Abstract
The role of c-erbB-2 protooncogene status in feline invasive mammary carcinomas (FMCs) was assessed through the HER-2 receptor immunohistochemical expression. The HER-2 overexpression was then correlated with some relevant histologic parameters and with the clinical course of the disease during a 2-year follow-up. Forty-seven FMCs from surgically treated queens were considered. Tumors were classified according to the WHO criteria and stromal or lymphatic invasion (or both) and histologic grading were recorded. The immunohistochemical staining was performed on paraffin sections and a well-defined scoring system based upon numbers of HER-2 receptors expressed on the cell surface was applied according to standard guidelines. Overall survival (OS) distributions were generated with the Kaplan-Meier method. HER-2 overexpression was detected in 28 of the 47 carcinomas (59.6%). This parameter was demonstrated to be significantly correlated with the shorter OS (
Introduction
Feline mammary carcinomas (FMCs) are one of the most common feline neoplasms and represent an important cause of mortality. The incidence and the morbidity of these tumors has been demonstrated to be very high because their biologic behavior is characterized by rapid growth, high proliferation rates, and capability to metastasize to regional lymph nodes and distant organs. 9 , 13 , 23 In the past decades, there has been increasing interest in investigations for markers that may provide useful information about the course of these malignancies and help in therapeutic options. Among these markers, the role of oncogenes and tumor suppressor genes in the etiology and progression of several carcinomas has been considered in canine and feline mammary tumors. 14 , 16
Her-2/neu (c-erbB-2) is a transmembrane receptor with intrinsic tyrosine-kinase activity, and it is related to the epidermal growth receptor family, believed to play important roles in cancer development. 18 The overexpression of this protein is caused by the amplification of the HER-2/neu protooncogene. 7 The genomic amplification is in fact usually associated with an increased expression of the messenger RNA and of the encoded oncoprotein. The HER-2/neu gene amplification or protein overexpression occurs in about 10–40% of breast carcinomas and is shown to be associated with high clinical tumor aggressiveness and poor prognosis. 15 , 18 , 20 This large percentage spread can also be related to the variety and heterogeneity of the methods in use for analyzing the HER-2 status of a tumor. All methods either measure the degree of gene amplification, the receptor overexpression, or the amount of circulating protein. In practice, immunohistochemistry (IHC) is the most frequently used technique, and many attempts have been made to standardize the methods used for staining and to apply common interpretation criteria. 7 , 19 , 21
In veterinary oncology, the role of c-erbB-2 and of its receptor has been investigated in canine mammary carcinomas and tumor-derived cell lines by both hybridization and immunohistochemical investigations. These preliminary studies suggest that overexpression data of this oncogene show similar relative frequency as that in human mammary cancers, thus pointing out a possible role in the development of malignancy and suggesting its usage as a useful prognostic indicator. 1 , 16 Up to now, there are no reports of the overexpression of HER-2 in feline tissues. The aim of this study is thus to investigate by IHC the possible role of HER-2 status as a marker of malignancy in FMCs and to determine its prognostic value.
Materials and Methods
We report on a retrospective study of 47 queens bearing FMCs, which underwent surgical resection at the Department of Veterinary Pathology of our School. The queens were clinically followed after surgery for a period of 2 years by their referring surgeons and overall survival (OS) was recorded as the period of time from the primary surgery to death by causes related to the mammary tumors. Necropsy was adopted for demonstrating the relation.
Mammary specimens were fixed in buffered formalin and embedded in paraffin blocks. For the histologic examination, 4-µm-thick hematoxylin and eosin–stained sections were evaluated, and tumors were classified according to the WHO criteria 12 and graded according to the recommendation of Elston and Ellis. 4 Stromal or lymphatic (or both) invasion was also noted.
IHC for Her-2/neu was performed using the streptavidin–biotin–peroxidase method. From each block, 4-µm-thick sections were cut on coated slides (Superfrost plus, Menzel-Gläser, Germany) and dried at 37 C for 24 hours. The sections were deparaffinized and rehydrated in distilled water through a series of graded alcohols and were then treated with a solution of Endoblocker (Biømeda Corp., Foster City, CA, USA) to exhaust endogenous peroxidase activity. After microwave cycling (three exposures, 5 minutes each, 750 W), a preincubation of 30 minutes with normal horse serum (NHS) diluted 1 : 10 in phosphate-buffered saline (PBS) and 1% bovine serum albumin (BSA) was performed; then the primary mouse monoclonal antibody anti-human c-erbB-2 oncoprotein (Dako, Glostrup, Denmark) diluted 1 : 250 in PBS-1%BSA was applied for 1 hour at room temperature. The sections were reincubated with NHS-1%BSA for 30 minutes and subsequently with a biotinylated, pan-specific secondary antibody made in horse (Vector Lab., Burlingame, CA, USA). Furthermore, the sections were incubated with a streptavidin-biotinylated horseradish peroxidase complex (Biospa, Milan, Italy) diluted 1 : 250 in PBS for 10 minutes. The peroxidase reaction was developed using Nova Red kit (Vector Lab.) and the sections were lightly counterstained with hematoxylin and then mounted. Between each step in the staining procedure (except before the incubation with the primary and secondary antibody), the slides were rinsed three times in PBS. The positive controls for each run consisted of one section from a human breast carcinoma known to overexpress the receptor. As a negative control, the primary antibody was replaced with an irrelevant, isotype-matched antibody, to control for nonspecific binding of the secondary antibody.
Slides were independently submitted to blind scoring by two independent pathologists. Any discordant interpretation was settled using a multiviewer microscope. The results of the immunohistochemical and clinicopathologic findings underwent later evaluation for correlation. Overexpression of HER-2/neu was defined as membranous staining in more than 10% of neoplastic cells, and staining was examined over the maximum area of staining intensity in a semiquantitative fashion as previously described, 8 , 17 , 19 according to the following criteria: 0 = no staining; + 1 = weak, incomplete membranous staining; + 2 = moderate, complete membranous staining of at least 10% of tumor cells; and + 3 = strong membranous staining of at least 10% of tumor cells. Cytoplasmic staining was disregarded as nonspecific. Cases interpreted as 0 or + 1 were considered negative, whereas scores of + 2 or + 3 were considered to indicate HER-2 overexpression.
The statistical analysis was performed using the statistical package SPSS Advanced Statistics 7.5 (SPSS Inc., Chicago, IL, USA). Chi-square tests were used to assess associations between categoric variables. Statistical significance was based on a 5% significance level. OS curves were prepared by the Kaplan-Meier method and survival analysis was performed using the Tarone-Ware test.
Results
The mean age at the time of diagnosis of the 47 queens was 10.9 years ± 2.9 population SD (range: 4–18 years). At the end of the clinical follow-up, 15 queens (31.9%) were still alive and 32 (68.1%) had died of mammary carcinoma (i.e., total 100%, no deaths by extraneous causes).
Tubulopapillary carcinoma was the most common type (33 of 47, 70.2%), whereas solid carcinomas were less frequently observed (14, 29.8%). Histologic grading of the 47 carcinomas investigated revealed that 12 (25.5%) were well-differentiated carcinomas (WDCs; grade I), 27 (57.4%) were moderately differentiated carcinomas (MDCs; grade II), and eight (17%) were poorly differentiated carcinomas (PDCs; grade III). The relationship between the results of histologic classification and tumor grading is detailed in Table 1. All the tubulopapillary carcinomas were WDCs or MDCs, and solid carcinomas were MDCs and PDCs. The presence of lymphatic invasion was observed in 27 of the 47 carcinomas (57.4%), whereas in the other 20 carcinomas (42.6%) only a local stromal invasion was detected.
Relationship between the tumor histologic type according to the WHO classification12 and the grading of the invasive FMCs4 ∗.

∗ FMCs = feline mammary carcinomas; WDC = well-differentiated carcinoma; MDC = moderately differentiated carcinoma; PDC = poorly differentiated carcinoma.
The antibody we used highlighted membranous tumor cell staining. Only in a few cases was a weak cytoplasm staining detected. The cell membrane staining pattern was observed exclusively in neoplastic cells, and positive staining was mostly distributed uniformly throughout the tumors. No neighboring hyper-plastic or normal epithelial cells showed membrane staining for the HER-2.
The staining pattern characteristics of tumors scoring of + 1, + 2, and + 3 are shown in Fig. 1. The HER-2 expression was detected in 28 carcinomas (59.6%): 21 tumors scored + 2 and seven + 3. Essentially all cases with HER-2 scores of + 3 showed positive staining in the vast majority (> 90%) of tumor cells. Table 2 summarizes the immunohistochemical results and lists the incidence of HER expression for each tumor characteristic considered. HER-2/neu status did not significantly correlate with histologic type, tumor grading, or presence of lymphatic invasion. On the contrary, the status was significantly correlated with the shorter OS (
Correlation between HER-2 status, determined by IHC, and histologic type, tumour grading, lymphatic invasion, and OS.∗
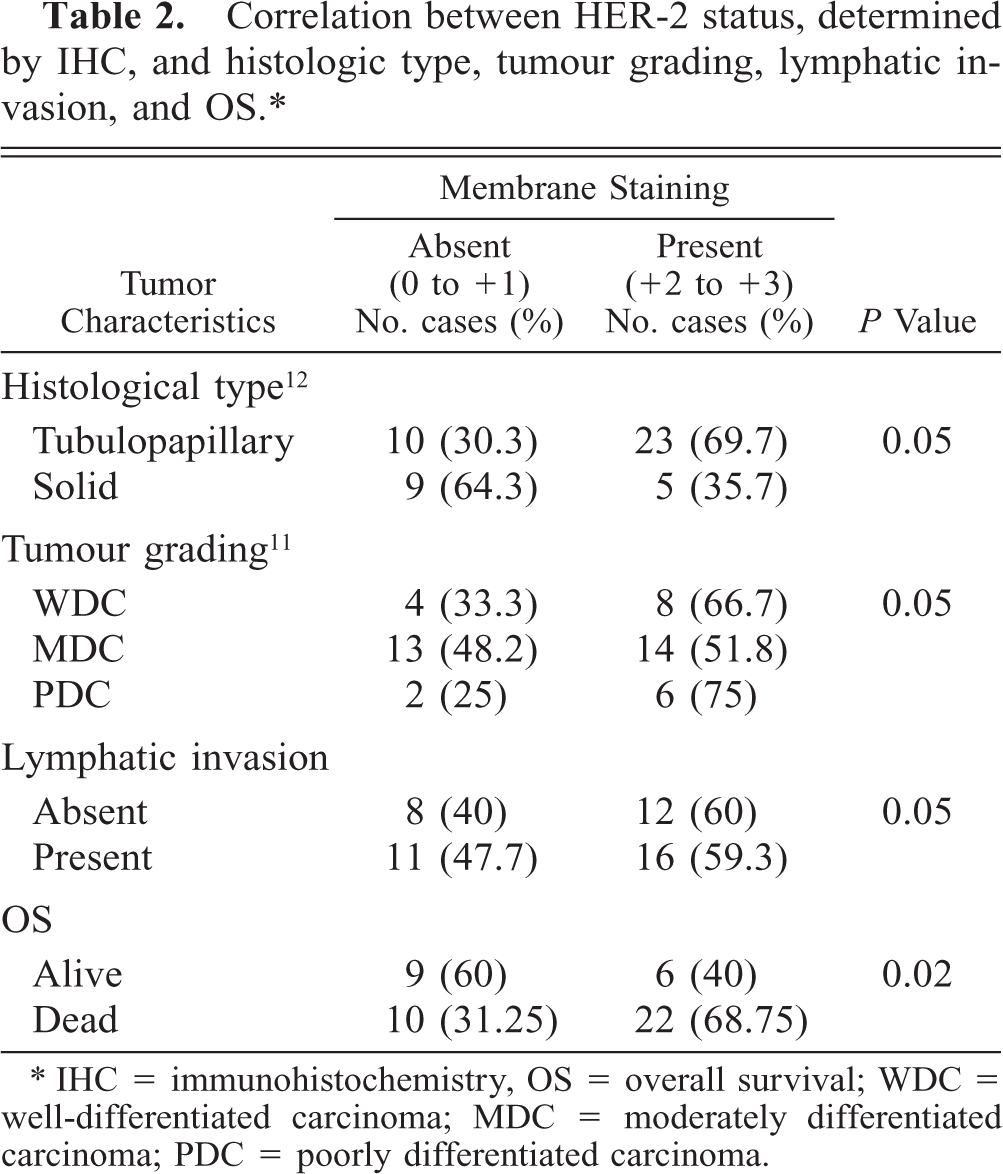
∗ IHC = immunohistochemistry, OS = overall survival; WDC = well-differentiated carcinoma; MDC = moderately differentiated carcinoma; PDC = poorly differentiated carcinoma.
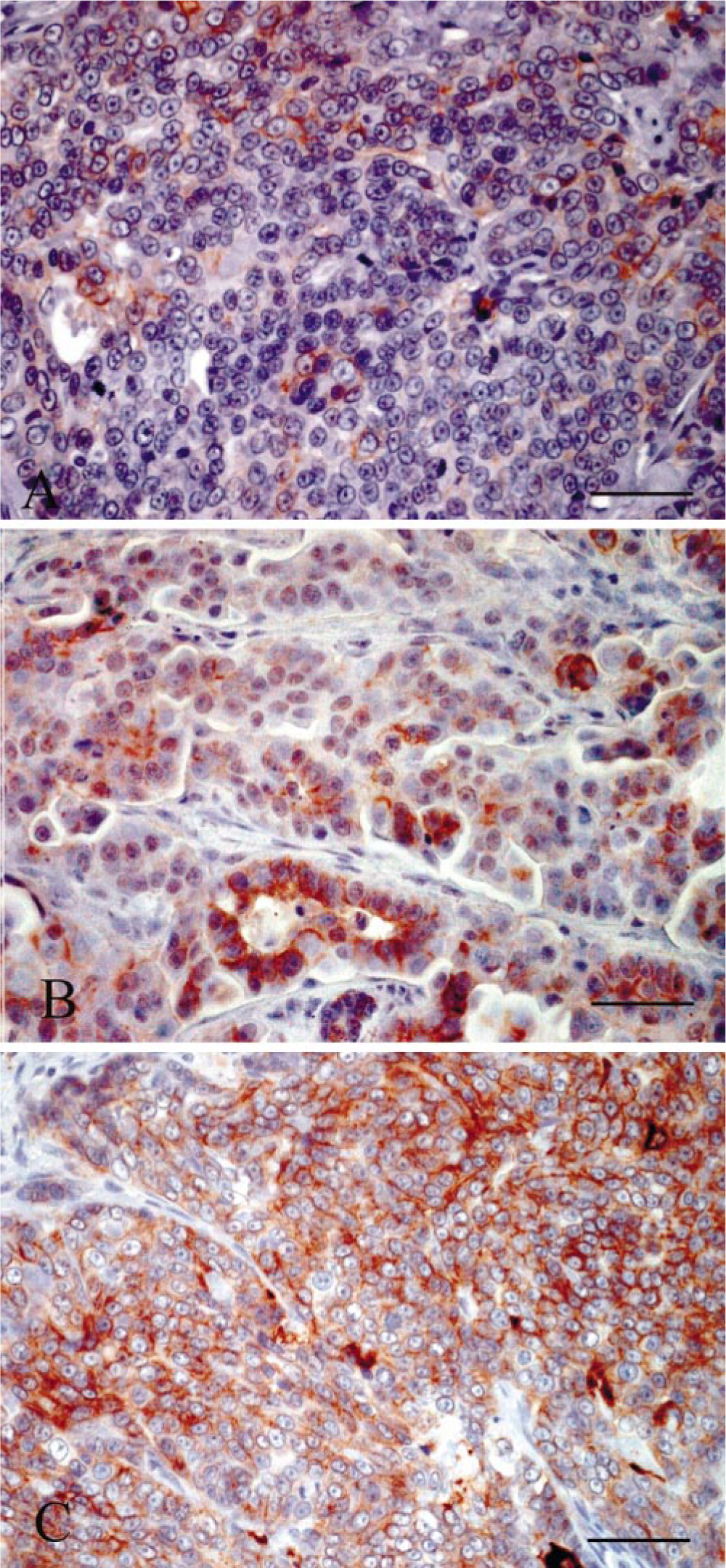
Mammary gland; Queens bearing invasive carcinomas, subjects Nos. 6, 20, and 31. HER-2 expression detected by IHC using a mouse monoclonal antibody anti-human c-erbB-2 oncoprotein. (A) Incomplete membranous staining of weak to moderate intensity (score 1). (B) Complete membranous staining of moderate intensity in at least 10% of the tumor cells (score 2). (C) Complete membranous staining of strong intensity in more than 10% of tumor cells (score 3). Anti–HER-2 immunoperoxidase histochemistry and hematoxylin. Bar = 100 µm.
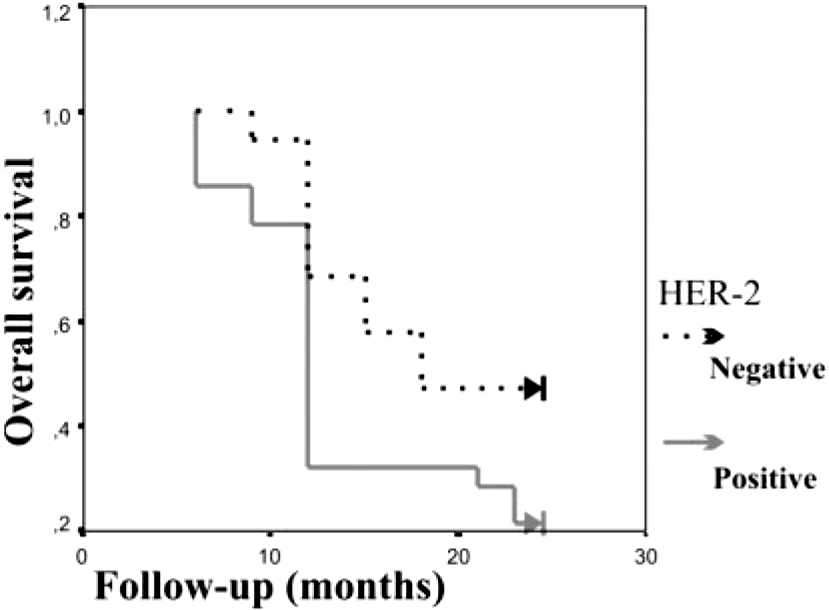
OS curves for the group of 47 queens with invasive mammary carcinomas scoring negative (→) and positive (→) for HER-2 overexpression. (
Discussion
In this study we have investigated the immunohistochemical status of HER-2 receptor and its prognostic potential in FMCs. The high death rate recorded (68.1%) confirms the malignant behavior of these tumors and suggests the importance of selecting appropriate markers having a potential to add useful information on the biologic behavior of the disease and on possible therapeutic options.
In human oncology, many studies have demonstrated that gene amplification of HER-2/neu or overexpression of its receptor (or both) are linked to shorter disease-free intervals, increased risk of metastasis, and resistance to many types of therapy. The receptor activation, in fact, potentiates tumor cell motility, protease secretion, and invasion and also modulates the cell cycle checkpoint function, the DNA repair, and the apoptotic responses. 3 The HER status of a tumor of the breast is of clinical importance; in fact, anticancer therapies targeted to specific tumor-associated gene products are expected to be effective in individualizing therapeutic regimens. Recently, a humanized anti–HER-2/neu monoclonal antibody has been developed as a drug that binds to the HER-2/neu protein and inhibits the enhanced tumor cell growth 2 : thus, it has been introduced for the treatment of carcinomas overexpressing HER-2/neu. 5 , 22 It is therefore important to accurately evaluate the status of HER-2/neu. In this sense, many studies have indicated that analyses at both DNA and protein levels are equally useful. 7 , 21 The IHC lends itself as a simple and reliable method for examining the protein expression status and it proves to be correlated with other standard methods such as the fluorescent in situ hybridization. 6 , 7
In this study, 56.9% carcinomas expressed HER-2 at the + 2 and + 3 levels. This percentage was higher than that of canine mammary tumors reported in a previous study, 16 but this discrepancy might originate from some lack of standardization of the methodologies. The authors, in the above reference, in fact, defined as positive the specimens showing more than 20% immunoreactivity and, in addition, considered any cytoplasmic staining as a sign of immunoreactivity. In this study, we have assessed the HER-2 expression using a standardized method already adopted in the routine evaluation of the HER-2 status in human oncology. 8 However, the percentage of feline carcinomas overexpressing the receptors was higher than that in humans. 18 These preliminary results definitely support the malignant behavior of this tumor in the feline species and suggest a possible important role of HER-2 overexpression as a marker of malignancy of FMCs. Thus, standardizing the staining methods appears to be of critical importance and common interpretation criteria should be defined to enable direct comparison of the results. The statistically significant correlation between HER-2 overexpression and shorter OS also suggest that HER-2 status could provide valuable prognostic and predictive information. As suggested by Martin de las Mulas et al. with respect to HER-2 expression in canine mammary tumors, 10 FMCs can be also considered as a suitable natural model of the subset of human breast carcinomas that overexpress the HER-2 protein. Although a larger study is required to strengthen the predictive ability of this marker, the assessment of HER-2 overexpression can be viewed as a possible tool for the selection of patients who are likely candidates for specific anti–HER-2 therapy. 11 , 21
Footnotes
Acknowledgements
This work was partially supported by the Department of University and Scientific and Technological Research of the Italian Government, Rome, Italy.
