Abstract
Scrapie, a transmissible spongiform encephalopathy (TSE), is a naturally occurring fatal neurodegenerative disease of sheep and goats. The current study documents incubation periods, pathologic findings, and distribution of abnormal prion proteins (PrPSc) by immunohistochemistry and Western blot in tissues of genetically susceptible and resistant neonatal lambs inoculated with pooled brain homogenates from 13 U.S. origin scrapie-affected ewes. Nine Suffolk lambs with genotypes AA/RR/QQ (n = 5) and AA/RR/QR (n = 4) at codons 136, 154, and 171, respectively) were orally inoculated, within 12 hr of birth, with 1 ml of a 10% (w/v) brain homogenate prepared from scrapie-affected sheep brains. Inoculated animals were euthanized when advanced clinical signs of scrapie were observed. All QQ sheep developed clinical signs of scrapie, with a mean survival time of 24 months. Spongiform lesions in the brains and PrPSc deposits in the central nervous system and lymphoid tissues were present in these sheep. None of the QR sheep succumbed to the disease. A previous study that used a larger volume (30 ml of 10% brain suspension) of the same inoculum in 4-month-old Suffolk lambs of susceptible genotype documented longer survival periods (average 32 months), and only 5 of 9 inoculated sheep developed scrapie. Findings of this study suggest that orally exposed neonatal lambs of a susceptible (QQ) genotype exhibit a higher attack rate and shorter incubation period than older (4-month-old) lambs exposed to a larger dose (30×) of the same inoculum.
Introduction
Transmissible spongiform encephalopathies (TSE) or prion diseases are fatal neurologic diseases of animals and humans. Susceptibility to TSEs, including scrapie, appears to be age dependent. 9 In a recent study, 3 it was shown that 4-month-old lambs orally inoculated with scrapie inoculum succumbed to the disease in an average of 32 months after inoculation (MPI). 3 Although these lambs were inoculated with a large dose of inoculum (3 g of scrapie-infected brain tissue), only 5 of 9 recipients developed scrapie. Recent studies on PrPSc-expressing reproductive tissues from scrapie-infected pregnant sheep showed that PrPSc accumulates at the fetal-maternal interface, which indicates that scrapie may be transmitted from infected ewes to their scrapie-susceptible fetus during the perinatal period by exposure to infectious placental tissues 10 or milk. 5 Therefore, it would appear that, under natural conditions, neonatal exposure to scrapie would be common. The current study documents results of oral scrapie inoculations in neonatal lambs.
Materials and methods
Animals, housing, and inoculum
Nine 1-day-old Suffolk lambs (6 females, 3 males), and their dams were obtained from a scrapie-free sheep flock at the National Animal Disease Center (NADC; Ames, IA). Five lambs were AA/RR/QQ and 4 were AA/RR/QR at codons 136, 154, and 171 of the PRNP gene, respectively (Table 1). All lambs received the scrapie inoculum by the oral route. The inoculated animals and their dams were housed in an outside pen at NADC. They were fed pelleted growth and maintenance rations that contained no ruminant protein, and clean water was freely available. At 4 months, the lambs were weaned by removal of the ewes from the pen. The inoculum (no. 13–7) was prepared from pooled brain homogenates derived from 13 scrapie-affected ewes from 7 different flocks. 3 The brains were ground in a mechanical grinder, and a final concentration of 10% (w/v) was prepared with phosphate buffered saline solution. The animals were each orally administered 1 ml of inoculum.
Samples
The lambs were euthanized when they showed advanced clinical signs consistent with scrapie. A detailed necropsy was carried out on each animal, and 2 sets of tissue samples (nasal turbinate, tongue, palatine and pharyngeal tonsils, spleen, retropharyngeal and mesenteric lymph nodes, ileum, liver, masseter muscle, diaphragm, eye, and the whole brain) were collected. The brain was divided longitudinally into 2 halves. One set of tissues was fixed in 10% buffered formalin, embedded in paraffin wax, sectioned at 5 μm, and stained with hematoxylin and eosin for light microscopy. The second set of tissues was frozen.
. Genotypes, incubation (survival) times, and pathologic findings in 9 neonatal Suffolk lambs orally inoculated with U.S. scrapie inoculum (no. 13–7).*
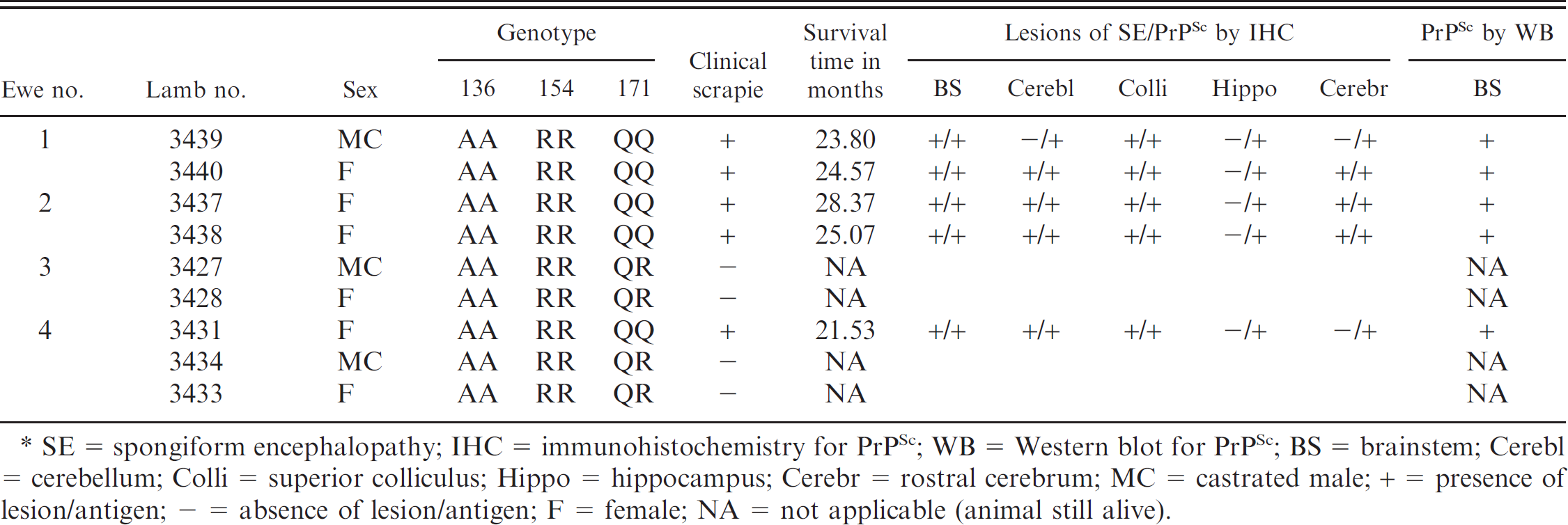
SE = spongiform encephalopathy; IHC = immunohistochemistry for PrPSc; WB = Western blot for PrPSc; BS = brainstem; Cerebl = cerebellum; Colli = superior colliculus; Hippo = hippocampus; Cerebr = rostral cerebrum; MC = castrated male; + = presence of lesion/antigen; – = absence of lesion/antigen; F = female; NA = not applicable (animal still alive).
All paraffin-embedded tissues were also stained by an automated immunohistologic (IHC) method for detection of PrPSc, as previously described. 3 Briefly, after deparaffinization and rehydration, tissue sections were autoclaved for 30 min in an antigen retrieval solution a and were stained with an indirect avidin–biotin system b designed for an automated immunostainer. c The primary antibody was a cocktail of 2 monoclonal antibodies, F89/160.1.5 8 and F99/97.6.1, 7 each used at a concentration of 5 μg/ml, and incubation was carried out at 37°C for 32 min. The secondary antibody was biotinylated anti-mouse, d diluted 1:200 and incubated for 8 min at 37°C.
The brain was cut longitudinally, and one half of the brainstem and medulla were fixed in formalin for histopathology and IHC. The remaining one half of the brain was frozen for immunodetection of PrPSc by a modified Western blot (WB) technique as previously described. 3 Briefly, the modification consisted of the use of P4 as primary antibody and a secondary biotinylated anti-mouse antibody (dilution 1:10,000), e followed by a streptavidin–horseradish peroxidase conjugate (dilution 1:10.000). f The immunoblot was developed with an ECL Plus chemiluminescence solution g before being exposed to film or scanned by an imaging system.
Results
Spongiform lesions in the brains and PrPSc deposits in the central nervous system (CNS) and lymphoid tissues were present in all sheep with clinical prion disease (Table 1; Fig. 1). All QQ sheep (5/5) were clinically affected with the disease, and their mean survival time was 24 MPI, whereas the QR sheep did not succumb to the disease at 50 MPI. At the time of writing, all QR sheep were still alive and appeared healthy.
The distribution of histologic lesions (Fig. 1), and the results of PrPSc IHC (Fig. 2) and WB assays (Fig. 3), are given in Table 1. Microscopic spongi-form lesions characteristic of prion disease were present in all clinical animals (Table 1). The lesions were severe and consisted of vacuolations in the neuropil and neuronal perikarya (Fig. 1). They were most severe in the dorsal motor nucleus of the vagus. No significant differences were observed in the degree and distribution of morphologic lesions between individual sheep. Neither increased numbers of glial cells nor neuronal cell degeneration was seen in the affected areas of the examined sections.
Immunohistochemical labeling of PrPSc in CNS and lymphoreticular system (LRS) tissues was present in 5 of 5 scrapie-affected QQ sheep. The PrPSc labeling intensity and distribution in the CNS were similar in all sheep. The intensity of PrPSc labeling was greatest in the brainstem (area of obex; Fig. 2), cerebellum, and midbrain (level of superior colliculus), with lesser labeling in the hippocampus and rostral cerebrum on average. The PrPSc accumulations coalesced, becoming nearly confluent in substantial areas of the brainstem, midbrain, and cerebellar folia and basal nuclei. Multifocal punctate PrPSc labeling of CNS neuropil with coalescing perineuronal labeling were the more typical patterns seen in the hippocampus and cerebral cortex. Labeling of neuronal perikarya tended to be less intense and was punctuate in character, and was the only localization site of PrPSc in the trigeminal ganglion. The plexiform layers of retina contained confluent accumulations, and receptor, nuclear, and ganglion cell layers had multifocal punctuate PrPSc labeling.
Localization of PrPSc in the peripheral nervous system was limited to a moderate amount of punctuate labeling of the perikaryon of several neuron groupings in the myenteric plexus. Relatively scant punctuate PrPSc labeling of adrenal cortical and medullary cells (1/5 sheep) and of adrenal medullary cells only (1/5 sheep) was noted. The majority of LRS tissues examined contained PrPSc labeling in 4 of 5 sheep. Moderate autolysis of tissues of sheep no. 3439 with loss or partial effacement of lymphoid follicles made IHC interpretation of many LRS organs from this animal difficult; however, moderately strong PrPSc labeling was present in submucosal lymphoid tissue (Peyer's patches) in sections of ileum. PrPSc labeling in the LRS was present in germinal centers of lymphoid follicles (Fig. 3) with lesser amounts in the interfollicular/paracortical areas. PrPSc labeling was present in the spleen of 3 of 5 sheep, localized to periarteriolar lymphoid sheaths. Lymphoid follicles in sections of the third eyelid were inconspicuous or absent in 4 of 5 sheep; the 3 well-developed follicles present in sheep no. 3438 were devoid of PrPSc labeling.
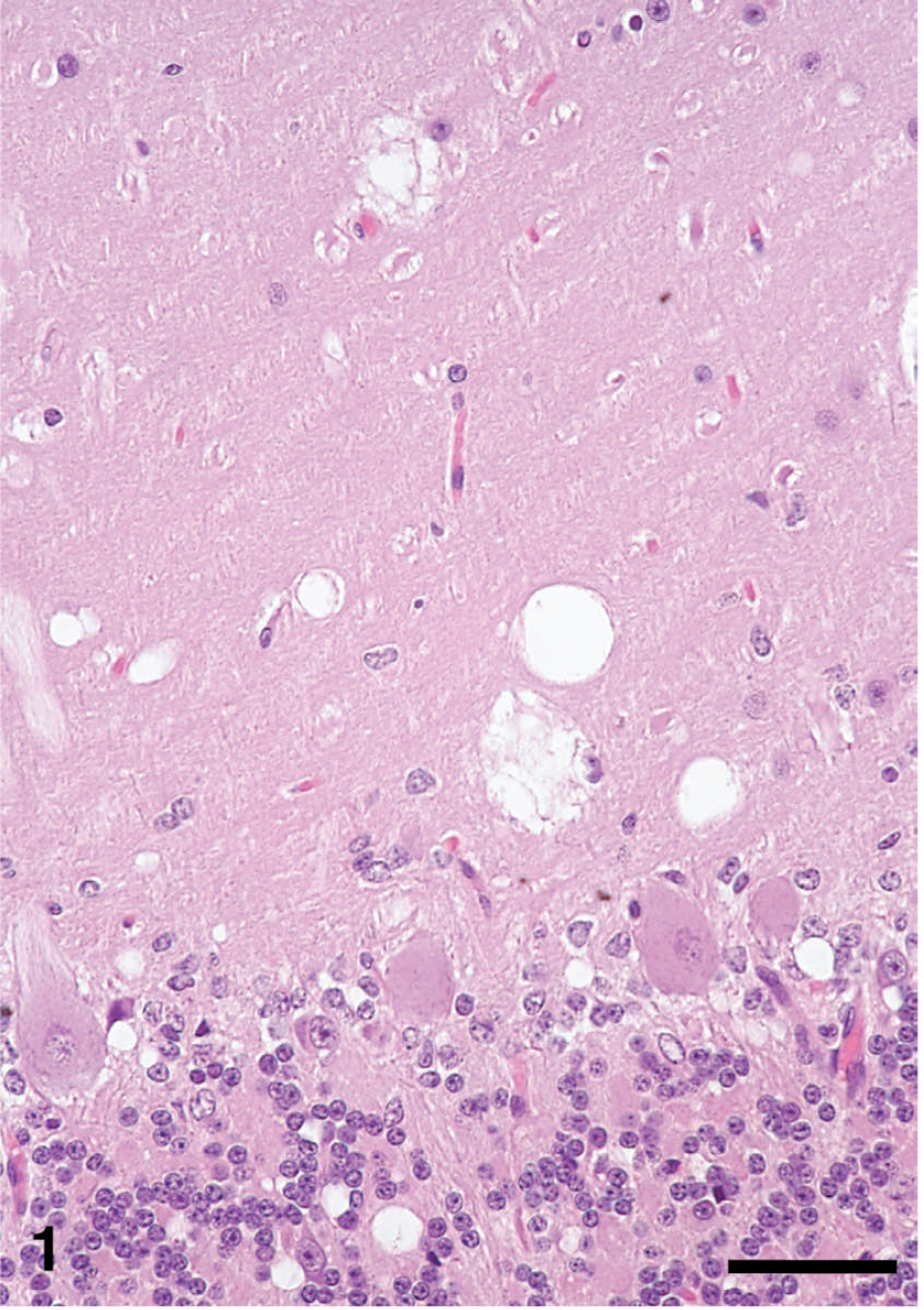
Cerebellum of sheep no. 3440 with clinical scrapie, showing area of spongiform encephalopathy. There are variably sized vacuoles in molecular, Purkinje, and granular cell layers. Hematoxylin and eosin. Bar = 50 μm.
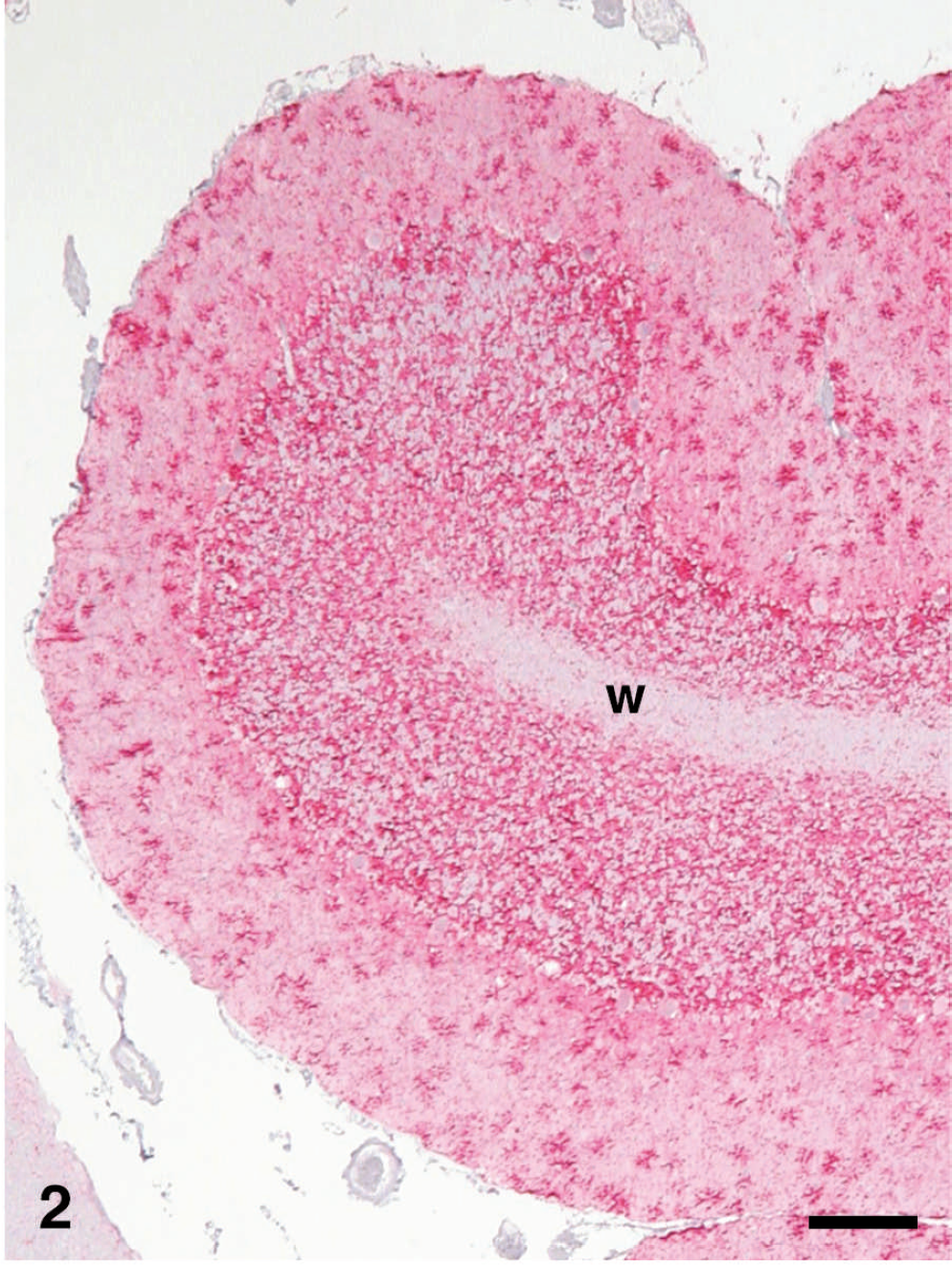
Cerebellum of sheep no. 3440, with clinical scrapie, showing PrPSc immunohistochemical (IHC) staining (red). There is extensive diffuse PrPSc labeling in the molecular, Purkinje, and granular cell layers. Cerebellar white matter (w) does not show the presence of PrPSc. Stained for PrPSc labeling (red) by IHC (immunoalkaline phosphatase) and counter-stained with hematoxylin. Bar = 200 μm.
In the WB analysis with monoclonal antibody P4, brainstem samples from all 5 sheep were positive for PrPSc, which showed the typical profile of 3 bands of proteinase K–resistant isoforms of PrPSc (Fig. 4), which represents the diglycosylated, monoglycosylated, and unglycosylated polypeptides. No difference in molecular weight and glycoform profile was noted between the individual sheep when compared to the inoculum.
Discussion
A previous study that used a large volume (30 ml of 10% brain suspension) of the same inoculum (no. 13–7) in 4-month-old QQ Suffolk lambs documented a longer survival (incubation) times (average 32 MPI) and a lower attack rate (only 5 of 9 inoculated sheep developed scrapie). 3 In the present study, the lambs were given only 1 ml of the same inoculum (10% solution of no. 13–7), and all QQ sheep developed clinical scrapie within an average of 24 MPI. This higher susceptibility with a relatively short incubation time could be related to the inoculation of neonatal animals.
The present study supports the hypothesis that younger animals (neonatal QQ lambs) have increased susceptibility to scrapie, have higher attack rates, and require smaller doses of inoculum to induce the disease when compared with older animals. These results are in agreement with observations of others 1,6 who noticed that sheep exposed to scrapie at older ages appeared to be less susceptible to the disease than when animals were exposed earlier in life. In a previous study, inoculation of neonatal lambs with scrapie had failed to show infectivity. 2 In that study, the lambs were not specified as susceptible (QQ), were inoculated at older age (3–12 days), and were euthanized earlier after infection (5–7 MPI). The early manifestation of scrapie in the present study could be because of the susceptibility of lambs (QQ at 171) and the inoculation during the neonatal period (within 12 hr of birth). The neonatal intestinal mucosa has been shown to allow relatively free passage of protein molecules (e.g., immunoglobulins and others) from luminal contents into the lymphatic system for up to 24 hr after birth. Because the scrapie prion is a protein molecule, it may use the same route of translocation from the gut mucosa to the blood as do immunoglobulins. The question still remains whether the gut mucosa of neonatal QR lambs supports the PrPSc translocation. In our study, we do not know whether the PrPSc in QR animals passed through the gut mucosa or did not. In a recent study by using gut-loop experimentation, it was shown that there was no significant difference in tissue distribution of PrPSc in the gut or in the draining lymph node of QQ and QR sheep in early stages after inoculation (up to 24 hr after infection). However, PrPSc was not found in QR sheep but was seen in QQ sheep at 30 days after infection. 4 This indicates that, in QR sheep, the host's microenvironment in intestines and mesenteric lymph nodes prevents maintenance and/or replication of scrapie prions.
Four years after inoculation, the 4 remaining QR sheep remain alive and apparently healthy. These animals will be kept alive for another 2 years (total = 6 years after inoculation) and will be observed for clinical signs of scrapie. At that time, they will be euthanized, and their tissues will be examined for the presence of PrPSc.
Acknowledgements
The authors thank the late Dr. L. Nusz and Dr. J. Laufer for clinical assistance, Dr. Marcus Kehrli Jr. for constructive comments and for his enthusiastic support, and James Fosse for the photomicrographs. Expert technical assistance was provided by Martha Church, Dennis Orcutt, Joseph Lesan, Trudy Tatum, Deb Clouser, Kevin Hassall, Ginny Montgomery, Micky Fenneman, and the TSE animal caretakers. This study was carried out under the guidelines of the institutional Animal Care and Use Committee at NADC. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture.
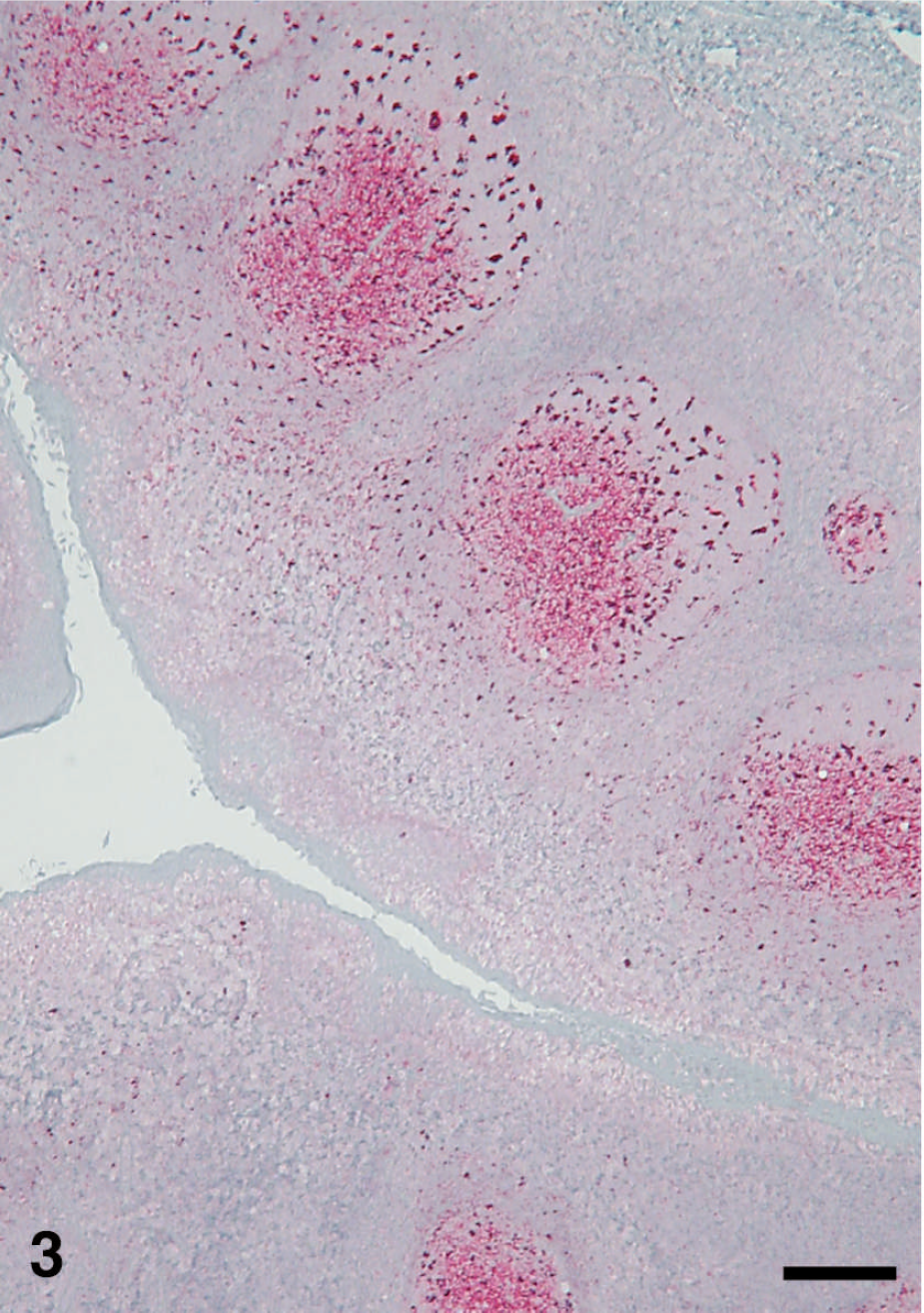
Palatine tonsil of sheep no. 3440, showing PrPSc immunohistochemical (IHC) staining (red), which is mainly in the germinal centers of follicles. Stained for PrPSc labeling (red) by IHC (immunoalkaline phosphatase) and counter-stained with hematoxylin. Bar = 200 μm.
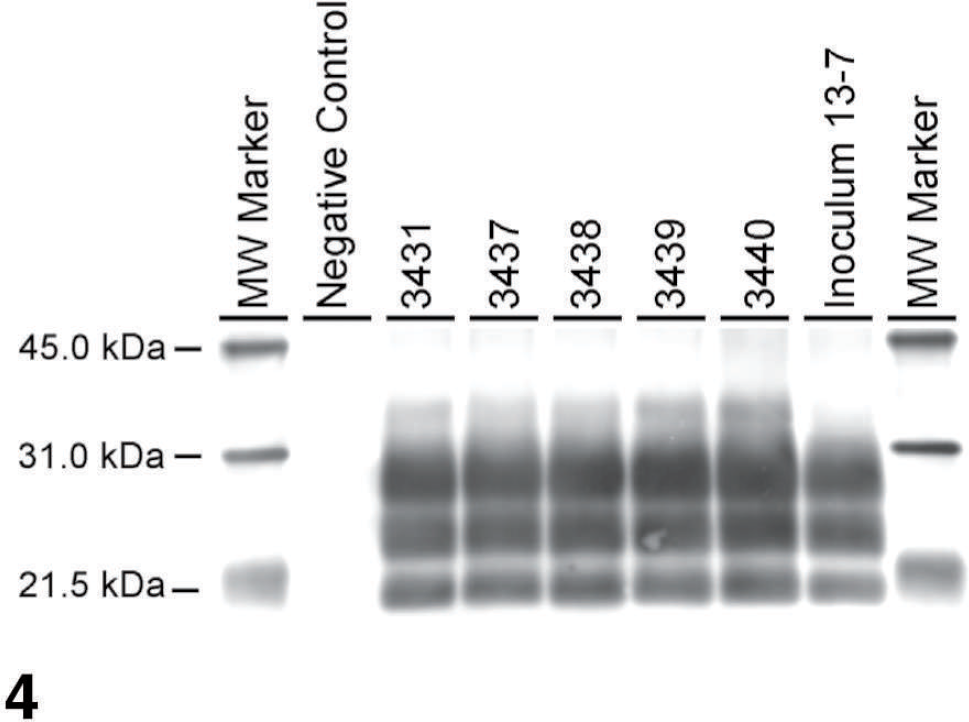
Western blot analysis with monoclonal antibody P4, showing distinct profile of PrPSc in the brainstem of 5 QQ sheep and the inoculum. Samples were loaded at 0.06 mg per lane. Molecular weight (MW) markers in kDa are indicated on both sides of the blot.
Footnotes
a.
DAKO™ Target Retrieval Solution, Dako North America Inc., Carpinteria, CA.
b.
Basic Alkaline Phosphatase Red Detection Kit, Ventana Medical Systems Inc., Tucson, AZ.
c.
NexES IHC module, Ventana Medical Systems Inc., Tucson, AZ.
d.
Biotinylated anti-mouse IgG (made in horse), Vector Laboratories, Burlingame, CA.
e.
Biotinylated anti-mouse IgG, Amersham Biosciences, Piscataway, NJ.
f.
Streptavidin–horseradish peroxidase conjugate, Amersham Biosciences, Piscataway, NJ.
g.
ECL Plus, Amersham Biosciences, Piscataway, NJ.
