Abstract
Diagnostic records of 30 primary and one metastatic follicular stem cell carcinomas in 30 dogs were reviewed. Neoplastic cells had a clear cytoplasm and formed lobules and nests surrounded by a basement membrane. Trichoepitheliomatous and apocrine differentiations were noted in 22 of 30 (73%) and 21 of 30 (70%) primary tumors, respectively. Glycogen was present in 20 of 20 (100%) tumors tested, suggesting tricholemmal differentiation. Antibodies against AE1/AE3 cytokeratin, vimentin, and melanA/MART1 stained 29 of 30 (97%), 29 of 30 (97%), and 12 of 27 (44%) primary tumors, respectively. Small amounts of melanin were identified in 14 primary tumors, either on the hematoxylin and eosin-stained section (
Keywords
Cutaneous tumors with ballooning degeneration of neoplastic cells are uncommon in man and animals. They include balloon cell nevus, 11 balloon cell melanoma, 1 , 28 clear cell basal cell carcinoma, 3 and clear cell acanthoma. 5 , 12 Immunohistochemistry and electron microscopy are often required to definitively categorize these tumors. The aim of this paper is to characterize histologically, immunohistochemically, and ultrastructurally a type of cutaneous carcinoma unique to the dog. This tumor was previously referred to as sebaceous carcinoma 39 in the veterinary literature. We propose this tumor to be named follicular stem cell carcinoma.
Materials and Methods
The database of IDEXX Veterinary Services, West Sacramento, was searched for the descriptive term “balloon” diagnosed from 1 January 1997 to 15 April 2001. All tumors with the term “balloon” in the microscopic description, diagnosis, or comments were reviewed, and those meeting the criteria described below were included in the study. Tissue samples for histology had been fixed in 10% neutral buffered formalin, processed by conventional methods, embedded in paraffin, sectioned at 5 µm, and were stained with hematoxylin and eosin (HE). In addition, slides were stained with Fontana-Masson silver method (
Immunohistochemistry was performed using a standard immunohistochemical method 9 based on tissue availability. It was performed on 30 primary and two metastatic tumors for cytokeratin (1 : 100 dilution cytokeratin cocktail AE1/AE3 mouse monoclonal antibodies; Biogenex, San Ramon, CA) and for vimentin (1 : 100 dilution mouse monoclonal antibody, clone Vim3B4; Dako, Glostrup, Denmark). AE1/AE3 anti-cytokeratin antibodies recognize human cytokeratins 1, 5, 8, 10, 14, 16, and 19. 40 Seven primary and three metastatic tumors were labeled for CD18 (1 : 200 dilution rabbit polyclonal antibody; Dako) and lysozyme (ready-to-use solution, mouse monoclonal antibody; Davis, University of California, CA), using the same technique. Twenty-seven primary tumors and one metastatic tumor also were stained for Melan A/MART-1 (1 : 50 dilution, mouse monoclonal antibody, clone A103; Novoacastra, Newton upon Tyne, UK), and 14 tumors were stained for S100a (1 : 1,000 dilution, rabbit polyclonal antibody; Dako), using techniques described elsewhere. 15 The “a” designation in the S100a antibody denotes the clone and not the “A” antigen of S100. This antibody is reported by the manufacturer to label determinants that are common to the alpha and beta chains of S100a (α/β) and S100b (β/β). Ten tumors were stained for α-smooth muscle actin (ready-to-use solution, mouse monoclonal antibody; Biogenex, San Ramon, CA). The antibody reaction products were observed with 3.3′-diaminobenzidine tetrachloride and counterstained with Mayer's hematoxylin for cytokeratin, vimentin, CD18, lysozyme, and α-smooth muscle-actin–labeled sections. The antibody reaction products were observed with streptavidin conjugated to alkaline phosphatase, and the color reaction was accomplished using the Histomark red kit (Kirkegaard & Perry Laboratories, Gaithersburg, MD) for slides stained with Melan A/MART-1 and S100a. The specificity of the immunologic reactions was verified by labeling control skin sections. A section of each tumor was also processed under identical conditions replacing specific antiserum with phosphate-buffered saline (PBS) to detect nonspecific labeling.
The distribution of cytokeratin labeling of the normal epidermis and of the hair follicles and their adnexae was evaluated in noninflamed skin adjacent to the tumors. It was variable among hair follicles and individuals, an observation that has also been made in cat. 27 The structures that were labeled most consistently with cytokeratin were the stratum corneum, the stratum basale, the basal layer of sebaceous glands, the luminal cells of the epithelium of apocrine sweat gland ducts, and the medulla of the hair shafts (Table 1).
Labeling of the epidermis and hair adnexae with AE1/AE3 anticytokeratin antibodies in 10 dogs.
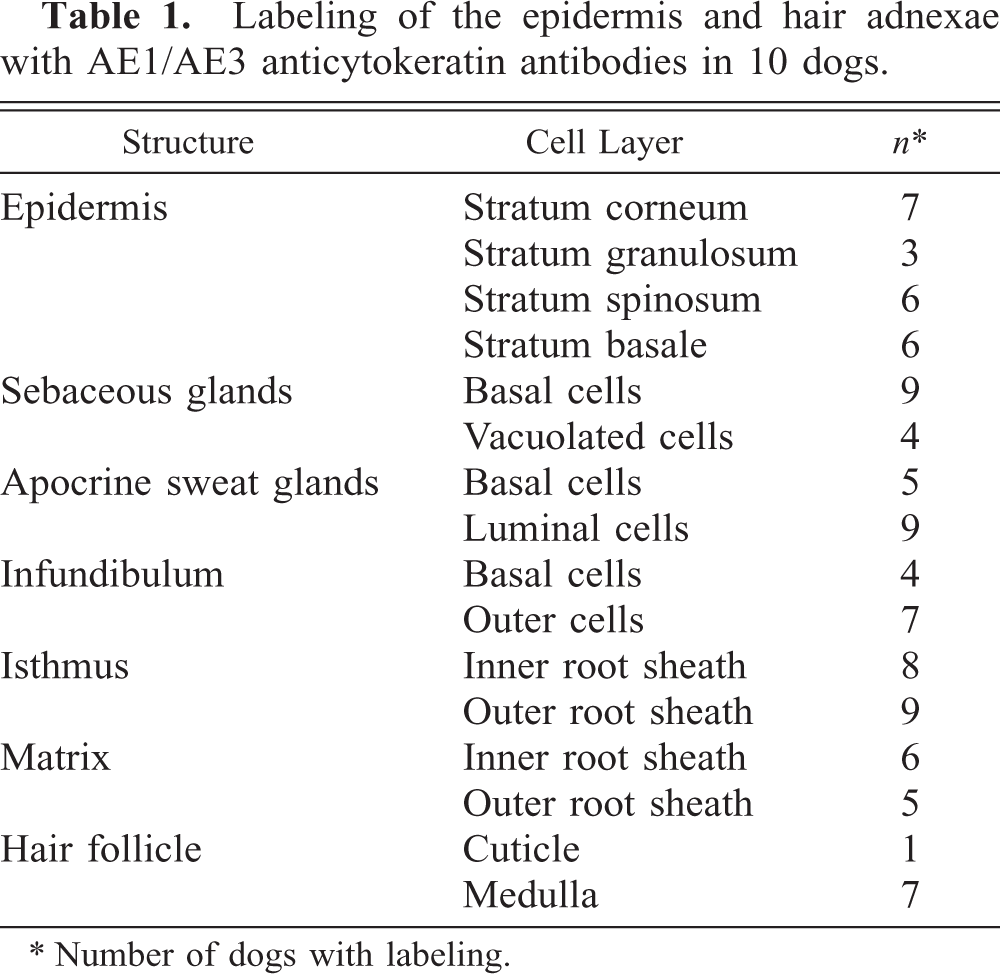

∗ Number of dogs with labeling.
Formalin-fixed material from four tumors (Nos. 12, 21, 24, and 30) was used for electron microscopy. Tissue samples were postfixed in 2% glutaraldehyde fixative, stored in 0.085 M sodium cacodylate (pH 7.2), and postfixed in 1% OsO4 buffered with 0.1 Zetterqvist buffer (pH 7.2). After rinsing three times for 10 minutes each with Zetterqvist wash, tissue specimens were dehydrated, stained for 2 hours with 0.5% uranyl acetate in 70% ethanol at room temperature, dehydrated in a graded series of acetone, and embedded in Spurr's resin. Silver to silver-gold ultrathin sections were stained with 3% uranyl acetate for 15 minutes followed by 10 minutes in lead citrate. These were then examined with a Zeiss 10A electron microscope.
A follow-up questionnaire was sent to the referring veterinarians. The presence and date of onset of any local recurrence, the occurrence and date of detection of any metastases, the date of last follow-up, the date the patient died, and the presence of other conditions, were evaluated. Necropsies were not performed.
Results
Signalment
A total of 30 primary follicular stem cell carcinomas were identified in 29 dogs (3–13 years; mean = 7.3 years), including one dog with two tumors. Three metastatic tumors were examined: one metastatic follicular stem cell carcinoma was identified in one dog that had been diagnosed by another laboratory with a “sebaceous carcinoma” approximately 2 years earlier; one follicular stem cell carcinoma was associated with a metastasis to the draining lymph node at the time of surgery; and finally, one follicular stem cell carcinoma from this series metastasized to the draining lymph node, and the metastasis was submitted 953 days after the primary tumor was excised.
Females (
Histology
The histologic appearance of the tumors is illustrated in Figs. 1–5. With the exception of the metastatic tumor for which the primary neoplasm was not available for review, the primary tumors were located in the dermis (
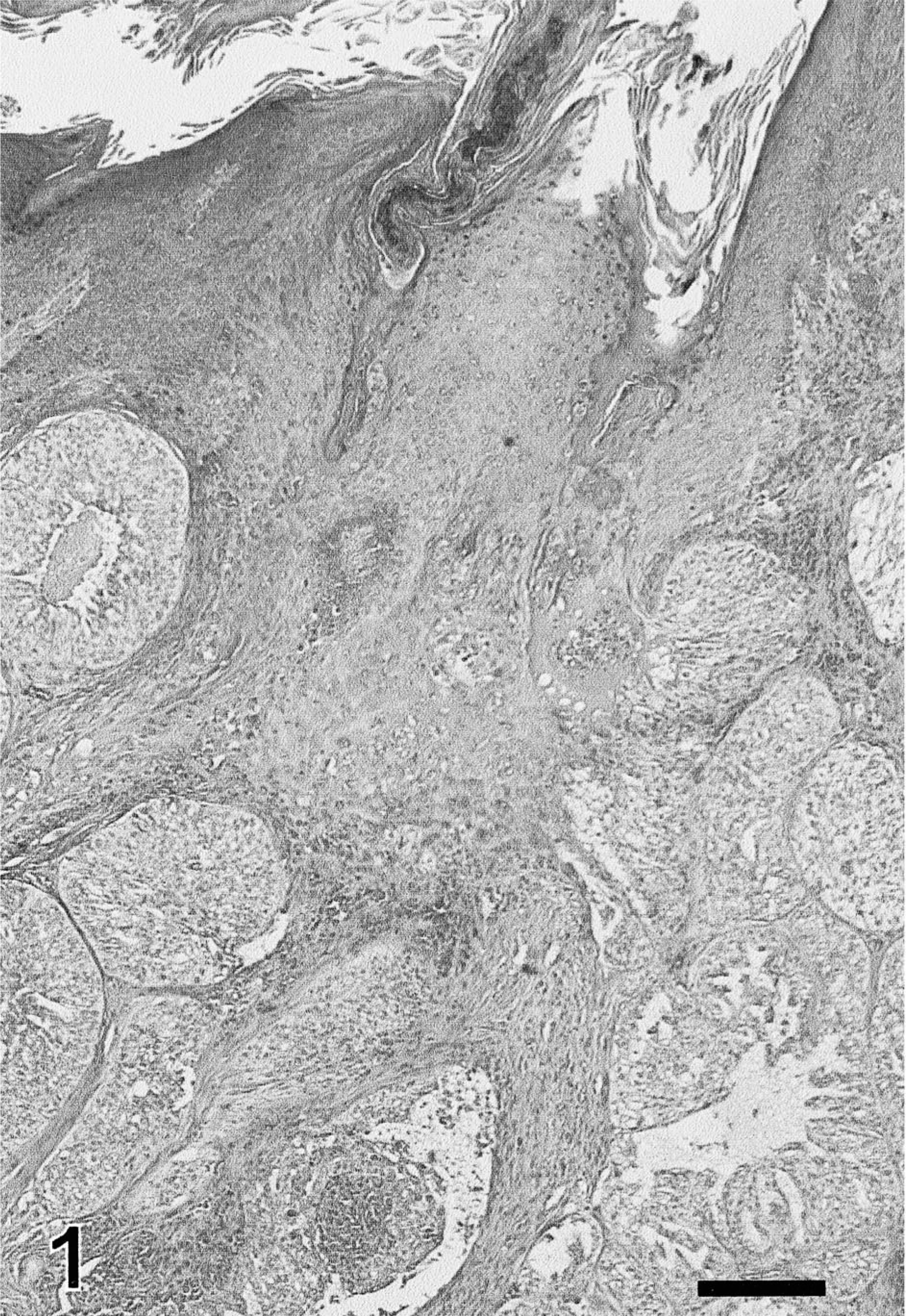
Follicular stem cell carcinoma; dog. The neoplasm forms large lobules connected with the overlying epidermis. Neoplastic cells have a pale cytoplasm. HE. Bar = 120 µm.
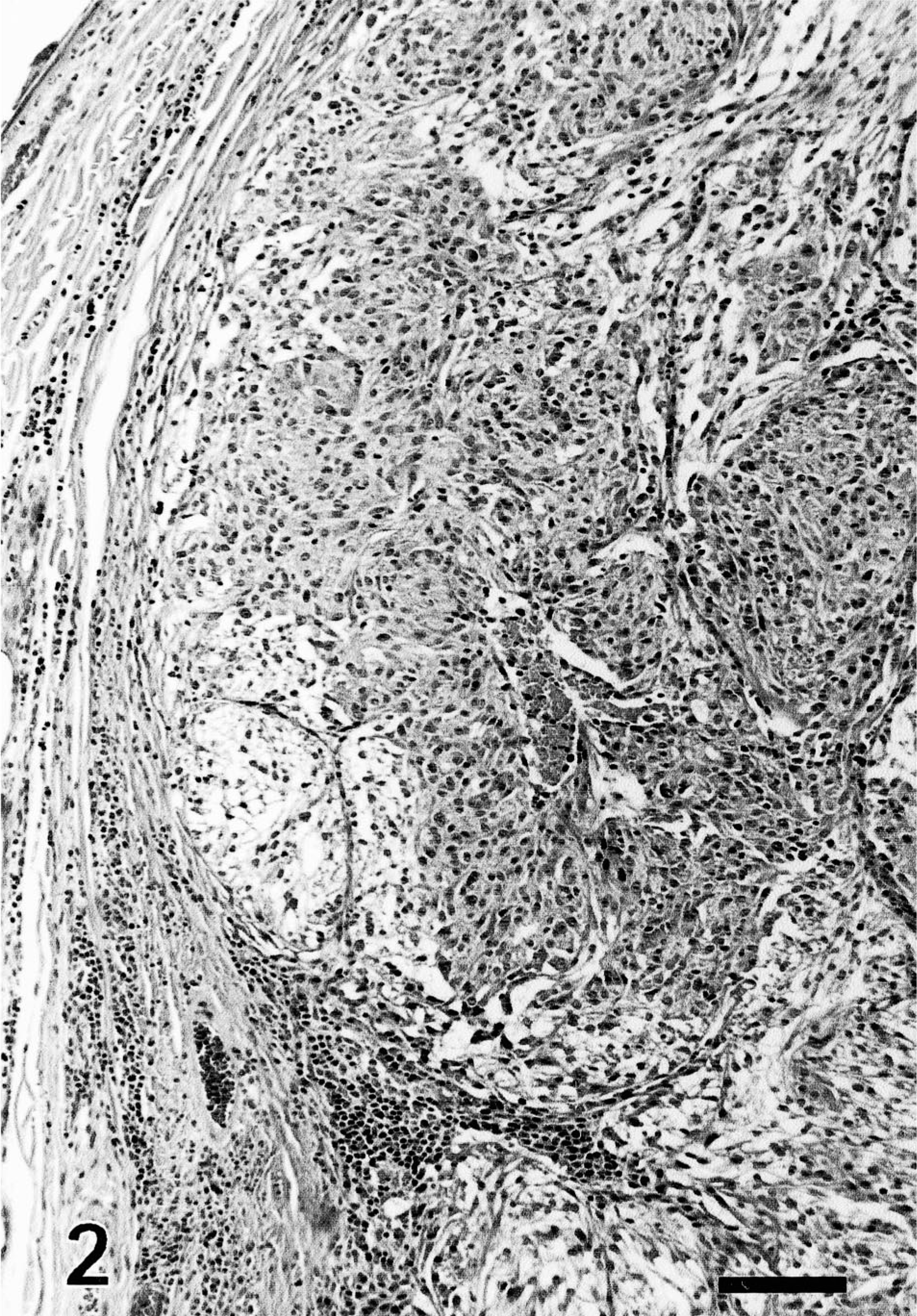
Follicular stem cell carcinoma; dog. Neoplastic cells form large nest. They have a variable amount of pale cytoplasm. Swelling of the cytoplasm differs among neoplastic cells and is more prominent at the periphery of the nests. HE. Bar = 120 µm.
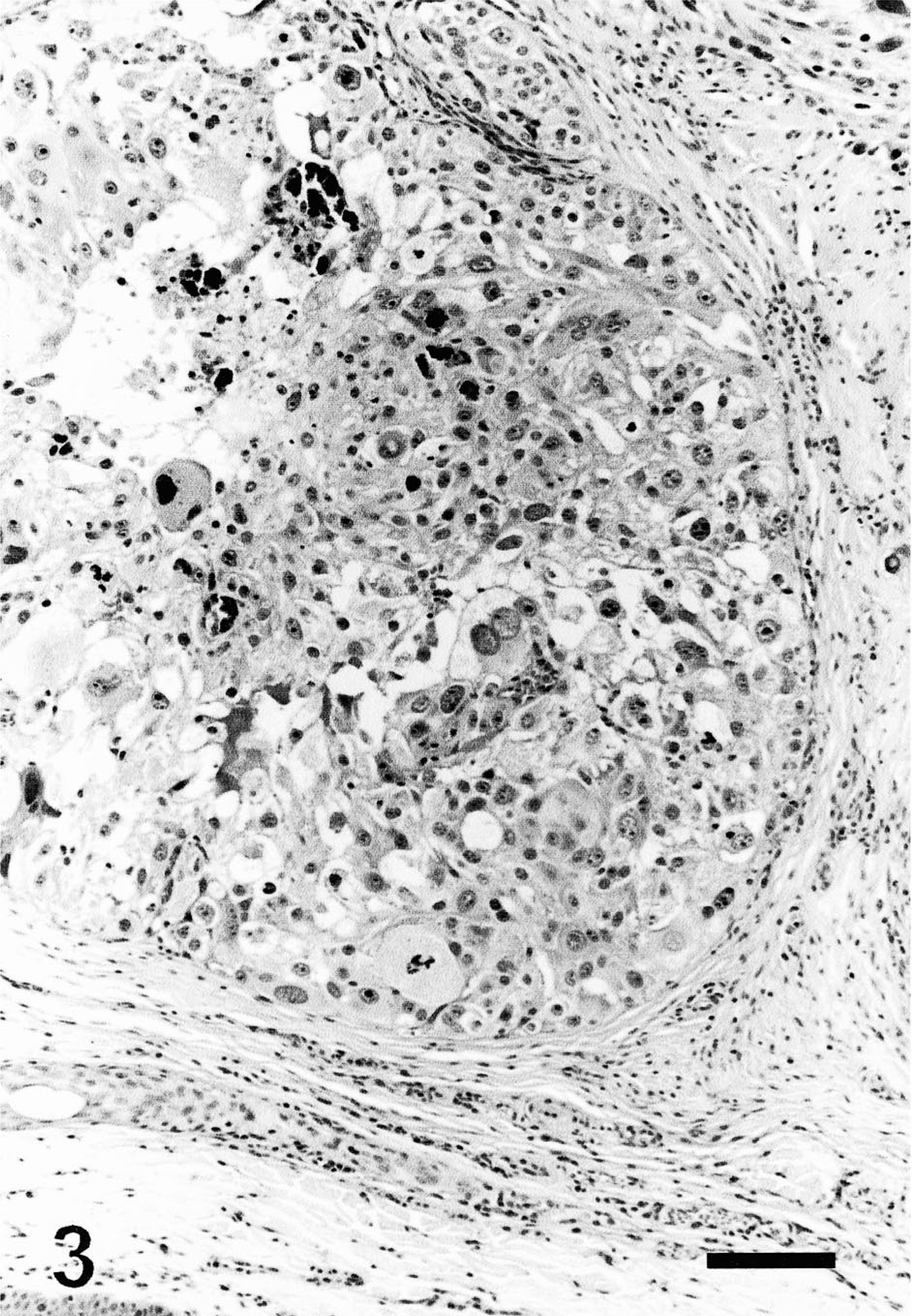
Follicular stem cell carcinoma; dog. Neoplastic cells form a large nest. They have a large amount of pale cytoplasm and some are multinucleated. Cellular and nuclear atypia, in this case, are prominent. The center of the nest is necrotic. HE. Bar = 60 µm.
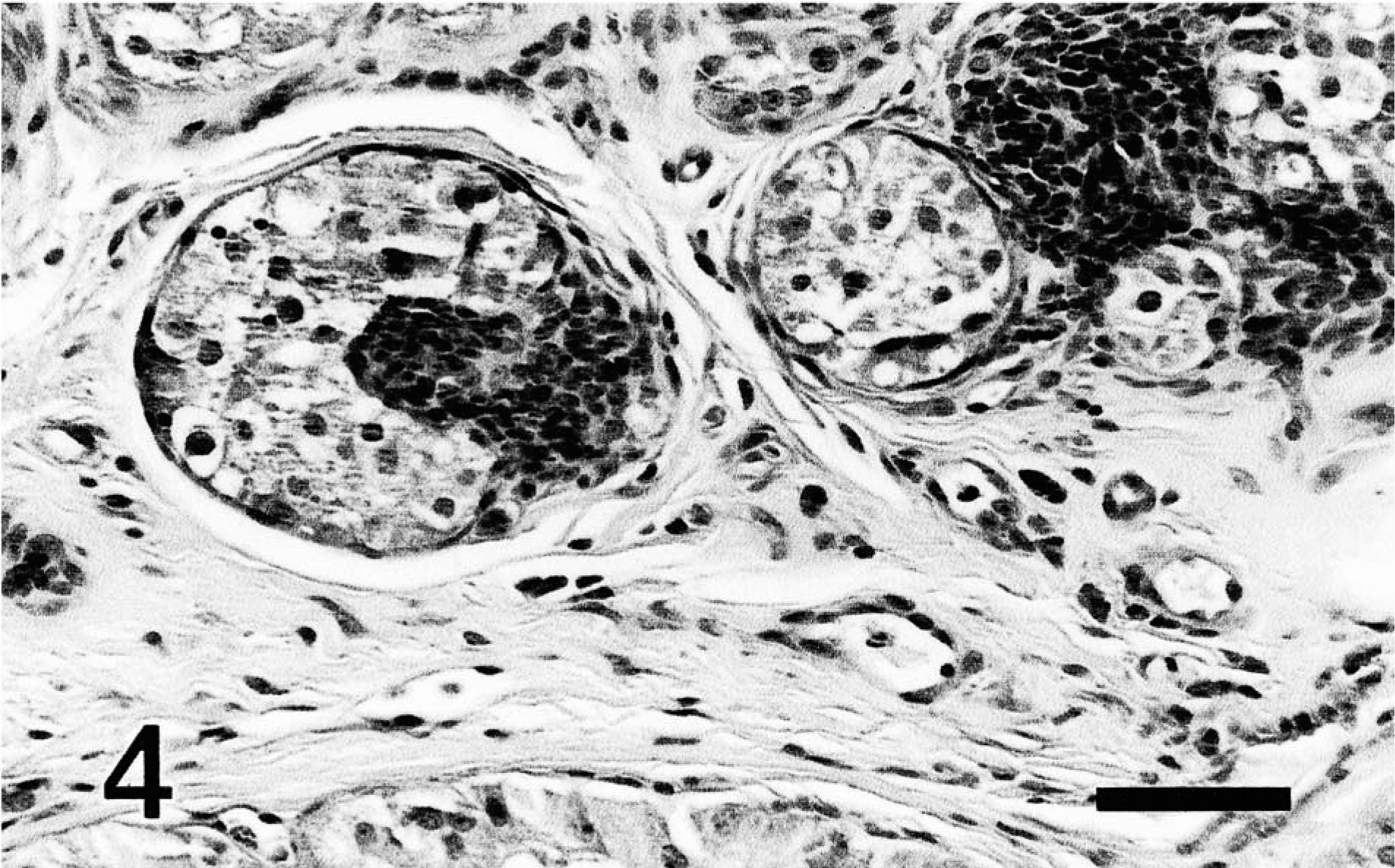
Follicular stem cell carcinoma; dog. The stroma of the tumor contains an aggregate of plump cells mimicking a follicular papilla (trichoepitheliomatous differentiation). HE. Bar = 60 µm.
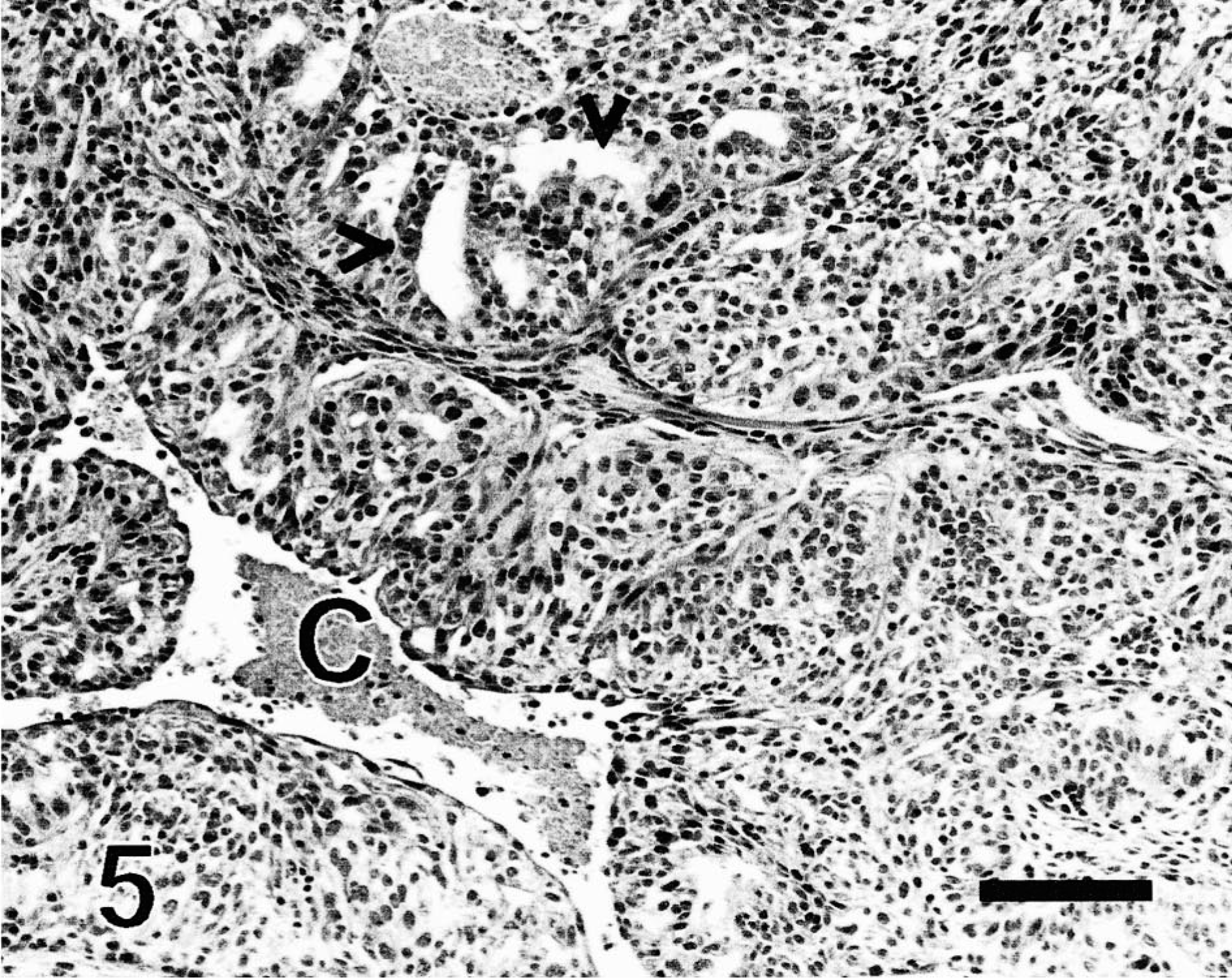
Follicular stem cell carcinoma; dog. Neoplastic cells form tubules (apocrine differentiation; arrowheads). Necrosis at the center of this lobule resulted in the formation of a large cleft (C). The stroma contains a few bundles of spindle cells reminiscent of follicular papillae. HE. Bar = 120 µm
The tumors measured 6–28 mm in diameter (mean = 14 mm) and were well delineated and densely cellular. They were composed of variably sized nests and solid areas separated by a small to moderate amount of fibrovascular stroma (Figs. 2, 3). Neoplastic cells often formed streams within the nests.
Inconspicuous tubules lined by an epithelium of 1–3 cells thickness were present in 21 of 30 primary tumors and two of three metastatic tumors (Fig. 5). Cells lining some of these tubules occasionally had apical blebs. The formation of tubules was interpreted as an evidence of apocrine differentiation.
Nuclear palisading was present at the periphery of a few neoplastic nests in all tumors. Neoplastic cells were medium-sized to large, polygonal to fusiform, with indistinct cell borders and a moderate to large amount of pale eosinophilic cytoplasm. Melanin was identified in the cytoplasm of a small to moderate number of neoplastic cells in a few neoplastic lobules in 14 tumors, either on the HE-stained section (
The nucleus was medium-sized to large, central, oval to slightly elongated, hypochromatic, with a lacy chromatin pattern and a small central basophilic nu-cleolus. Anisokaryosis and anisocytosis were prominent in five tumors. Intranuclear cytoplasmic inclusions were present in these five tumors. Multinucleated neoplastic cells with up to 10 nuclei were identified in 22 tumors. Mitotic rate was 0–20 mitoses per 10 high-power fields (mean = 3.3).
A variable number of small structures comprising fusiform cells resembling follicular papillae populated the stroma in 22 of 30 primary tumors (Fig. 4) and in one of three metastatic tumors. The formation of follicular papillae structures was interpreted as an evidence of trichoepitheliomatous differentiation. 23 Clefting and cavitation at the center of the largest lobules were present in 24 primary tumors. Mild multifocal chondromatous metaplasia was noted in the stroma of two primary tumors and one metastatic tumor.
Lymphatic invasion and lymphatic emboli were present in five tumors. Necrosis was identified in all but two tumors and generally affected the center of the largest nests. Mineralization was present at the center of the necrotic lobules in 15 tumors and, generally, was associated with a moderate granulomatous inflammation. Lymphoplasmacytic inflammation was present in 26 tumors and usually consisted of lymphoid aggregates at the periphery of the neoplasm.
Immunocytochemistry and immunohistochemistry
PAS staining revealed a moderate to large number of purple granules in the cytoplasm of a small to moderate number of neoplastic cells in all tumors tested (
Immunohistochemical staining results are summarized in Table 2. Labeling for cytokeratin was present in 1–100% of neoplastic cells in 30 of 31 tumors. It was cytoplasmic, intense, and enhanced the angular contours of neoplastic cells (Fig. 6). Labeling was present in scattered individual cells, portions of neoplastic nests and lobules, or entire neoplastic lobules. Cells forming tubules consistently expressed cytokeratin (Fig. 7).
Major clinical, histologic, and immunohistochemical features of follicular stem cell carcinoma in 30 dogs.
∗ P = Primary; M = metastatic.
† Years.
‡ F = female; M = male.
§ Formation of tubules reminiscent of apocrine glands.
∥ Presence of bundles of stromal cells reminiscent of follicular papillae.
# = Melanin was identified on the HE section; +∗ = melanin could be identified only after staining with the Fontana-Masson silver method; - - = melanin was not identified on the slide stained with the Fontana-Masson silver method; - = melanin was not identified on the HE section, and unavailability of tissues prevented the use of the Fontana-Masson silver staining method.
¶ - = <1% cells were positive; - - = 0–1% cells were positive; + = 1–10% cells were positive; ++ = 11–50% cells were positive; +++ = >50% cells were positive; ND = not determined.
∗∗ + = presence; - = absence.
†† NS = not specified.
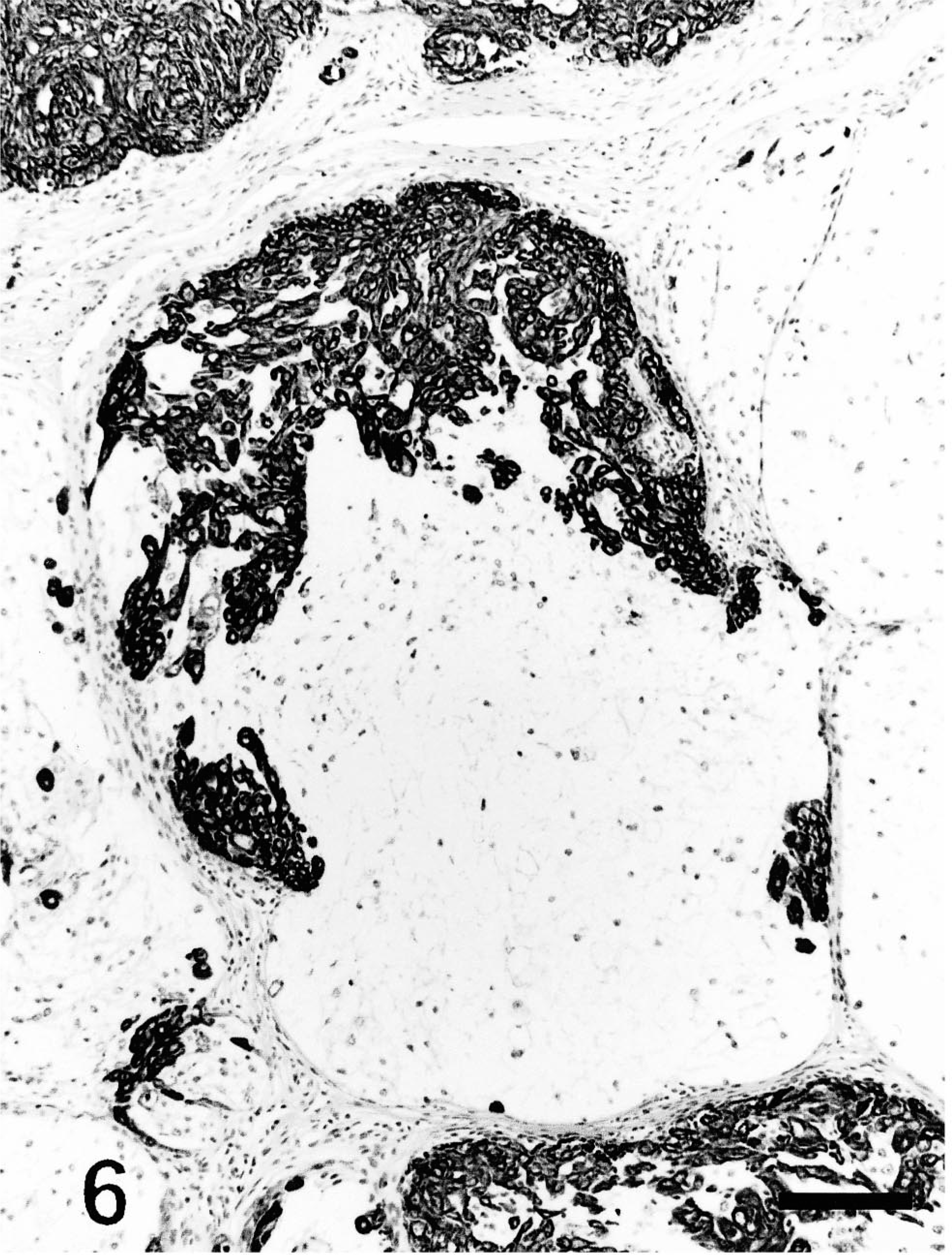
Follicular stem cell carcinoma; dog. Approximately 30% of neoplastic cells in this lobule react intensely with cytokeratin. Immunohistochemical labeling for cytokeratin accentuates the angular contours of neoplastic cells. Cytokeratin, avidin–biotin–peroxidase complex. Mayer's hematoxylin counterstain. Bar = 120 µm.
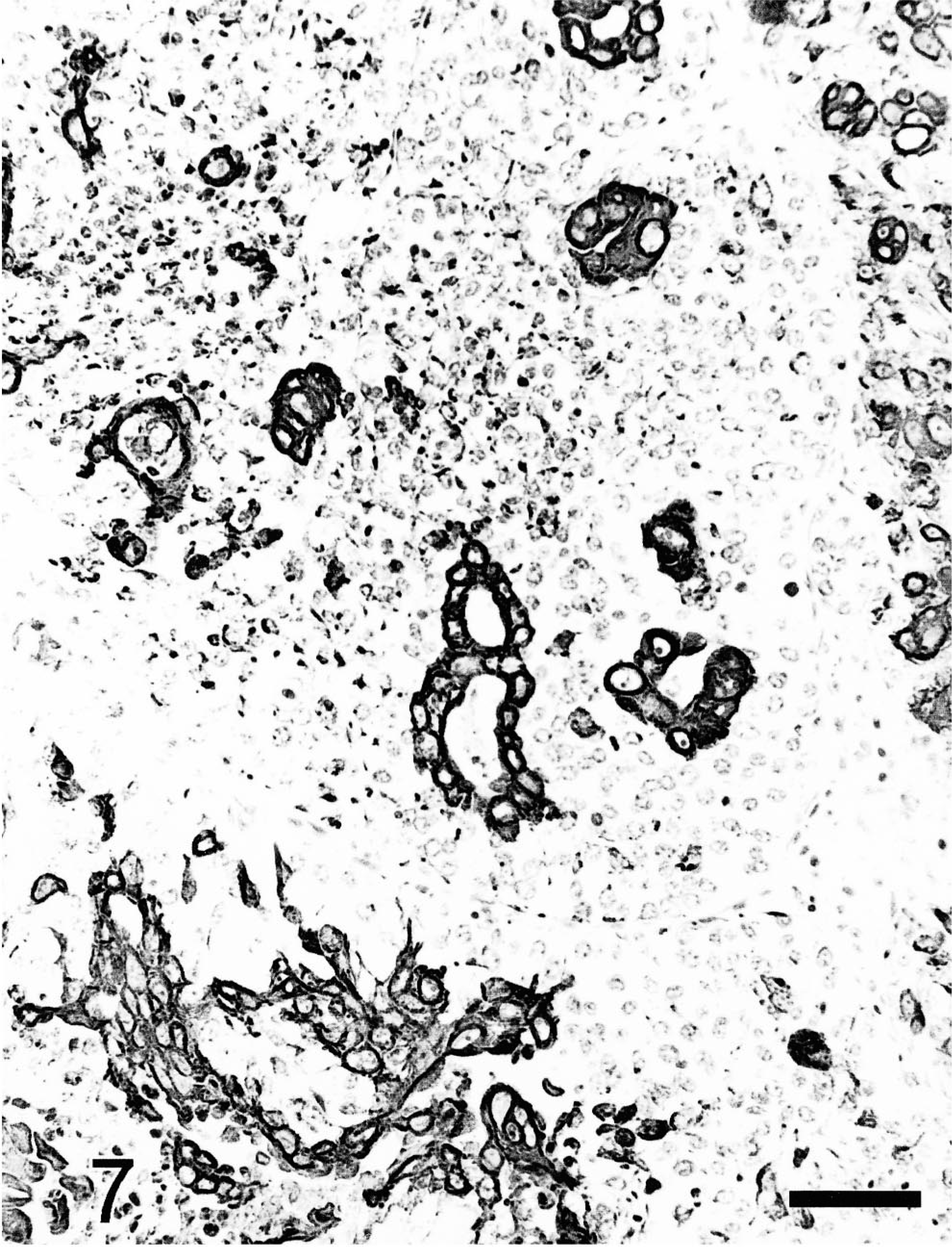
Follicular stem cell carcinoma; dog. Neoplastic cells which react with cytokeratin occasionally form tubules. Cytokeratin, avidin–biotin–peroxidase complex. Mayer's hematoxylin counterstain. Bar = 60 µm.
Labeling for vimentin in neoplastic cells was present in 5–100% of neoplastic cells in 30 of 31 tumors. It was cytoplasmic and moderate in intensity. Numerous neoplastic cells coexpressed vimentin and cytokeratin (Fig. 8a, b). Many neoplastic cells in most neoplasms did not stain with either of these markers.
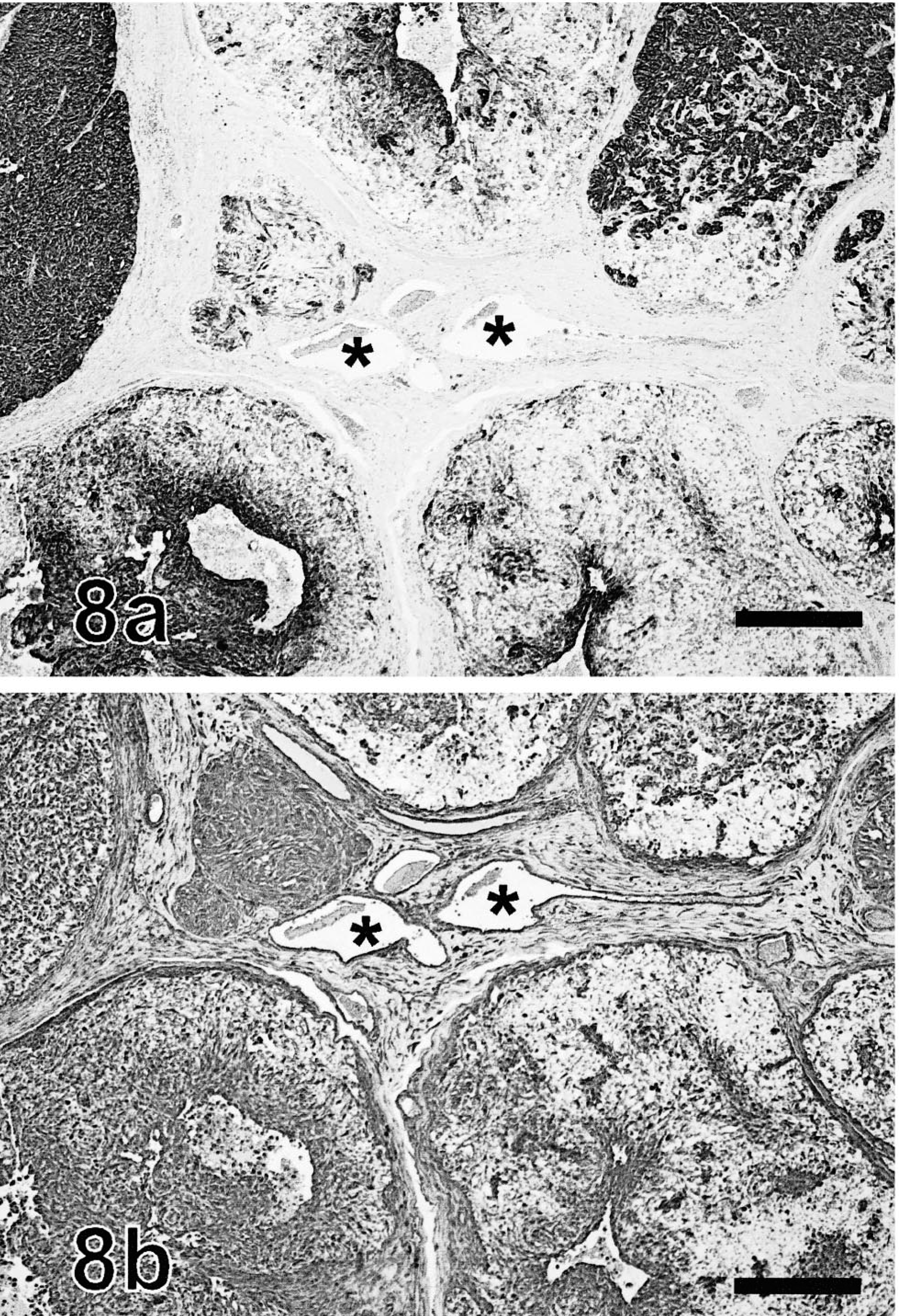
Follicular stem cell carcinoma, sequential immunolabeled sections; dog. Veins (∗) located at the center of these figures are used as position indicators. Fig. 8a. Some neoplastic lobules label diffusely for cytokeratin, whereas some others are partially unlabelled. Cytokeratin, avidin–biotin–peroxidase complex. Mayer's hematoxylin counterstain. Fig. 8b. Most neoplastic cells react moderately for vimentin, although some neoplastic lobules are partially negative. Coexpression of cytokeratin and vimentin is widespread. Mayer's hematoxylin counterstain. Bar = 200 µm.
Strong labeling for vimentin was observed in numerous stellate cells with long and delicate dendritic projections interspersed among neoplastic cells. These cells were present in all tumors, including the three metastatic tumors. These vimentin-labeled stellate cells were best visualized in the areas where neoplastic cells did not stain for vimentin (Fig. 9). Most of these interstitial cells labeled strongly with CD18 and lysozyme in 10 primary and three metastatic tumors. They were thus interpreted as dendritic cells.
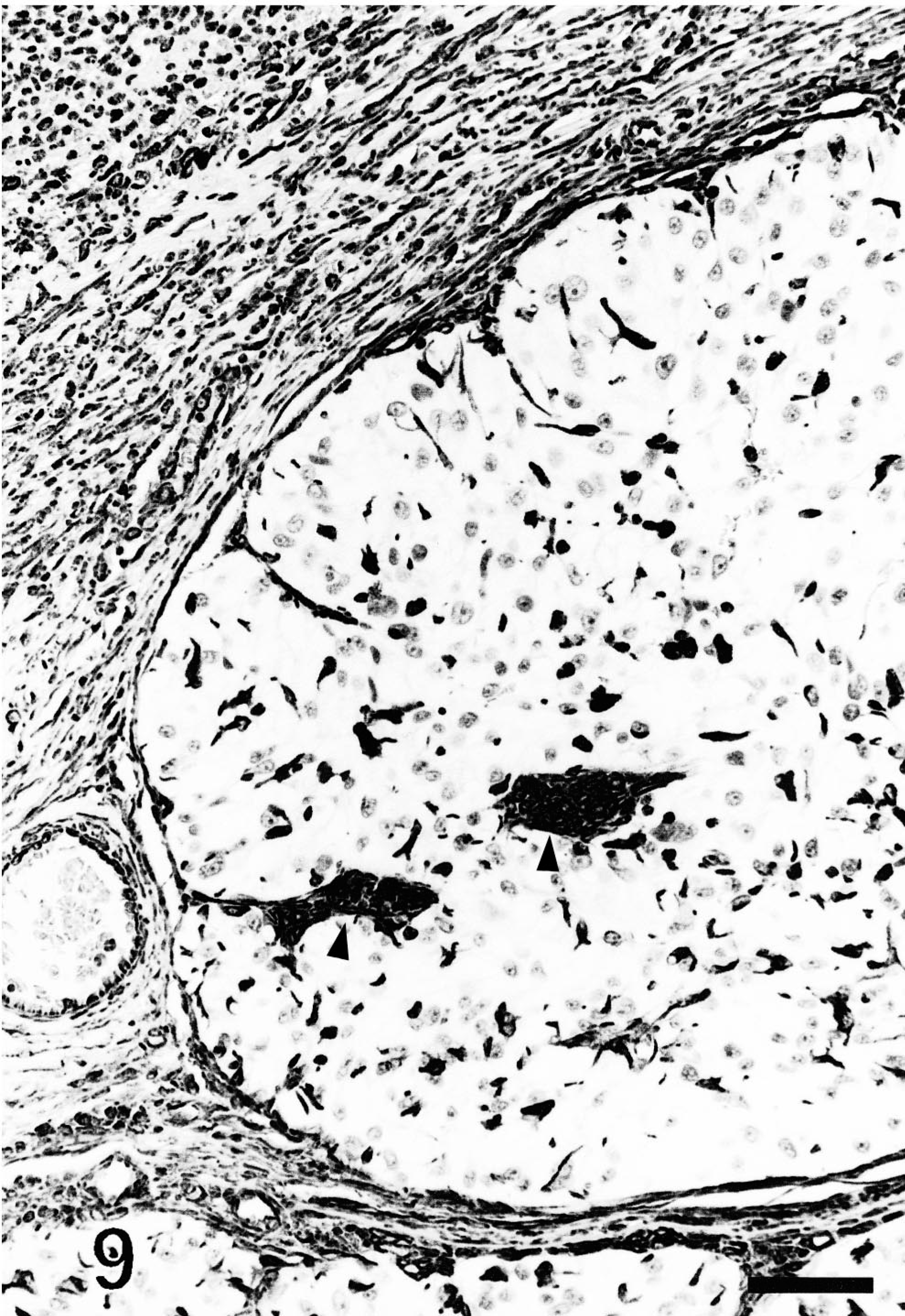
Follicular stem cell carcinoma; dog. A delicate network of dendritic cells populates a neoplastic lobule. Two bundles of spindle cells reminiscent of follicular papillae are present (arrowheads). Vimentin, avidin–biotin–peroxidase complex. Mayer's hematoxylin counterstain. Bar = 60 µm
Labeling for Melan A/MART-1 was strong, coarsely granular, and cytoplasmic. It was present in neoplastic cells in 12 of 27 primary tumors and in one metastatic tumor. It was detected in less than 1% of neoplastic cells in eight of 13 positive tumors. In the five other positive tumors, the staining was strong and was present in 5–100% of neoplastic cells. The tumor with 100% of cells positive for Melan A/MART-1 was heavily pigmented with melanin.
Neoplastic cells did not stain for S100a. A small population of dendritic cells scattered throughout neoplastic nests in five of 13 tumors were tested positive for S100a. The stain was cytoplasmic, strong, and coarsely granular. These cells were interpreted as melanocytes. Neoplastic cells did not express α-smooth muscle actin.
Electron microscopy
Neoplastic lobules were larger and were surrounded by an unevenly thick basement membrane (Fig. 10). Intercellular lumina were present in three tumors (Figs. 11, 12), indicating apocrine differentiation. Cells lining these lumina generally had microvilli or thick apical blebs (Fig. 11). Intracellular ducts with numerous microvilli were present in the cytoplasm of a few cells in one tumor (Fig. 13), indicating apocrine differentiation. Many neoplastic cells in this tumor were surrounded by numerous microvilli and some contained mucopolysaccharide granules (Figs. 11, 13). The cytoplasm of some neoplastic cells was markedly expanded by large non–membrane-bound areas made of a pale electron-dense material interpreted as proteinaceous fluid (Figs. 12, 13). Neoplastic cells had a moderate number of hemidesmosomes and a few, often extremely long desmosomes (Figs. 11, 14). Tight junctions were occasionally identified around tubules. Neoplastic cells had numerous large and swollen, round to elongated mitochondria with tubular cristae. They had a moderate amount of rough endoplasmic reticulum, glycogen granules, and occasionally a Golgi apparatus. They contained variable numbers of round to oval granules resembling neuroendocrine granules. These granules were most prominent and larger in cells located at the periphery of the lobules (Fig. 10). They measured 90–400 nm and consisted of electron-dense granular material surrounded by an electron-lucent halo and a cell membrane (Fig. 14). They resulted from the coalescence of smaller granules located at the periphery of the nucleus. Some cells contained closely entangled bundles of microfilaments, which generally were located at the vicinity of the nucleus. Nuclei tended to be central, round to oval, with peripherally located small clumps of heterochromatin.
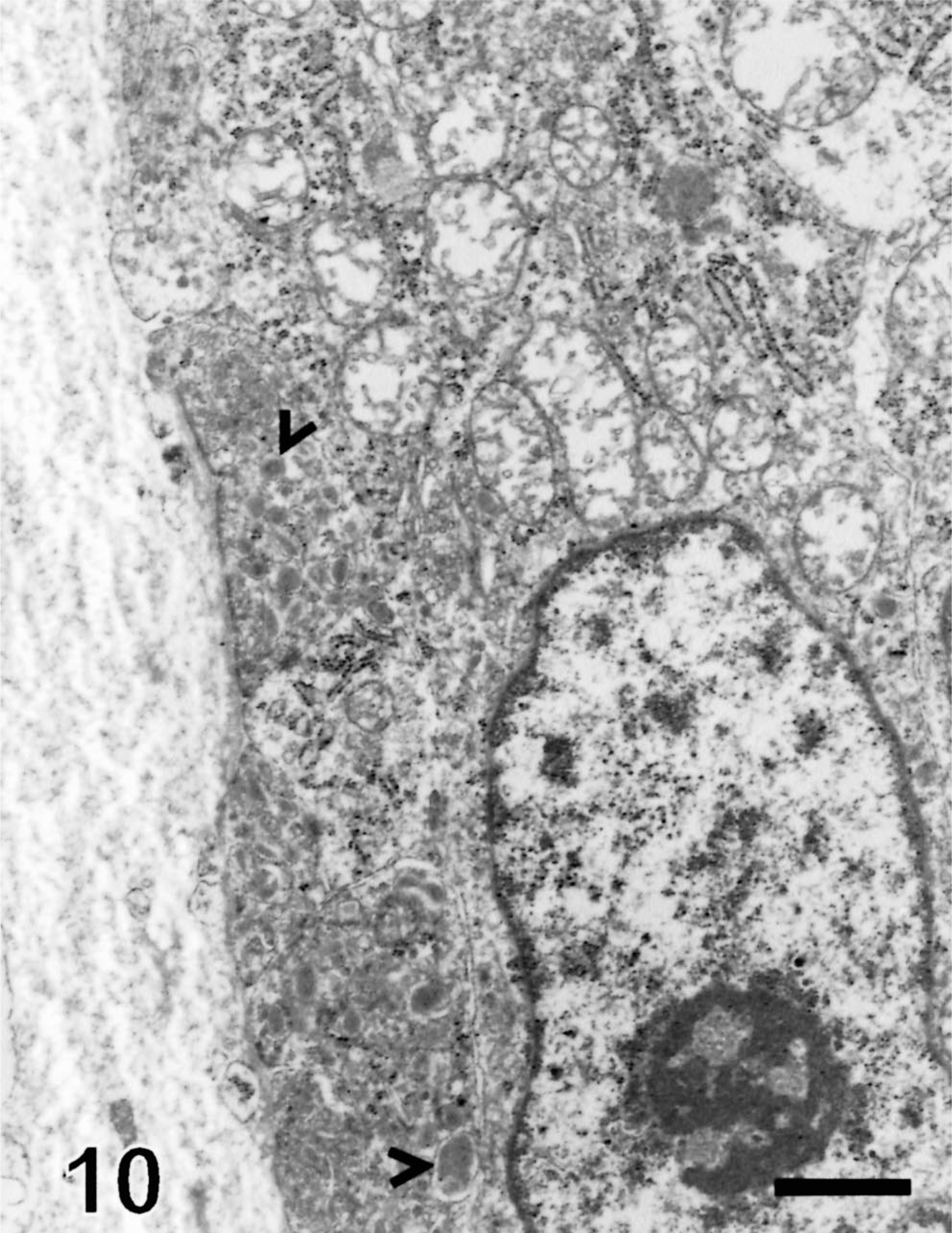
Electron micrograph, follicular stem cell carcinoma; dog. The neoplastic lobule is surrounded by an unevenly thick basement membrane. Neoplastic cells contain numerous neuroendocrine-like granules (arrowheads), which are more numerous at the vicinity of the basement membrane. Neoplastic cells contain numerous swollen mitochondria, aggregates of ribosomes, rough endoplasmic reticulum, and a few glycogen granules. Uranyl acetate and lead citrate. Bar = 2.1 µm.

Electron micrograph, follicular stem cell carcinoma; dog. Neoplastic cells form an intercellular lumina (L) filled with a mucoid substance and are surrounded by a cytoplasmic membrane, which forms blebs. One cell contains numerous mucuslike granules (arrowheads). Cell junctions include tight junctions and long desmosomes. Uranyl acetate and lead citrate. Bar = 2.1 µm.
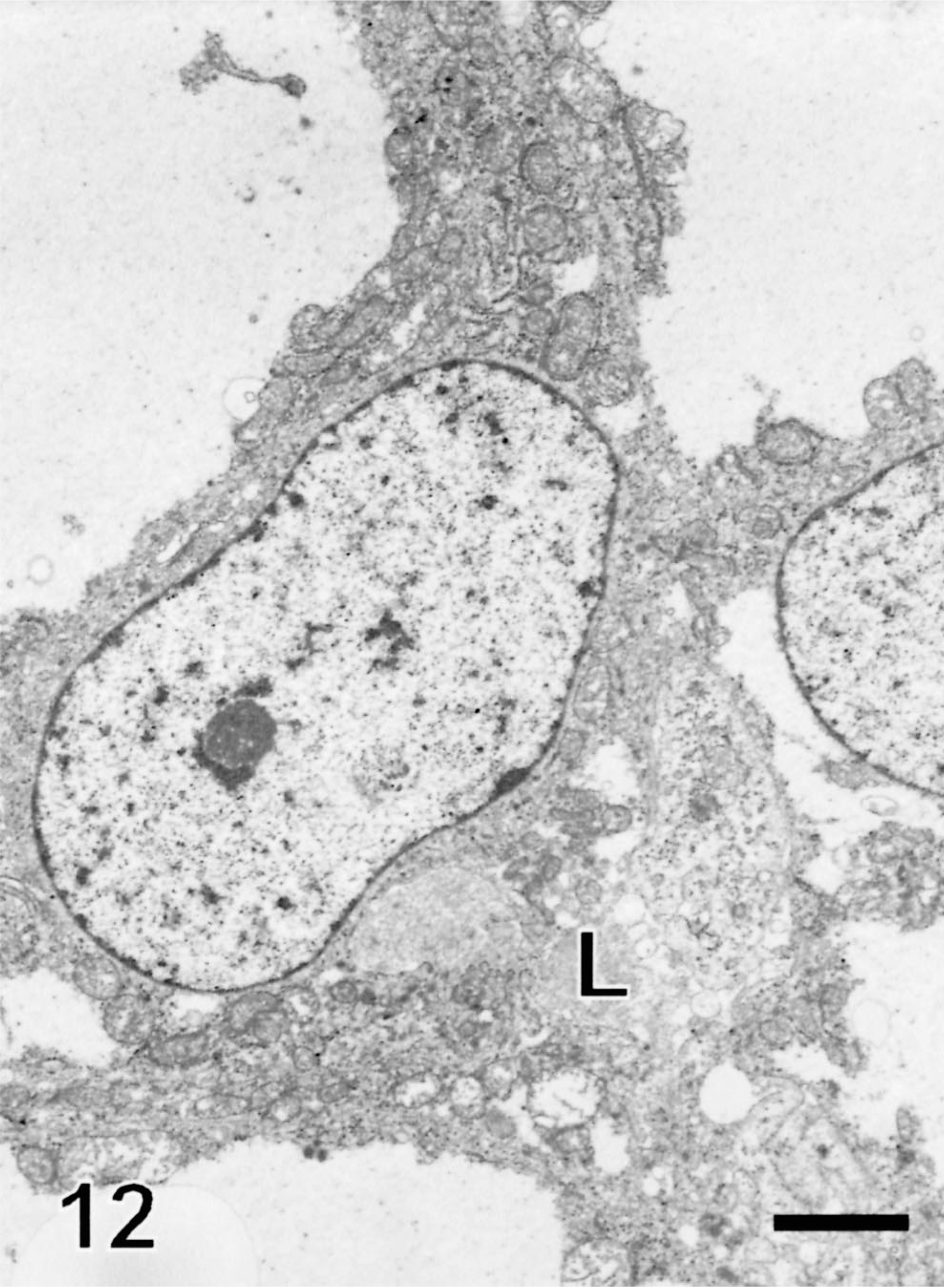
Electron micrograph, follicular stem cell carcinoma; dog. The cytoplasm of neoplastic cells is markedly expanded by non–membrane-bound vacuoles filled with a proteinaceous material. Neoplastic cells form an ill-defined intercellular lumina (L), which contains mucus material. Uranyl acetate and lead citrate. Bar = 5.2 µm.
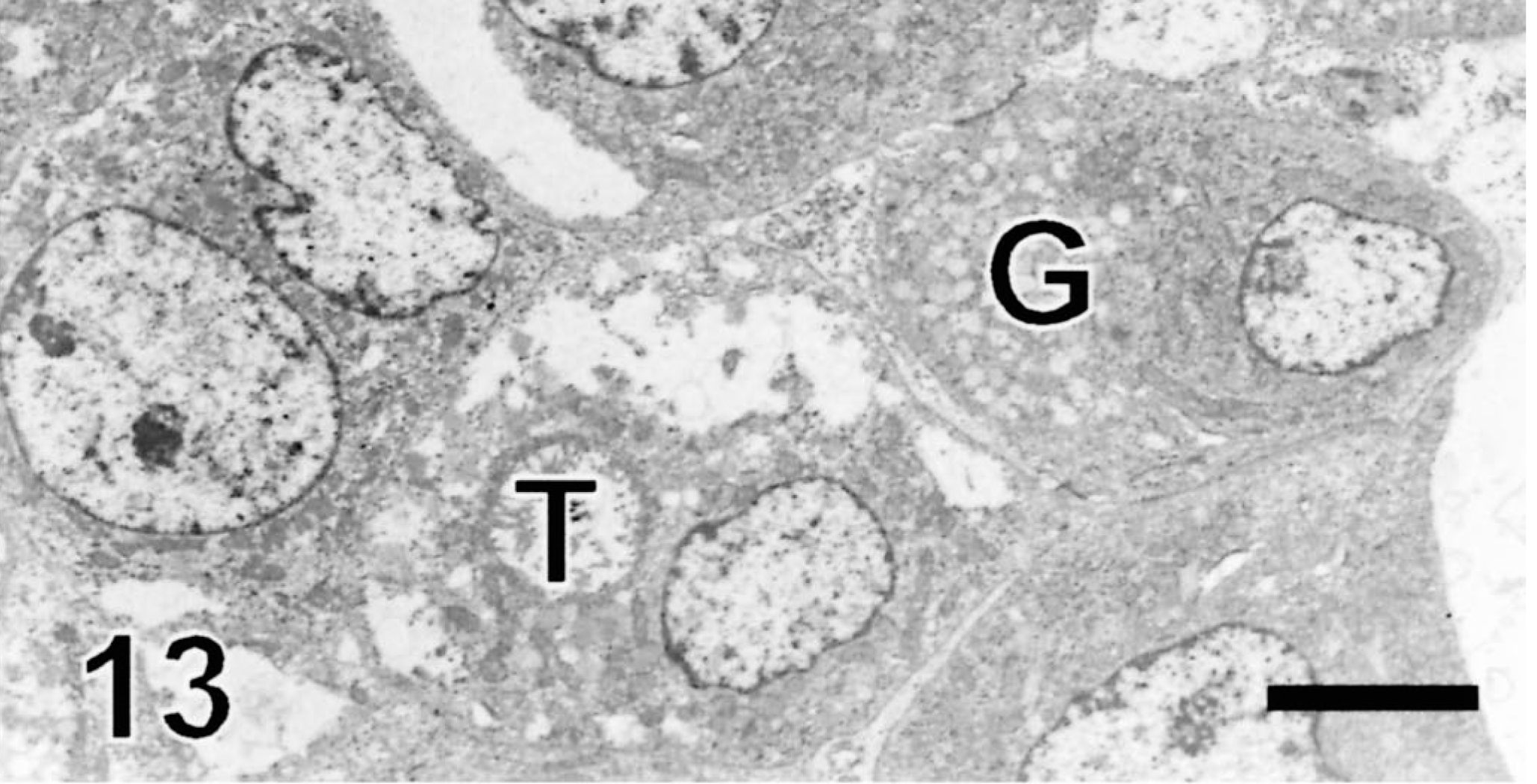
Electron micrograph, follicular stem cell carcinoma; dog. One neoplastic cell contains an intracellular tubule (T) lined by microvilli and shows moderate cytoplasmic swelling. One neoplastic cell contains mucopolysaccharide granules (G). Uranyl acetate and lead citrate. Bar = 9.75 µm.
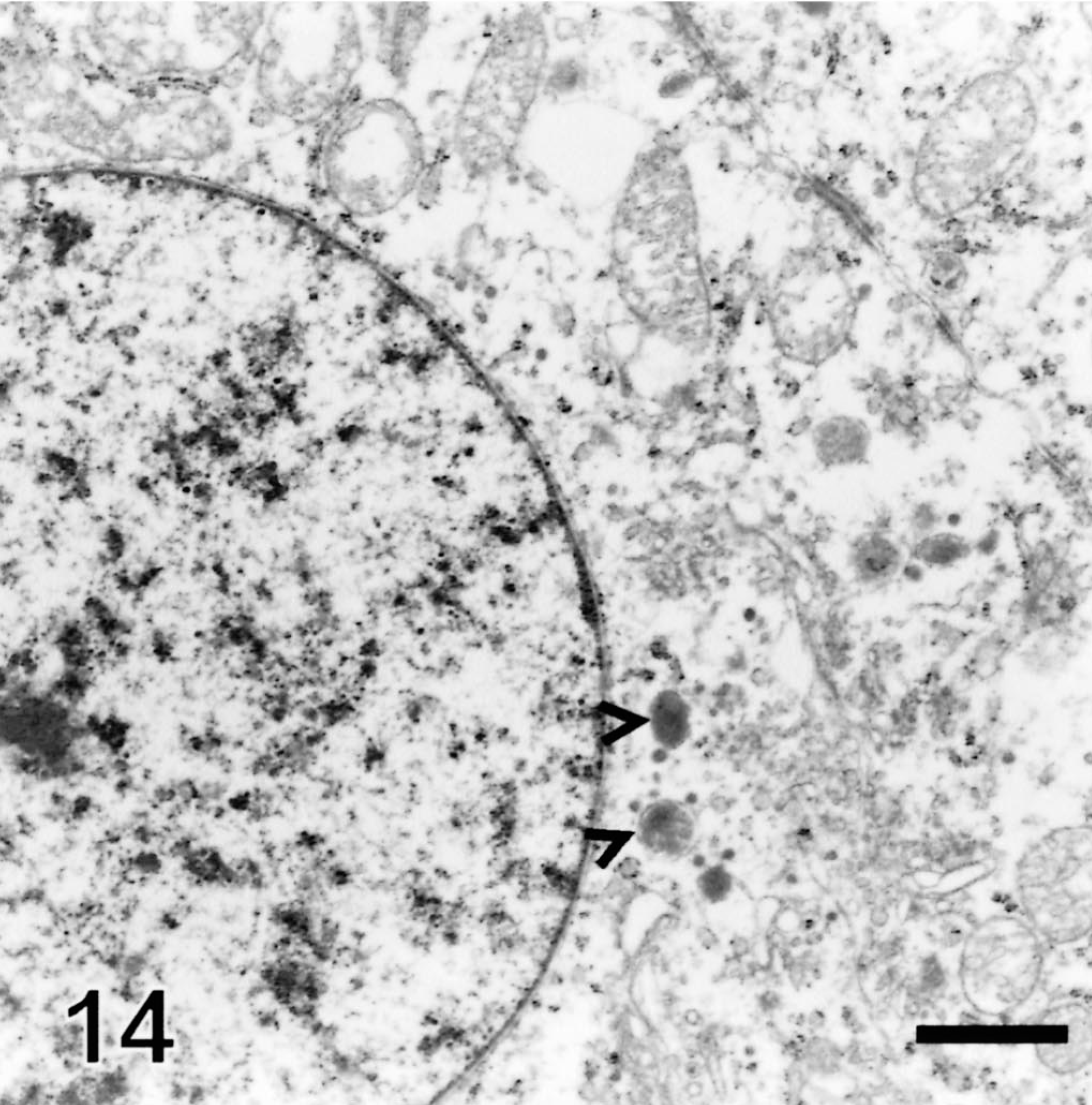
Electron micrograph, follicular stem cell carcinoma; dog. Neuroendocrine-like granules are formed at the vicinity of nucleus as the result of fusion of smaller granules (arrowheads). Mitochondria have tubular cristae and are swollen. Long desmosomes are present. Uranyl acetate and lead citrate. Bar = 1.76 µm
Dendritic cells with clear cytoplasm were present in neoplastic lobules in all four neoplasms examined ultrastructurally. They were characterized by long cytoplasmic projections and a scant pale cytoplasm with a few organelles. These organelles included a small amount of rough endoplasmic reticulum, a few mitochondria with tubular cristae, a few free ribosomes, and a small amount of glycogen. They lacked desmosomes, Birbeck's granules, and melanosomes. Their nucleus was oval with prominent clumps of marginally located heterochromatin. Melanocytes and Merkel cells were not found in the neoplasms examined.
Clinical study
Follow-up information was available for eight patients (10–1,095 days follow-up, mean = 450 days). Three tumors metastasized to the draining lymph node, and the metastases were detected on days 0, 253, and 1,095 after excision of the primary tumor. Metastases to other organs were not reported. One additional dog was euthanatized 877 days after excision of the primary tumor because of unidentified internal masses.
Discussion
This article identifies a new type of epithelial tumor in the dog that has not been reported in other species, including man. Most of the neoplasms presented here showed areas of apocrine or trichoepitheliomatous (hair follicle) differentiation or both, whereas the presence of significant amounts of glycogen in neoplastic cells was suggestive of tricholemmal differentiation. The presence of cells with coexpression of vimentin and cytokeratin and areas of stromal chondromatous differentiation supported a myoepithelial differentiation, 41, although labeling for α-smooth muscle actin was not detected. Combined together, these features suggest that the tumors described herein originate from a primitive epithelial cell capable of differentiating into different portions of the hair follicle and apocrine gland; hence, the proposed name of follicular stem cell carcinoma. A cell capable of giving rise to the entire hair follicle and the epidermis has been identified in mice. 26 , 36 This cell is located in the bulge region of the hair follicle and has been named “follicular stem cell,” and we propose that this cell is the origin of the tumors described herein.
The number of cells which stained with the AE1/AE3 anti-cytokeratin antibody differed among the neoplasms of this series. This feature is also observed in canine and feline basal-cell carcinomas (I. Mikaelian, personal observation) and in human basal cell carcinomas. 2 , 34 Partial staining in follicular stem cell carcinomas was expected because the normal canine (this study) and feline 27 epidermis, hair follicles and their adnexae stain unevenly with the AE1/AE3 anti-cytokeratin antibodies. Immunohistochemical staining of follicular stem cell carcinomas and of the normal skin of dogs with a panel of specific anti-cytokeratin antibodies may allow better characterization of the cell of origin of follicular stem cell carcinomas.
Follicular papillae were noted in 23 of 33 tumors, including a metastatic tumor. Follicular papillae are occasionally observed in a small number of canine and feline matrical carcinomas (I. Mikaelian, personal observation) as well as in a few human basal cell carcinomas, 10 apocrine poromas, 8 and spiradenocylindromas. 23 Hair follicle development is the result of complex and poorly understood interactions between the follicular papilla and the epidermis. 21 , 25 The presence of follicular papillae in most follicular stem cell carcinomas, including a metastatic tumor, suggests that follicular stem cells play a pivotal role in the development of the follicular papilla.
Differential diagnoses for follicular stem cell carcinomas include balloon cell melanoma and sebaceous carcinoma. Balloon cell melanomas are differentiated from follicular stem cell carcinomas by the absence of nesting, follicular papillae, nuclear palisading, tubules, clefts, and of direct connection with the epidermis.
Immunohistochemistry is useful in differentiating follicular stem cell carcinomas from melanomas. Cytokeratin was expressed in all but one follicular stem cell carcinomas, whereas it is not expressed in canine melanomas. 4 Vimentin was expressed by a limited number of cells in follicular stem cell carcinomass, whereas it is generally expressed by all cells in melanomas. 29 Finally, S100a was not expressed in follicular stem cell carcinomas, whereas it is expressed by a large proportion of canine melanomas. 4 , 29
Electron microscopy further confirmed a diagnosis of epithelial tumor and also differentiated follicular stem cell carcinomas from melanomas. Unlike follicular stem cell carcinomas, melanomas lack desmosomes, neuroendocrine-like granules, intercellular lumina, and intracellular ductules. Although melanomas may form basement membranes, 33 they are not as continuous and thick as those observed in follicular stem cell carcinomas. Also, neoplastic melanocytes consistently contain premelanosomes or melanosomes, a feature absent from all four follicular stem cell carcinomas studied by electron microscopy.
Microvilli are an ultrastructural feature of stem cells 18 and were identified in three of four follicular stem cell carcinomas studied under electron microscopy. However, microvilli may also be observed in apocrine sweat glands 17 and their tumors. 22
Sebaceous carcinomas are differentiated from follicular stem cell carcinomas by the presence of lipid vacuoles that encroach on the nucleus, the common presence of areas of squamous differentiation, and the absence of follicular papillae, tubules, and pigmentation. Cytochemistry and electron microscopic evaluation of the tumors confirmed the absence of lipids in the cytoplasm of follicular stem cell carcinomas.
Melan A/MART-1 was expressed in 12 of 27 follicular stem cell carcinomas. Expression of this marker in an epithelial neoplasm is in contradiction with the current human and veterinary literatures. 15 , 29 However, Melan A/MART-1 is expressed in the epithelium of the ducts of salivary glands, prostatic acinar cells, urethral epithelium, and intrapulmonary epithelial cells. 29 Our observations establish that Melan A/MART-1 can stain some cutaneous epithelial neoplasms and that it should not be considered a specific marker for canine melanomas.
A large proportion of neoplastic cells in all four follicular stem cell carcinomas examined ultrastructurally contained membrane-bound granules. These granules have been described in human basal cell tumors 13 , 16 , 35 , 38 , 42 and primary epithelial stem cells. 16 They have been interpreted as lysosome-like structures, 42 pinocytic granules, 13 zymogen granules for collagenases, 16 or evidence of neuroendocrine differentiation of basal cell carcinomas. 35 , 38 Their presence in larger numbers at the periphery of neoplastic lobules is consistent with the distribution expected for zymogen granules.
The cytoplasmic granules were not interpreted as neuroendocrine granules of Merkel cells. Merkel cell tumors differ histologically from follicular stem cell carcinomas in that neoplastic cells do not form conspicuous nests and have a small amount of cytoplasm. Merkel cell tumors immunohistochemically differ from follicular stem cell carcinomas in that staining for vimentin is absent or minimal 31, whereas staining for cytokeratin is confined to the paranuclear zone. 7 Finally, Merkel cells ultrastructurally differ from follicular stem cell carcinomas in that neoplastic Merkel cells have cytoplasmic processes and paranuclear aggregates of intermediate filaments 32, whereas they lack basement membranes and desmosomes.
Two types of interstitial cells populated follicular stem cell carcinomas: a population of CD18- and lysozyme-positive dendritic cells, and melanocytes. The CD18- and lysozyme-positive cells may represent Langerhans or dermal dendritic cells. These cells, as expected in the dog, 14 , 24 lacked Birbeck's granules, and thus, it was not possible to determine whether they represented Langerhans cells or dermal dendritic cells. However, their location in neoplastic nests is consistent with that of Langerhans cells.
Melanocytes were detected upon histologic evaluation of 15 tumors but were not detected upon ultrastructural evaluation of four follicular stem cell carcinomas. This may be the result of the overall scarcity of melanocytes as determined by labeling for S100a.
Langerhans cells, melanocytes, and occasionally Merkel cells, are present in a variety of human epithelial cutaneous tumors. 6 , 20 , 30 , 37 The occurrence of cells consistent with Langerhans cells in metastatic tumors is an important observation. Currently, it is not known whether epidermal dendritic cells are components of epithelial skin tumors or whether they represent an immune reaction targeted at neoplastic cells. 20 Their presence in the three metastatic carcinomas warrants further evaluation of their role in the biology of canine epithelial tumors.
Follow-up identified only three dogs with metastases. However, lymphatic invasion was noted in five tumors, one of which was metastatic at the time of excision of the primary tumor, which suggests that metastatic rate is probably higher than reported here. Additional follow-up is needed to adequately document the biological behavior of follicular stem cell carcinomas.
Footnotes
Acknowledgements
This work would not have been possible without the expertise and the kind encouragements of Dr. Culbertson, Dr. Liu, Dr. McCullough, Dr. McElhaney, Dr. Gross, and Dr. Smith, who have recognized this entity for many years. Dr. Odin, Dr. Molon-Noblot, Dr. Ramos-Vara, Dr. Walder, and Dr. Wojcieszyn provided constructive ideas for this manuscript. We are thankful to R. Havens, M. Litton, S. Puerner, and A. Whittington for technical assistance. We are grateful to the referring veterinarians of IDEXX Veterinary Services for the follow-up on these cases.
