 150 μm in width, with marginal tegumental spines, parenchymous body cavity, and indistinct reproductive and digestive tracts.
Differences in C forsteri–Associated Lesions Among Study Groups
The prevalences of C forsteri lesions were not evenly distributed among study groups (Table 2). C forsteri life stages were not microscopically identified in any fish from control group A (wild caught). In contrast, life stages of C forsteri were microscopically identified in all remaining study fish and were always found in association with branchitis and myocarditis of varying severity and chronicity. Fish of group C had mixed chronicity of branchitis and myocarditis, whereas fish of group D all had subacute branchitis and myocarditis, and fish of group B all had chronic branchitis and myocarditis. Five of 7 case fish (mortalities) had severe acute branchitis and mild myocarditis. One of 7 had moderate subacute branchitis and mild subacute myocarditis, and 1 of 7 had severe subacute branchitis and mild subacute myocarditis.
Lesions Identified in Fish of Each Study Group: n (%)
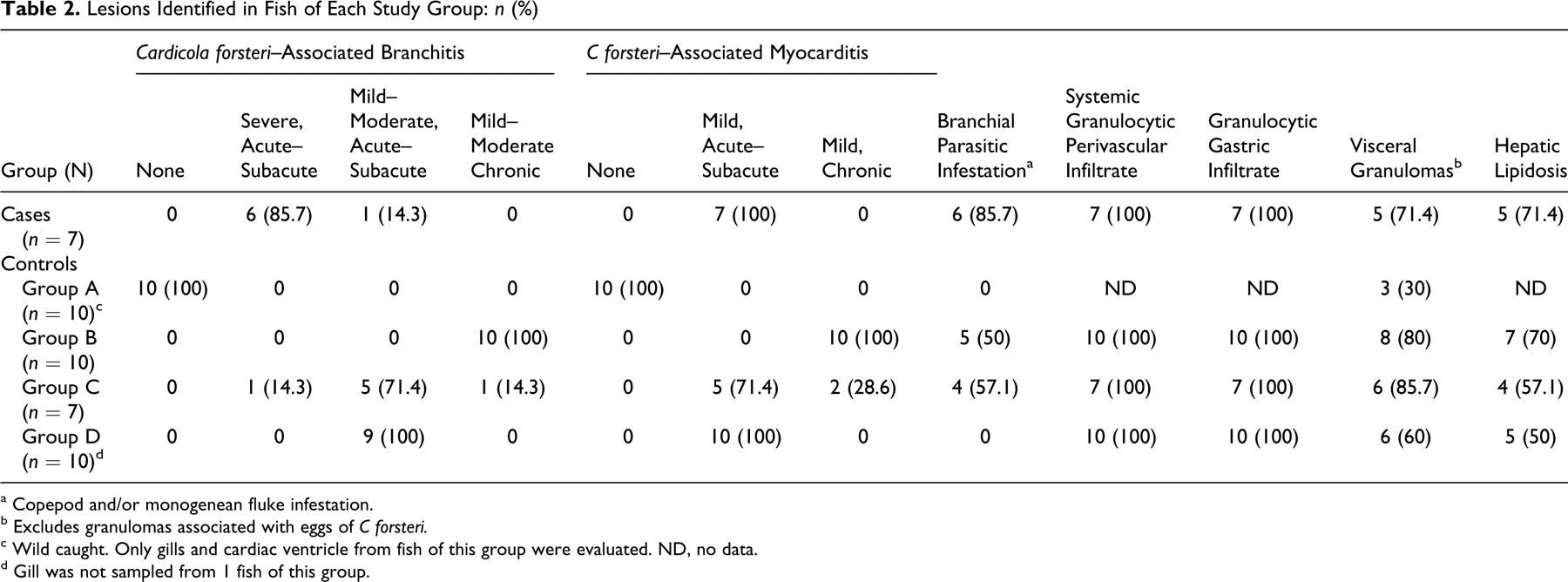
a Copepod and/or monogenean fluke infestation.
b Excludes granulomas associated with eggs of C forsteri.
c Wild caught. Only gills and cardiac ventricle from fish of this group were evaluated. ND, no data.
d Gill was not sampled from 1 fish of this group.
Severe C forsteri branchitis occurred more frequently in case fish (mortalities) than in fish that were sampled live during mortality spikes (62 to 65 days poststocking) (P < .0005). When comparing C forsteri gill lesions among fish that were sampled during mortality spikes (62 to 65 days poststocking), the odds of severe acute or subacute branchitis were 90 times greater for fish that died than for fish that were sampled live (95% confidence interval, 5 to 1,684).
The occurrence of acute–subacute C forsteri-associated myocarditis was similar among case fish (mortalities) and fish that were sampled live during mortality spikes (62 to 65 days poststocking) (P = 1.0).
Other Lesions
Study fish had several other findings that were apparently not related to C forsteri. Monogenean flukes and/or copepods infested the gills of several fish (Table 2). Occasionally, adjacent to attachment sites, there was focal proliferation of lamellar epithelial cells and infiltration of granulocytes. Although these parasites were low in number and associated with minimal tissue damage or response, gill infestation was associated with mortality (P < .05).
Systemic granulocytic perivascular infiltrate occurred in all cultured fish of the study. Cuffs of granulocytes admixed with fewer mononuclear leukocytes were around small arteries and veins (Fig. 12). The infiltrate often extended into surrounding tissues, frequently involving peripheral nerves. It was most severe around vessels and nerves of the intestine, stomach, and myocardium and was milder but consistently noted within pyloric cecae, liver, and serosal vessels of other viscera. Occasionally, affected arteries had villous endothelial hyperplasia, fibroplasia of the tunica adventitia, or smooth muscle hypertrophy and hyperplasia of the tunica media. The degree of infiltrate ranged from mild (n = 10) to moderate (n = 24) and was not associated with mortality (P = .64).
Granulocytic gastric infiltrate also occurred in all cultured study fish (Fig. 13); variably dense infiltrates of granulocytes were diffuse throughout the gastric submucosa and, to a lesser extent, tunica muscularis. The degree of infiltrate was mild (n = 12), moderate (n = 20), and severe (n = 2). Moderate to severe granulocytic gastric infiltrate was not associated with mortality (P = .38).
The majority of study fish had granulomas in a variety of tissues that were not associated with C forsteri eggs. Affected tissues included gastric submucosa (n = 18), pyloric cecae submucosa or serosa (n = 5), gastric serosa (n = 3), intestinal submucosa or serosa (n = 3), bulbus arteriosus adventitia (n = 1), and epicardium (n = 1). In addition, 3 fish of control group A (wild caught) had myocardial granulomas that were devoid of C forsteri eggs. Granulomas of 14 fish contained intralesional life stages of the digenean Didymocystis sp. No other etiologic agents were identified in the remaining granulomas with HE, Ziehl Neelsen acid fast, Fite-Faraco acid fast, and gram Twort stains. Mortality was not significantly associated with granulomas (P = .81) or visceral didymozoidiaisis (P = 1.0).
Hepatic lipidosis was observed in most cultured fish, ranged from mild (n = 16) to moderate (n = 11), and was not associated with mortality (P = 1.0).
Six of 7 case fish (mortalities) and 17 of 18 fish of control groups B–D that had spleen available for evaluation had distention of splenic sinusoids with red blood cells and eosinophilic homogenous material consistent with proteinaceous fluid. Three fish of group C had pyloric cecal coccidiosis.
Discussion
The present study demonstrates the potential for C forsteri to result in fatal lesions in cultured SBT. Adult C forsteri reside within the heart and, to a lesser extent, branchial arteries. 1,5 The eggs produced by C forsteri reach the gills hematogenously, become lodged in afferent filament arteries and lamellar capillaries, and result in considerable tissue damage where blood supply is disrupted or where eggs emerge and rupture from lamellae. In some fish, the ensuing branchitis may become severe enough to result in death, especially when eggs are produced in great numbers in a short period. The lesion can account for death through several mechanisms, including respiratory insufficiency, blood loss, and/or loss of osmoregulation and homeostasis. 8,11 In the present study, fish dying during mortality events were significantly more likely to have severe branchitis relative to control fish sampled from similar pontoons at similar time points. The only mortality examined that did not have severe branchitis had moderate branchitis, which theoretically had the potential to result in death if the fish had been amply stressed. Furthermore, apart from lesions associated with C forsteri infection, there were no other lesions identified in examined mortalities that were of sufficient severity or physiologic significance to have accounted for death. Although brain and kidney were not evaluated by the present study, a diagnostic investigation in 2008 failed to identify lesions in these tissues of SBT dying during mortality outbreaks (data not shown). Taken together, the severity of gill disease, the strong statistical association between severe branchitis and mortality, and the lack of ancillary lesions support the conclusion that C forsteri–associated branchitis is the key lesion and the pathologic basis for observed mortality.
Most of the mortality occurring on the studied culture site appears to be due to C forsteri infection. In concurrence with other studies, 2,3 cultured SBT appeared to become infected with C forsteri after being introduced into pontoons, given that no histologic evidence of infection was identified in wild-caught SBT. The life cycle of C forsteri is not known; however, a terebellid polychaete is a suspected intermediate host. At the studied culture site, peak mortality occurred between 6 and 9 weeks following introduction of fish into pontoons, a period similar to the prepatent period of other sanguinicolids 18 and a period when adult fluke intensity and prevalence are reportedly at their maximum. 2,3 In pontoon 4, the cumulative mortality curve was prolonged and bimodal; however, the second mortality peak coincided with 6-week poststocking mortality for fish that were introduced late into this pontoon. Cumulative mortality for the study pontoons reached only 2.1% (173 fish) during the 2009 season. This figure is much lower than cumulative mortalities affecting this farm in the previous few years; however, the culture site used by this farm was moved in 2009 to a remote area that had not been leased. Thus, it is likely that C forsteri infection pressure was much lower than previous years. The pathologic findings of SBT mortalities in the present study are similar to the findings of a diagnostic investigation conducted by FFVS and the University of Sydney in 2008, which examined 13 mortalities from 4 culture sites where cumulative mortality reached 10% (data not shown), suggesting that mortality due to C forsteri may be an industry-wide problem. Further structured epidemiologic research is needed to confirm the role of C forsteri in SBT mortality epidemics that occur annually throughout the South Australian industry.
Severe branchitis resulting from other sanguinicolid blood flukes has caused mortality outbreaks in other freshwater and marine fish. 9,11,15 Additionally, a new species of Cardicola was recently recognized in cultured Pacific bluefin tuna and is a serious concern to the Japanese industry. 16 Despite C forsteri infections being recognized in cultured South Australian SBT for more than a decade, 5 only slightly increased mortality has been ascribed to C forsteri infection, 13 and severe branchitis has not been described. Colquitt et al described lesions that were similar to chronic branchitis and chronic myocarditis of the present study and that were not considered to be sufficient to cause mortality. 4 The reported branchitis did not feature the large numbers of eggs and associated lamellar necrosis, lamellar epithelial hyperplasia, and branchial thrombosis seen in the present study. However, Colquitt et al evaluated fish that had not died and had been held in pontoons for longer than 4 months. Therefore, differences in sampling strategy relative to temporality of infection and health status of fish may account for failure to observe fatal severe branchitis.
It is unclear if fatal C forsteri infection is a new occurrence in cultured South Australian SBT. In the present study, severe branchitis was the result of large numbers of destructive eggs reaching lamellae. All SBT of the present study that died with severe branchitis had acute–subacute lesions, indicating that the early stage of infection, when eggs are produced in apparently great numbers, is the most life-threatening. Other factors that influence the level of egg production by adult flukes could include intensity of adult fluke infection, immunocompetency of the host, stress, and concurrent disease. The intensity of adult C forsteri infection declines 2 to 3 months after fish are introduced into pontoons, 1,3 a period that corresponds to the decline of the mortality curve. However, Aiken et al described epidemics of C forsteri infection with high intensities of adult fluke infection that were not accompanied by mortality outbreaks. 2,3 It could be that intensity of adult fluke infection does not necessarily correlate with egg production, and because gills were not histologically evaluated, it is possible that potentially fatal gill lesions did not occur in the fish of those studies. 2,3 Presumptively, egg production could be hampered by an immune response, but this has yet to be demonstrated in C forsteri–infected SBT. Although none of the SBT of the present study were known to be immunosuppressed, many had concomitant abnormalities that may have affected immune function. It is not known if concurrent disease is needed for severe C forsteri branchitis to develop or become fatal. The only other abnormality associated with mortality besides severe C forsteri branchitis was mild branchial parasitic infestation, which probably had little independent impact on the host but may suggest debilitation of the host. We hypothesize that years of culturing SBT in the same grow-out area has led to increased environmental C forsteri load and infection pressure, as well as more rapid and efficient infection of SBT when introduced to pontoons, leading to synchronous maturation of a greater number of adult flukes, the production of greater egg burden, and therefore increased incidence of severe branchitis and mortality among SBT cultured in this area. Primary debilitating conditions may exist in cultured SBT that facilitate fatal C forsteri infection, but this was not evident in the present study.
Branchial parasitic infestation, hepatic lipidosis, systemic granulocytic perivascular infiltrate, granulocytic gastric infiltrate, visceral granulomas, and visceral didymozoidiaisis also occurred in farmed SBT. Except for branchial parasitic infestation, none of these findings were associated with mortality. Monogenean fluke and copepod gill infestations are common in SBT and are considered innocuous at low levels because individual parasites elicit little tissue damage. 6,7,17 Hepatic lipidosis could reflect either mobilization of body fat stores during times of stress or anorexia or feeding of high-fat diets, a widespread industry practice aimed at increasing the lipid-content and flavor of tuna meat. Didymozoidiasis is known to be prevalent among farmed SBT and is generally considered to have little pathogenicity. 6,12,17 The significance of the perivascular and gastric granulocytic infiltrate is unknown. The degree of the infiltrate suggests that it is not physiologic, and similar infiltrates have been described in tunas infected with Didymozoidae. 10 Evaluation of a set of control tissues from noncultured fish would be helpful in determining the physiologic significance of these infiltrates and their relationship with parasite infections.
In conclusion, C forsteri infection appears to produce fatal lesions in farmed SBT and may account for a substantial proportion of mortality affecting SBT following introduction to pontoons. It is presently unknown to what extent mortality due to C forsteri is affecting the Australian SBT aquaculture industry; however, several companies representing more than half the industry report mortality patterns analogous with those described here. Further research is needed to identify factors that favor the development of fatal disease and to elucidate mitigations for C forsteri infection.
Footnotes
Acknowledgements
We thank Mark Thomas and Simon Firestone for assistance with illustrations and Jan Slapeta for help with parasite descriptions. The authors also thank the staff of Veterinary Pathology Diagnostic Services, University of Sydney, for technical assistance.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
This work was part of a diagnostic investigation funded by Blaslov Fishing Group Pty Ltd.
