Abstract
Samples from feline normal, dysplastic, and neoplastic mammary tissues were used to investigate the usefulness of MIB-1 labeling index (MIB-1 I) as a prognostic indicator. Forty-eight queens bearing invasive carcinomas were included in a 2-year follow-up study. Mammary lesions were classified according to the World Health Organization system, and invasive carcinomas were further graded on the basis of the degree of tubule formation, the degree of nuclear and cellular pleomorphism, and mitotic count. Additional sections were immunostained using MIB-1 antibody, and MIB-1 I was expressed as a percentage of positive nuclei. In normal mammary gland tissues, the mean MIB-1 I was <1%. A low proliferation rate was found in all mammary adenosis and in situ carcinomas, and the highest rates were observed in feline mammary hypertrophy and invasive carcinomas. Twenty-one (43.7%) of the queens bearing invasive carcinomas were still alive at the end of the trial, and 27 (56.2%) had died. The MIB-1 I was not significantly correlated with clinical outcome, age, histologic type, or grading of the tumors, but a borderline correlation was observed with invasion of lymphatic vessels. Univariate analysis showed that high MIB-1 I was also not associated with decreased overall survival, whereas the grading system of the tumors had high predictive value (P = 0.0040) for postsurgery survival. The lack of correlation between MIB-1 I and postsurgery survival suggests that this marker alone is not sufficient to determine a correct prognosis in feline mammary carcinomas, even if it is a useful proliferation marker.
Mammary tumors are commonly obsreved in female cats. 1 Between 80% and 96% of these tumors are malignant and very aggressive 10,18, and can grow rapidly and metastatize to regional lymph nodes, spleen, liver, and lung. 11,17 For these reasons, these tumors are an important cause of mortality, especially in middle-aged and elderly females. The interval between feline mammary carcinoma (FMC) detection and death varies from <6 to 12 months. 18 Several factors, including age, diameter of the primary lesion, the presence of histologically tumor-positive lymph nodes, mitotic count, extent of necrosis, and complete surgical excision, are independently correlated with postsurgery survival time. A radical surgical treatment instead of a conservative one is significantly related to the disease-free interval. 16,23 In the last decades, the interest of veterinary pathologists has been focused on the assessment of markers that may improve the prognosis of FMC, such as morphologic features 2,23 and the proliferative activity of the tumor. 3,20 In human oncology, the immunohistochemical detection of cell proliferation–associated antigens has been widely investigated. One of the most useful reagents in this field is the antibody to Ki-67 antigen, a nonhistone, highly protease-sensitive nuclear protein, whose expression is strictly associated with cell proliferation. 8,22 Ki-67 antigen is expressed in all phases of continuously cycling cells, except G0 and G1. 7 Thus, it is an excellent marker for determining the so-called growth fraction of a given cell population. Antibodies directed against this protein are widely used in evaluation of tumors, especially mammary tumors. The Ki-67 labeling index (percentage of positive nuclei) is often correlated with the clinical course of the disease. 12,22 The MIB-1 monoclonal antibody (MIB-1) directed against this antigen produces a cross-reaction in frozen and paraffin-embedded feline tissue sections. 13
The few studies on the determination of cell proliferation activity of canine and feline mammary tumors by the immunohistochemical detection of Ki-67 antigen have produced contradictory results. 4,15,19 We used immunohistochemistry to analyze MIB-1 labeling in a series of feline dysplastic and neoplastic mammary tissues to determine whether this antibody would give more information on the clinical behavior of invasive FMC in a 2-year follow-up study. We also considered the usefulness of tumor grading as a prognostic indicator.
Materials and Methods
All the mammary gland specimens used in this study were retrieved from the archives of the Tumour Registry at the Department of Animal Pathology, University of Pisa between January 1997 and December 1998. The samples were surgically obtained by simple mastectomy or block dissection from 75 female cats of different breeds (52 European Domestic Shorthair cats, 8 Siamese, and 15 Persians) ages 2–19 years (○ ± SD = 10.3 ± 2.6 years).
The queens included in the study were examined and surgically treated at the Veterinary Teaching Hospital (Clinic Department, School of Veterinary Medicine, University of Pisa) or by practitioners in the surrounding areas. After anamnesis collection, including age, breed, body size, history of ovariohysterectomy, and prevention of estrus with hormonal treatment, a complete physical examination was done. Information on tumor size (maximum length), adherence to underlying tissues, and skin ulceration were also recorded. All the cats included in the study were followed for at least 2 years after surgery by the referring veterinary surgeons to evaluate the postsurgery course of the disease. Overall survival was defined as the time from the day of diagnosis until the day of death or last follow-up.
Ten normal mammary tissues were also studied. These samples were from specific-pathogen-free cats (Iffa Credo, L'Asbrege, France) used as negative controls in a feline immunodeficiency virus (FIV) vaccination program and housed in a climate-controlled animal facility (Techniplast, Gazzada, Italy) of the Retrovirus Centre of the University of Pisa, under conditions stipulated by European Community law.
Representative portions of each mammary sample were fixed in 10% buffered formalin and routinely embedded in paraffin. Five-micrometer-thick sections were stained with hematoxylin and eosin (HE) for histologic evaluation; additional 5-µm sections were used for immunohistochemical studies. The lesions were classified according to the World Health Organization (WHO) classification, 9 and tumors displaying different features were classified according to the most pronounced histologic differentiation. Presence of lymphatic or stromal invasion was also recorded.
Histologic grading was performed on HE-stained sections according to the classification proposed by Elston and Ellis 5 and previously used to determine the grade of FMCs. 2 Histologic grading mainly involved invasive carcinomas; noninvasive tumors were excluded from this part of the study. According to the Elston and Ellis method, 5 the overall grade of an individual tumor was derived from the assessment of three morphologic features: degree of tubule formation, degree of nuclear and cellular pleomorphism, and mitotic count. To score the degree of tubular formation, two complete sections from different areas of each tumor were examined, and the proportion of tumor showing tubular formation was determined. When >75% of the examined area displayed definite tubular formation, a score of 1 point was given, from 10% to 75% was given 2 points, and less than 10% was given 3 points. Nuclear pleomorphism was scored qualitatively (1 point when nuclei were small and regular with uniform chromatin, 2 points when cells were larger than normal with vescicular nuclei and clearly visible nucleoli, and 3 points when cells differed in size and shape with prominent and multiple nucleoli). Mitotic count was evaluated quantitatively (1 point was given for >7 mitoses/10 fields; 2 points for 8–15 mitoses/10 fields, and 3 points for >15 mitoses/10 fields). After adding together the score of each category, the tumor grade was obtained as follows: well-differentiated carcinoma (grade I), 3–5 points; moderately differentiated carcinoma (grade II), 6 or 7 points; and poorly differentiated carcinoma (grade III), 8 or 9 points.
Five-micrometer-thick sections on poly-
Statistical analysis was performed using the statistical package SPSS Advanced Statistics 7.5 (SPSS Inc., Chicago, IL). The analysis of variance (ANOVA) was determined, and the difference between groups was evaluated with Bonferroni's test. Curves for overall survival were estimated by the Kaplan–Meier method.
Results
The 75 samples examined consisted of 57 of FMC, 11 of feline mammary hyperplasia (FMH), and 7 of mammary adenosis. Of the 57 tumors examined, nine (15.8%) were in situ carcinomas, and 48 (84.2%) were invasive carcinomas. Based on the WHO classification, papillary carcinoma was the most common type of invasive carcinoma (22/48, 45.8%), followed by solid carcinomas (14/48, 29.2%), tubular carcinomas (8/48, 16.7%), and papillary cystic carcinomas (4/48, 8.3%).
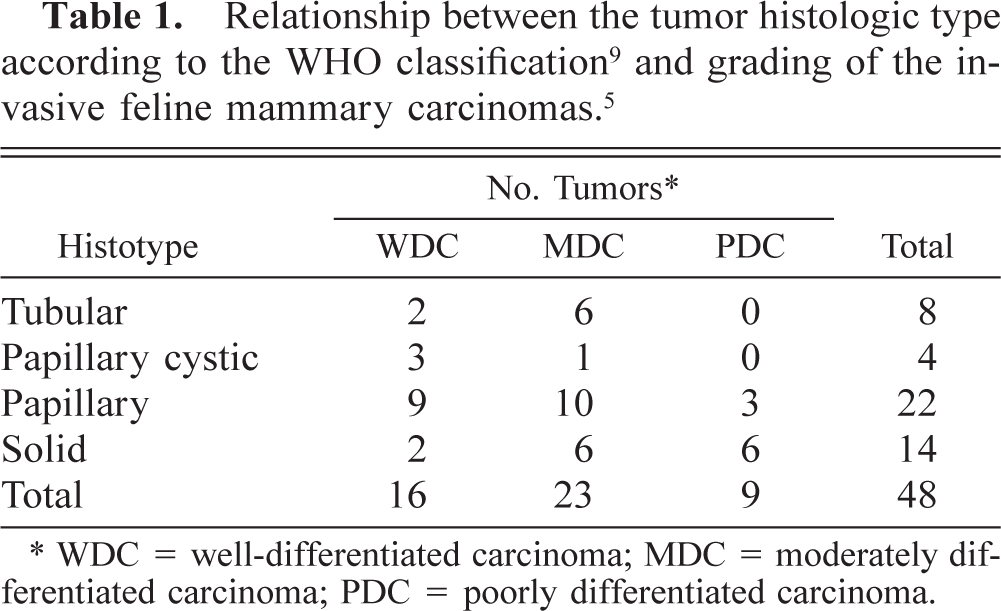
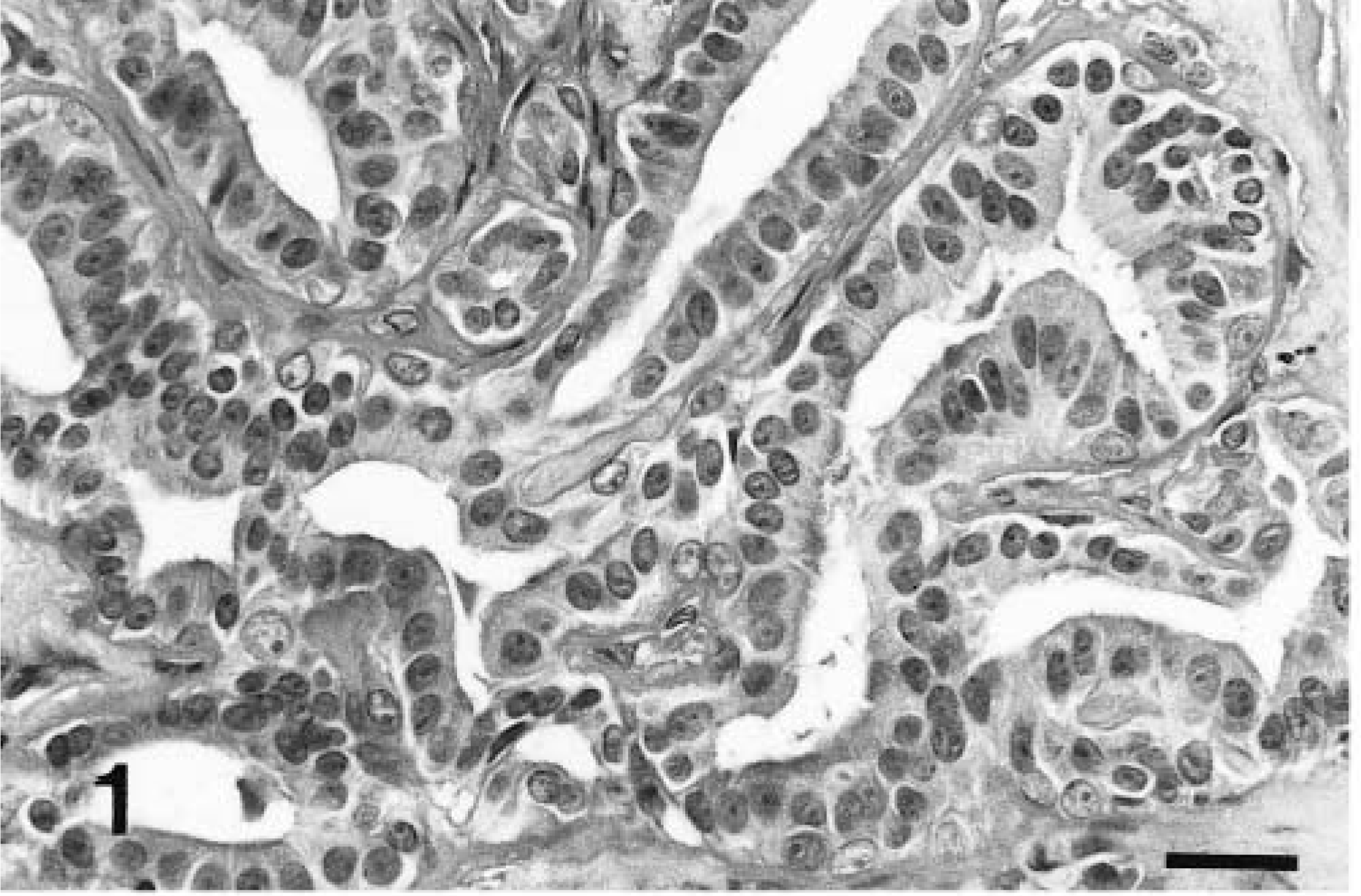
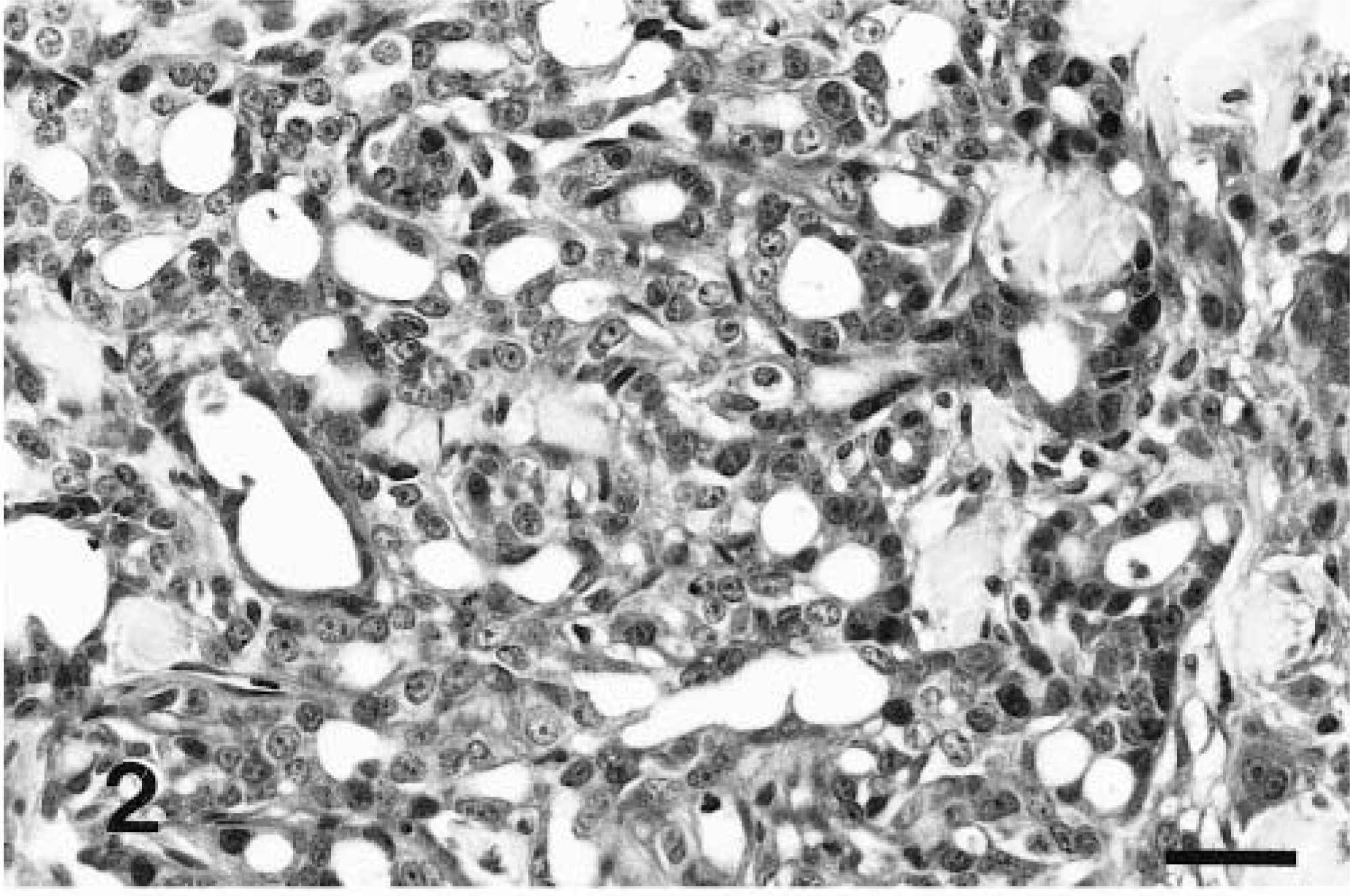
Histological grading of the 48 invasive carcinomas revealed that 16 (33.3%) were well-differentiated carcinomas (WDCs; grade I, Fig. 1), 23 (47.9%) were moderately differentiated carcinomas (MDCs; grade II, Fig. 2), and nine (18.7%) were poorly differentiated carcinomas (PDCs; grade III, Fig. 3). The relationship between the results of histologic classification and tumor grading is detailed in Table 1. All the tubular and papillary cystic carcinomas were WDCs or MDCs, and solid carcinomas were mostly MDCs and PDCs.
∗ WDC = well-differentiated carcinoma; MDC = moderately differentiated carcinoma; PDC = poorly differentiated carcinoma.
Mammary gland; cat No. 26. Well-differentiated carcinoma (grade I) with a high degree of tubule formation and relatively uniform cells and nuclei. HE. Bar = 50 µm.
Mammary gland; cat No. 25. Moderately differentiated carcinoma (grade II). The tumor has a high degree of tubule formation and shows cells larger than normal with vescicular nuclei and high mitotic activity. HE. Bar = 50 µm.
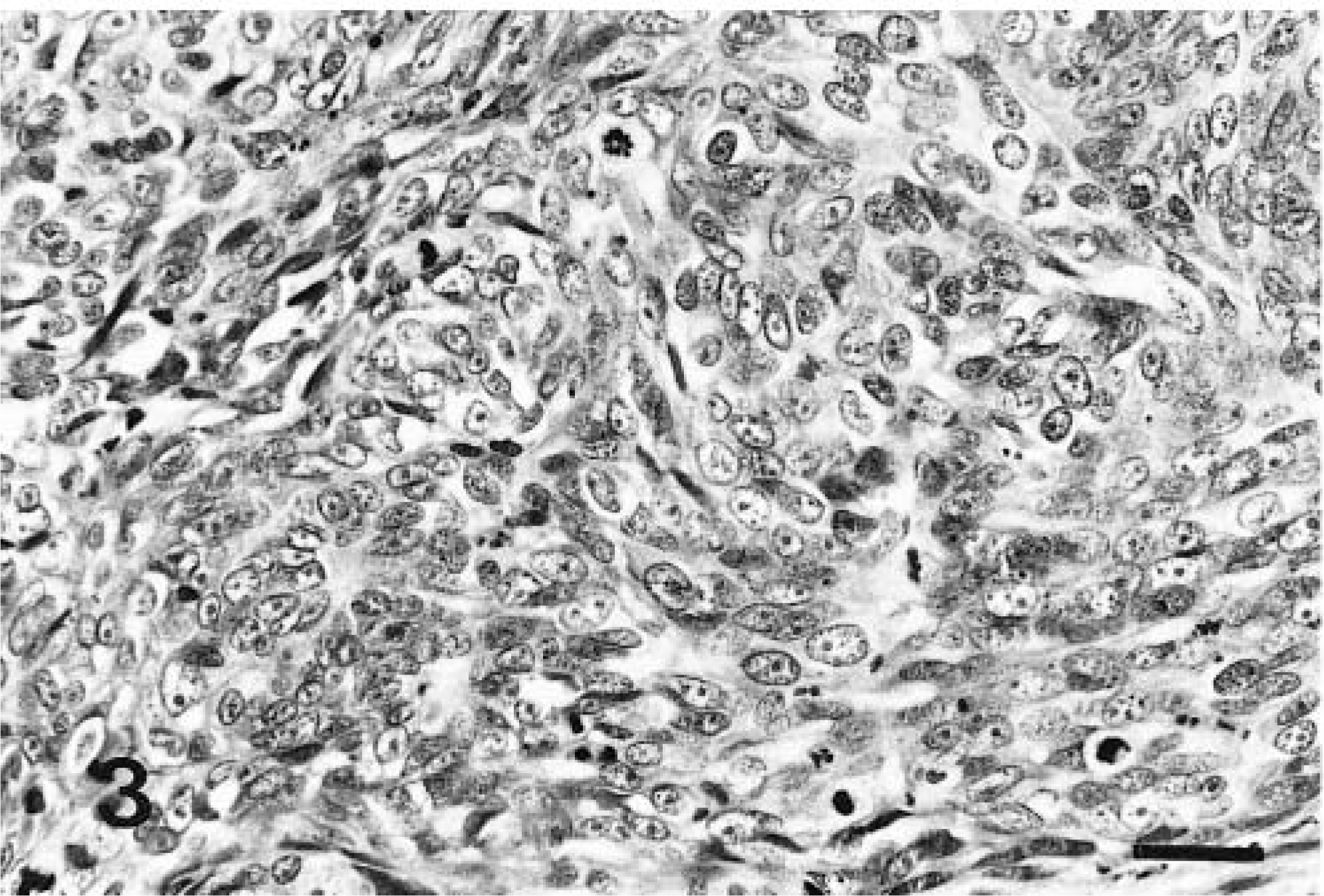
Mammary gland; cat No. 29. Poorly differentiated carcinoma (grade III) showing lack of tubule formation, large and vescicular nuclei with prominent nucleoli, and high mitotic index. HE. Bar = 50 µm.
In lymph node central sections, MIB-1 produced nuclear staining of germinal center cells, predominantly centroblasts. The staining pattern of MIB-1 antibody-positive cells in control and mammary gland tissue sections was nuclear (either diffuse or granular), and mitotic figures when present were always strongly labeled (Fig. 4).
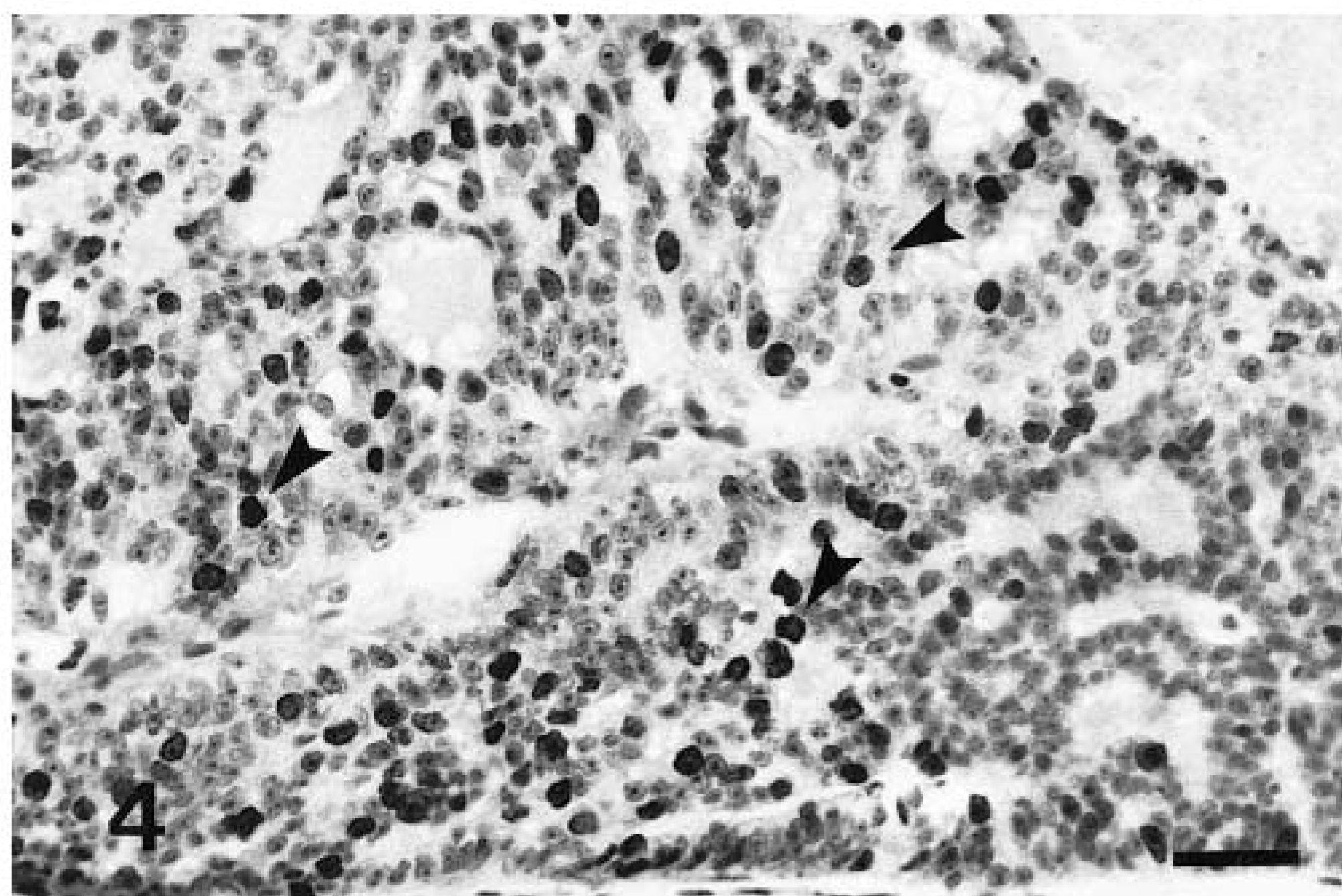
Mammary gland; cat No. 2. MIB-1 immunoreactivity is present in many nuclei (arrows). Streptavidin–biotin–peroxidase method, Harris' hematoxylin counterstain. Bar = 125 µm.
Normal mammary glands showed rare MIB-1–positive cells. The MIB-1 I in dysplastic and neoplastic mammary tissues is presented in Fig. 5. The MIB-1 I was higher in FMH and invasive carcinomas; the latter had an MIB-1 I significantly higher than that of adenosis (P = 0.005) and noninvasive (in situ) carcinomas (P = 0.006). No significant difference in MIB-1 I was found between invasive carcinomas and FMHs.
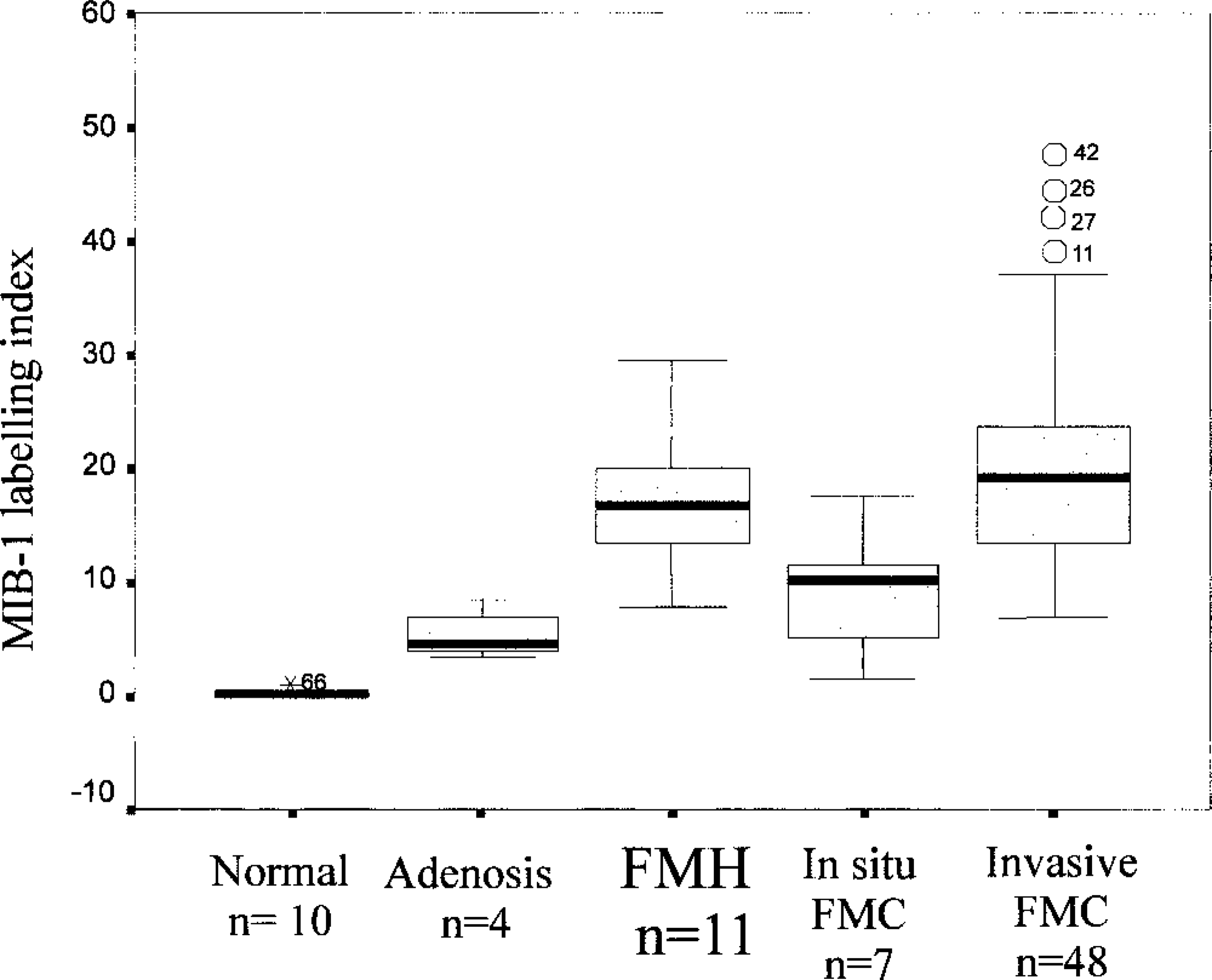
Mib-1 labeling index in normal mammary tissues, mammary adenosis, feline mammary hyperplasia (FMH), and in situ and invasive feline mammary carcinomas (FMC). In the boxplot, the box encloses cases within the 25th to 75th percentiles, the broad horizontal line within each distribution is the median, the upper bar represents the largest observed value that is not an outlier, and the lower bar represents the smallest observed value that is not an outlier. ∘ and identification number = value >1.5 box lengths from the 75th percentile (outlier); ★ and identification number = value >3 box lengths from the 75th percentile (extreme).
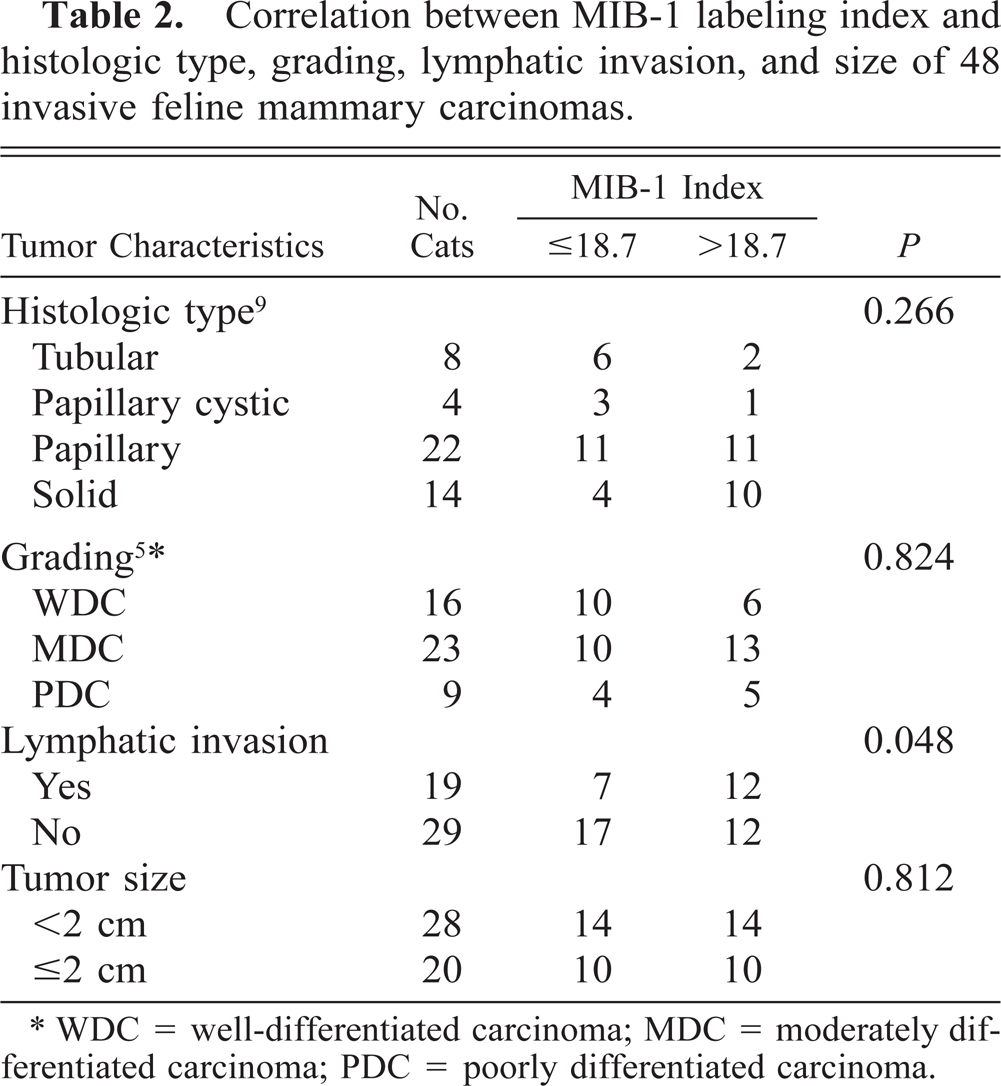
Invasive carcinomas were segregated by MIB-1 I below and above the median value (18.7%), and the relationship with histologic type, grading of tumor, lymphatic invasion, and tumor size is shown in Table 2.
Correlation between MIB-1 labeling index and histologic type, grading, lymphatic invasion, and size of 48 invasive feline mammary carcinomas.
∗ WDC = well-differentiated carcinoma; MDC = moderately differentiated carcinoma; PDC = poorly differentiated carcinoma.
High MIB-1 I showed a borderline significant association with lymphatic vessels invasion (P = 0.048) but no correlation with histologic type, grading, and size of tumor. Concerning histologic type, only 25% of tubular and papillary cystic carcionmas, 50% of papillary carcinomas, and 71.4% of the solid carcinomas had an MIB-1 I higher than 18.7% (Table 2). No differences were detected on the basis of grading or size of tumor.
All the queens with FMH and noninvasive (in situ) carcinoma were alive and well at the end of the 2-year trial, without any sign of local or metastatic recurrence. Twenty-one (43.7%) of the 48 queens with invasive carcinoma (group A) were alive and well at the end of the 2-year follow-up, and 27 (58.2%; group B) had died. At the end of the first year of study, 24 of the 27 queens of group B had already died. There was no significant difference in the age at tumor diagnosis between group A (○ ± SD = 10.3 ± 2.3 years) and group B (10.7 ± 2.8 years) (P = 0.24).
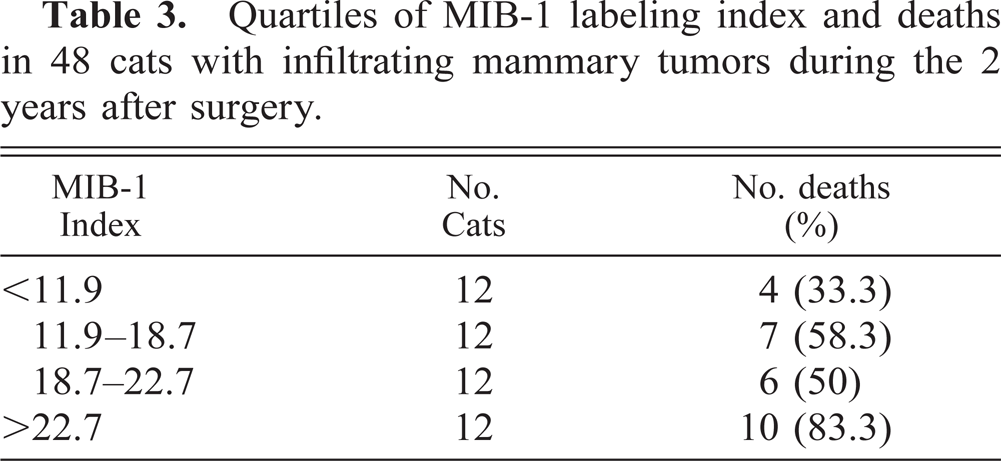
Neoplastic cells of group B queens showed higher MIB-1 I (22.4 ± 10.7) than did those of group A cats (18.7 ± 7.7), but this difference was not significant (P = 0.157). The relationship between MIB-1 I by quartiles and the postsurgery deaths within 2 years is presented in Table 3. Even if a significant association between the survival time and the MIB-1 I was not evident, the queens with an MIB-1 I of >27.2% had a poor prognosis compared with queens of the other quartiles.
Quartiles of MIB-1 labeling index and deaths in 48 cats with infiltrating mammary tumors during the 2 years after surgery.
The lack of correlation between higher MIB-1 I and an unfavourable prognosis was also demonstrated by the estimation of curves for overal survival. There was no significant association between overall survival and higher MIB-1 I (>18.7%) in the group of cats with invasive carcinomas (P = 0.0540; Fig. 6). No significant differences in the course of the disease were observed when the histotype of invasive carcinomas was considered (P = 0.1193).
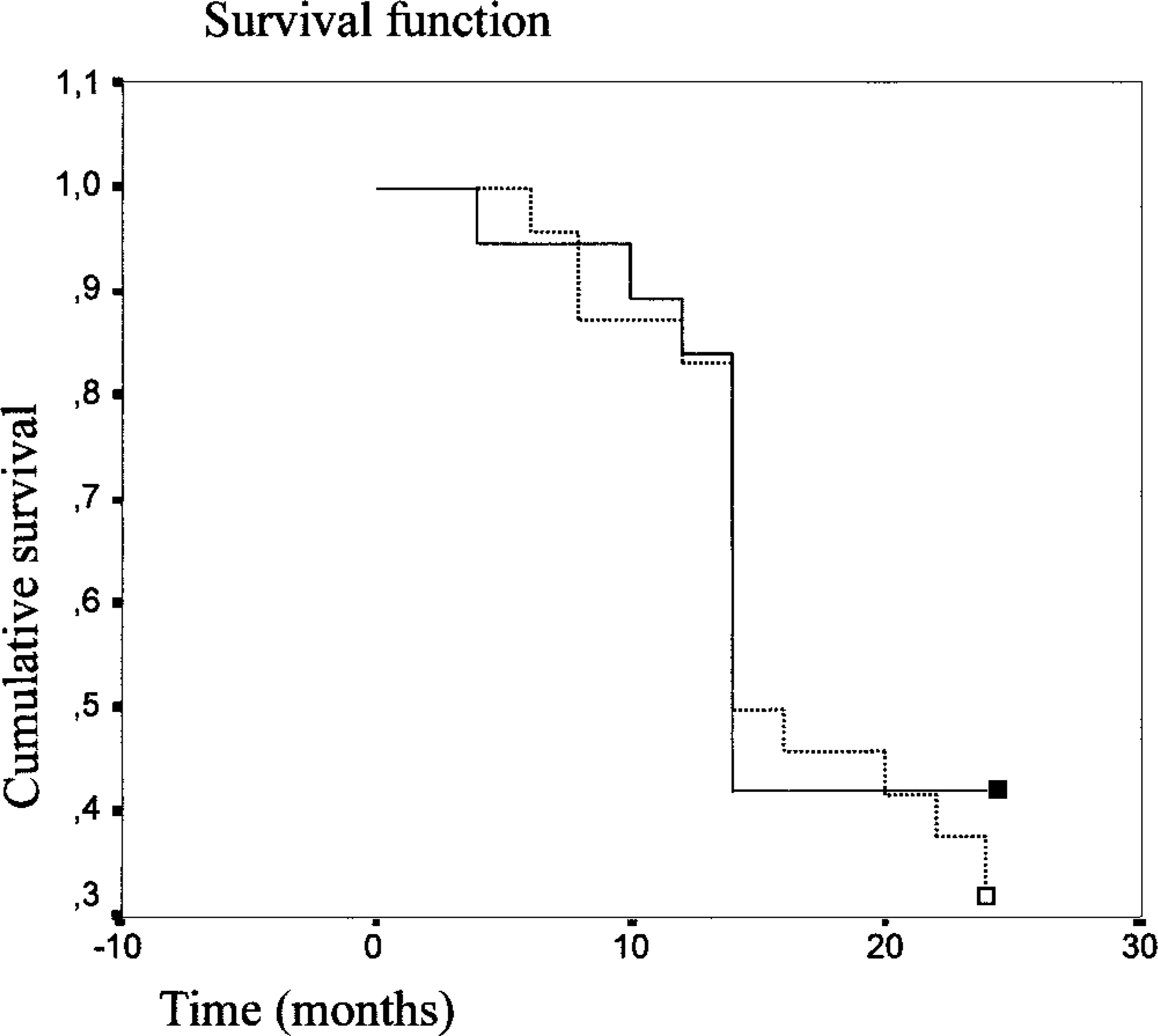
Overall survival curves for the group of 48 queens with invasive carcinomas with low MIB-1 labeling index (≤18.7% □) and high MIB-1 labeling index (>18.7% ▪) (P = 0.0540).
The number of tumor-related deaths at the end of the follow-up period was 6/16 (37.5%) in queens with WDCs, 12/23 (52.2%) in queens with MDCs, and 9/9 (100%) in queens with PDCs. The high predictive value of tumor grading was confirmed by estimation of curves for overall survival; PDCs were associated with a shorter overall survival (P = 0.0040; Fig. 7).
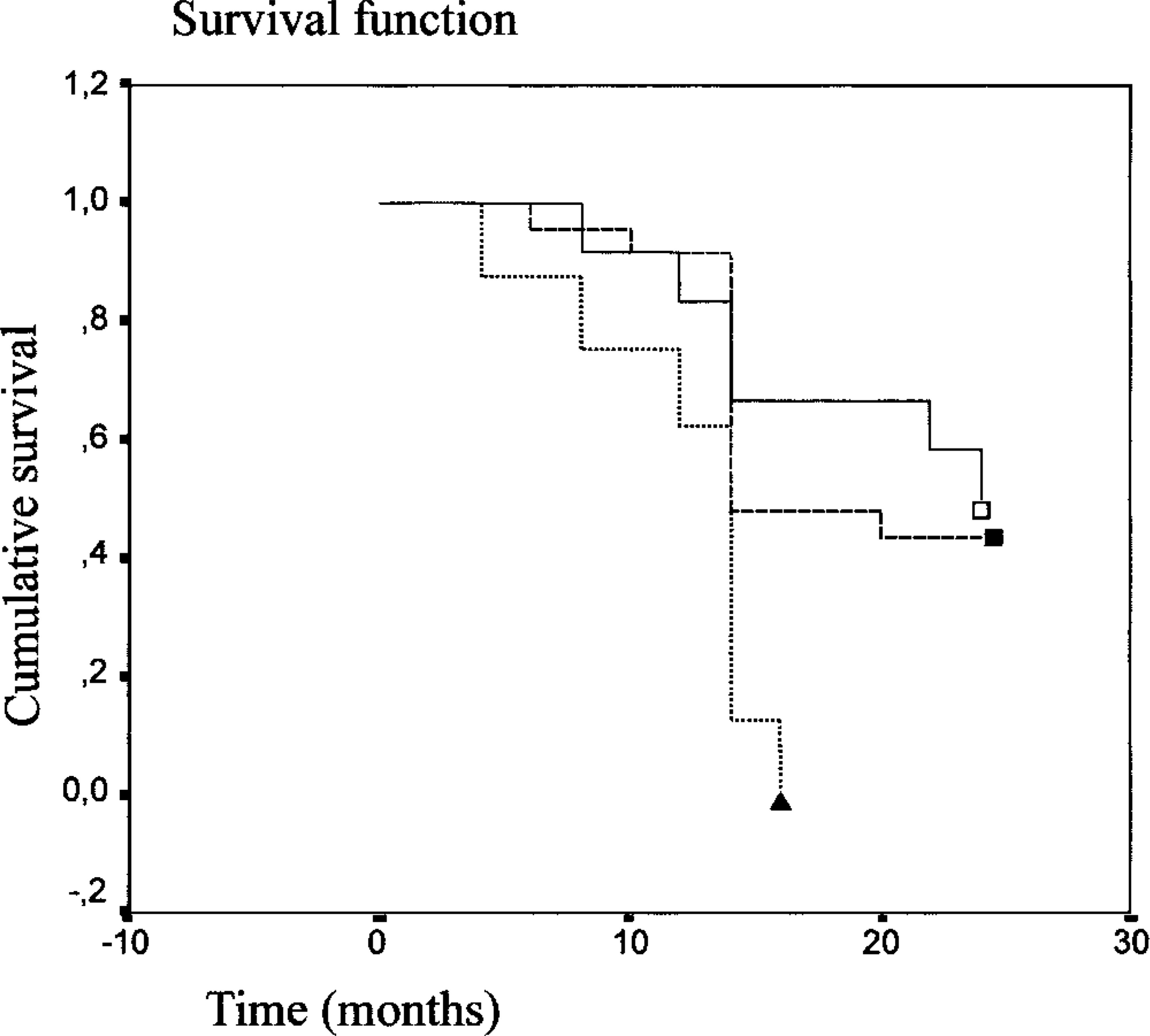
Overall survival curves for the group of 48 queens with invasive carcinomas divided according to the tumor grade. 5 Queens had poorly differentiated carcinomas (▴), moderately differentiated carcinomas (▪), or well-differentiated carcinomas (□) (P = 0.0040).
Discussion
In this study, we examined the relationship between the assessment of cellular proliferation and tumor grading and the clinical behavior of FMCs. All the cats with invasive carcinomas were kept under observation for 24 months after resection. As previously observed, most queens with progressive disease had an overall survival of <1 year. 23 Only 3 of the 24 queens that died during the follow-up survived for >12 months after the detection of a tumor.
Our study confirmed the usefulness of MIB-1 antibody for the assessment of cell kinetics by immunohistochemical localization of the Ki-67 antigen on feline formalin-fixed, paraffin-embedded tissues routinely processed for histochemistry.
Normal mammary gland tissues showed a low proliferative rate (<1%). Mammary tissues with adenosis maintained a proliferative rate similar to that of normal tissues, and noninvasive (in situ) carcinomas had low MIB-1 I. In contrast, MIB-1 I was higher in invasive FMCs and in FMHs.
In spite of these results, a lack of significant prognostic importance of MIB-1 I in mammary carcinomas was found, even though higher mortality was observed in queens with tumors displaying elevated MIB-1 I. As with canine mammary tumors, the prognostic relevance of kinetic parameters for FMCs remains to be determined. Previous studies demonstrated that mitotic index and argyrophilic nucleolar organizer region counts were significantly correlated with the course of mammary carcinomas in cats, 3,23 but few studies have investigated MIB-1 as a prognostic factor for these tumors. A previous investigation, carried out on FMCs, demonstrated that the Ki-67 index was a useful tool for identifying tumors with a more aggressive course. 4 Similar resutls were obtained in a study on canine mammary tumors: multivariate analysis concerning metastasis, disease-free survival, and overall survival revealed that the Ki-67 index had a prognostic value. 19 Notably, however, another study dealing with Ki-67 immunohistochemistry in canine mammary tumors reported a lack of prognostic information of this marker. 15 Even though the diagnostic and prognostic value of Ki-67 immunostaining of human tumors has been widely documented and accepted, 6,12 some researchers have not found an association between Ki-67 and disease-free survival. 14,21 This discrepancy in results has been explained based on the criteria employed in dividing the patients into groups. 12 In canine mammary tumors, the contrasting results concerning the prognostic information on the proliferative activity detected by Ki-67 immunohistochemistry were related to the heterogeneity in the prevalence and biologic behavior of the various histologic types. 4 In the present study, we used a population similar to that employed in the previous study on feline mammary tumors so that the results of the two studies could be compared.
In the present study, other factors such as cat age, tumor histologic type, and tumor grade were not significantly related to MIB-1 I, but MIB-1 expression had a borderline correlation with presence of lymphatic invasion. This finding is in agreement with those of other studies where no correlations were found between proliferation indexes and morphologic appearance of mammary carcinomas in cats 4 and dogs. 20
Animal age and tumor histologic type were also not correlated with survival, but tumor grading, determined using the Elston and Ellis method, 5 had high predictive value, as shown by survival curves. Comparable results have been found in a previous study, 2 especially with respect to grade I and grade III carcinomas.
MIB-1 I measured in paraffin-embedded sections is a useful method for assessing proliferation in a routine setting. However, the results of this and other studies on MIB-1 expression as a prognostic factor for survival in FMC have produced conflicting results, and at this time the data do not support the use of MIB-1 I in clinical decision making. To reach a definitive conclusion on the value of this index, it will be necessary to develop a standardized methodology to compare the results obtained in different studies. More information is needed to determine whether the use of this marker in association with histologic grading will add new prognostic information for the treatment of FMCs.
Footnotes
Acknowledgements
This work was supported by grants from Ministero dell'Universitá e della Ricerca Scientifica, Rome, Italy.