Abstract
Infection with Rift Valley fever phlebovirus (RVFV) causes abortion storms and a wide variety of outcomes for both ewes and fetuses. Sheep fetuses and placenta specimens were examined during the 2010–2011 River Valley fever (RVF) outbreak in South Africa. A total of 72 fetuses were studied of which 58 were confirmed positive for RVF. Placenta specimens were available for 35 cases. Macroscopic lesions in fetuses were nonspecific and included marked edema and occasional hemorrhages in visceral organs. Microscopically, multifocal hepatic necrosis was present in 48 of 58 cases, and apoptotic bodies, foci of liquefactive hepatic necrosis (primary foci), and eosinophilic intranuclear inclusions in hepatocytes were useful diagnostic features. Lymphocytolysis was present in all lymphoid organs examined with the exception of thymus and Peyer’s patches, and pyknosis or karyorrhexis was often present in renal glomeruli. The most significant histologic lesion in the placenta was necrosis of trophoblasts and endothelial cells in the cotyledonary and intercotyledonary chorioallantois. Immunolabeling for RVFV was most consistent in trophoblasts of the cotyledon or caruncle. Other antigen-positive cells included hepatocytes, renal tubular epithelial, juxtaglomerular and extraglomerular mesangial cells, vascular smooth muscle, endothelial and adrenocortical cells, cardiomyocytes, Purkinje fibers, and macrophages. Fetal organ samples for diagnosis must minimally include liver, kidney, and spleen. From the placenta, the minimum recommended specimens for histopathology include the cotyledonary units and caruncles from the endometrium, if available. The diagnostic investigation of abortion in endemic areas should always include routine testing for RVFV, and a diagnosis during interepidemic periods might be missed if only limited specimens are available for examination.
Keywords
Rift Valley fever phlebovirus (RVFV) of the family Phenuiviridae, genus Phlebovirus, is primarily transmitted by mosquitos and causes disease in both livestock and humans. Frequent epidemics occur in eastern and southern Africa with significant outbreaks in South Africa between 1950 and 2011. 22,24 Previously, we reported that a clear distinction can be made between the lesions and cellular tropism of natural RVFV infections in young lambs and adult sheep. 18,19 The disease is generally fatal in new-born lambs, and virtually all pregnant sheep abort when infected with a wild-type strain of RVFV. 24 Rift Valley fever (RVF) is also an important zoonosis with most human infections presenting as an acute self-limiting febrile illness. 2,11,14 However, in a minority of human patients, severe hepatic disease with widespread hemorrhages, renal impairment, encephalitis, or ocular lesions can develop. 2,11,14 Additionally, virus replication in the human placenta and vertical transmission of RVFV to human fetuses resulting in an increased risk of miscarriage have also been reported. 1,4,6,16
An outbreak of RVF in South Africa occurred over a 4-year period from 2008 to 2011. 22 Subsequently, an isolated outbreak occurred in 2018 with cases in sheep and humans. 13 No other outbreaks were reported between 2012 and 2017 in South Africa. However, interepidemic circulation of RVFV is suspected to occur based on seroprevalence studies. In a number of African countries, significant high-seroprevalence clusters exist in areas that experienced epidemics during the late 20th century, and significant low-seroprevalence clusters occur in contiguous areas of Western and Central Africa. 7 In South Africa, RVFV is endemic and may circulate without causing outbreaks in northern KwaZulu-Natal Province, which is a tropical coastal plain. 26 By comparison, occasional epidemics occur in the grasslands of the Free State Province of South Africa where IgG antibody against RVFV were detected among domestic ruminants, born after the most recent outbreak. 17 This suggests that the virus might be maintained at low levels in the environment and sporadic clinical cases and abortions may be overlooked or misdiagnosed.
Experimental RVFV infection in pregnant ewes causes a wide variety of outcomes. The susceptibility of European-breed ewes to RVFV infection was investigated during the first, second, and third trimesters of gestation. 3 Four of 11 pregnant ewes inoculated with RVFV developed a viremia and succumbed or had to be euthanized before the end of the experiment. The researchers reported that 3 of these ewes developed multifocal necrotizing hepatitis and the fourth had moderate to severe hepatitis. Four of the 7 surviving ewes, euthanized at 21 to 23 days post infection, developed neither detectable viremia nor clinical signs. These ewes also failed to seroconvert and lesions typical of RVF were absent at postmortem examination. In contrast, the other 3 surviving ewes developed detectable viremia and seroconverted but had no significant lesions at postmortem examination. Interestingly, 11/11 liver specimens and 9/11 brain specimens from ewes were positive for viral RNA. Lesions in the brain were not described.
Reports concerning the development of lesions and detection of viral RNA in fetal organs and placenta in experimental cases also varies in the literature. Viral RNA was detected in liver or brain samples of all 18 fetuses obtained from 9 experimentally infected ewes. 3 Multifocal hepatic necrosis was reported for one of the fetuses and placental lesions were identified in 3 of 11 specimens. There were 108.33 RNA copies per gram in the liver of one fetus, whereas viral RNA was undetectable in the liver of 5 other fetuses. Experimental infection of 11 pregnant ewes at either one third of gestation or at mid gestation with RVFV showed efficient replication of virus in maternal endometrial epithelial cells with subsequent infection of fetal trophoblasts. 21 Moreover, the virus directly targets fetal trophoblasts in the hemophagus region and abortion in ewes is associated with widespread necrosis of placental tissues. 21 Viral RNA was detected in 17 liver samples and 9 brain samples from 23 fetuses in the same study. 21 In another study, a ewe inoculated intradermally with the virulent van Wyk RVFV strain aborted after infection and a viral load of 106.2 virus per gram was measured in the placenta using the mouse intraperitoneal median lethal dose (mouse lethal doses) technique. 10 However, minimal virus was identified in fetal blood, and virus could not be detected in fetal liver, lungs, or spleen. In a RVFV vaccine study, a single vaccine dose did not protect ewes against challenge with virulent RVFV, and at necropsy all 58 ewes had either aborted or had dead fetuses. 28 A RVFV concentration of 102.5 to 104.5 mouse lethal doses per gram was obtained from fetal spleen, liver, and brain specimens.
The lesions and cellular tropism of naturally occurring RVF in sheep fetuses and placenta have not been described in previously published research. There is also the perception that RVF disappears in the interepidemic periods and only reappears suddenly when heavy rainfall occurs. However, as mentioned, experimental studies have demonstrated a wide variety of outcomes for pregnant ewes and fetuses infected with RVFV. Additionally, previous research suggests interepidemic circulation of virus. Therefore, excluding RVF from the list of possible differential diagnoses in endemic areas might be difficult given the paucity of information concerning the gross, histopathological and immunohistochemical findings in naturally occurring abortions caused by RVFV. Therefore, the principal aim of this study was to describe RVF in naturally infected fetal tissues and determine the diagnostic significance of the lesions. A second aim was to evaluate the diagnostic usefulness of the placenta relative to other organs and to determine which section of the placenta, if any, is most useful for diagnosis.
Materials and Methods
Case Selection
Tissue specimens were collected from sheep and fetuses as described previously. 18 Briefly, tissues from 1034 necropsied animals were collected by government officials and the principle investigator (LO), preserved in 10% formalin, embedded in paraffin, and hematoxylin and eosin–stained histologic sections were routinely prepared. Included were tissues from at least 122 ovine fetuses of which 99 tested positive for RVF. From this collection, cases were excluded when the level of autolysis was severe, a liver specimen was not available for study, or only a single organ was available. This work was done in agreement with the Animal Ethics Committee of the University of Pretoria (Clearance Certificate V096-16). Additional material in support of the results presented in this article is available from the authors upon request.
A total of 72 ovine fetus cases were suitable for study of which 58 cases tested positive for RVF based on results of histopathology, RT-qPCR (reverse transcriptase qualitative polymerase chain reaction), and/or IHC (immunohistochemistry). Also included in this study were placenta and uterus (caruncle) from 8 ewes that tested positive for RVF. Matching fetal organs (included in the 72 fetus cases) were available for 3 of these ewes. In total, placenta specimens were available for 35 of the cases. Animals that died as part of the outbreak and were shown to be infected with RVFV based on a parallel interpretation of one or more tests (histopathology, RT-qPCR, and IHC) were presumed to have died of RVF even if macroscopic or histologic lesions were absent. The negative control tissues for the study were 14 aborted fetuses from the same outbreak that tested negative for RVFV.
Diagnostic Tests
Nucleic acid extractions and RT-qPCR on fresh tissue specimens from 54 of the cases were performed at the Biotechnology PCR Laboratory of the Agriculture Research Council–Onderstepoort Veterinary Institute, as described previously. 20
Immunohistochemistry with additional controls for the detection of RVFV antigen was performed using polyclonal antibody to RVFV (mouse ascitic fluid) as described in the original validation study. 20 Briefly, the method included microwave antigen retrieval in citrate buffer (pH 6.0), blocking of endogenous peroxidases with 3% hydrogen peroxide, incubation with the anti-RVFV primary antibody (National Institute for Communicable Diseases, Johannesburg, South Africa) at 1:500 dilution for 1 hour, and rabbit anti-mouse secondary antibody (F0232, DakoCytomation) followed by detection with a standard avidin-biotin peroxidase system, Vectastain Elite ABC-HRP Kit (PK-6100, Vector Laboratories, Inc), NovaRED peroxidase substrate (SK-4800, Vector Laboratories, Inc), and hematoxylin counterstain. Additionally, to confirm that intravascular immunoreactivity observed in this study was due to RVFV antigen, sequential tissue sections were tested with antibodies to RVFV and Wesselsbron virus. The polyclonal antibody (mouse ascitic fluid) to Wesselsbron virus was also prepared at the National Institute for Communicable Diseases and IHC performed as described previously. 20 Other controls were RT-qPCR- and IHC-positive RVF cases from 2009, and additional negative tissue controls that had liver lesions that resembled RVF but were due to other causes. Other irrelevant antibodies that were used were polyclonal antibody (mouse ascitic fluid) to bovine herpesvirus-1 and rabbit polyclonal antiserum to equine herpesvirus-1.
Examination of Tissues
Histomorphological lesions in all available organs were systematically recorded. Two authors (LO, SJC) reviewed 20% of the cases together to validate data collection methods, and the remainder were examined by one author (LO). Images of representative findings were reviewed remotely by a third author (ASD).
Lesion severity in the liver, kidney, and spleen was scored using a qualitative scale as mild, moderate, or severe. A semiquantitative scale was used to score the number of primary foci in the liver specimens as isolated (<5), scattered (between 6 and 10), or widespread (>10); and immunolabeling for RVFV antigen as isolated, scattered, or widespread as previously described. 19
The severity of autolysis was qualitatively graded as mild, moderate, or severe as described prior. 20 Briefly, autolysis was mild when cells were intact and organ architecture was distinct. Autolysis was moderate when cytoplasmic borders were still distinct, but some cells showed fading of nuclei and/or tissues stained uniformly eosinophilic. Severe autolysis was classified by loss of basophilia, indistinct cytoplasmic borders, nuclear fading, complete lysis of red blood cells, and the presence of many putrefactive bacteria in the tissues.
The number and types of fetal organs available for study varied with liver (n = 72; inclusion criterion), lung (n = 61), spleen (n = 59), and kidney (n = 58) being the specimens most often submitted. Additional tissues included 47 heart, 17 gastrointestinal tract, 4 lymph node, 32 brain, 27 thymus, 6 adrenal, and 3 skin specimens. Gastrointestinal tract specimens included tongue (n = 7), forestomach (n = 7), abomasum (n = 5), small intestine (n = 15), and large intestine (n = 2).
Placenta specimens were available for 30 of the 72 fetuses included in this study. Another 5 placenta specimens had matching maternal organ samples but no other fetal tissues. For the 35 available placenta specimens, cotyledon was included in 30/35 with a portion of the chorioallantoic membrane connected to cotyledonary villi in 21/35 cases. Intercotyledonary chorioallantois was available in 11/35 cases and in 2 cases this was the only placenta specimen available. For 7 of the 8 ewes classified as positive for RVF, caruncle with myometrium and varying amounts of chorioallantoic villi were available. Additionally, there was one caruncle specimen with myometrium attached that had matching fetal organs, but other tissues from the ewe were not available. Uterus specimens with intact endometrium, myometrium, and perimetrium were available for 4/8 ewe cases. For 4/30 of the fetal cases, maternal caruncle with myometrium and varying numbers of cotyledonary villi were also available. The negative control specimens included placenta for 4 of the 14 RVFV-negative fetuses.
Statistical Analysis
Dichotomous data were described using proportions and 95% mid-P confidence intervals. Scalar data were descriptively presented using the median and interquartile range (25th to 75th percentiles). Data from ordinal scales were compared between the liver and other organs using Wilcoxon signed rank tests. Bivariate correlations between scaler data variables were evaluated using Spearman’s ρ. Agreement between dichotomous variables was estimated using the κ statistic. Freeware (Epi Info, version 6.04, US Centers for Disease Control and Prevention) and commercial statistical software (IBM SPSS Statistics Version 25, International Business Machines Corp) were used to perform statistical analyses. Statistical results were evaluated at the 5% level of significance.
Results
Overview
Macroscopic lesions most often reported in examined fetuses included edema and hemorrhages in visceral organs (Fig. 1). Reported postmortem changes were increased friability of tissues with occasional moderate to advanced autolysis and hemoglobin imbibition that made it difficult to discern lesions. Effusions, possibly attributable to endothelial injury or postmortem changes, were also present in body cavities. Before excluding cases due to severe autolysis or lack of sufficient organs for examination, autolysis was classified in histological specimens as mild, moderate, or severe in 48, 36, and 15 of the total number of RVFV-positive cases (n = 99), respectively. After exclusions there were 58 RVFV-positive cases included in the study for which autolysis was classified as mild or moderate, 31 (53%) and 27 (47%) cases, respectively.
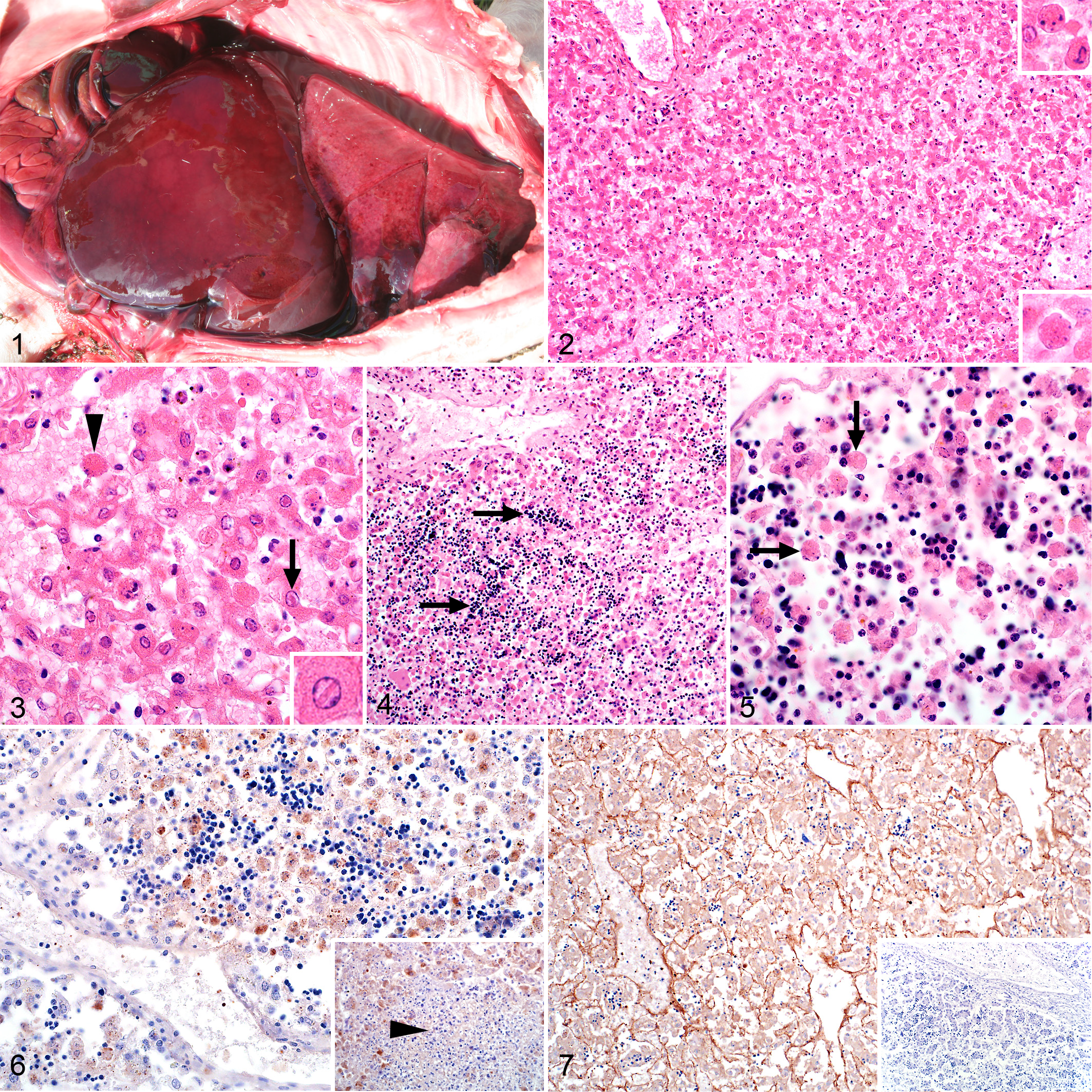
Rift Valley fever virus (RVFV), sheep fetuses.
Other than lung edema, typical histological lesions for RVF were absent from the organs of the fetuses included as RVFV-negative controls. Placental lesions in the negative controls included multifocal hemorrhages, varying degrees of karyorrhexis and karyolysis of trophoblasts, and the presence of cellular debris between the chorionic villi.
Viral antigen was easily observed and widespread in most organs including liver, kidney, lymphoid organs, lung, heart, brain, and placenta (Table 1). Positive labeling was typically fine diffuse to coarse granular cytoplasmic labeling or non-cell-associated antigen. In blood vessels, labeling was especially prominent as non-cell-associated antigen but also as cell-associated antigen in endothelial cells (Table 2). Viral antigen was also commonly present in vascular smooth muscle cells in multiple organs but particularly in the kidney (Table 3).
Frequency and Intensity of Immunolabeling in Different Organs Compared to Intensity of Immunolabeling in Liver, for 58 Sheep Fetuses With Rift Valley Fever Virus Infection.
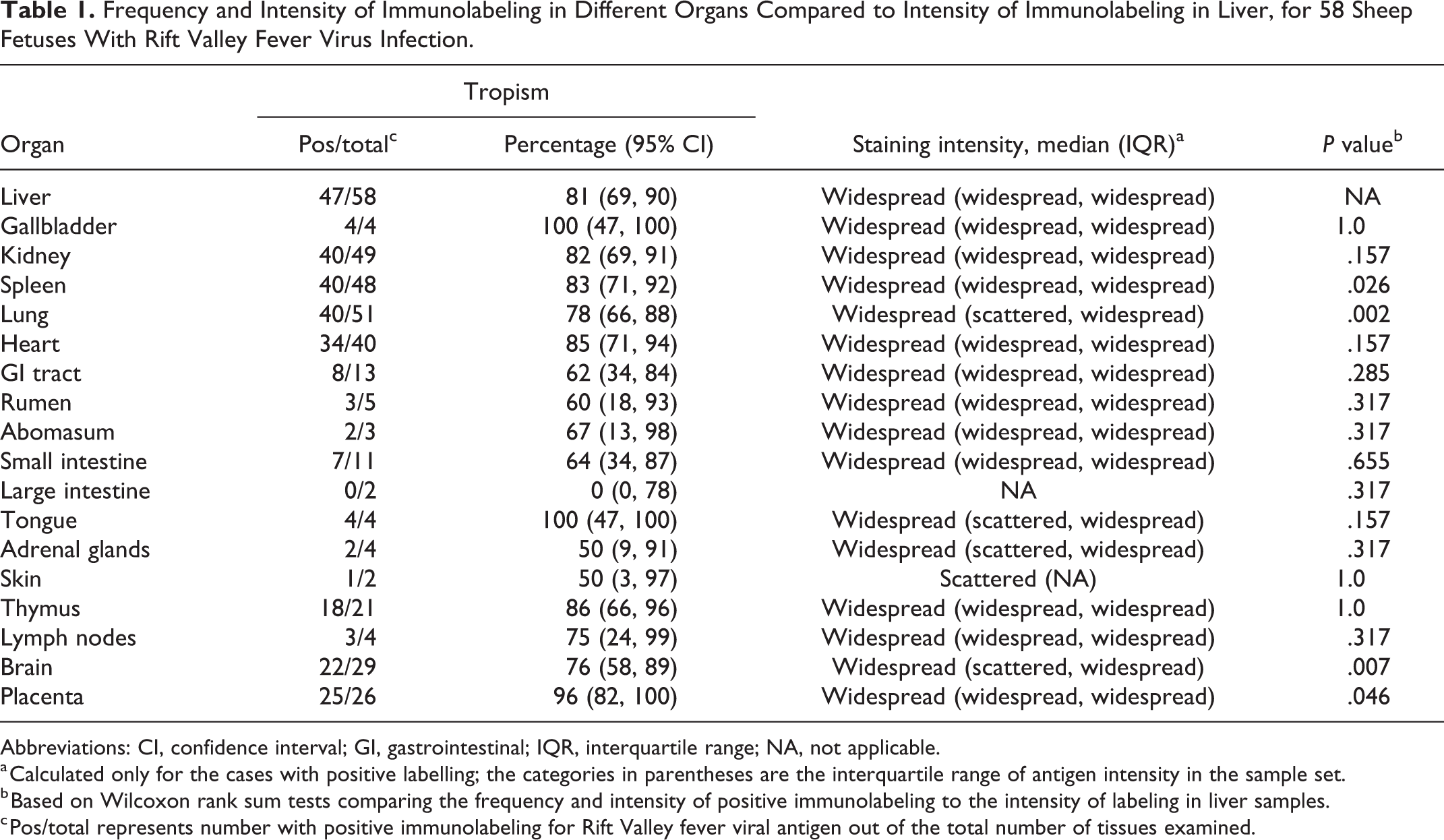
Abbreviations: CI, confidence interval; GI, gastrointestinal; IQR, interquartile range; NA, not applicable.
a Calculated only for the cases with positive labelling; the categories in parentheses are the interquartile range of antigen intensity in the sample set.
b Based on Wilcoxon rank sum tests comparing the frequency and intensity of positive immunolabeling to the intensity of labeling in liver samples.
c Pos/total represents number with positive immunolabeling for Rift Valley fever viral antigen out of the total number of tissues examined.
Frequency of Non-Cell-Associated Viral Antigen in Blood Vessels or Antigen in Vascular Endothelial Cells or Intravascular Cellular Debris for 58 Sheep Fetuses With Rift Valley Fever Virus Infection.
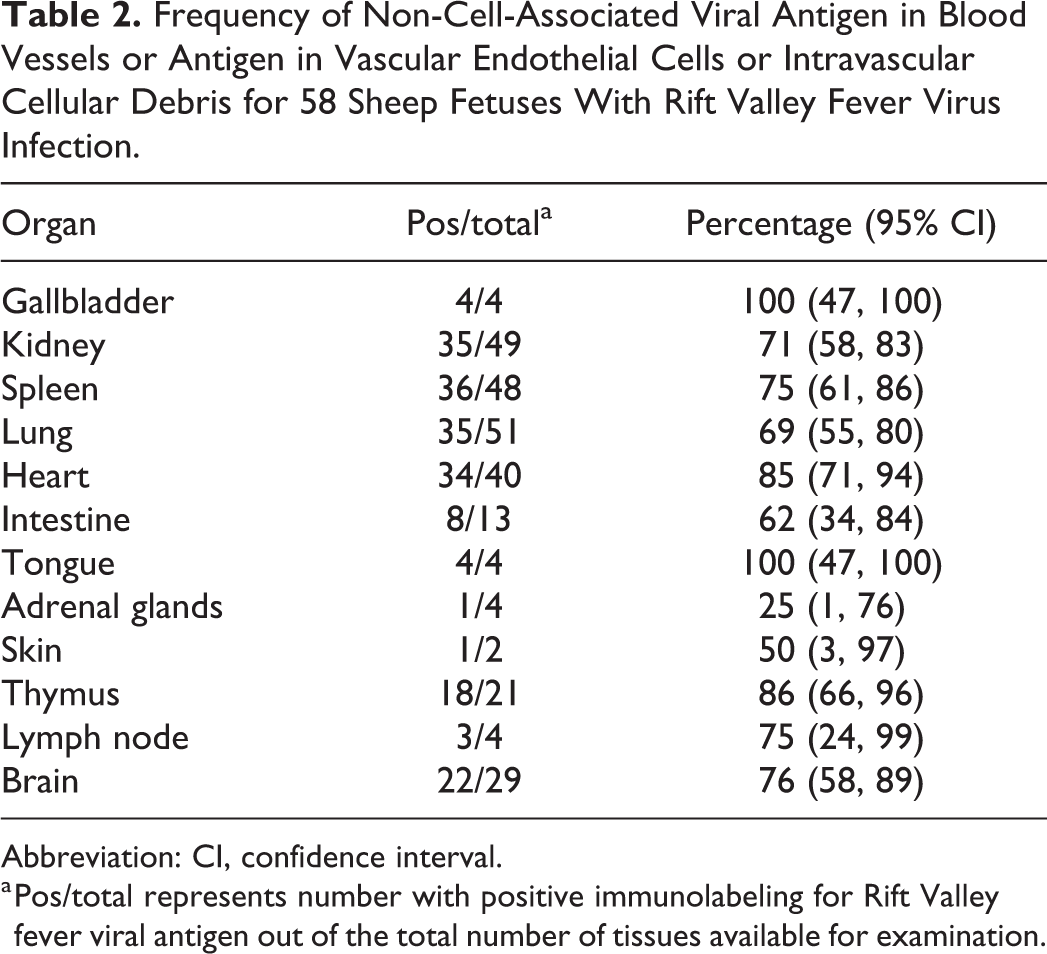
Abbreviation: CI, confidence interval.
a Pos/total represents number with positive immunolabeling for Rift Valley fever viral antigen out of the total number of tissues available for examination.
Frequency of Viral Antigen in Vascular Smooth Muscle in the Organs of 58 Sheep Fetuses With Rift Valley Fever Virus Infection.
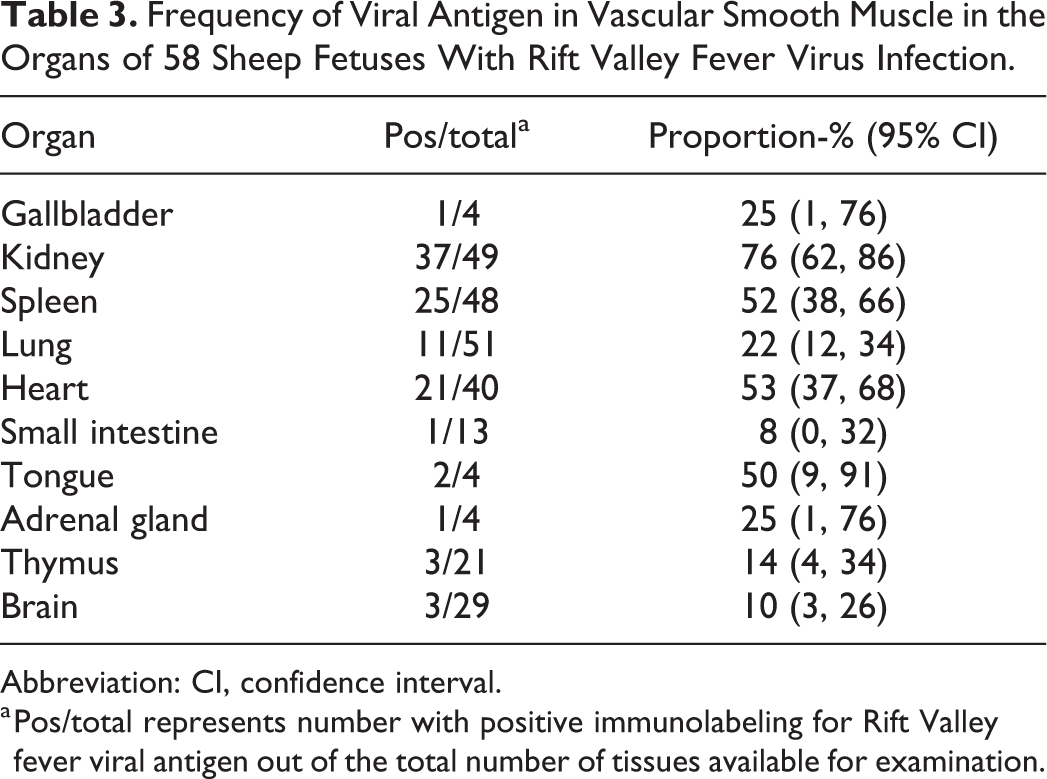
Abbreviation: CI, confidence interval.
a Pos/total represents number with positive immunolabeling for Rift Valley fever viral antigen out of the total number of tissues available for examination.
Non-cell-associated antigen was also often present in the capsules of organs or in the loose connective tissue around the gall bladder, intestines, adrenals, and thymus. The reticulin framework in the liver and spleen, and the basement membranes in the kidney cortex, apex of the renal papilla, and the heart were also occasionally delineated by the presence of non-cell-associated antigen. This was not considered to be an IHC artefact since immunolabeling was not observed in sequential slides incubated with antibody to Wesselsbron virus instead of RVFV (Supplemental Figs. S1–S4). Furthermore, viral antigen was always absent from epithelial cells, and their associated basement membranes, in the medulla of the kidney, the thymic cortex, T-cell areas of the gut-associated lymphoid tissue (GALT) in the ileum, the brain parenchyma, and mesenchymal connective tissues of the placenta. Additionally, RVFV antigen was not present in any of the tissues from the 14 cases included as negative tissue controls (Supplemental Figs. S5–S8).
Liver
Macroscopically, affected livers were often swollen and friable and either yellow or dark red (Fig. 1). Foci of necrosis or hemorrhage were not discernible. Microscopically, the severity of hepatic necrosis varied considerably among cases with 10 of 58 (17%), 18 of 58 (31%), and 20 of 58 (34%) specimens classified as mild, moderate, or severe necrosis, respectively. Necrosis in the liver was positively correlated with necrosis in the spleen (ρ = 0.511; P < .001). Descriptively, severity of liver necrosis was also correlated with necrosis in lymph nodes (ρ = 0.949) but the sample size was only 4 and the association was not significant (P = .051).
There was no discernible zone of necrosis in the liver. Instead, random hepatocyte necrosis or dropout of hepatocytes from the reticulin framework was present. Apoptotic bodies (Councilman bodies or acidophilic bodies), with condensed hypereosinophilic cytosol and marginalized chromatin, pyknosis or karyorrhexis, were also often present (Fig. 2). In mild cases, the architecture of the liver was largely intact with hepatocyte plates and sinusoids easily distinguishable. Necrotic hepatocytes with karyolysis or karyorrhexis were sparse and randomly scattered throughout the liver parenchyma. Occasional dropout of hepatocytes from the reticulin framework was also present in mild cases. Inflammation was either absent or sparse and predominantly neutrophilic. In moderate cases, histomorphologically normal-appearing hepatocytes were present in clumps or short strings of up to 10 cells, with scattered intervening hepatocyte necrosis and aforementioned hepatocyte dropout (Figs. 2, 3). The orderly arrangement of hepatocytes into plates was indistinct and sinusoids were sometimes difficult to distinguish. Macrophages were inconspicuous whereas neutrophils were often present (Fig. 3). In severe cases, surviving hepatocytes were present in small clumps or short strings of up to 5 cells with most nuclei in the majority of the high-power fields lost due to karyorrhexis or karyolysis accompanied by hepatocyte dropout and the presence of degenerate neutrophils (Figs. 4, 5). Occasionally there was complete dissociation of all the remaining hepatocytes and the sinusoids were difficult to distinguish from the empty spaces resulting from hepatocyte dropout. Portal areas were also difficult to identify in these severe cases. Irrespective of the degree of necrosis, hemorrhage or pooling of blood in spaces depleted of hepatocytes was rare, whereas edema was often present in the portal areas and hematopoiesis (Fig. 4) was present in most cases.
Foci of liquefactive hepatic necrosis (primary foci) were present in the liver specimens from 26 of 58 (45%) cases and were either isolated (20 of 58; 34%), scattered (3 of 58; 5%), or widespread (3 of 58; 5%). Necrosis in the liver was positively correlated with the number of primary foci (ρ = 0.296; P = .024). Intranuclear inclusion bodies (Fig. 3) were identified in 36 of 58 (62%) cases and mineralization of hepatocytes in 2 of 58 (3%). Unequivocal inclusion bodies were rod-shaped or filamentous with a halo, and the chromatin of the nucleus was marginalized. Depending on the plane of section through the nucleus other inclusion bodies were round or oval. Primary foci and inclusion bodies were absent from 5 of 58 (9%) cases that had mild (n = 1), moderate (n = 1), or severe (n = 3) necrosis. In a further 10 of 58 (17%) cases there were no lesions in the liver. For 5 of these 10 cases, matched placenta specimens were IHC-positive. For another case, the placenta was positive by RT-qPCR but negative by IHC. There were no placenta samples for the other 4 cases.
RVFV antigen was widespread and easily identified in the liver of 47 of 58 (81%) cases. Hepatocytes were the predominant target of RVFV (Fig. 6; Suppl. Figs. S1a and S2a). Viral antigen was sparse in primary foci and when present, found in scattered cellular debris (Fig. 6). Occasionally, the entire reticulin framework was delineated by the presence of non-cell-associated viral antigen accompanied by intense labeling of the tunica intima of hepatic veins (Fig. 7 and Suppl. Fig. S2a).
Gall Bladder
Microscopic lesions were absent in the available specimens. However, moderate to severe autolysis was present in all 4 specimens. Viral antigen was widespread in the capillaries and small blood vessels either as non-cell-associated viral antigen or as antigen in endothelial cells or intravascular cellular debris. Labeling was present in vascular smooth muscle cells in blood vessels of the adventitia and serosa in one case. Viral antigen was absent from the gall bladder epithelium in all the cases.
Spleen
Microscopically, lymphocytolysis was present in 42 of 48 (88%) RVF cases and was either mild (n = 14), moderate (n = 16), or severe (n = 12). Generally, the periarteriolar lymphatic sheaths comprised a narrow zone of lymphocytes, and follicular germinal centers and mantle zones were not developed, which was considered age-appropriate in fetuses. Lymphocytolysis and occasional tingible-body macrophages were present in both the red and white pulp in all spleen specimens that tested positive for RVFV antigen by IHC.
Splenic infection with RVFV was present in 40 of 48 (83%) cases and antigen was either widespread (n = 33), scattered (n = 3), or isolated (n = 4). Viral antigen was predominantly present in the red pulp and mostly non-cell-associated or in cellular debris. Positively labeled mononuclear cells, morphologically consistent with macrophages, were seen within the red pulp in 31 of 40 (78%) and the white pulp in 19 of 40 (48%) cases. Tingible-body macrophages labeled only rarely.
In 36 of 40 (90%) RVFV-positive cases, labeling was prominent in small blood vessels and capillaries as non-cell-associated antigen and antigen in endothelial cells and cellular debris (Fig. 8). In 25 of 40 (63%) cases, RVFV antigen was also present in smooth muscle cells in blood vessels in the capsule or in the central arteries (Figs. 8, 9). The serosa, smooth muscle cells in the capsule and the reticulin framework of the red pulp labeled intensely in some cases (Suppl. Fig. S3a).
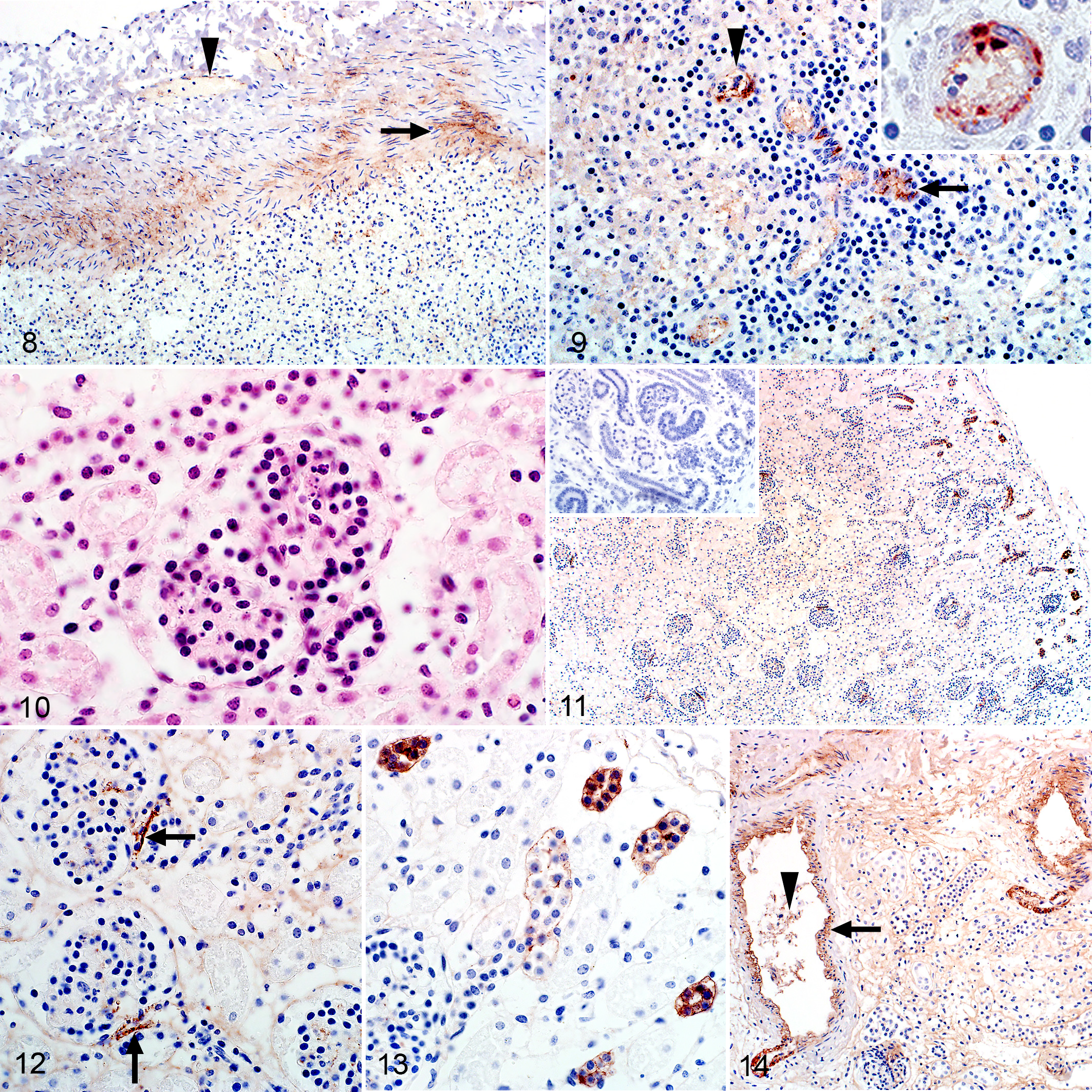
Rift Valley fever virus (RVFV), sheep fetuses.
Lymph Nodes
There were no gross lesions in the lymph nodes. Microscopically, there were varying degrees of lymphocytolysis in 3 of the 4 available lymph node specimens. In these, cellular debris was present in both the cortex and medulla. Similar to the spleen, follicular germinal centers were not developed.
Viral antigen was present in all 3 cases with lymphoid necrosis. Labeling was present in the capillaries and small blood vessels either as non-cell-associated viral antigen or as antigen in endothelial cells or intravascular cellular debris. In the sinusoids, mononuclear cells, morphologically consistent with macrophages, were also positive for viral antigen. In 2 specimens, non-cell-associated virus was present in the capsular blood vessels, and in one of these cases the loose connective tissue surrounding the capsule was also diffusely positive.
Thymus
No gross or microscopic lesions were observed in the thymus. The lymphocytolysis present in other lymphoid organs was absent. Viral antigen, present in 18 of 21 (86%) available specimens, was often present in capillaries and small blood vessels as non-cell-associated viral antigen or as antigen in endothelial cells or intravascular cellular debris (Suppl. Fig. S4a). Viral antigen was also present in vascular smooth muscle cells in 2 of 21 (10%) cases.
Kidney
Gross lesions were not present in the kidney. Microscopically, severe multifocal acute tubular epithelial injury lacking significant associated inflammation was present in only 3 of the 49 (6%) cases with available kidney specimens. This was characterized by karyolysis of proximal tubular epithelial cells and the presence of cellular debris and round desquamated cells with homogenous eosinophilic cytoplasm in the lumens of tubules, particularly in the superficial cortex and medullary rays. Hemorrhage was present in the cortical interstitium or below the capsule in some cases. Glomeruli often appeared less densely cellular than normal with pyknosis or karyolysis in 43 of 49 (88%) cases (Fig. 10). Nuclear debris was often present in interstitial capillaries in 37 of 49 (76%) cases, and the amount of pyknosis and karyorrhexis in the glomeruli was positively correlated with the quantity of nuclear debris in the interstitial capillaries (ρ = 0.617, P < .001). Pyknosis and karyorrhexis in the glomeruli was also significantly associated with the presence of viral antigen in the glomeruli (ρ = 0.705, P < .001).
RVFV antigen was present in 40 of 49 (82%) cases with available kidney specimens. In the positive cases, labeling was present in the cortex in 40 of 40 (100%) cases (Fig. 11) and in both the cortex and the medulla in 26 of 40 (65%) cases. RVFV antigen was most prominent in the glomeruli and was present in 39 of 40 (98%) positive cases. Interpretation of cellular tropism in fetal glomeruli was often hindered by the presence of intense granular labeling in most of the glomerular tufts; however, in some cases antigen could be localized specifically to either vascular endothelial cells or mononuclear cells. Viral antigen was often present at the vascular pole of the glomerulus opposite the macula densa, in a small group of cells that are likely juxtaglomerular cells and extraglomerular mesangial cells (Fig. 12). In 11 of 40 (28%) cases, labeling was also present in tubules in the cortex (Fig. 13 and Suppl. Fig. S5a). In contrast, labeling was absent in tubules in the medulla.
Labeling was also occasionally present in smooth muscle cells in the efferent or afferent arterioles or in small blood vessels in the cortex including arcuate blood vessels in the corticomedullary junction in 37 of 40 (93%) cases (Fig. 14 and Suppl. Fig. S6a). Non-cell-associated antigen or antigen in vascular endothelial cells or cellular debris was often present in blood vessels or interstitial capillaries in both the cortex and the medulla.
Viral antigen was more prominent in the interstitial capillaries of the cortex compared to the medulla. Viral antigen was present in the cortical interstitial capillaries in 32 of 40 (80%) positive cases and labeling in the glomerulus was significantly associated with labeling in these interstitial capillaries (ρ = 0.551; P < .001). Viral antigen was also present in the medullary interstitial capillaries in 23 of 40 (58%) cases, and the presence of viral antigen in the cortical interstitium was significantly associated with the presence of viral antigen in the medullary interstitium (ρ = 0.609; P < .001). Finally, viral antigen was occasionally present in the capillaries and small blood vessels of the perirenal adipose tissue.
Adrenal
Two of 4 cases for which specimens were available were positive for RVFV antigen. However, significant histological lesions were not discernible in either case. One sample was in good condition, and viral antigen was in cortical epithelial cells in all 3 zones as well as in vascular endothelial cells in the capsule, cortex, and medulla. Autolysis was advanced in the second specimen and only the capsule and cortex were available for examination; however, labeling was clearly discernible in scattered cortical epithelial cells and in vascular smooth muscle cells in the capsule. Labeling was also present as non-cell-associated antigen or antigen in endothelial cells in the periadrenal adipose tissues.
Lung
Macroscopic hemorrhages were occasionally present in the lungs and a serosanguineous effusion was common in the thoracic cavity. Microscopically, pulmonary interstitial edema was present in 44 of 51 (86%) cases with available specimens and was classified as moderate to severe in 29 of 51 (57%) cases (Fig. 15). Edema expanded the connective tissue surrounding blood vessels, bronchi, or bronchioles and was also present in the pulmonary septa. Intra-alveolar edema could not be assessed since a mild accumulation of liquid in fetal airways is considered to be age-appropriate. A few lung specimens were clearly aerated (6 of 51; 12%). Mild multifocal expansion of the alveoli was often noted but interpreted as attributable to the loss of liquid during processing and not the presence of air. Hemorrhage was present in the pulmonary septa or the visceral pleura in 21 of 51 (41%) cases. Mild pyknosis and karyorrhexis were often present in the alveolar septa and pulmonary blood vessels.
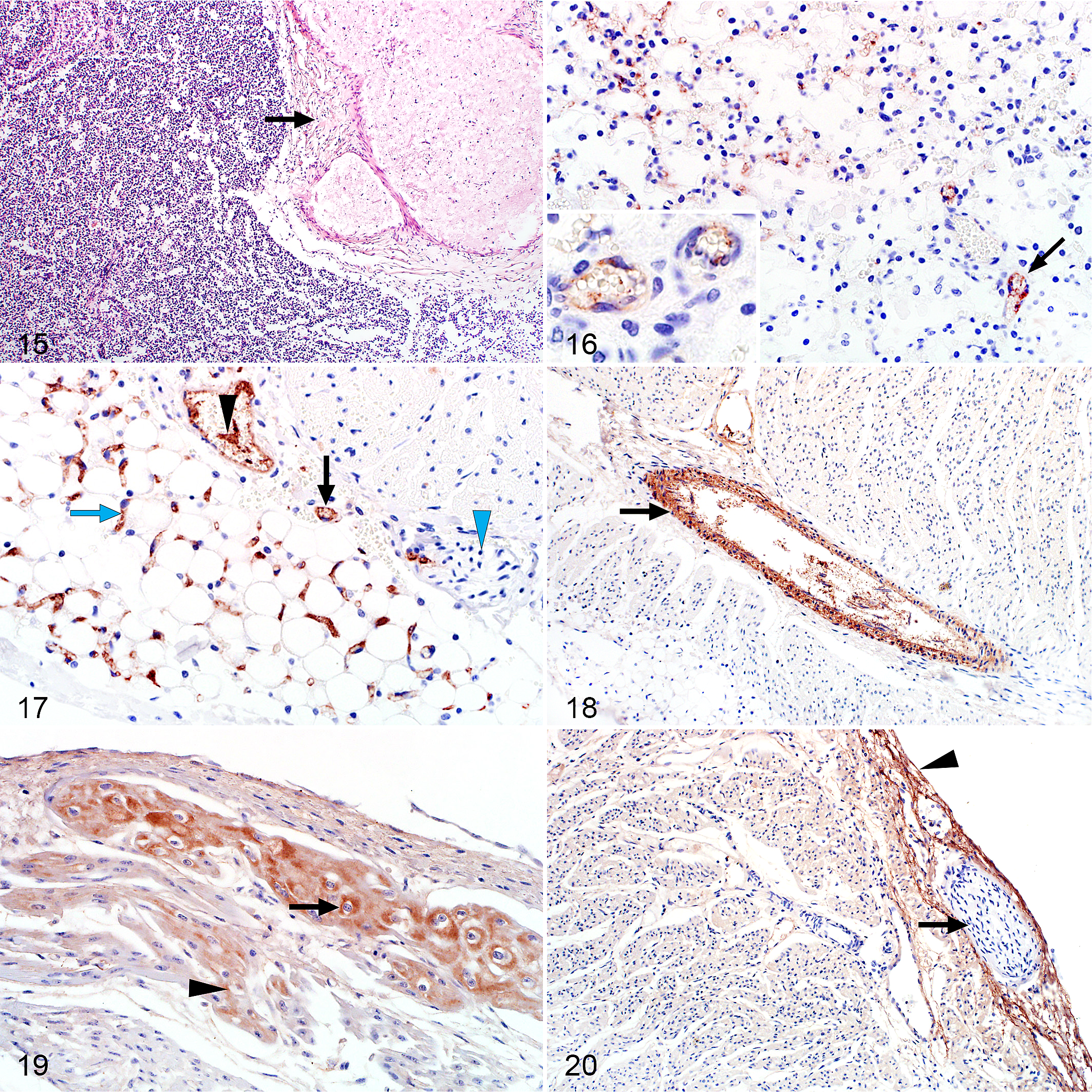
Rift Valley fever virus (RVFV), sheep fetuses.
Labeling was present in 40 of 51 (78%) cases (Fig. 16). Viral antigen was present in mononuclear cells, histomorphologically consistent with macrophages, in the interstitial capillaries in 38 of 40 (95%) of RVFV-positive cases. Labeling was also present as non-cell-associated antigen or antigen in endothelial cells or cellular debris in small blood vessels and capillaries in 35 of 40 (88%) positive specimens (Suppl. Fig. S7a). In 11 of 40 (28%) positive specimens, viral antigen was present in vascular smooth muscle cells.
Heart
Macroscopic and histopathological lesions were absent in the cardiac parenchyma apart from one case where subepicardial hemorrhage was present. RVFV antigen was present in heart specimens of 34 of 40 (85%) cases. Viral antigen was present as non-cell-associated antigen or antigen in endothelial cells or cellular debris in small blood vessels and capillaries in all RVFV cases (Fig. 17) and widespread in most instances (31 of 34; 91%). Labeling was also present in vascular smooth muscle cells in 21 of 34 (62%) RVFV-positive specimens (Fig. 18 and Suppl. Fig. S8a). Purkinje fibers were available for examination in 35 of the heart samples. In 22 of these (63%) the Purkinje fibers labeled intensely positive (Fig. 19 and Suppl. Fig. S9a). Viral antigen was also present in cardiomyocytes in 13 of 34 (38%) positive cases (Fig. 19). Diffuse labeling of the myocardium, not considered to be artefactual immunolabeling, and intense subepicardial and subendocardial labeling was occasionally observed (Fig. 20; Suppl. Figs. S8a and S9a). Rarely, viral antigen was also present in capillaries in the pericardial adipose tissue (Fig. 18 and Suppl. Fig. S8a).
Gastrointestinal Tract
Macroscopically, blood or blood-stained mucoid fluid was present in the abomasum. A serosanguineous effusion was often present in the peritoneal cavity. Microscopically, rare foci of necrosis were present in the lamina propria of the small intestine. Cellular debris, likely due to lymphocytolysis of GALT, was observed sporadically in the lamina propria of the small intestine. Karyorrhexis was also present in the connective tissue between the Peyer’s patches whereas lymphocytes within the Peyer’s patches were not affected (Fig. 21).
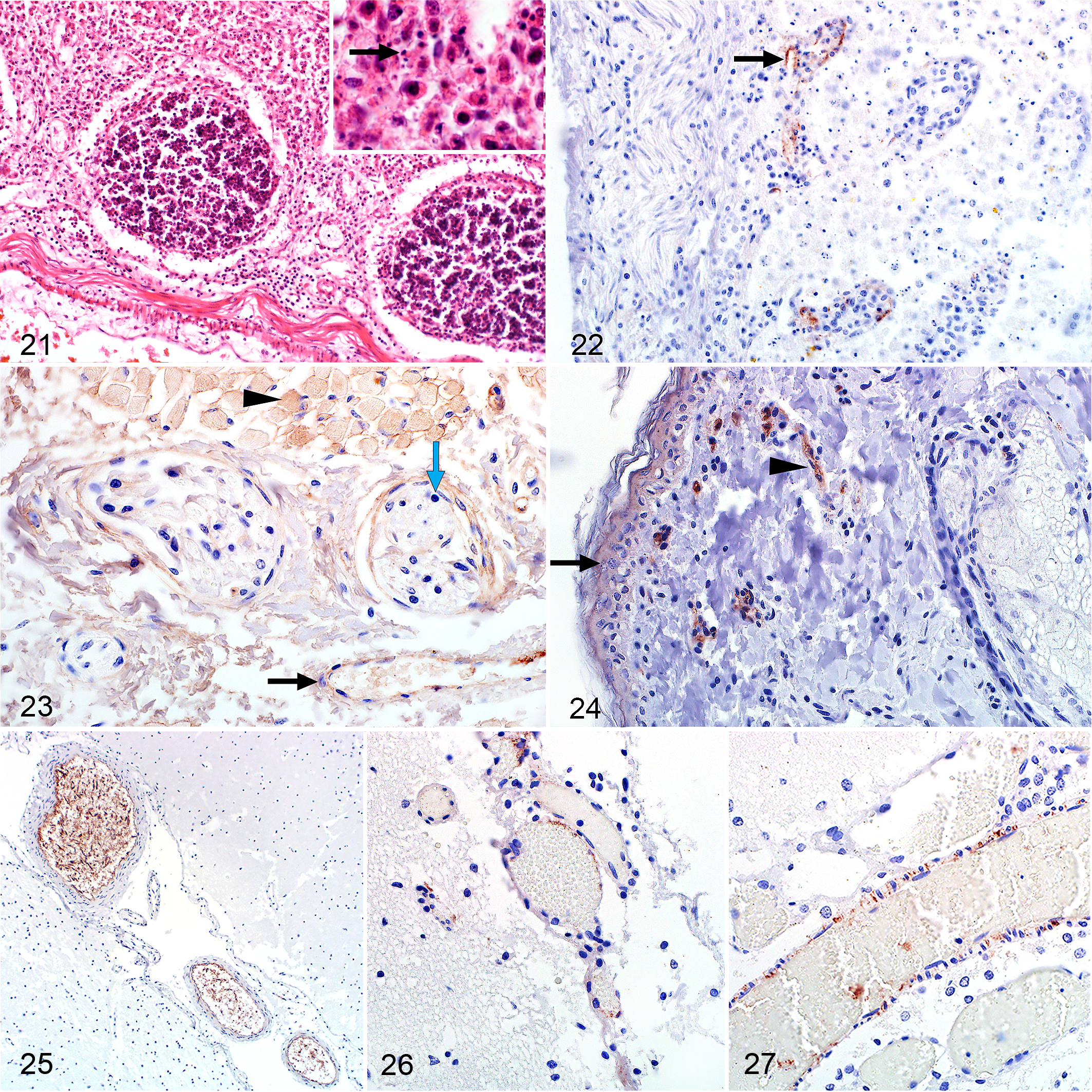
Rift Valley fever virus (RVFV), sheep fetuses.
Viral antigen was present in 8 of 13 (62%) cases with specimens from the intestinal tract. Commonly, labeling was present in the vascular endothelial cells of small blood vessels and capillaries in the rumen, abomasum, small intestine, and tongue (Figs. 22, 23). Labeling within the lamina propria was more pronounced in the small intestine. Occasional labeling was also present as non-cell-associated antigen or antigen in cellular debris in small blood vessels in the muscularis mucosae or the serosa. In one case, labeling was also observed in vascular smooth muscle cells in a small blood vessel in the serosa. In another case, fine diffuse labeling was present throughout all layers of the small intestine including the connective tissue adjacent to the serosa and smooth muscle cells in the tunica muscularis. Positively labeled cellular debris was also present in the lumen of the small intestine in this case and in the rumen of another case.
RVFV antigen was present as non-cell-associated antigen or antigen in endothelial cells or cellular debris in small blood vessels of 4 of 4 (100%) cases with tongue specimens. In 2 of these cases, fine diffuse labeling was present in the connective tissue and in the skeletal myofibers (Fig. 23).
Skin
Only 2 samples were available for histological examination of which one was positive for RVFV antigen. Labeling was prominent in vascular endothelial cells or cell debris in capillaries (Fig. 24). There was also diffuse viral antigen labeling in the epidermis.
In the case where the skin sample was RVFV antigen negative, all the other available visceral organs from the fetus (liver, spleen, lymph node, thymus, kidney, lung, heart, and intestines) also tested negative. However, samples from the placenta and visceral organs of the ewe (liver, gall bladder, spleen, lymph node, kidney, adrenal, lung, and skin) were positive while the uterus specimen was negative.
Nervous System
Other than edema, no lesions were present in any tissues from the central nervous system. Labeling was present in capillaries and small blood vessels of the meninges in 22 of 29 (76%) cases either as non-cell-associated antigen or antigen in vascular endothelial cells or cellular debris (Figs. 25, 26; Suppl. Fig. S10a). Occasionally, viral antigen was present in vascular smooth muscle cells (3 of 11; 27%) or in capillaries in the white or gray matter but never within the brain parenchymal cells (Fig. 27). Peripheral nerve bundles, occasionally encountered in tissue sections, had viral antigen in the capillaries. However, antigen was absent from nerve fibers (Figs. 20, 23).
Placenta
Macroscopically, there was intercotyledonary edema and the cotyledons were congested and necrotic (Fig. 28). Edema was confirmed microscopically and was prominent in the cotyledonary chorioallantois (Fig. 29). However, the most significant histological lesion was necrosis of trophoblasts and endothelial cells in both the cotyledonary and intercotyledonary chorioallantois. In the cotyledonary villi, necrosis of trophoblasts was generally diffuse with multifocal cellular debris between the villi (Fig. 30). Other histological changes in the placenta, possibly normal physiological changes, included hemorrhage and mineralization. In the RVFV-positive cases, hemorrhage was present in 8 of 26 (31%) placenta specimens with matching fetal tissues, and 4 of 5 (80%) placenta specimens where other maternal tissues were available but no fetal tissues. Multifocal hemorrhage was most often adjacent to chorioallantoic villi (10 of 12; 83%). Occasionally hemorrhage was present in the myometrium (3 of 9; 33%) or the perimetrium (1 of 4; 25%). Fine basophilic granules, possibly mineral, were present in multiple single trophoblasts or in aggregates of trophoblasts in the chorioallantoic villi in 9 of 31 (29%) cases. Occasionally, a basophilic finely granular to amorphous material was present in the chorioallantoic villi (4 of 30; 13%) that was possibly glycosaminoglycans or mineral and was not considered to result from disease. Intravascular cellular debris and severe congestion were often present. Necrotizing vasculitis was rare (n = 1) and there were no thrombi in any of the placenta specimens.
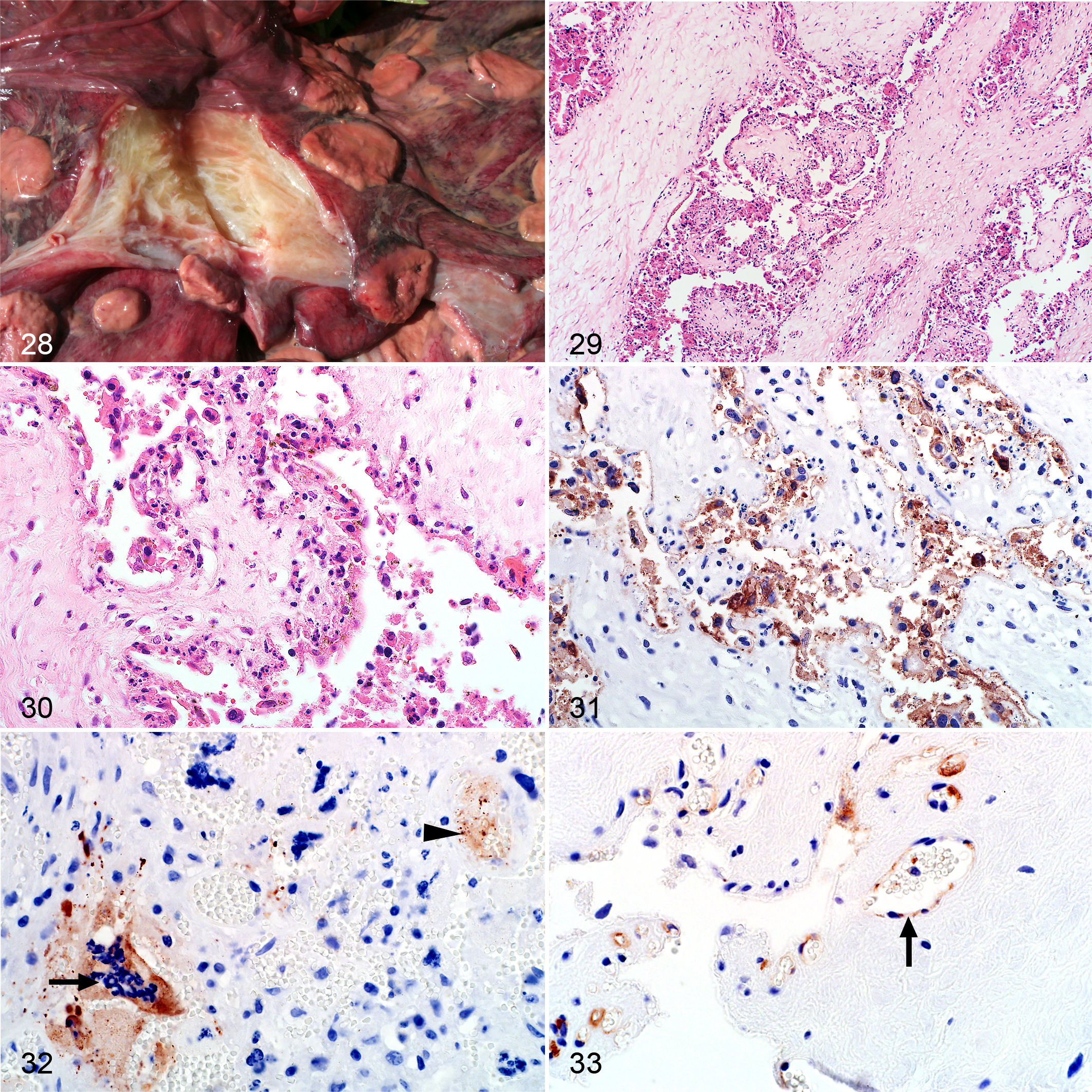
Rift Valley fever virus (RVFV), sheep placenta.
RVFV antigen was present in 28 of 31 (90%) RVFV-positive fetus and ewe cases where placenta was available. Labeling was predominantly in trophoblasts and cellular debris in the cotyledonary chorioallantois (Fig. 31 and Suppl. Fig. S11a). Occasionally binucleate and multinucleated maternal syncytial cells were also positive (Fig. 32). However, not all necrotic trophoblasts were positive for RVFV antigen. Labeling was generally sparse in the intercotyledonary placenta, with rare intercotyledonary trophoblasts positive in 4 of 10 (40%) cases.
Viral antigen was also in vascular endothelial cells, intravascular cellular debris, or non-cell-associated in 16 of 31 (52%) positive fetus and ewe cases. Labeling was present in fetal blood vessels in the chorioallantoic membranes in 15 of 26 (58%) of available specimens and also prominent in the cotyledonary chorioallantois in 4 of these cases (Fig. 33 and Suppl. Fig. S12a). Viral antigen was also present in blood vessels in 3 of 9 (33%) myometrium specimens. Labeling was always absent from the endometrium, squamous mesothelial cells lining the allantoic cavity, and chorionic and allantoic mesenchyme.
In 11 of 58 (19%) fetal cases, all available organ specimens were IHC-negative. Placenta was available for 7 of these cases of which 6 tested IHC-positive. In one case, all available fetal organs were negative, but the placenta was PCR-positive and IHC-negative. In this case, only a small portion of the chorioallantoic membrane was available to examine histologically. Karyorrhexis and karyolysis of trophoblasts were present, although only a few trophoblasts could be reliably identified.
Viral antigen was present in the placenta specimens of 6 of 8 (75%) RVFV-positive adult sheep. For one adult case, liver tested negative by both IHC and RT-qPCR but the placenta and the adult kidney labeled positive by IHC. A fetus was not available for testing in this case. In another adult case the liver tested positive, but the placenta was negative. For a further 3 adult cases fetal tissues were also available. Liver specimens from the ewes tested positive in all 3 cases. Fetal liver specimens tested positive by RT-qPCR but were IHC-negative, and the placenta specimens were IHC-positive. Another 2 adult ewes were RT-qPCR and IHC-positive. In one case liver was IHC-positive but intercotyledonary placenta and uterus specimens that included caruncle were all negative. In the third case maternal liver, kidney, spleen, and lung tested IHC-positive but the perimetrium, myometrium, endometrium and severely autolyzed caruncle were IHC-negative.
Discussion
Infection with RVFV has a wide variety of outcomes for both ewes and fetuses and limited information is available concerning the lesions in fetal organs, including the placenta. Therefore, the principle aim of this study was to summarize gross, histopathological, and immunohistochemical findings and evaluate the diagnostic utility of different tissues and specific histologic features of RVFV in fetuses and placenta. A further aim was to compare the distribution of viral antigen in fetal organs and cells with previous results in adult sheep and young lambs from the same outbreak. 18,19 As with the results for adult sheep and young lambs, a limitation of this study is that the frequencies of lesions and immunolabeling could be an overestimate of the general population of RVF cases. 18,19 This is due to the fact that organs were not consistently sampled in these field cases and nonspecific macroscopic lesions in most fetal organs might have exacerbated this shortcoming. Another limitation was that the stage of gestation or the length of fetuses was not known and usually fetuses could not be paired with the dams or placentas. It was not uncommon for multiple dead ewes, lambs, fetuses, and the occasional placenta from the same flock to be presented for necropsy on the same day. In the present study, only 4 fetuses could be matched with the dam and a placenta specimen.
In the 2010–2011 RVF outbreak in South Africa, farmers typically reported nearly simultaneous abortion of large numbers of pregnant ewes. This was accompanied by dystocia, the death of up to 100% of young lambs, and the deaths of a significant number of adult ewes. Many pregnant ewes were found dead, or died after having aborted. Other pregnant ewes aborted without showing clinical signs and recovered. During the outbreak in Kenya in 1931, it was reported that the ewes had been bred to lamb in July and August and that abortions started to occur in the latter part of June (probably when many ewes were in mid to late pregnancy). 9 By mid-July (when many ewes were probably in late pregnancy) there was a marked increase in the number of abortions on the farm and also high mortality of ewes with coincident high mortality in lambs. 9 By the end of July, mortality in ewes and lambs was more extensive but abortions among the ewes had almost ceased with the situation much the same by mid-August. 9 Ewes were either found dead without having shown clinical signs or succumbed a few hours after having a mucopurulent nasal discharge, inappetence, and occasional vomition and hematochezia. However, some ewes that aborted were not visibly ill with the exception of a few that developed metritis or peritonitis. Additional experiments demonstrated that recovery was also a common sequel to abortion in pregnant animals. 9 In a -experimental study in the Netherlands to determine the susceptibility of pregnant animals (n = 11) of a native breed of sheep to RVFV, 3 of 4 (75%) ewes infected in the third trimester had to be euthanized due to severe clinical signs and 1 of 3 ewes in the second trimester of gestation died. 3 Four other ewes in the study, euthanized at 21- and 23-days postinfection, did not develop clinical signs or viremia but postmortem examination revealed 1 dead fetus in a third trimester ewe, and live twin fetuses in the uterus of 2 first trimester ewes and 1 second trimester ewe. In 2 of 4 (50%) first trimester ewes euthanized at 22 days postinfection, there were remnants of gestation in the uterus. 3 The study was terminated, and the 7 surviving ewes euthanized at 21 to 23 days postinfection. Therefore, it is unknown how many of the ewes may have aborted and survived the infection. However, it would seem from the data provided by Daubney et al 9 and the experiment by Antonis et al 3 that ewes in the later stages of pregnancy are more susceptible to lethal disease and might die before aborting whereas ewes in the earlier stages of pregnancy might abort or resorb the fetus and survive the infection. Additional studies would be needed to validate these speculations.
In the present study, macroscopic lesions in fetuses with RVF were nonspecific and autolysis was mild or moderate in most cases (84 of 99; 85%). Similar observations were made in other field cases and in experimental studies. 3,5,9 In a trial of a RVF vaccine candidate, 7 fetuses were recovered from 4 ewes that aborted following inoculation with a wild-type strain of RVFV, and none of the 7 had gross lesions. 5 Autolysis was also described as insufficient to impair histological examination of tissues. Apparently, some RVFV-infected ovine fetuses might be born alive, since the lungs of at least 6 of 58 (10%) fetuses in the present study were aerated. Another study, examining the susceptibility of pregnant sheep to a strain of virus from the 1974 RVF outbreak in the Free State Province of South Africa, reported that macroscopic lesions were absent in 18 fetuses recovered and autolysis was present in 6 of 12 (50%) fetuses that had died in utero. 3 Therefore, discharged fetuses or fetuses recovered from the uteruses of dead ewes are not always severely autolyzed and diagnostically useful samples might still be available.
In agreement with the present study, fetal malformations have not been reported in natural cases of RVF or in experimental cases using wild-type virus. 3,5,9,21 However, a number of live-attenuated RVF vaccine strains cause fetal malformations of the central nervous system and the musculoskeletal system that include spinal hypoplasia; hydranencephaly; hypoplasia of the cerebrum, cerebellum, and spinal cord; brachygnathia; arthrogryposis and scoliosis; brachygnathia inferior and arthrogryposis. 8,12,15 Therefore, natural RVFV infection with wild-type virus is not a differential diagnosis when fetal malformations are encountered in sheep. However, ill-advised use of live attenuated vaccines in ewes in the first or second trimester of pregnancy should be investigated if fetal malformations are reported.
Microscopically, hepatocellular single cell death with foci of liquefactive hepatic necrosis (primary foci) and intranuclear inclusions in hepatocytes were useful diagnostic features in fetuses, but were not present in all cases in this study. Liver lesions were absent in 10 of 58 (17%) cases and were subtle in another 10 of 58 (17%) cases. However, in most of the cases where necrosis was present, either primary foci or intranuclear inclusions were also present (43 of 48; 90%) with intranuclear inclusions observed in 62% of fetal liver specimens. Cases where both primary foci and intranuclear inclusions are absent might present a diagnostic challenge if other specimens, particularly fetal cotyledon or maternal caruncle, are not available. There were 4 such cases identified in this study. Therefore, absence of necrosis, primary foci, or nuclear inclusions in the liver does not necessarily exclude a diagnosis of RVF. Additionally, whereas intranuclear inclusions were detectable in HE-stained sections in 62% of fetal liver specimens, it was previously reported that they were present in 38% of young lambs and only in 7% of adult sheep cases. 18,19 Whereas mice experimentally infected with RVFV may develop pleomorphic intranuclear inclusions with variably distinct borders and no or only an equivocal halo, distinct inclusions in sheep (including ovine fetuses) are rod-shaped or filamentous with a halo. 18,19,25
The lack of a fully developed adaptive and innate immune system in young or unborn animals may also lead to higher morbidity and mortality in young animals and fetuses compared to older animals. 29 Previous research demonstrated that neonatal mice exposed to toll-like receptor stimuli (lipopolysaccharide, polyinosinicacid: polycytidylic acid, and murine hepatitis virus-A59) suffer from uncontrolled proinflammatory innate immune responses related to their developmentally normal low numbers of T lymphocytes. 29 Additionally, after LPS treatment, the proinflammatory cytokines TNF, MCP-1, and IL-6 in serum gradually decreased as the age of the mice increased. 29 Large numbers of naive T lymphocytes, present in adult animals, might be required to temper the early innate responses and reduce the secretion of proinflammatory cytokines. 29 Moreover, children infected with dengue hemorrhagic fever virus often have minimal bleeding manifestations but develop severe shock due to excessive plasma leakage and severe depletion of intravascular volume. 27 In the present study hemorrhages in fetuses were minimal but plasma leakage with effusions in body cavities, accompanied by brain and lung edema, was very common. This study and previous research demonstrated that endothelial cells are a target of RVFV, and lesions suggestive of vascular injury were reported in adult sheep and lambs. 18,19,21 Consequently, young or unborn animals might suffer severe microcirculatory dysfunction and shock due to a lack of sufficient numbers of T lymphocytes to keep the proinflammatory innate responses under control.
Other histological lesions in fetuses were generally far subtler than those previously reported in young lambs and adult sheep, and could be easily overlooked. 18,19 Lymphocytolysis was present in all lymphoid organs except for the thymus and Peyer’s patches where resident T lymphocytes were unaffected. In the spleen, follicular germinal centers and mantle zones were undeveloped, deemed to be age-appropriate, and necrosis was generally most obvious in the red pulp. Acute renal tubular injury was rare but nuclear pyknosis and karyorrhexis in the glomeruli, and to a lesser extent in the interstitial capillaries, were useful diagnostic features. Additionally, mild pyknosis and karyorrhexis were often present in the alveolar septa and pulmonary blood vessels.
Fetal liver specimens were not consistently positive for RVFV antigen. It was previously observed for adult sheep and young lambs that most RVFV immunolabeling was in the liver. 18,19 In fetuses, the cotyledon and caruncle seem to be the more reliable specimens for diagnostic testing, with viral antigen present in the trophoblasts of 28 of 30 (93%) RVF cases (Table 4). A recent study investigating how RVFV crosses the ovine placenta showed that epithelial (syncytial) cells of the maternal villus and fetal trophoblasts are highly susceptible and permissive for infection and that widespread necrosis of these cells causes abortion. 21 Occasionally, the rapid progression of placental necrosis caused fetal mortality before fetuses could be infected. 21 Another study concerning vertical transmission of RVFV in rats also found that the placenta had a higher viral load than the liver and other maternal organs. 16 High levels of infectious virus were obtained from the placenta regardless of the time of infection. The same study also reported that RVFV has a preference for reproductive tissues including the uterus, ovary, placenta, and mammary glands. 16 Therefore, in RVFV-infected fetuses or pregnant ewes, specimens from the cotyledonary placenta or endometrial specimens (caruncle), if available, are more likely to yield a positive test result than fetal liver samples.
Preferred Fetal Diagnostic Specimens for Investigation of RVF Using Histopathology and Immunohistochemistry.

Previous research has shown that brain specimens from fetuses test positive by PCR and virus isolation, more often than the liver. 3,23 However, in the present study the heart was more frequently positive by IHC than brain specimens. In the brain, viral antigen was most often present in the meningeal blood vessels, occasionally in capillaries in the white or gray matter but never within neurons or glial cells. In the heart, viral antigen was often present in myocardial Purkinje fibers, cardiomyocytes, and vascular smooth muscle cells. It was previously reported that labeling was occasionally present in these cells in young lambs whereas in adult sheep viral antigen was only sporadically found in vascular smooth muscle cells and never in cardiomyocytes or myocardial Purkinje fibers using IHC. 18,19 We therefore recommend that heart samples should be collected in studies involving fetuses or very young lambs rather than brain samples, and when specimens from the brain are collected these must include meninges.
Remarkably high viremias of 108.33 RNA copies per gram in the liver using RT-qPCR, and 106.2 virus per gram in the placenta using the mouse lethal doses technique have been reported in fetuses, and in the present study viral antigen was present in capillaries and blood vessels as non-cell-associated virus or within endothelial cells. 3,10 A recent study also reported viral antigen, detected using IHC, in blood and endothelial cells in the brain or umbilical cord of 8 of 23 (35%) ovine fetuses recovered from experimentally infected ewes. 21 Moreover, in the present study, viral antigen appeared to have diffused into the capsule and connective tissue stroma of organs in some fetuses giving the impression that effusions in the thorax and abdomen might have contained extremely high viral loads. A study in rats demonstrated RVFV in the amniotic sac. 16 Viral RNA were also detected by RT-qPCR in the amniotic fluid of 15 of the 23 (65%) ovine fetuses recovered from experimentally infected ewes. 21 Therefore, amniotic fluid might contain high viral loads in some cases. Further support for this included diffusely present viral antigen in the epidermis of one fetus. However, further studies are needed to investigate if these assertions are accurate.
Conclusion
Although many species are susceptible to RVFV infection, sheep are often the most severely affected. This is particularly true for pregnant sheep. RVFV infection in pregnant ewes caused a variety of outcomes for both ewes and fetuses, and it may be impossible to rule out RVF even if sampling is adequate. Some ewes show no clinical signs, do not seroconvert, and do not have a detectable viremia. 3 Macroscopic lesions in most fetuses were also absent or nonspecific. As expected with wild-type RVFV infections, we observed no fetal malformations. Microscopically, the liver often had characteristic lesions but in a small percentage of cases the necrosis was mild and diagnostic features such as primary foci and inclusion bodies were absent.
RVFV antigen-positive cells in fetal tissues included hepatocytes, renal tubular epithelial cells, renal juxtaglomerular cells, renal extraglomerular mesangial cells, vascular smooth muscle cells, skeletal muscle cells, cardiomyocytes, myocardial Purkinje fibers, adrenocortical cells, vascular endothelial cells, macrophages, and occasionally neutrophils. However, while cotyledonary placenta or endometrial samples are often positive the fetal organs can be IHC-negative. Therefore, for routine histopathology and IHC, liver specimens should be examined together with specimens from at least the cotyledonary placenta with fetal spleen and kidney being useful additional samples. If available, specimens from the endometrium (caruncle) are also very useful. Additional testing should be performed in endemic areas even if histological lesions are absent and RVFV IHC is negative. The recommended samples for PCR are heart in addition to liver and brain since heart was positive by IHC more frequently than the brain in the present study. However, even when multiple samples are tested using multiple techniques and negative results are obtained, RVF should remain a differential diagnosis, especially when abortions continue to occur.
Supplemental Material
Combined_supplemental_materials-Odendaal_et_al - Ovine Fetal and Placental Lesions and Cellular Tropism in Natural Rift Valley Fever Virus Infections
Combined_supplemental_materials-Odendaal_et_al for Ovine Fetal and Placental Lesions and Cellular Tropism in Natural Rift Valley Fever Virus Infections by Lieza Odendaal, Sarah J. Clift, Geoffrey T. Fosgate and A. Sally Davis in Veterinary Pathology
Footnotes
Acknowledgements
We acknowledge the staff of the histopathology laboratory of the Department of Paraclinical Sciences, Section of Pathology, especially Rephima Phaswane and Naomi Timmerman, for their assistance, knowledge, input, and support. We thank colleagues at the National Department of Agriculture, Fisheries and Forestry, Directorate of Veterinary Services, for providing access to original diagnostic data and colleagues at IDEXX Laboratories (South Africa) for the specimens. We also thank Antoinette Odendaal for preparing all the panels for this study and Mal Rook Hoover, CMI, KSU, for her assistance. The authors are also grateful for the support of Professor Estelle Venter from the College of Public Health, Medical and Vet Sciences, James Cook University, Australia, and the Department of Veterinary Tropical Diseases, Faculty of Veterinary Science of the University of Pretoria.
Declaration of Conflicting Interest
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Agricultural Sector Education Training Authority of South Africa. Publication of this study was funded by the Department of Paraclinical Sciences of the Faculty of Veterinary Science, University of Pretoria, and the Department of Diagnostic Medicine/Pathobiology, College of Veterinary Medicine, Kansas State University.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
