Abstract
Gills from Atlantic salmon with experimentally induced amoebic gill disease (Neoparamoeba spp.) were examined with transmission electron microscopy to assess pathology and host-cell responses. Amoebae were found either on the surface epithelium or with pseudopodia extending deeply into invaginations of epithelial cells. The amoebae had various densities along the plasma membrane and contained electron-dense deposits within their cytoplasm. Surface epithelial cells sloughed from the gills and had features consistent with apoptosis, including rounded shape, loss of surface microridges, and hypercondensation of nuclear chromatin. Affected areas of gills had fusion of secondary lamellae with interlamellar spaces occupied by mitotic epithelial cells and eosinophils. Eosinophils contained abundant fusiform-shaped granules that measured approximately 1 μm long and 360 nm wide. The granule consisted of an electron-dense matrix with a central inclusion that was less electron-dense, consisting of particulate and fibrillar material. In many instances, the central inclusion appeared empty and 90% of the eosinophils had morphology suggestive of piecemeal degranulation. Also observed within affected areas were a few neutrophils, mucous cells releasing mucus, and a small number of dendritic-like cells.
Amoebic gill disease (AGD) is a common problem for the culture of Atlantic salmon, Salmo salar, within seawater net pens in Tasmania, Australia. Disease is associated with the colonization of the amphizoic amoeba Neoparamoeba spp. (comprising at least 2 species: Neoparamoeba pemaquidensis and Neoparamoeba branchiphila) on the surface of the gills. 10, 16 A distinguishing feature of amoebae in the Neoparamoeba genus is the presence of an endosymbiotic Perkinsiella amoebae–like organism, also referred to as the parasome. 10 AGD has been described as affecting Atlantic salmon and rainbow trout, Oncorhynchus mykiss, from around the world, including Australia, North America, France, Spain, Ireland, Chile, and New Zealand. 24 The disease has also been described in nonsalmonids including turbot, Scophthalmus maximus; sea bream, Sparus aurata; and sea bass, Dicentrarchus labrax. 9 AGD is of economic significance to Australia, whereby the cost of the disease (which, although potentially fatal to fish, is treated with freshwater baths) incurs an estimated cost to the salmonid aquaculture industry in the range between 10 and 20% of the annual gross production value estimated to be equivalent to approximately $17–19 million Australian dollars annually. 2, 23 Clinical signs attributed to AGD include raised white mucoid patches on gill filaments, lethargy, and flared opercula, and pathophysiologic changes include respiratory and acid-base disturbances and systemic hypertension. 18, 19, 22, 27, 28 There is a significant metabolic cost associated with the disease, which translates to reductions in growth if the disease is not controlled by freshwater bathing (the only effective treatment for AGD). Mortalities attributed to AGD in untreated net pens have been reported to reach up to 2% per day and 50% total. 22
Infection results in a host-cell response leading to the fusion of secondary lamellae, focal hyperplasia of the filament epithelium, and a leucocytic infiltration of the lesion. 1, 23 Clinical outbreaks of AGD appear to be associated with water temperatures between 12–20°C, although other risk factors have been identified including algae, site characteristics, salinity, and water chemistry. 4, 7, 8 Amoebic gill infections have also been suggested to be a secondary complication of bacterial infections, as seen in salmonids with nodular gill disease (NGD); In Ireland, AGD can occur concurrently with ciliate infections. 4, 35
There have been several reports on the host-cell responses associated with AGD. 1, 33 The most prominent change in infected gills was degeneration of the surface epithelium with hyperplasia of the underlying epithelial cells expressing proliferating cell nuclear antigen histologically, as well as down-regulation of the tumor-suppressor protein p 53 in affected gills. 1, 21 It has been reported that during disease outbreaks, gills are infiltrated with neutrophils, while gills recovered from disease are infiltrated with macrophages and lymphocytes. 33 A light microscopic study of the pathogenesis of AGD showed that early attachment of amoebae caused hypertrophy and desquamation of the epithelium, followed by hyperplasia leading to the fusion of the secondary lamellae. These areas were infiltrated with predominantly neutrophils, which emigrated from the central venous sinus. Large numbers of eosinophilic granule cells (EGC) were reported surrounding the primary filament cartilage of infected fish, although this is likely to reflect the inherent population of these cell types in the gill. 1
Understanding host cellular responses is important in understanding the pathogenesis of disease. Examination of cell types in fish gills responding to pathogens provides information about defense mechanisms in the gills. The associated inflammatory response with AGD may be more detrimental to the host rather than curative, and treating the inflammation may be a viable approach to treating fish during disease outbreaks. With an understanding of the nature of the inflammatory response associated with AGD, one can propose therapy strategies aimed at host responses. This study describes the host-gill cellular responses and ultrastructural pathology of AGD-affected Atlantic salmon.
Materials and Methods
Source of AGD-affected gill tissue
At the University of Tasmania, seawater-acclimated Atlantic salmon (approximately 100 g each) were introduced to an active infection tank containing about 20 AGD-affected conspecifics for at least 7 days to allow for infection by cohabitation. The infection tank consisted of an 1,800-L fiberglass Rathburn tank, biologic filter, and recirculating pump (total system volume of ∼3,000 L). Water was maintained at a salinity of 35 parts per thousand, temperature of 15–17°C, dissolved oxygen in excess of 90% saturation, and total ammonia nitrogen less than 0.25 mg L−1. Moribund salmon with clinical signs of AGD were removed from the tank and euthanized with an overdose of clove oil (0.03 mL L−1). Rapidly, individual gill arches were excised, placed into 3 consecutive washes of 20 ml of chilled 2.5% glutaraldehyde in sodium cacodylate buffer (0.1 M), and finally placed into 25-ml chilled fresh fixative and refrigerated overnight (4°C). Under a dissecting microscope, macroscopic white raised patches, characteristic of AGD lesions, were excised from the gill arches in 5–10 lamellae sections and placed in chilled fixative. The fixative was replaced daily, and the samples were shipped 48 hours after initial collection to the Atlantic Veterinary College, Prince Edward Island, Canada.
Transmission electron microscopy
Upon arrival, samples were washed in 2 changes of cacodylate buffer for 10 minutes each and postfixed in 1% osmium tetroxide in cacodylate buffer for 1 hour at room temperature. Tissue was then dehydrated through a series of ascending concentrations of ethanol including two 10 minute changes of 50%, 75%, and 95% and, lastly, 2 changes of 100% ethanol for 15 minutes each. Tissue was then cleared in propylene oxide (PO) (2×, 10 min each) and infiltrated with Epon (Canemco-Marivec, Quebec, Canada). Infiltration included 1 change in a 1 : 1 ratio of Epon to PO, then to 1 change in a 3 : 1 ratio of Epon to PO for 1 hour each, and, lastly, to 100% Epon overnight in a desiccator under vacuum. For each fish, 5 pieces of gill tissue were embedded in Epon. Semithin sections (0.5 μm) were cut from each piece of gill tissue and stained with toluidine blue to examine with light microscopy. Areas where lesions were observed were trimmed, and ultrathin sections were cut (90 nm) for transmission electron microscopy. Ultrathin sections were stained with uranyl acetate and lead citrate and examined and photographed with a Hitachi-7500 transmission electron microscope (Hitachi High-Technologies Canada Inc.) operated at 80 kV. Measurements of cell organelles were made directly from electron micrographs and reported as the mean ± SE.
Results
Gills infected with Neoparamoeba spp. displayed characteristic pathology, which included hyperplasia of the epithelium leading to the fusion of secondary lamellae, around which amoebae containing clearly identifiable parasomes (a diagnostic feature of the genus Paramoeba) could be seen on the surface of the epithelium. Bacteria were present in areas that were heavily infected with the amoebae.
The interface between the amoebae and the host tissue was examined, and various forms of interaction were observed. In many instances, amoebae were observed without direct contact with the epithelial membrane, but the shape of the amoebae appeared to conform to the shape of the surface epithelium, suggesting the amoebae had been lying on the surface but lifted off during fixation or tissue processing. Some amoebae, with intimate contact with the host epithelium, extended their pseudopodia into invaginations of the epithelial layer (Fig. 1). The plasma membrane of amoebae had a heterogeneous density where the membrane appeared increasingly electron-dense in some areas. In one instance, an area of increased membrane density was accompanied by increased cytoplasmic density (Fig. 2). The surface epithelial cells adjacent to areas of the host-parasite interface were sloughed away from the underlying tissue, much of their microridge surface patterning was lost, and the cells assumed a rounded shape (Fig. 3). The nuclei of these cells appeared rounded and had hypercondensation of chromatin (Fig. 3). Many of the epithelial cells below the surface layer were in various stages of mitosis.
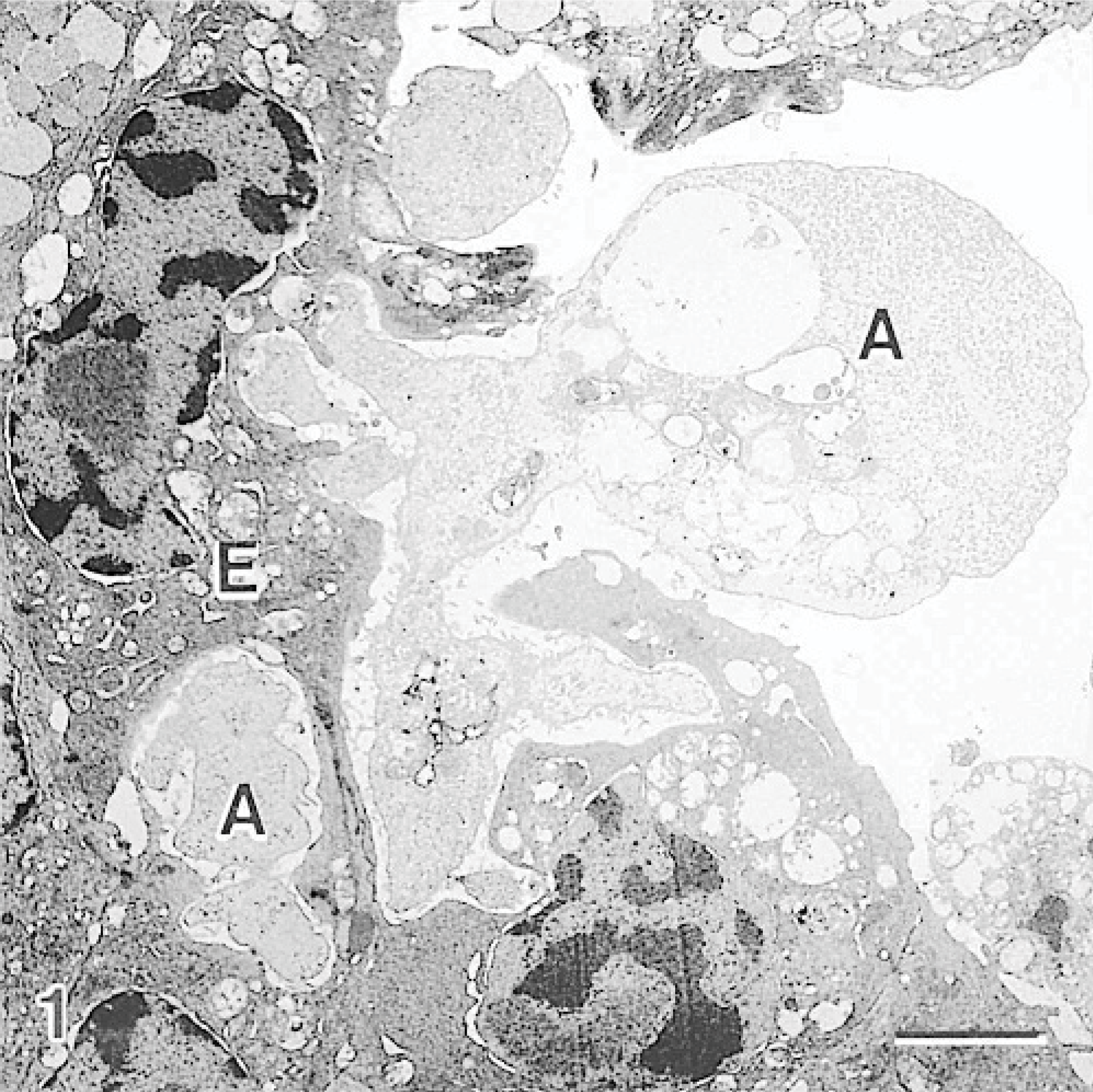
Gill; Atlantic salmon. Amoeba (A) has pseudopodia within the surface epithelial layer (E), where the epithelial cells affected by the amoeba have a distorted shape and loss of surface microridges. Uranyl acetate and lead citrate. Bar = 3 μm.
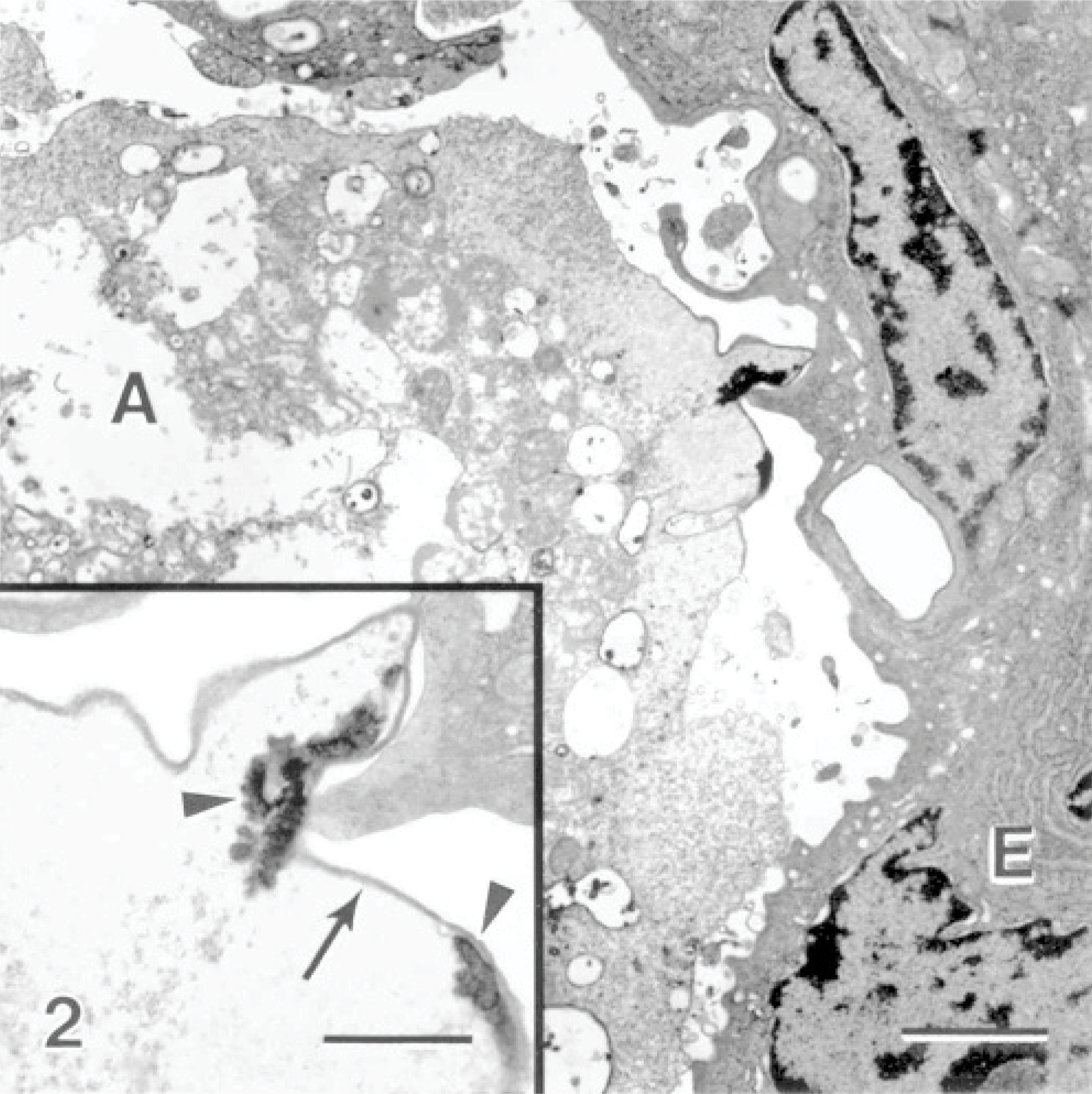
Gill; Atlantic salmon. Amoeba (A) are located near the surface epithelium (E) of the gills. Bar = 2 μm. Inset: An area of the amoeba near the epithelium where the amoeba plasma membrane is increasingly electron-dense (arrow) and electron-dense deposits are seen within the amoeba (arrowheads). Uranyl acetate and lead citrate. Bar = 700 nm.
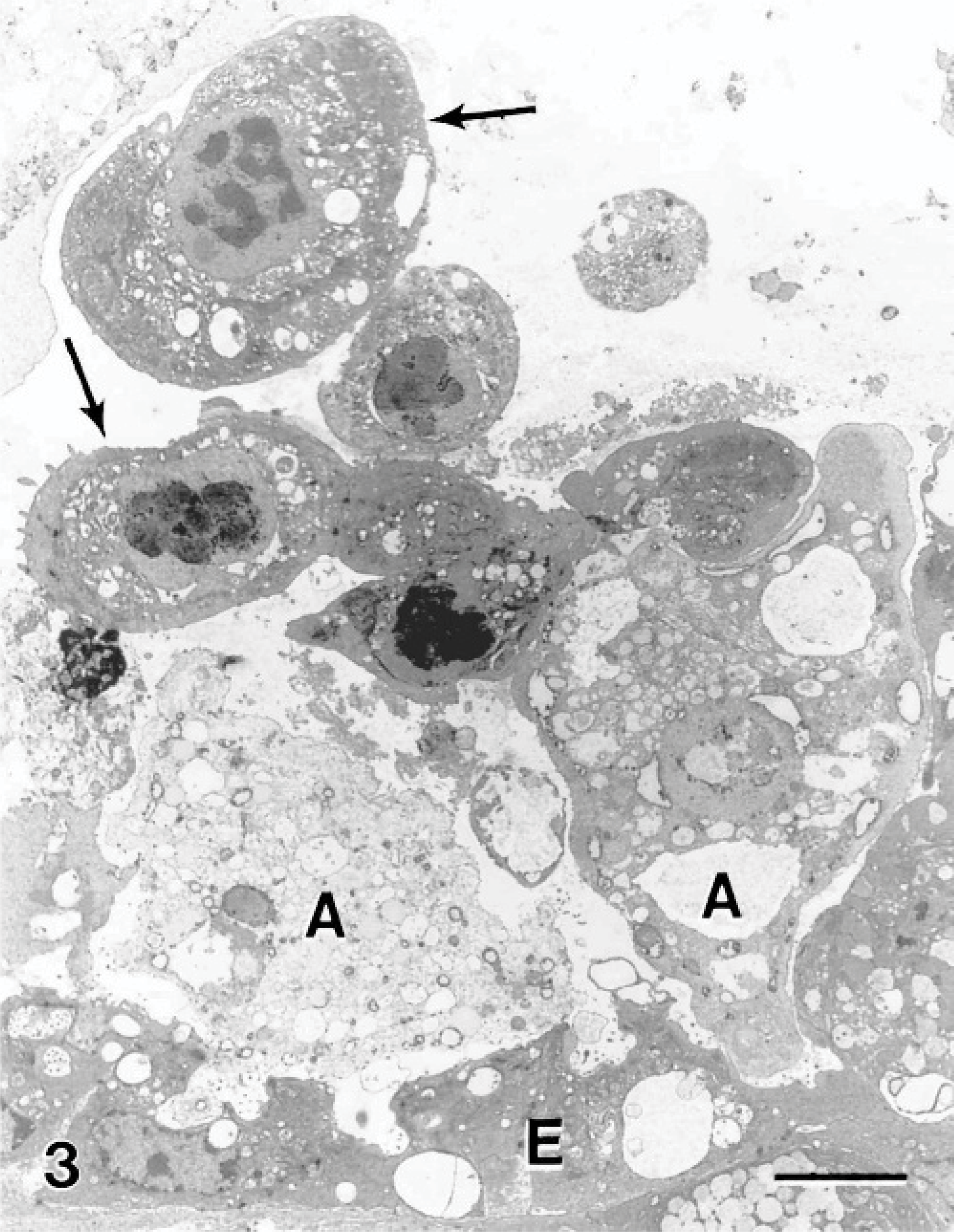
Gill; Atlantic salmon. Two amoebae (A) near the epithelium (E) have several epithelial cells (arrows) that have sloughed from the tissue. The sloughed cells have a rounded shape, loss of most surface microridges, and hypercondensation of the chromatin within the nucleus. Uranyl acetate and lead citrate. Bar = 5 μm.
Within the hyperplastic epithelium of the secondary lamellae was a heavy infiltrate of inflammatory cells. The infiltrate predominantly consisted of cells resembling eosinophils (Fig. 4). These cells measured between 6 and 15 μm along their long axis, depending on their plane of section. The nuclei appeared rounded in most cells, and occasionally an indentation was seen on 1 side (Fig. 4). The cells contained an abundance of unique granules; up to 50 could be seen within a cell in a single section plane. When cut in longitudinal sections, the granules appeared fusiform in shape (Fig. 5). Frequently running along the long axis of the granules was a density composed of particulate and fibrillar material, which was less electron-dense than the rest of the granules (Fig. 5). Similar granules that contained electron-lucent inclusions were observed (Fig. 6). When cut in cross-section, the granules were spheric, and the electron-lucent inclusion was seen in the center or off-center (Fig. 6). The granules measured 1.00 ± 0.09 μm long (range of 0.64–1.6 μm) and 0.36 ± 0.03 μm wide (range of 0.22–0.51 μm), based on measurements of 10 granules appearing in longitudinal section. Measurements of the diameter of 12 granules cut in cross-section had a mean of 0.42 ± 0.02 μm (range of 0.32–0.56 μm). Granules of 30 cells were examined, and 90% of the cells contained granules with completely electron-lucent inclusions. From a total of 666 granules, 32% appeared with an electron-lucent inclusion and the remaining 68% contained the particulate and fibrillar material. All lesions that were associated with the amoebae were significantly infiltrated with this cell type; the cells were observed within areas of fusion and at the surface epithelium, where the amoebae were also present. Neutrophils were present, and goblet cells were commonly observed to be releasing their contents in areas of the lesions (Fig. 7).
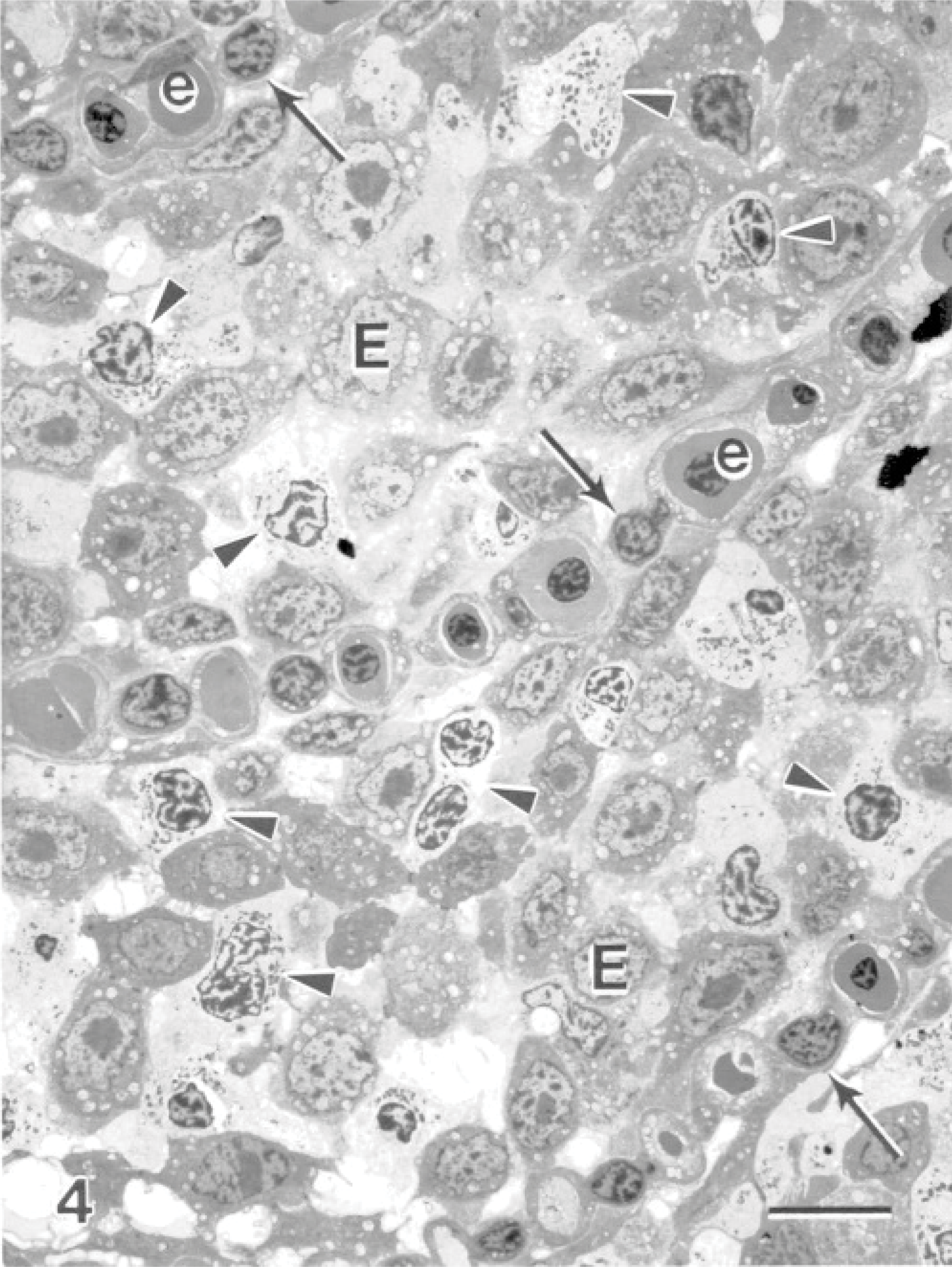
Gill; Atlantic salmon. Fusion of gill lamellae are seen adjacent to areas infected with amoebae. Three secondary lamellae are identified by pillar cells (arrows) with intermittent blood channels often containing erythrocytes (e). The interlamellar areas contain epithelial cells (E) and cells that resemble eosinophils (arrowheads). Uranyl acetate and lead citrate. Bar = 10 μm.
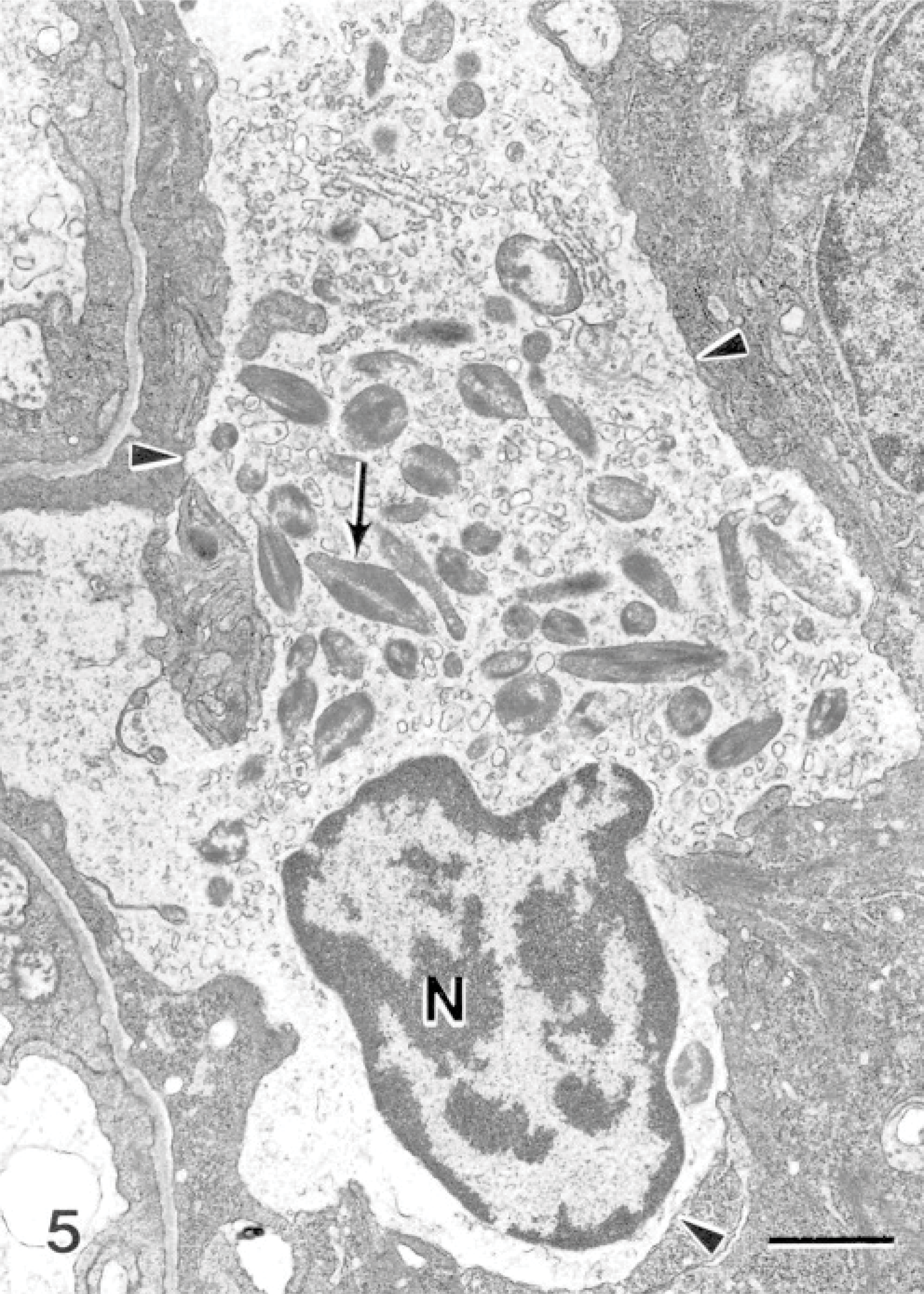
Gill; Atlantic salmon. Eosinophil-containing electron-dense fusiform granules (arrow) have fibrillar inclusions that are less electron-dense. Arrowheads indicate plasma membrane. N = nucleus. Uranyl acetate and lead citrate. Bar = 1 μm.
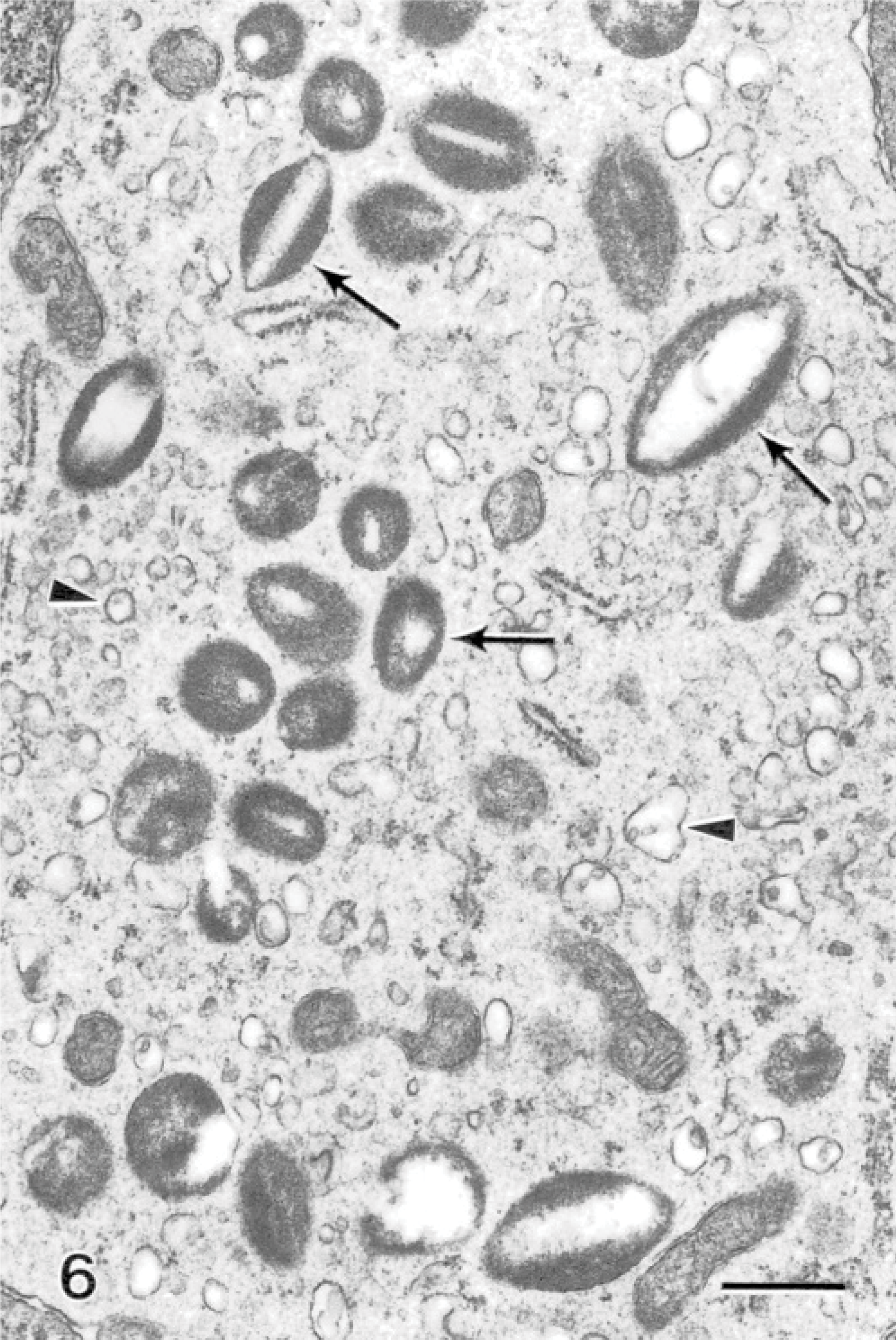
Gill; Atlantic salmon. The cytoplasm of eosinophil-containing granules has electron-lucent crystalloid inclusions (arrows) and an abundance of cytoplasmic vesicles (arrowheads). Uranyl acetate and lead citrate. Bar = 500 nm.
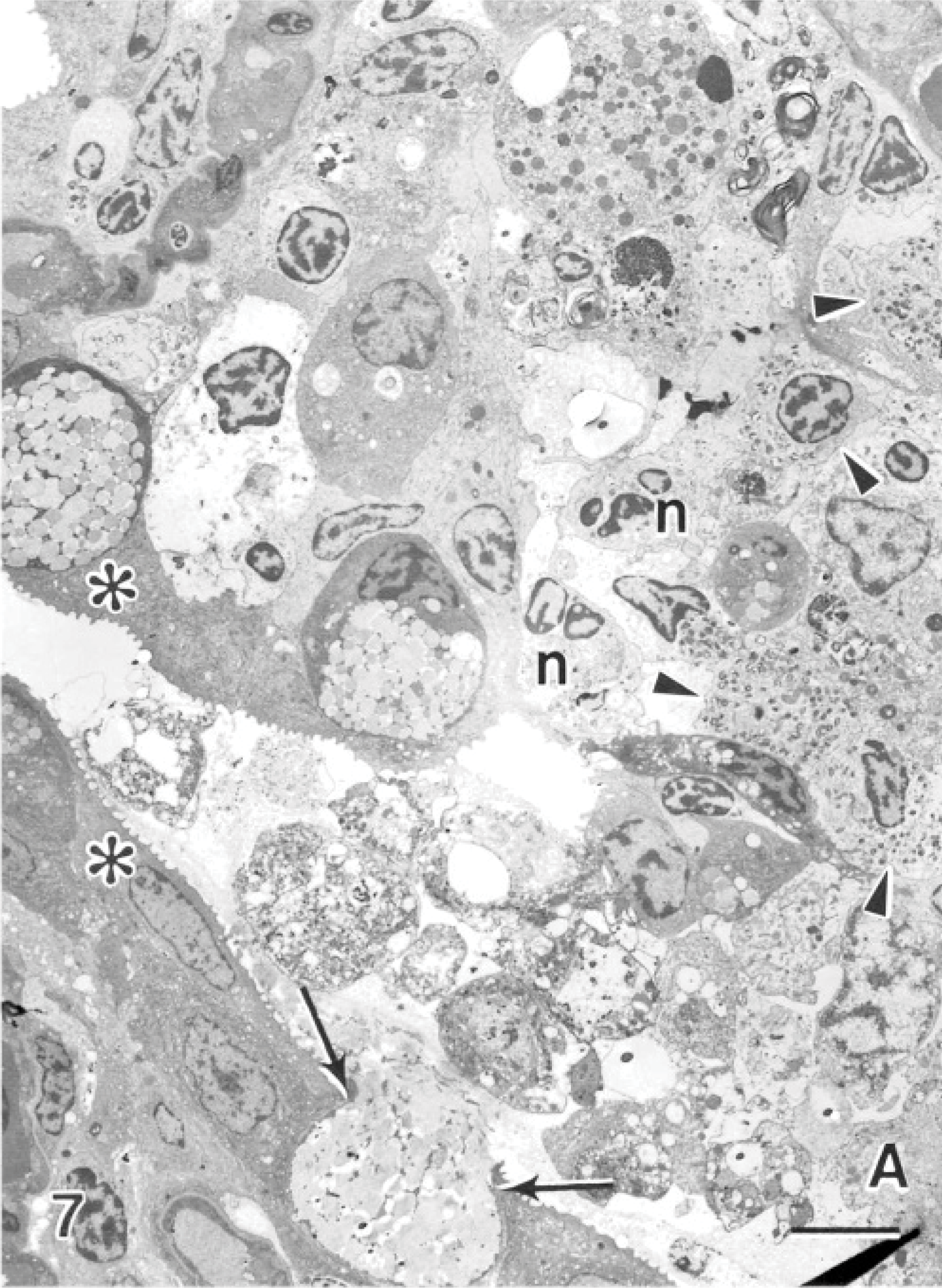
Gill; Atlantic salmon. Two apposing primary gill lamellae have amoeba (A) and sloughed cells in the interlamellar space. A goblet cell is releasing its mucus (arrows) on 1 primary gill lamellae, and the apposed lamella has lost much of its surface epithelium and is infiltrated with neutrophils (n) and eosinophils (arrowheads). Normal epithelial cells are seen with surface microridges (asterisk). Uranyl acetate and lead citrate. Bar = 8 μm.
Examination of all the material revealed 2 cells with large rounded nuclei and large rod-shaped granules within their cytoplasm (Fig. 8). The granules contained particulate material forming a density adjacent to the granule membrane forming an electron-dense lining along the inner surface of the granule membrane (Fig. 8, inset). Spheric granules were observed with a similar density on the luminal side adjacent to the granule membrane, forming a concentric ring (Fig. 8, inset). The lumen of the granules often contained a square-lattice structured material (Fig. 9). This cell type is identical to previously described dendritic-like cells. These cells were observed in 2 instances and not consistently encountered in the lesions.
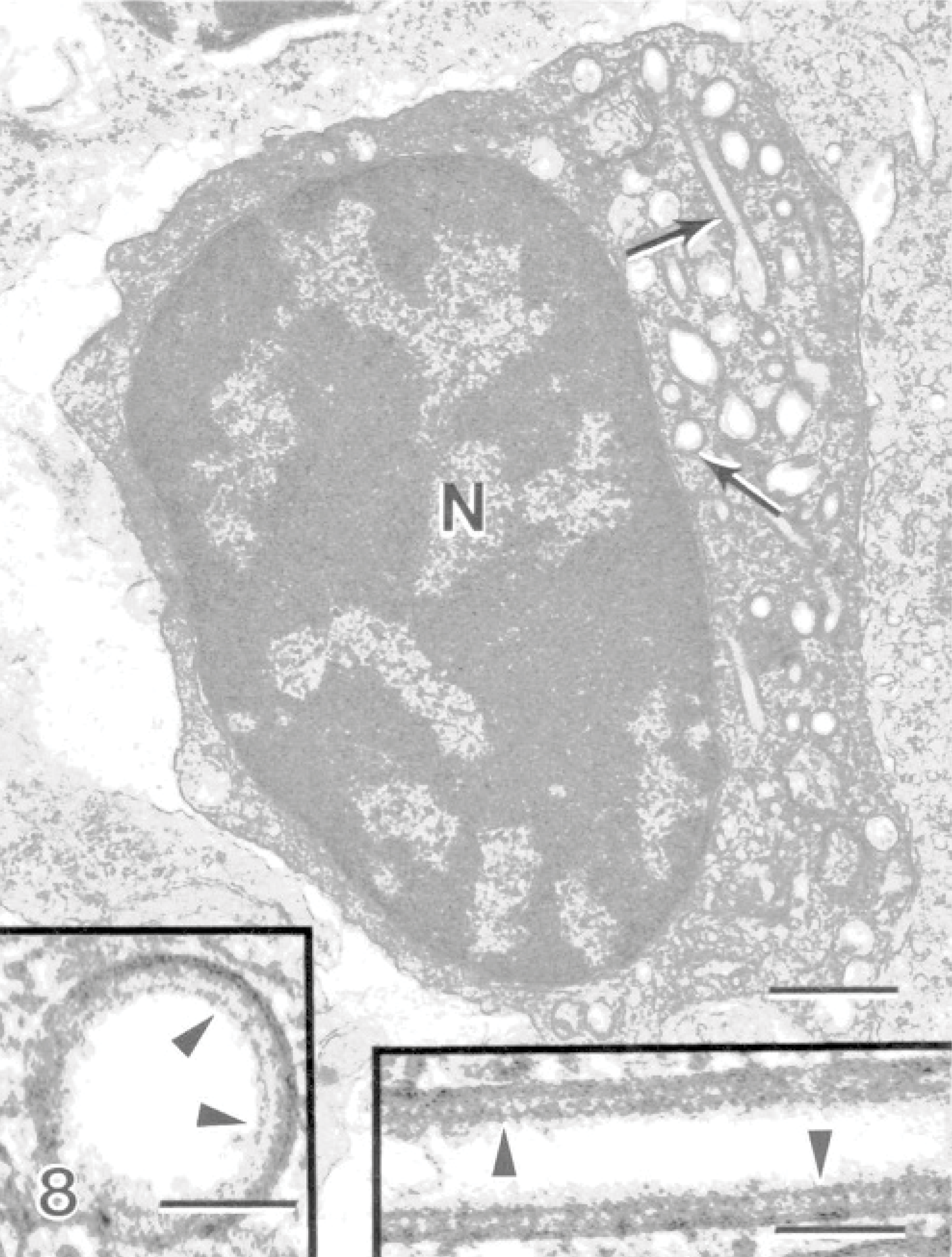
Gill; Atlantic salmon. A dendritic-like cell has a large rounded nucleus (N) containing cytoplasmic rod-shaped granules (arrows). Bar = 700 nm. Insets: The granules in cross-section and in longitudinal section, where particulate material within the granule forms a density on the luminal side adjacent to the granule membrane. Uranyl acetate and lead citrate. Bar = 80 nm.
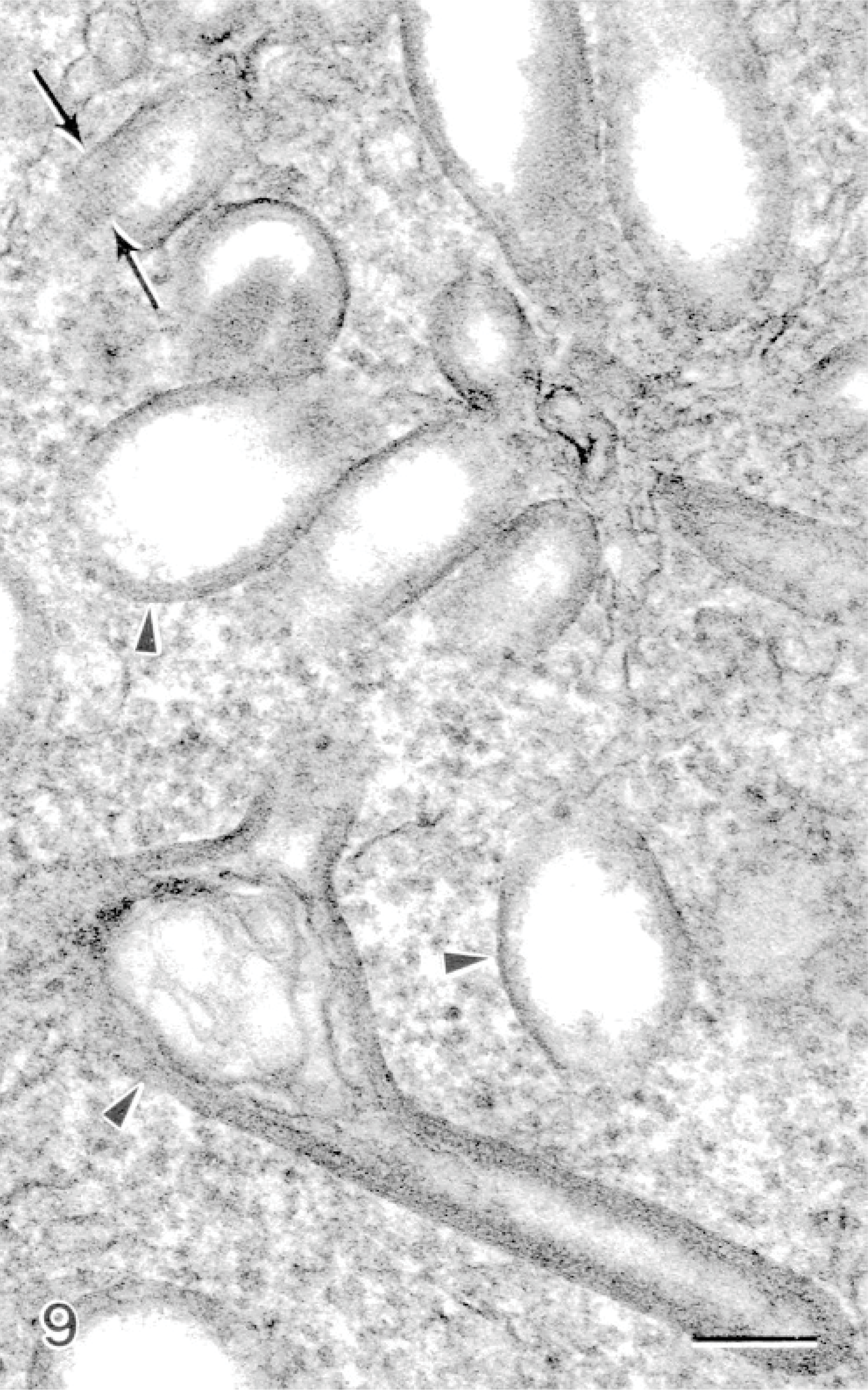
Gill; Atlantic salmon. Rod-shaped granules within dendritic-like cells that have a vacuolated portion (arrowheads), which contains particulate material with a square-lattice structure (arrows). Uranyl acetate and lead citrate. Bar = 100 nm.
Discussion
An important finding in this study was the observation of cells that resembled eosinophils in all the lesions examined resulting from this acute experimental infection. These cells represented the majority of the infiltrated cells and were characteristic in the AGD lesions. The eosinophils were distinct from previously described EGCs, which are commonly observed in the gills, skin, and gut of salmonids. 13 EGCs, which are thought to be similar to mammalian mast cells, contain large spheric membrane-bound granules containing a dense homogenous matrix. 12, 31, 32 The morphology of the eosinophil granules in this study very closely resembled that of mammalian eosinophil granules, which are elliptic and contain a crystalline inclusion within the center of the granule, confirming that these cells are indeed not EGCs but actual eosinophils of perhaps a different lineage to EGC/mast cells. 25, 34 The crystalline cores of mammalian eosinophil granules can be electron-dense or lucent. Eosinophils have not been previously described for salmonids, although they have been described for other fish species. Previously identified granules of eosinophils in carp, Cyprinus carpio, have a central zone called the “internum,” which is surrounded by a dark wrapper, often elongated at 1 end. 17 A different description given for carp eosinophil granules was oval to elongate in shape, containing 1 or more, dense bar-shaped crystalloids. 36 The salmonid eosinophil granules (SEG) described in this study showed similarities and differences when compared with the carp eosinophil granules (CEG). The inclusions within the SEGs contained material that was less electron-dense than the rest of the granule. The density of the inclusion was similar to the internum described for the CEG, but the shape was different; in carp, internums are localized on 1 side of the granule and are more robust than the cores seen in the SEG. The core of the SEG was located centrally within the granule and sometimes had a crystalloid appearance. None of the cores of the SEGs were electron-dense, as described for some mammalian and carp eosinophils.
Many of the granules seen in this study contained electron-lucent cores, while the remaining parts of the granule remained unchanged. This feature has also been observed in carp eosinophils. 17 The empty cores could be a result of degranulation of the core portion of the granule. A type of degranulation, called piecemeal degranulation (PMD), has been reported in eosinophils, which involves selective release of granule proteins. 11, 15 The process of PMD is characterized by an increase in vesicles within the cytoplasm of the cell and loss of material from granules without granule-granule or granule-plasma membrane fusion. 6 The eosinophilic granules in this study fit the morphologic criteria for PMD, and the granules with fibrillar cores are likely intact granules, while the granules with empty cores have released their contents by PMD. Cells that have granules with empty cores also have abundant vesicles within their cytoplasm (Fig. 6). In eosinophilic diseases, degranulation of eosinophils is considered to be a major pathogenic factor. It has been shown that eosinophils from the blood of human patients with active allergic disease show signs of PMD, while the eosinophils from asymptomatic allergy patients appear similar to those of healthy individuals. 15 Indeed in fish, although true eosinophil PMD has not been reported, EGC-tissue eosinophilic cells appear to undergo a similar degranulation in the gut in response to systemic neurotransmitter exposure and bacterial exotoxins. 26, 29, 30
Severe tissue eosinophilia is not always associated with PMD; it has been shown that the colonic mucosa of individuals with inflammatory bowel diseases and the airway mucosa of individuals with airway diseases have similar tissue eosinophilia, but the eosinophils in the airway diseases show extensive PMD, while those in inflammatory bowel diseases do not. 11 This finding shows that when determining the pathogenesis of diseases with eosinophilia, both the degree of eosinophilia and the extent of PMD must be assessed. In this study of AGD in salmon, 90% of eosinophils showed signs of PMD and 34% of all granules had the central core degranulated, leading to the conclusion that tissue eosinophilia was extensively accompanied by PMD.
Gill eosinophilia and degranulation of the central core contents of eosinophils in the gills of Atlantic salmon with AGD likely had a large role in the pathogenesis of the disease. Eosinophils are commonly associated with allergic diseases and parasitic infections in mammals, though they have also been thought to act as antigen-presenting cells and to aid in tissue repair. 2 The proteins within the central core of eosinophils in fish have not been examined to date, but it is known that the central core of mammalian eosinophils is composed mainly of major basic protein. 25 Major basic protein is known to efficiently destroy parasites and also has been shown to damage the resident population of cells exposed to the protein around the tissue eosinophilia. 14 There are known differences in the central core of eosinophils in fish and mammals though. The central core internum of carp eosinophils contains activity of both acid phosphatase and peroxidase, while neither of these has been demonstrated in the central core of mammalian eosinophils. 17
A small number of dendritic-like cells with rod-shaped granules containing square-latticed structured material, which were occasionally vacuolated on 1 end, were encountered in this study (Figs. 8,9). These cells very closely resembled previously described dendritic-like cells from the gills of chinook salmon, Oncorhynchus tshawytscha, suffering from microsporidial gill disease caused by the inflammation-inducing parasite, Loma salmonae. 20 These cells were not typical in the lesions caused by amoebae but were encountered in 2 lesions that were examined. The role of this cell type may not be major in the pathogenesis of AGD, but this is the first finding of this cell type in Atlantic salmon gills. Morphologically, this cell type contains granules very similar to Birbeck granules found in mammalian Langerhans cells. Birbeck granules from Langerhans cells and the granules in the salmonid dendritic-like cells are both rod-shaped, containing square-lattice structured material and commonly vacuolated at 1 end. 20 The granules in the salmonid dendritic-like cells also bear close resemblance to those found in mammalian monocyte-derived dendritic cells, which contain granules with a ring-like shape and comprise 2 concentric membranes. 20 The finding of this cell type in a different species with a different gill disease further suggests that it is an important cell type responding to pathogens in the gills. These cells were located in connective tissue of fused secondary lamellae, which also contained eosinophils, few neutrophils, mitotic epithelial cells, and mucous cells.
There has been interest in examining the interaction of amoebae and host tissue in order to elucidate some mechanisms of attachment and pathogenesis leading to disease. The interactions between amoebae and the host epithelium, which result in disease, remain a mystery. Previous work has shown that gill-attached amoebae were distinct from in vitro cultured amoebae in that the gill-attached amoebae had more pseudopodia extensions and contained electron-dense deposits within vesicles in the cytoplasm. 16, 33 The composition and origin of the electron-dense deposits are unclear, but similar deposits were observed in this study. Also in this study, portions of the amoebae membrane appeared more electron-dense and contained cytoplasmic electron-dense deposits. Although many amoebae were observed without direct contact to the host epithelium in this study, it was common to observe amoebae with pseudopodia inserted within invaginations of epithelial cells. This may be a mechanism to better anchor itself into the tissue or a way to increase the surface area of the amoebae contacting host tissue, perhaps to increase efficiency of factors the amoebae may release to affect the host tissue.
In this investigation, it is clear that the host surface epithelial cells are severely affected by the presence of the amoebae. Normal surface epithelial cells contain elaborate fingerprint-like microridges along their surface, which aid in increasing the respiratory surface area and aid in the attachment of mucus. 13 An example of normal surface microridges on gill epithelial cells can be seen inFig. 7. The affected epithelial cells in the present study have lost much of their surface microridges, and additionally, the cells have sloughed from the surface and have a rounded shape and hypercondensed nuclear chromatin. These morphologic features are consistent with those of apoptosis. Cells undergoing apoptosis lose surface differentiations and round up, and chromatin becomes hypercondensed due to DNA fragmentation. 25 It has been shown that culture of N. pemaquidensis with an Atlantic salmon gill epithelium cell line resulted in complete cytolysis of the cell monolayer within 5 days. 5 The cytopathic effect that amoebae have on the epithelial cells or the presence of inflammatory cells in vivo may initiate apoptosis of epithelial cells.
Footnotes
Acknowledgements
This research formed part of a project of the Aquafin Cooperative Research Centre (CRC), the Australian Governments CRC Programme, and received funds from Fisheries Research & Development Corporation and other CRC participants (MDP). Additional funding was received by Contributing to Australian Scholarship and Science foundation (JAB).
