Abstract
This case report describes a placental tumor from a stillborn East African bongo fetus. On gross examination, the tumor was reminiscent of a chorangioma; however, based on histomorphology, histochemical and immunophenotypic characterization, comparison to 21 human cases of chorangioma, and embryogenesis, this tumor is hypothesized to be a placental stem cell tumor. Histologically, the tumor had 2, often comingled morphologies with one area having dense cellularity and scant vascularity and a second area having scant cellularity and prominent vascularity. Pleomorphic mesenchymal cells were arranged in intersecting fascicles, often whorled around, or were contiguous with vascular channels and had features of skeletal muscle, smooth muscle, pericyte, endothelial, fibrous, and undifferentiated mesenchymal cells. Phosphotungstic acid-hematoxylin outlined cross-striations in some cells. cells variably were immunoreactive for vimentin, CD31, von Willebrand factor VIII, desmin, muscle specific actin, alpha-smooth muscle actin, and skeletal myosin and negative for cytokeratin antigens. The relationship of this tumor to the presence of fetal musculoskeletal and omental anomalies is discussed.
This case report describes a placental tumor from a full-term stillborn East African bongo fetus (Eurycerus isaaci). On gross examination, the tumor was reminiscent of a chorangioma as described in humans and bovids; however, based on histomorphology, histochemistry, immunohistochemistry (IHC), comparison to 21 human cases of chorangioma, and embryogenesis, this tumor is hypothesized to be a placental stem cell tumor. 1– 7, 11 The relationship of this tumor to the presence of fetal musculoskeletal and omental anomalies is discussed.
A 3-year-old primiparous dam had dystocia and delivered, with assistance, a stillborn male fetus on 24 February 2005. The dystocia was caused by a left shoulder lock. The dam recovered uneventfully, and the placenta and fetus were submitted for necropsy. On gross examination, the placenta weighed 3,770 g (normal range 1,300–1,600 g) and had the normal number and arrangement of cotyledons for this species, which is between 125 and 150 cotyledons arranged in 4 rows (K. Benirschke, personal communication). Cotyledons ranged in sized from 3.2 × 3.0 × 0.2 cm to 15.0 × 6.0 × 0.2 cm. Approximately 50% of the cotyledons had diffusely tan maternal surfaces. The intercotyledonary chorioallantoic membrane was expanded and covered 4, partially demarcated, variably sized (up to 975 g), irregular, smooth, fleshy, lobulated, red to purple masses (Fig. 1). The chorionic membrane was thickened, firm, and translucent to opaque white. The intercotyledonary areas also were translucent to opaque with thick, opaque, yellow to white foci. The fetus had a wet body weight of 21.5 kg, 79-cm crown to rump length, was covered in dirt, had intact fetal hooves, appeared flaccid and large, and had nonaerated lungs. The scapular and humeral regions of the thoracic limbs had markedly asymmetrical muscling, with the left limb larger than the right. Skeletal asymmetry was excluded by radiographic examination. The sternum had pectus excavatum. The manubrium at the left thoracic inlet had a 5.0 × 4.0 × 3.0 cm, gray to tan mass of cartilaginous consistency that was contiguous with the costo-sternal junction. The entire length of the trachea was flattened laterally. A blind-ended, 49.0 × 29.0 × 1.0 cm, smooth, tan, membranous mass filled one third of the abdominal cavity and was contiguous with the serosa of the forestomach. This mass was interpreted as malformed omentum.
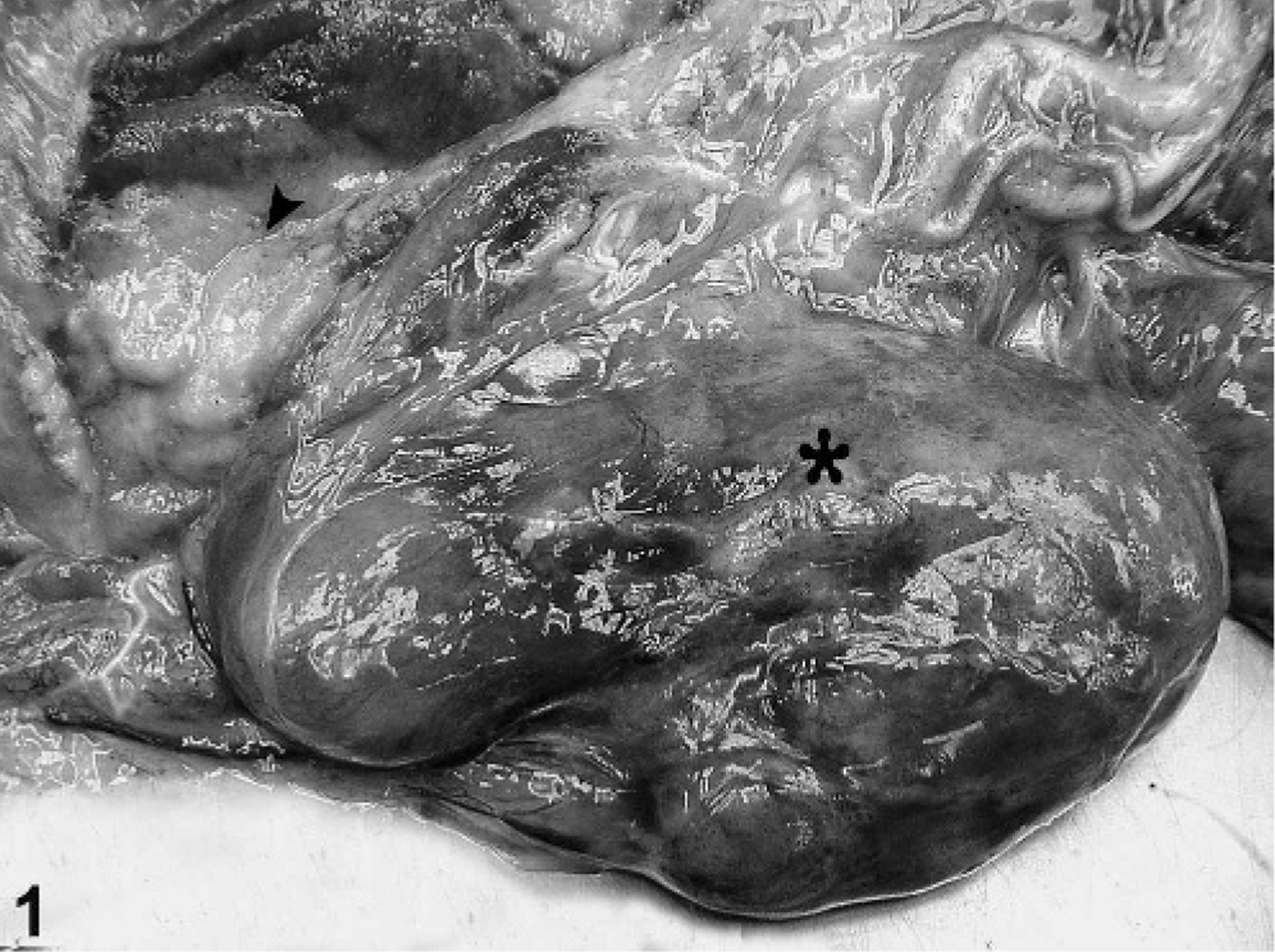
Placental tumor; bongo. The intercotyledonary chorioallantoic membrane is expanded by a partially demarcated, smooth, fleshy, lobulated tumor (asterisk), which grossly resembles chorangioma. The intercotyledonary areas also have multifocal, thick, opaque foci (arrowhead).
Sections of placenta and fetus were immersed in 10% neutral-buffered formalin, routinely processed for paraffin embedding, sectioned at 5 μm, and stained with hematoxylin and eosin (HE). Select serial sections of placenta and placental tumor were stained with phosphotungstic acid-hematoxylin (PTAH) and Masson's trichrome. 9 IHC was performed on serial sections of placental tumor and, as internal controls, on placenta and fetal liver, skeletal muscle, lymph node, peripheral nerve, and adipose tissue. Normal bovine tissue served as positive external control, and omission of the primary antibody served as a negative control. The technique was a standard streptavidin–biotin–horseradish peroxidase procedure as previously described with similar cited modifications. 8, 10 Primary antibodies (mouse monoclonal anti-human unless otherwise specified) were AE1/AE3, clone AE1 and AE3 (1°1000; BioGenex Corp, San Ramon, CA); α-smooth muscle actin (SMA), clone 1A4 (1°200; BioGenex); CAM5.2, predilute clone CAM5.2 (Becton-Dickinson Corp, San Jose, CA); CD31, clone JC/70A (1°50; Dako Corp, Carpinteria, CA); desmin, clone 33 (1°100; BioGenex); MAK6, clones KA4 and UCD10.11 (1°250; Zymed Labs, San Francisco, CA); muscle-specific actin, clone HHF35 (1°300; Dako); rabbit polyclonal myoglobin, clone A0324 (1°800; Dako); antibovine vimentin, clone 3B4 (1°200; Dako); UCD10.11, clone UCD10.11 (1°250; Zymed); rabbit polyclonal von Willebrand Factor VIII (F8), clone A0082 (1°50; Dako). For each antibody, the external and internal controls were appropriately immunoreactive, except in the case of myoglobin, and were weakly immunoreactive in the case of UCD10.11. HE slides of 21 human cases of chorangioma from the personal archives of one author (KB) were reviewed.
Histologically, the chorioallantois of the intercotyledonary placenta was expanded by multiple, poorly demarcated, variably sized, mesenchymal masses that were pleomorphic in cellular and stromal composition, of which 2 distinct morphologies often blended indiscernibly (Fig. 2). One morphology had moderate cellularity with scant vascularity (arbitrarily referred to as area No. 1; Fig. 3), and a second morphology had scant cellularity with prominent vascularity (arbitrarily referred to as area No. 2; Fig. 4). Area No. 1 comprised a pleomorphic population of spindle-, stellate-, and oval-shaped cells arranged in intersecting fascicles and bundles supported by minimal intervening, pale eosinophilic, fine fibrillar stroma. Cells sometimes assumed a storiform pattern or were contiguous with or whorled around vascular channels, which variably contained erythrocytes. Most cells had distinct cell borders, tapering or blunt ends, a small to moderate amount of intensely eosinophilic, granular to amorphous, sometimes lightly vacuolated cytoplasm, and a peripherally placed, thin to plump, hyperchromatic to open-faced oval nucleus. Some cells had cytoplasmic cross-striations (Fig. 5), multiple nuclei, and central nuclear alignment (Fig. 3 inset). Area No. 2 comprised scattered oval- and spindle-shaped cells supported by a large amount of pale eosinophilic, fine fibrillar stroma. Cells sometimes were contiguous with and whorled around variable-sized, usually blood-filled and often distended, blood vessels. Cells had indistinct cell borders, a small amount of pale eosinophilic cytoplasm, and an oval to polygonal, basophilic nucleus with variably distinct fine chromatin stippling. In both regions, mitotic figures were rare, and there were mild, multifocal hemorrhage, occasional acute fibrin thrombi, and interstitial edema. The chorion had mild, multifocal interstitial fibrosis and mineralization, which are not uncommon findings in ruminant placentas in the experience of this institution (Zoological Society of San Diego). Chorionic villi also had mild, multifocal, neutrophilic placentitis, which was attributed to prolonged parturition.
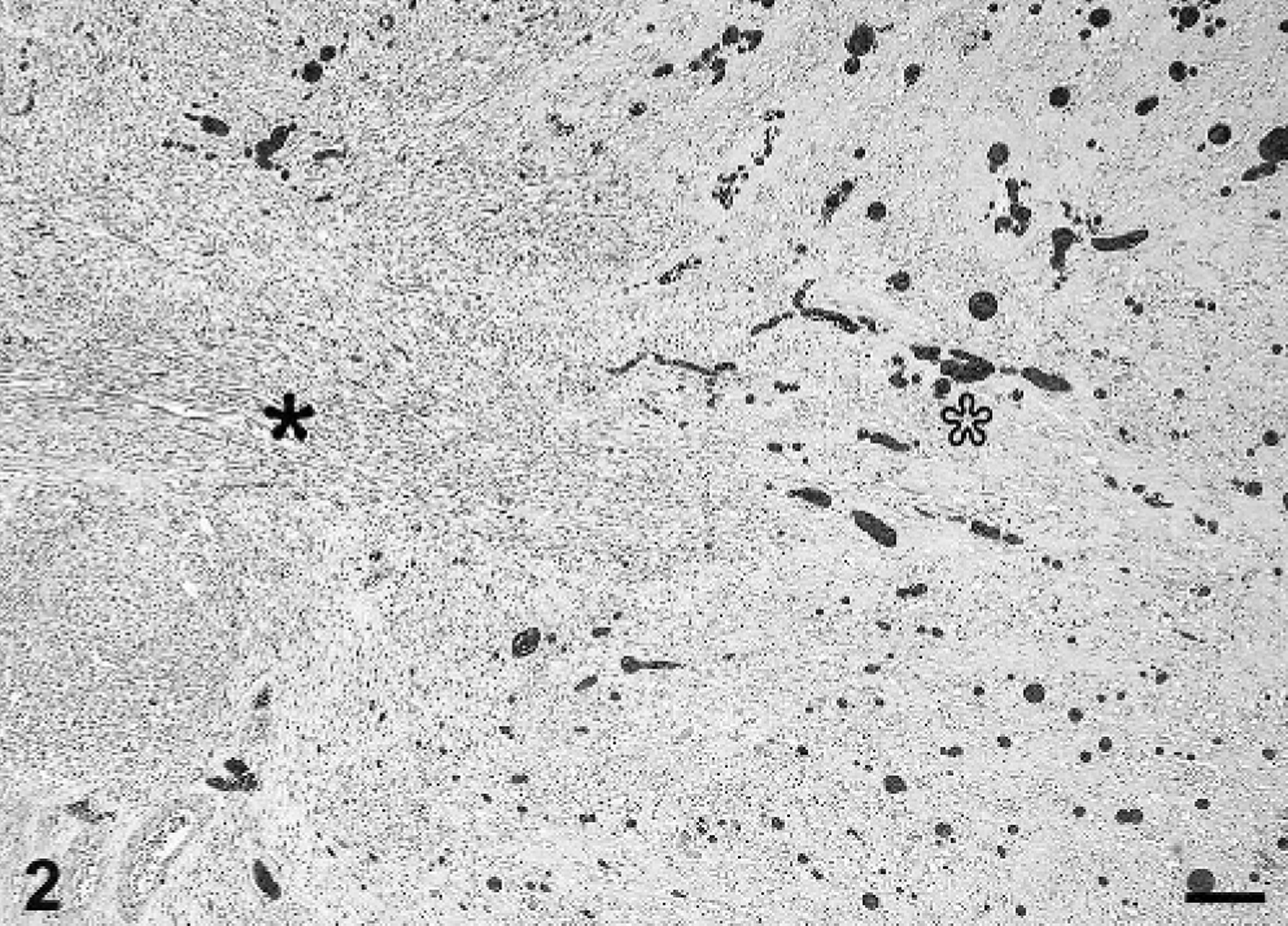
Placental tumor; bongo. The placental tumor is pleomorphic in cellular and stromal composition, having 2 distinct morphologies that periodically blend into each other. Area No. 1 (black asterisk) has moderate cellularity and scant vascularity and transitions into area No. 2 (white asterisk) that has scant cellularity and prominent vascularity. HE stain. Bar = 200 μm.
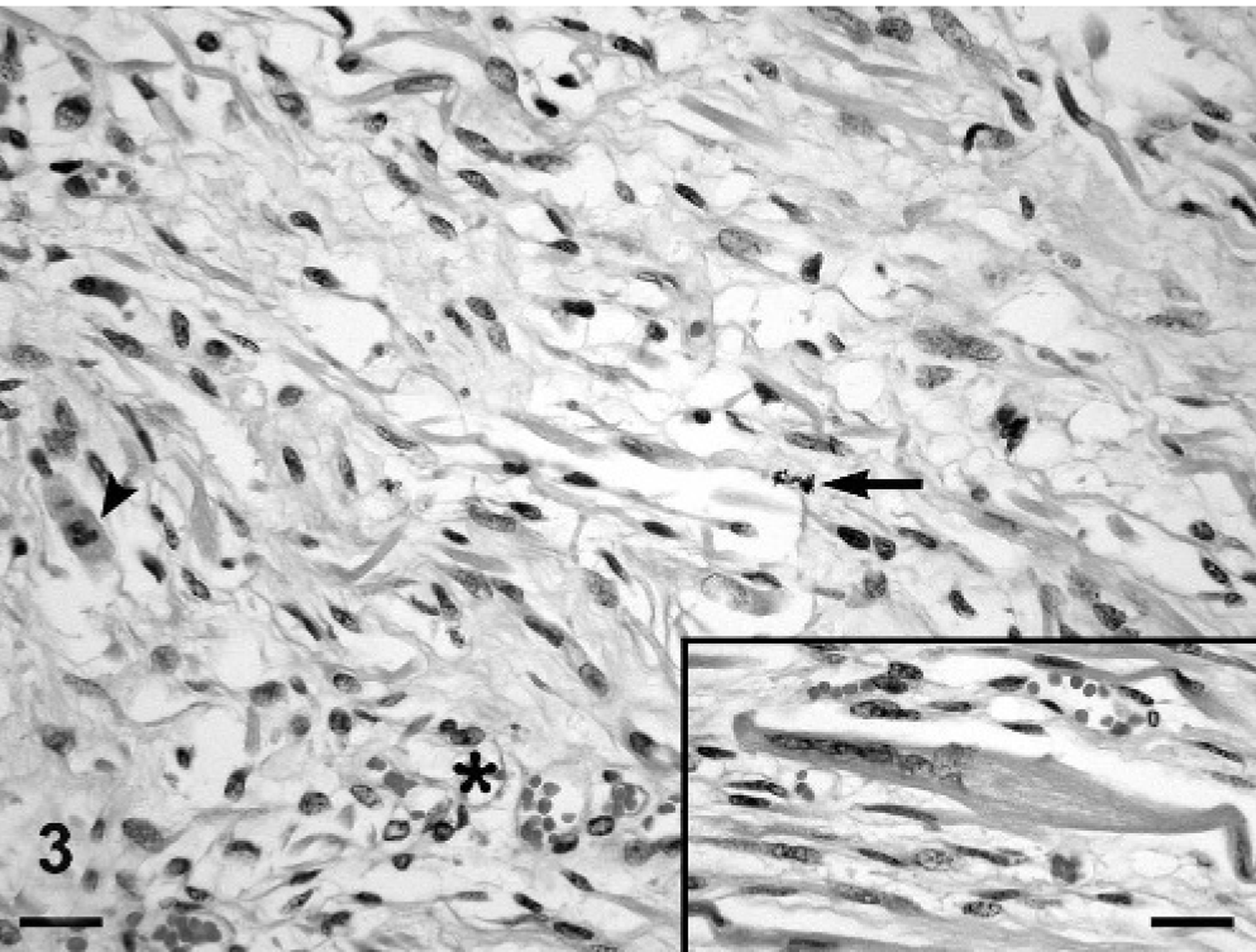
Placental tumor; bongo. Area No. 1 with moderate cellularity and scant vascularity is composed of intersecting fascicles of discrete spindle-, stellate-, and oval-shaped cells that sometimes are contiguous with vascular channels variably filled with erythrocytes (asterisk) and are supported by minimal fine fibrillar stroma. Some cells have multiple nuclei, central nuclear alignment, and cytoplasmic cross-striations (arrowhead and inset), suggestive of skeletal muscle. Mitotic figures are rare (arrow). HE stain. Bar = 20 μm, inset = 20 μm.
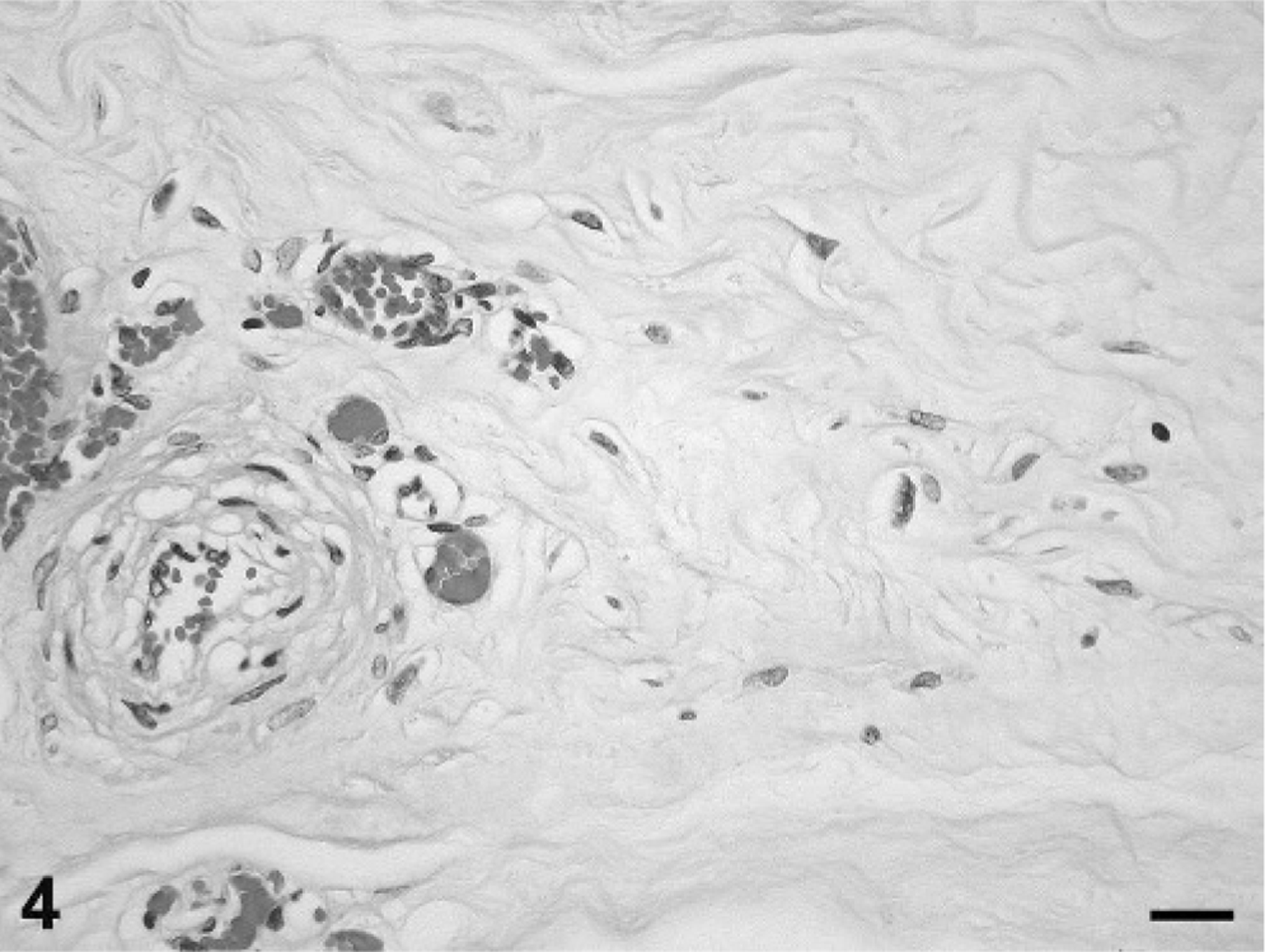
Placental tumor; bongo. Area No. 2 with scant cellularity and prominent vascularity is composed of scattered oval- and spindle-shaped cells that have poorly demarcated cell borders, sometimes are contiguous with variably sized, blood-filled vessels, and are supported by a large amount of fine fibrillar stroma. Mitotic figures are not present. HE stain. Bar = 20 μm.

Placental tumor; bongo. In area No. 1, a stellate-shaped cell with distinct cell margins, central nuclei (partially out of the plane of section), and a moderate amount of cytoplasm with cross-striations that are darkly stained by phosphotungstic acid-hematoxylin stain. These features are indicative of skeletal muscle origin. Bar = 10 μm.
Histology of the fetus showed that the manubrial mass comprised lobules of cartilage dissected and surrounded by thin bands of fibrous connective tissue and small peripheral aggregates of adipocytes and undifferentiated mesenchymal cells. The malformed omentum comprised a proliferation of adipocytes with multifocal infiltration of the serosa and wall of the forestomach. The asymmetrical muscling of the thoracic limbs was likely caused by skeletal muscle hypertrophy, but definitive interpretation would require comparison to an age-matched control, which was not available. The lateral tracheal flattening was not associated with histological changes.
Table 1 provides a summary of results for antibodies immunoreactive with cells in the placental tumor. Overall, the tumor had endothelial, smooth muscle, skeletal muscle, undifferentiated muscle, and fibrous components. In addition, area No. 1 often contained less mature cells than area No. 2, and area No. 1 contained skeletal muscle cells unlike area No. 2.
Comparison of the presence and relative number of immunoreactive cells of the blood vessel endothelium and wall (including pericytes) and the interstitium of areas No. 1 and No. 2. To facilitate interpretation of immunohistochemistry, tumor cells were divided into 3 main regions: vascular endothelium, vascular wall and pericytes, and interstitial cells. The terms refer to the subjective assessment of the number of immunoreactive cells in that region of tissue.∗
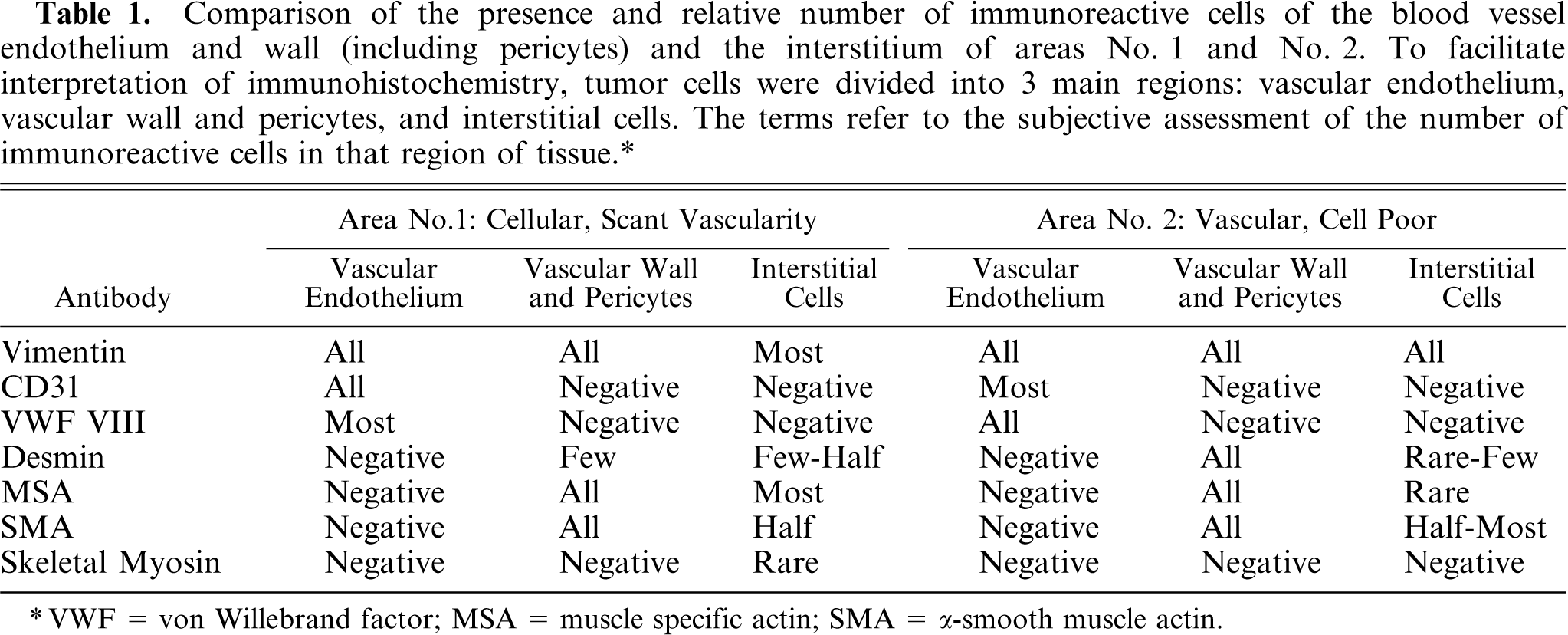
∗ VWF = von Willebrand factor; MSA = muscle specific actin; SMA = α-smooth muscle actin.
On gross examination, the placental tumor resembled a chorangioma, as described in humans. 1, 2 Chorangiomas (also referred to as chorioangiomas) are ill-defined, benign placental tumors that are hypothesized to be of fetal blood vessel origin or a hamartoma of primitive chorionic mesenchyme. Chorangiomas can occur in the placenta, usually toward the fetal side, or in the umbilical cord and can present as 1 to multiple, usually well-circumscribed, thinly encapsulated, fleshy, red tumors that sometimes have tan foci indicative of infarction. The histologic appearance can vary depending on the contribution of vascular and stromal components and the degree of vascular engorgement and can appear malignant, but metastasis has not been reported. The stromal component is not well described, but chorangiomas have been referred to as hemangioblastoma, fibroangiomyxoma, fibroma, myxoma, and pericytoma. In veterinary medicine, placental masses of similar description have been documented only in 5 bovine placentas, of which 1 case had concurrent fetal cutaneous and lingual hemangiomas. 4, 6, 7
The gross similarity of the bongo placental tumor to a chorangioma was not fully supported by findings on histology or ancillary diagnostics. Most notably, in addition to fibrous cells, endothelial cells, pericytes, smooth muscle, and undifferentiated mesenchymal cells, the tumor contained skeletal muscle cells, as confirmed by PTAH-stained cross-striations (Fig. 5) and cells immunoreactive with skeletal myosin. The presence of skeletal muscle cells was not described in previously reported or reviewed cases of chorangioma and is not consistent with their proposed origin. Thus, combining information of placental tumor morphology, histochemistry, immunophenotype, and embryogenesis (as described below), the placental tumor in this East African bongo is hypothesized to be a stem cell tumor.
During embryogenesis, embryoblastic-derived mesenchyme and vessels contribute to formation of the placenta. 3 Later in placental development, hemangioblastic progenitor cells differentiate from local fetal mesenchyme, develop into fetal capillaries and hematopoietic stem cells, and subsequently are involved in villous differentiation and placental maturation. Fetal mesenchyme is derived from progenitor cells in somites. The dermatomyotome compartment has cells that can remain undifferentiated or differentiate into endothelium (also from the sclerotome compartment), epidermis, muscle (skeletal, cardiac, or smooth muscle), muscle satellite cells, adipose tissue, and cartilage. 5 Determination and differentiation of the progenitor cells occur through a complex cascade of events that involves a network of signaling proteins and transcriptional factors to mediate the sequential transcription of distinct genes during different stages of development. These regulatory factors have a paracrine or autocrine effect.
Recent studies suggest that at least some satellite cells of skeletal muscle are not derived from somite cells but from the endothelium and/or pericytes of embryonic vessels, such as the dorsal aorta. 5 Cells competent to generate satellite cells can express both myogenic markers, including desmin and α-smooth muscle actin, and endothelial markers, including CD31 (PECAM) but not von Willebrand Factor VIII. They can differentiate, depending on the local regulatory factors, into skeletal muscle during embryonic and postnatal growth and regeneration, into vascular components, or perhaps into other mesenchyme. Cells competent to generate satellite cells migrate to their destination early in development, likely through the circulation. These cells, however, do not acquire the competence to differentiate until later in development and upon maintenance of interaction with neighboring cells. These progenitors may remain demonstrable only in the bone marrow, therefore they may be related to or representative of the multi-potent mesenchymal cells that have been identified in the bone marrow and are capable of producing chondroblasts, adipocytes, skeletal muscle, osteoblasts, and possibly endothelial cells. 5, 11
The proposed diagnosis of placental stem cell tumor from this East African bongo suggests that there is an alteration in the local microenvironment or a dysregulation of autocrine- or paracrine-mediated proliferation and differentiation of dormant fetal multi-potent cells resulting in a tumor composed of endothelial, pericyte, smooth muscle, skeletal muscle, fibrous, and undifferentiated mesenchymal cells. This hypothesis leads to the question of whether this stem cell tumor represents a variant of chorangioma and therefore, whether chorangioma and chorangioma-like tumors as described in humans and bovids actually are stem cell tumors of which the final cell type is dictated by the alteration in the local microenvironment. It also raises the question of whether the presence of multiple structural anomalies noted in this East African bongo fetus represent additional regional anomalies in morphogenesis or is an unrelated pathogenesis.
Footnotes
Acknowledgements
We thank Yvonne Cates for histochemistry and Diane Nayden for immunohistochemistry.
