Abstract
Feline mammary tumors are usually malignant and aggressive carcinomas. Most cases are simple monophasic carcinomas (1 epithelial population), and additional phenotyping is usually not needed. In this study, we describe 10 malignant mammary tumors from 9 female cats that had unusual histomorphology: they appeared biphasic, with 2 distinct cell populations. Initially, they were morphologically diagnosed as either carcinosarcoma (1/10) or malignant pleomorphic tumor (9/10) of the mammary gland, as the latter did not match any previously described histological subtype. Immunohistochemistry (IHC) was performed for pancytokeratin, cytokeratins 8 and 18, cytokeratin 14, cytokeratins 5 and 6, vimentin, p63, calponin, alpha-smooth muscle actin, Ki-67, ERBB2, estrogen receptor alpha, and progesterone receptor. In 7 of 10 cases, the biphasic nature was confirmed and, on the basis of the IHC results, they were classified as carcinoma and malignant myoepithelioma (4/10), ductal carcinoma (1/10), and carcinosarcoma (2/10). The other 3 of 10 cases were monophasic based on IHC. In the cases of carcinoma and malignant myoepithelioma, the malignant myoepithelial cells were 100% positive for vimentin (4/4) and variably positive for p63, calponin, and cytokeratins (4/4). These findings show that, although rare, biphasic mammary carcinomas do occur in cats. In dogs and humans, tumors composed of malignant epithelial and myoepithelial cells have a less aggressive behavior than certain simple carcinomas, and therefore, their identification might also be clinically significant in the cat.
Mammary tumors are the most common neoplasms of female dogs and the third most common of female cats. 5,20,41,50 In dogs, half of mammary tumors are malignant, whereas 80% to 96% of feline mammary tumors are aggressive carcinomas. 41,50,61 Morphologic classification has prognostic value in both human breast cancer 26 and canine mammary tumors, 35,37 and therefore, standardization with detailed description of histologic subtypes has been progressively updated for these species. 13,14,26,31,48,49 Mammary tumors in cats are much less heterogeneous than those in dogs and humans, and identification of subtypes is less problematic. A specific prognostic importance of histopathologic classification in the cat 14,31,60 has not been determined. 61 However, new feline histologic subtypes have been described and may have prognostic significance. 15,44,45,59
One of the major differences between canine and feline mammary tumors is the predominance of monophasic (only 1 cell population) tumors in the cat and of biphasic and triphasic tumors in the dog, in which 2 or 3 cell populations proliferate simultaneously. More specifically, a frequent feature of canine mammary tumors is the concomitant proliferation of luminal epithelial cells and interstitial myoepithelial cells and at times also in association with mesenchymal cells (triphasic tumors). 14
Myoepithelial cells are contractile elements manifesting both epithelial and smooth muscle characteristics. In the normal mammary gland, they reside on the basement membrane below epithelial cells. A classification of myoepithelial cell subtypes has been suggested in the dog by some authors, 2,6,7 and it has been proposed in the recently published CL Davis Foundation classification of feline and canine mammary tumors. 60 Resting and proliferative suprabasal myoepithelial cells and spindle-to-stellate and round-to-oval interstitial myoepithelial cells have been identified. Suprabasal myoepithelial cells can be recognized on hematoxylin and eosin (HE)–stained sections as cells located above the basement membrane with either flattened (resting) or bulging (proliferative) morphology. 2,60 They typically express cytokeratins (CK; eg, CK19, CK5/6, CK14), p63, alpha smooth muscle actin (SMA), calponin, and vimentin, whereas luminal epithelial cells normally face the glandular lumen and express CK8/18 and multifocally also express CK5/6, CK14, and—only in the cat, both in the normal and in the neoplastic gland—vimentin. 3 Spindle-to-stellate interstitial myoepithelial cells are instead typically intermingled with the basophilic mucinous matrix and expand into the interstitium. 2,60 They are found only in canine complex and mixed tumor, and they can lose the expression of suprabasal myoepithelial cell markers, progressively becoming mesenchymal-like and retaining only vimentin expression (triphasic tumors). 6,7,42 In human breast cancer, normal or near-normal myoepithelial cells have a protective role in suppressing tumor aggressiveness, 17,33 and in the dog complex and mixed carcinomas, they show a better prognosis than other carcinomas. 31,37,40,42 Round-to-oval interstitial myoepithelial cells have been identified in ductal adenomas and carcinomas in the cat and in the dog. 59,60 These myoepithelial cells retain the morphology and phenotype of suprabasal myoepithelial cells but extend into the interstitium, where they can form solid areas and are not associated with the mucinous matrix of spindle-to-stellate interstitial myoepithelial cells. 59,60 Because of their morphology and phenotype, which is identical to that of suprabasal myoepithelial cells, they have been previously classified as basal cells, even if they were extending into the interstitium. 13,14 Ductal tumors have been previously classified as basaloid adenoma in the dog 31 and as feline complex tumors in the cat. 43,45
A third type of interstitial myoepithelial cells has been identified only in dogs until now: they become pleomorphic and variably express suprabasal myoepithelial cells markers. 39 They will be named here as pleomorphic interstitial myoepithelial cells, and they have been described as malignant myoepithelial cells in both biphasic (carcinoma and malignant myoepithelioma) and monophasic (malignant myoepithelioma) tumors. 37,60
Neither carcinoma and malignant myoepithelioma nor malignant myoepithelioma have been described in the cat, in which, in addition to ductal tumors, the only other biphasic mammary tumor is the carcinosarcoma. 14,31,60
In this study, we investigated 10 feline mammary tumors that had an unusual morphology on HE-stained sections with a biphasic malignant appearance. Therefore, immunohistochemistry (IHC) was performed to ascertain the nature of the cell populations and to better classify the tumors. The aim was to determine if, in the cat, there are biphasic tumors with malignant epithelium and malignant myoepithelium. Literature reports of biphasic and triphasic tumors in humans and dogs were also reviewed to better define their cellular components.
Materials and Methods
Samples and Histopathology
Mammary gland samples analyzed in this retrospective work were selected from the archives of the Diagnostic Service of the Veterinary Anatomical Pathology unit of the University of Padua, Italy. The samples were received between 2011 and 2017. They were from 9 female cats that presented to referring veterinarians for 1 or more mammary nodules (Table 1). Surgery was performed for both diagnosis and therapy, and no additional sampling was carried out for this study. Owners signed an informed consent form to use these samples for research. The cases included in this study were cats that had mammary tumors in which the routine histopathology examination suggested 2 neoplastic cell populations. Histologic examinations and classification were performed by 3 European College of Veterinary Pathologists–certified veterinary pathologists (R.R., D.C., V.Z.) following the updated World Health Organization classification. 14 Concomitant mammary tumors of other subtypes and/or hyperplastic/dysplastic mammary lesions were also assessed as well as specific tumor-associated morphological features (Table 1). Lymphovascular invasion was evaluated both within and external to the tumor. Infiltration of peripheral tissues was identified as an irregular contour of the tumor margins where the neoplastic cells had invaded the periphery, eliciting a stromal (scirrhous) reaction. The presence of peripheral lymphocytes was also determined. The size of each lesion was measured at histology as the greatest diameter of the sectioned nodule. The mitotic count was calculated as recently suggested 30 as the sum of mitoses in 10 consecutive high-power fields (HPFs; diameter of the field of view = 0.55 mm; HPF = 237 mm2; 40× magnification; Olympus BX40 microscope) starting from the most mitotically active areas. Only fields with representative tumor cells were assessed. If, after counting mitoses in the first HPF, no mitoses were found in the next 3 consecutive HPFs, the process was repeated by moving to another mitotically active area. One single value was calculated for each nodule; that is, the mitotic count was obtained by counting the mitoses in both of the presumptive neoplastic cell populations. Tumor malignancy was determined based on the following criteria: anisocytosis and anisokaryosis of the cells ranging from mild to severe (10/10 cases), mitotic count ≥3 (10/10 cases), partial/total loss of tubular arrangement (6/10 cases), presence of multifocal necrosis (6/10 cases), peripheral infiltration (7/10 cases), and vascular invasion (2/10 cases; Table 1; Suppl. Fig. S1, inset). These criteria were chosen by the authors based on previous prognostic studies. 23,43,44,61 Since there is no grading system for feline biphasic mammary tumors, grading was performed according to the work by Castagnaro and coworkers. 4 Tubule formation was assessed as an overall percentage of the tumor area, whereas nuclear pleomorphism was assessed in both presumptive populations. Regional lymph nodes were available for only 3 of 9 cases.
Signalment and Pathologic Features in 9 Feline Biphasic Mammary Tumors.

Abbreviations; CS, carcinosarcoma; DSH, domestic short hair; F, female; NR, nonreported; PT, malignant pleomorphic tumor; S, spayed.
a In 10 high-power fields. Consecutive fields are selected at the periphery or the most mitotically active parts of the sample. Diameter of the field of view = 0.55 mm; high-power field = 0.237 mm2.
b One focal infiltrate of less than 30 lymphocytes.
On routine histology, 2 malignant neoplastic cell populations were observed in at least 1 nodule per case. In all 10 cases, evidence of malignancy was present in both cell populations (Suppl. Table S1).
In all tumors, 1 cell population was clearly arranged in tubules and nests (Figs. 1a and 2a; Suppl. Figs. S1, S5), cordons (case 7; Fig. 3a; Suppl. Figs. S9 and S10), or papillae (case 9) and was therefore designated as a luminal epithelial population (Suppl. Table S1). The second cell population expanded the interstitial space in a looser nonadherent fashion and was designated as an interstitial population. Morphological details of both populations are described in Supplemental Table S1.
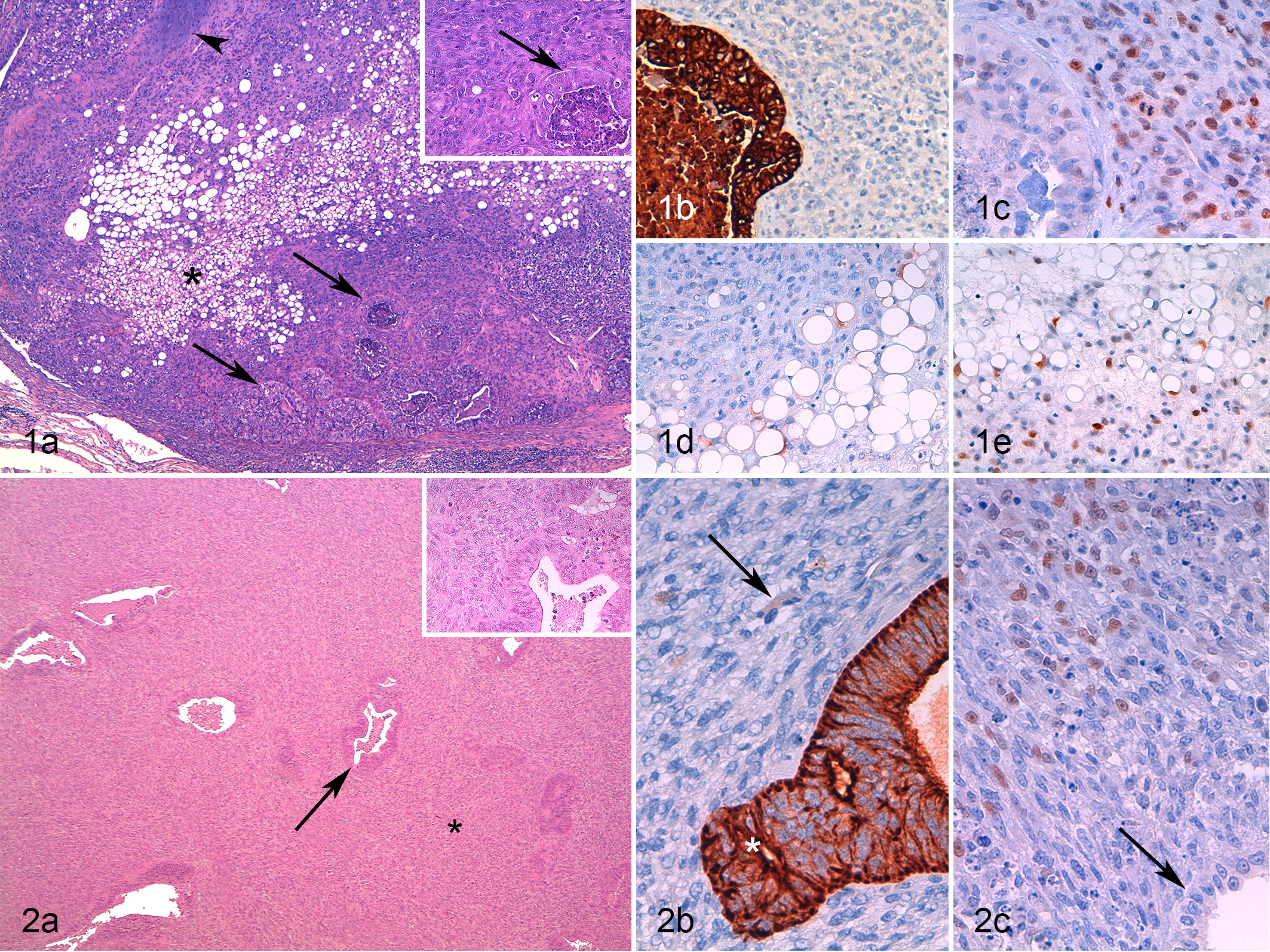
In case 4, 1 large (3-cm-diameter) nodule contained a cuboidal to columnar cell population arranged in irregular tubules and nests associated with a spindle to round-oval pleomorphic interstitial cell population intermingled with variable amounts of multifocally chondroid extracellular matrix. The interstitial cells also had areas of adipose tissue-like differentiation, with cells containing well-defined empty vacuoles ranging from 20 to 100 µm in their cytoplasm (Suppl. Table S1). This lesion was considered morphologically unusual, and a diagnosis of carcinosarcoma was suggested (Fig. 1a).
In the remaining 9 of 10 cases, a diagnosis of malignant pleomorphic tumor of the mammary gland was given, including a comment that explained the possible biphasic malignant nature of the tumor, similar to the description of carcinoma and malignant myoepithelioma in the dog. In all 9 of 10 cases, the interstitial cells showed variable pleomorphism and usually minimal association with an extracellular matrix (Suppl. Table S1; Figs. 1, 2, 4a, 5a; Suppl. Figs. S1, S5, S10). None of these feline cases had morphological features resembling spindle-to-stellate interstitial myoepithelial cells of complex and mixed mammary tumor of the dog.
In addition to the biphasic nodules, other nodules with more usual morphological features were diagnosed in 5 cases as simple tubular carcinoma, simple tubulopapillary carcinoma, intraductal papillary carcinoma, and cribriform carcinoma. In 1 cat (case 4), there were 7 nodules of simple tubular carcinoma. In 1 of 3 available lymph nodes, a metastasis of a presumptive pleomorphic tumor was observed.
Immunohistochemistry
IHC was performed on serial sections (4 μm) mounted on Superfrost Plus glass slides (Menzel GmbH, Braunschweig, Germany). A panel of antibodies was applied to all of the mammary nodules and to 1 lymph node metastasis (case 4). The panel included antibodies raised against human antigens that had been already tested in the cat. 3,38,59 The following antigens were analyzed: pan-CK (AE1/AE3, 1 in 100 dilution, Dakocytomation, Glostrup, Denmark). CK8/18 (NCL-L-5D3, 1 in 30 dilution, Novocastra, Newcastle Upon Tyne, UK), CK 14 (NCL-LL 002, 1 in 20 dilution, Novocastra), CK5/6 (D5/16 B4, 1 in 100 dilution, Dakocytomation), calponin (1 in 200 dilution, Dakocytomation), alpha-SMA (1A4, 1 in 100 dilution, Dakocytomation), vimentin (V9, 1 in 150 dilution, Dakocytomation), p63 (4A4, 1 in 200 dilution, Santa Cruz Biotechnology, Heidelberg, Germany), Ki-67 (MIB-1, 1 in 50 dilution, Dakocytomation), ERBB2 (A0485, 1 in 250 dilution, Dakocytomation), estrogen receptor alpha (ER alpha; NCL-ER-6F11, 1 in 40 dilution, Novocastra), and PR (1E2, ready to use, Ventana Medical System, Tucson, AZ). An automated immunostainer (BenchMark, Ventana Medical System) was used, and the protocols were performed as previously described. 3,38,59
The haired skin and the nonneoplastic mammary tissue adjacent to the nodules were used as positive internal controls for all of the antibodies, except for ERBB2, for which an external technical positive control was adopted (ERBB2 3+ human breast cancer) and for which the species-specific cross-reactivity had been previously described, 38 and ER and PR, for which feline uterus and ovary sections were immunolabeled.
For tumors in which the presence of 2 different neoplastic cell populations was confirmed, the IHC results were assessed separately for each population. Nuclear immunolabeling for p63, Ki-67, ER, and PR and cytoplasmic immunolabeling for pan-CK, CKs 8/18, CKs 5/6, CK14, SMA, calponin, and vimentin were assessed as the percentage of positive cells, counting 1000 cells in at least 10 random HPFs. The intensity of staining was classified as strong or mild. ERBB2 immunolabeling was scored as 0, 1+, 2+, or 3+ according to the American Society of Clinical Oncology 2018 recommendations (10% cutoff), with 2+ and 3+ cases considered weakly and strongly positive for complete membrane staining, respectively. 54
Follow-up Data
Referring veterinarians were contacted (June 2018) by both phone and email to monitor any significant clinical outcomes such as the development of new nodules in the remaining glands, the development of local recurrence, distant metastases, or tumor-associated death. Because of the low number of cases with data, no statistical analyses were performed.
Results
Immunohistochemistry
Results of IHC analyses are summarized in Table 2. Since calponin and SMA staining overlapped in 9 of 10 cases, results are shown only for calponin.
Immunohistochemistry (IHC) of the Mammary Tumors Included in the Study.a
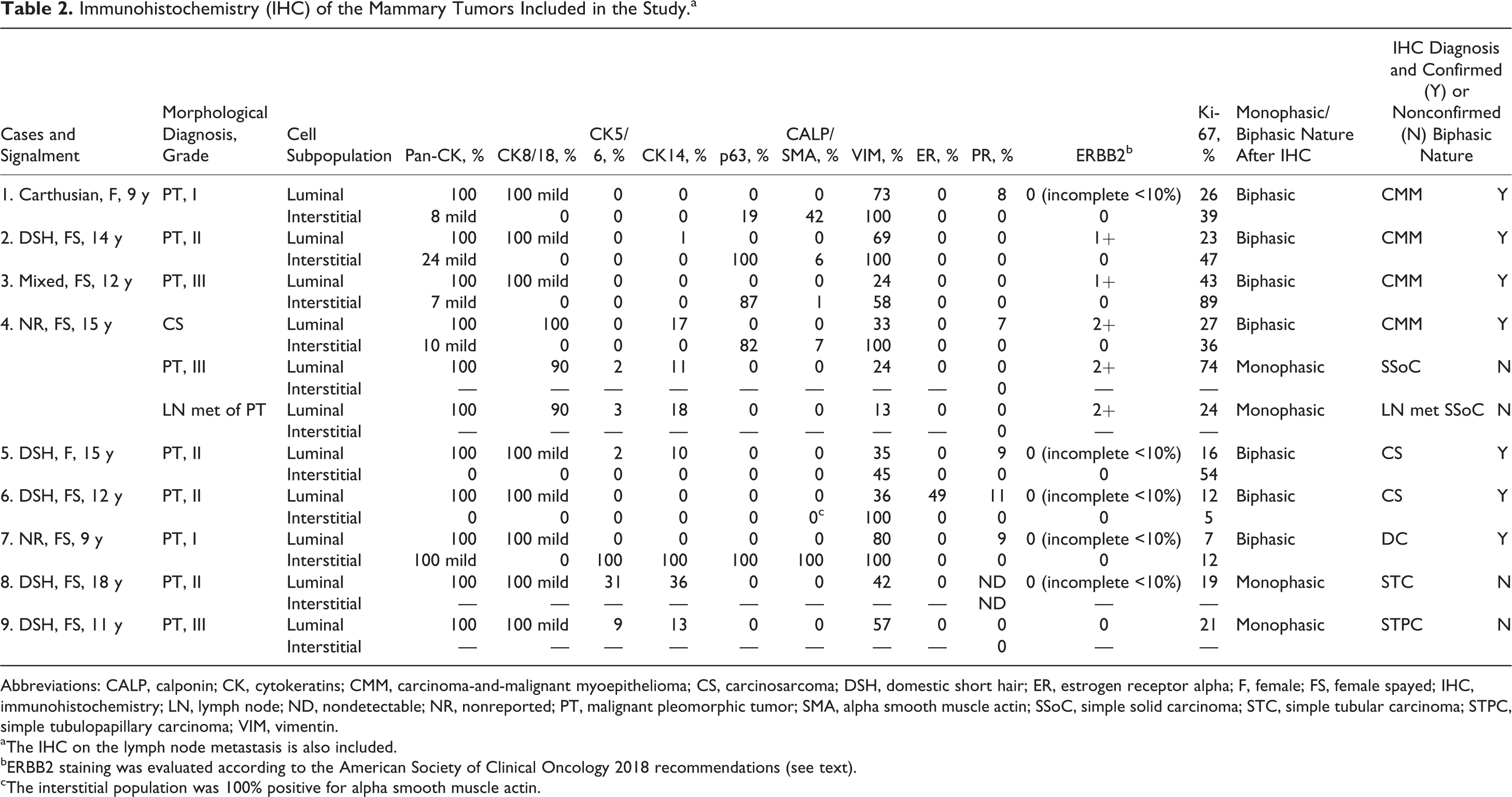
Abbreviations: CALP, calponin; CK, cytokeratins; CMM, carcinoma-and-malignant myoepithelioma; CS, carcinosarcoma; DSH, domestic short hair; ER, estrogen receptor alpha; F, female; FS, female spayed; IHC, immunohistochemistry; LN, lymph node; ND, nondetectable; NR, nonreported; PT, malignant pleomorphic tumor; SMA, alpha smooth muscle actin; SSoC, simple solid carcinoma; STC, simple tubular carcinoma; STPC, simple tubulopapillary carcinoma; VIM, vimentin.
aThe IHC on the lymph node metastasis is also included.
bERBB2 staining was evaluated according to the American Society of Clinical Oncology 2018 recommendations (see text).
cThe interstitial population was 100% positive for alpha smooth muscle actin.
In all cases (10/10), luminal epithelial cells were immunolabeled for pan-CK (100% positive cells; Figs. 1b, 2b, 4b, 5b; Suppl. Figs. S2, S6, S13), CK8/18 (90%-100% positive cells), and vimentin (13%-80% positive cells; Suppl. Figs. S4 and S7). In 7 of 10 cases, luminal epithelial cells showed positivity for CK14 (1%-36% positive cells) and in 5 of 10 cases for CK5/6 (2%-31% positive cells; Suppl. Fig. S12).
In 3 cases (cases 1, 6, and 7), the luminal epithelial population was the only neoplastic cell population detected on IHC. This result invalidated the presumptive morphological diagnosis of biphasic tumors based on HE (Fig. 4a), and a diagnosis of monophasic (simple) tumors was made. Based on morphology and IHC, 1 simple solid carcinoma, 1 simple tubular carcinoma, and 1 simple tubulopapillary carcinoma were diagnosed in these cases. The lymph node metastasis from case 4 was diagnosed also as a monophasic simple solid carcinoma based on IHC.
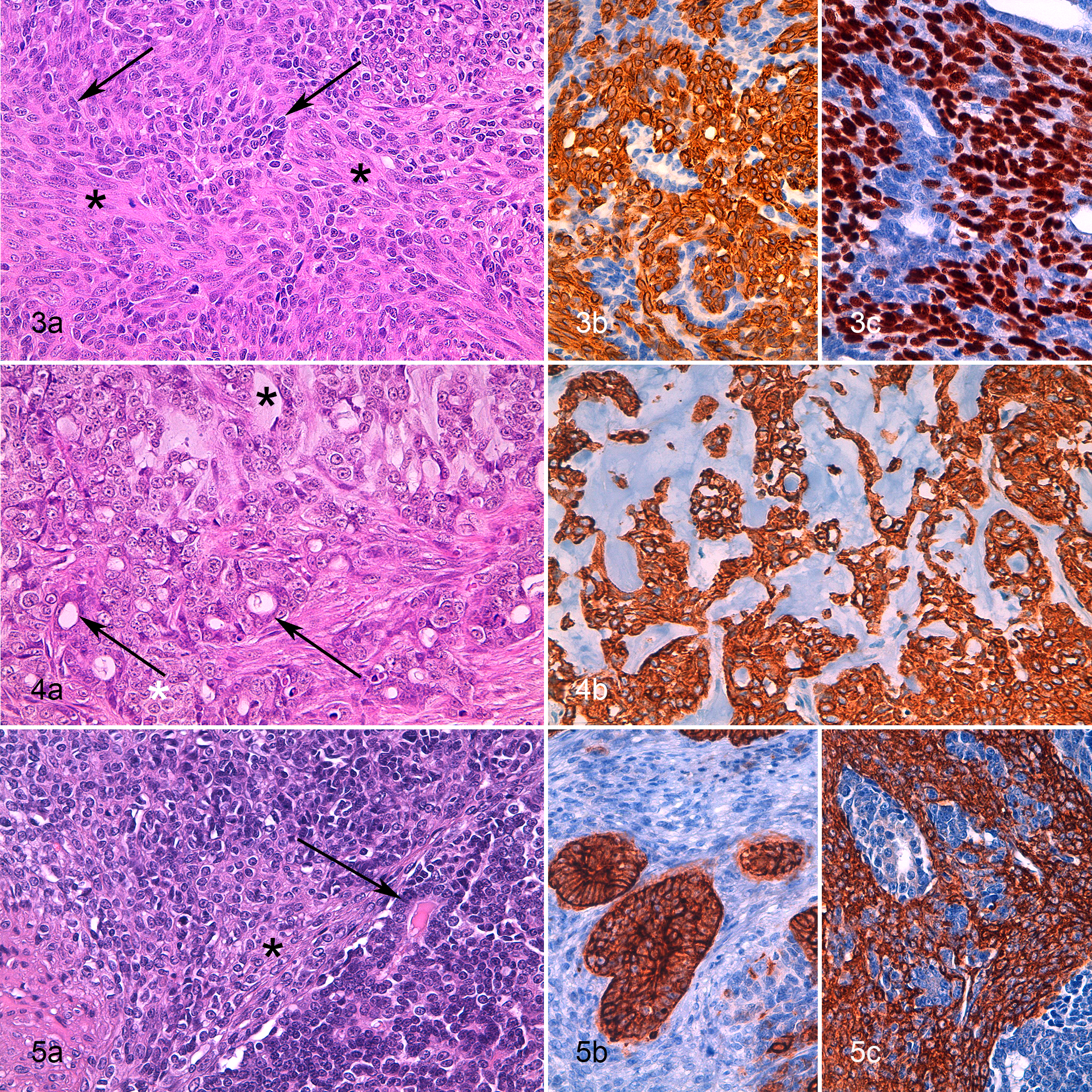
In 7 of 10 cases, the biphasic nature identified on HE evaluation was confirmed on the basis of a differential immunolabeling for the luminal epithelial and the interstitial population. In all 7 cases, the interstitial population was negative for CK8/18. In 1 case (case 5), the interstitial population expressed only vimentin (45% cells). In 1 other case (case 6), the interstitial component expressed only vimentin (100%) and SMA (100%; Table 2; Fig. 5c). Based on these results, these 2 cases were diagnosed as carcinosarcomas (Fig. 5a).
In case 7, interstitial cells were 100% positive for pan-CK (mild), CK5/6, CK14, vimentin, p63, calponin, and SMA (Suppl. Figs. S9–14). These interstitial cells were intimately associated with the luminal epithelial cells in a cordlike/solid manner (Fig. 3b, c; Suppl. Figs. S9–14). Based on IHC, they were classified as round-to-oval interstitial myoepithelial cells. The lesion was diagnosed as a ductal carcinoma.
In the remaining 4 cases, the pleomorphic interstitial population presented a myoepithelial cell IHC phenotype characterized by the expression of vimentin (58%-100% positive cells; Suppl. Figs. S4 and S7) and p63 (19%-100% positive cells; Fig. 1c, 1e, 2d, 3c; Suppl. Figs. S3 and S8) and to a lesser extent of calponin and SMA (1%-42% positive cells; Table 2). These cells also weakly expressed pan-CK (7%-24% positive cells; Fig. 1d, 2b, 5b; Suppl. Figs. S2 and S6). Based on the IHC results, a diagnosis of carcinoma and malignant myoepithelioma was given for all 4 cases.
IHC analysis also enabled the identification of suprabasal myoepithelial cells (vimentin+, p63+, CK14+, CK5/6+, calponin+, SMA+, pan-CKlow+, CK8/18–) at the periphery of the nodules. A continuous layer of suprabasal myoepithelial cells was detected only around an intraductal papillary carcinoma in case 9.
All cases except for 1 carcinosarcoma (case 6) were negative for ER (Table 2). Specifically, the luminal component of the carcinosarcoma was positive for ER (49%), whereas the interstitial cells were ER negative. Interestingly, this was 1 of the 2 cases with a Ki-67 index below 14%. PR was expressed in the luminal component of 5 of 9 cases (2 carcinoma and malignant myoepitheliomas, 2 carcinosarcomas, and the ductal carcinoma). All cases except 3 lesions from the same case (case 4) were also ERBB2 negative (Table 2). In case 4, the luminal epithelial cells of all lesions (carcinoma and malignant myoepithelioma, simple solid carcinoma and a lymph node metastasis of simple solid carcinoma) were scored as 2+.
Ki-67 expression was always higher than 14%, except for case 7 (ductal carcinoma) and case 6 (carcinosarcoma). Apart from case 6, the interstitial component always scored higher than the luminal component in biphasic lesions (Table 2).
Follow-up Data
The referring veterinarians did not have any additional information regarding cases 5 (carcinosarcoma), 8 (simple tubular carcinoma), and 9 (simple tubulopapillary carcinoma). Case 4 (simple solid carcinoma with lymph node metastases and carcinoma and malignant myoepithelioma) died 6 months after surgery due to lung metastases diagnosed at histology as simple solid carcinoma. Case 7, diagnosed with a grade I ductal carcinoma, showed neither relapses nor metastases 43 months after surgery and subsequently died of an invasive nasal carcinoma. Cases 1 (grade I carcinoma and malignant myoepithelioma) and 2 (grade II carcinoma and malignant myoepithelioma) showed neither relapses nor metastasis at 67 and 20 months after surgery, respectively. Case 3, which was diagnosed with a grade III carcinoma and malignant myoepithelioma with dirty margins and vascular invasion, relapsed 2 months postsurgery. Following a second excisional surgery, no further disease progression was observed in 11 months. Case 6, which had a carcinosarcoma, also did not show any relapse or metastasis by 21 months after surgery.
Discussion
In this study, 10 feline mammary tumors with unusual histomorphologies were analyzed. Among them, we identified 7 biphasic mammary tumors composed of 2 different cell populations that were classified as ductal carcinoma (1/7), carcinosarcoma (2/7), and carcinoma and malignant myoepithelioma (4/7) based on IHC. Three additional tumors that were initially suspected to be biphasic based on histopathology were classified as simple monophasic tumors composed of only malignant luminal epithelial cells based on IHC examination.
Mammary carcinoma and malignant myoepithelioma have been described in dogs and is composed of malignant luminal epithelial cells and malignant pleomorphic interstitial myoepithelial cells, which show variable morphology and variable expression of myoepithelial cell markers. 13,14,39,42,60 In our study, we identified, for the first time, 4 feline biphasic mammary tumors that resembled the canine carcinoma and malignant myoepithelioma both morphologically and by IHC. 39,58,60 One of these cases was histologically classified as a carcinosarcoma based on the presence of an interstitial chondroid and adipose tissue-like differentiation. The myoepithelial origin of the interstitial cells (including those with the chondroid and the adipose-like differentiation) was identified with IHC based on the diffuse expression of p63 (82%) and the labeling for pan-CK (10%) and calponin (7%). Mesenchymal metaplasia derived from myoepithelial cells is typical of canine mixed mammary tumors 14,42 but has never been described in canine carcinoma and malignant myoepithelioma. 39,58 This case was still classified as carcinoma and malignant myoepithelioma based on the morphological features that did not reveal well-differentiated cartilage or adipose tissue, as is usually described for mixed mammary tumors. 60 In this case, there was no evidence of chondrocyte differentiation, but only a chondroid-like matrix intermingled with the cells, and the adipose-like aspects were represented by the presence of empty vacuoles within the cells. The myoepithelial origin of the cells was identified based on their expression of pan-CK and p63, which is not a feature of mesenchymal tissue such as cartilage or adipose tissue.
Analysis of protein expression by IHC is the most reliable approach to identifying myoepithelial cells, particularly when they are not well differentiated. 39,58 In our carcinoma and malignant myoepithelioma cases, p63 was the most reliable marker for identifying myoepithelial cells. In addition, and similar to the dog, 39 the phenotype of nonneoplastic myoepithelial cells (vimentin+, p63+, CK14+, CK5/6+, calponin+, SMA+, pan-CK mild+, CK8/18–) was not completely retained in our carcinoma and malignant myoepitheliomas, since CK5/6 and CK14 were not detected. The most reliable markers for myoepithelial cells in the tumors were pan-CK, vimentin, and p63, and their concomitant expression helps to identify pleomorphic interstitial myoepithelial cells. All 4 carcinoma and malignant myoepithelioma cases were ER negative in both components, and PR was present (7% and 8% of positive cells) in the luminal component of 2 cases. Similar low to negative hormone receptor expression has also been described in canine carcinoma and malignant myoepithelioma. 28 ERBB2 was expressed (2+) in only 1 of 4 carcinoma and malignant myoepithelioma cases (case 4), in which a concomitant solid carcinoma with metastasis was also ERBB2+. ERBB2 is rarely expressed in feline mammary tumors; however, data in the literature are inconsistent. 29,38,46,47,53 No data are available for ERBB2 expression in canine carcinoma and malignant myoepithelioma. In women, the ERBB2-overexpressing human breast cancer molecular subtype is more aggressive than the ER+/PR+ luminal subtype. 25,27,36 In cats and dogs, the prognostic and predictive value of the molecular subtypes is still controversial because of variable results most likely related to the nonstandardized analytical protocols or variable sample sizes and tumor subtypes included. 1,3,11,24,46,52 Further analyses are needed to better specify the role of the molecular classification in cats and dogs. In case 4, the monophasic solid ERBB2+ carcinoma had already metastasized at time of diagnosis and caused the death of the patient, whereas the biphasic ERBB2+ carcinoma and malignant myoepithelioma was not metastatic at time of diagnosis. Also, the carcinoma and malignant myoepithelioma of cases 1 and 2 neither relapsed nor developed metastases. Only case 3, which had a carcinoma and malignant myoepithelioma, both of which had dirty surgical margins and vascular invasion, experienced recurrent disease. These data suggest that feline carcinoma and malignant myoepitheliomas might behave less aggressively than the more common simple carcinomas. However, further studies are needed to definitively characterize the behavior of these tumors.
Among the biphasic tumors in our series, we also observed a single ductal carcinoma with round-to-oval interstitial myoepithelial cells. 59,60 The diagnosis was not clearly evident on evaluation of HE-stained sections, and the myoepithelial cell phenotype became evident only with IHC. This tumor was negative to both ER and ERBB2, and 9% of luminal epithelial cells expressed PR. The Ki-67 index of this case was one of the lowest among our samples and less than 14%. This percentage of Ki-67 expression has recently been proposed as a potential cutoff to identify mammary tumors at risk for disease progression in the cat. 47 According to the referring veterinarian, this lesion did not progress after 4 years, indicating a possibly less aggressive behavior for ductal tumors that have been described as being of low-grade malignancy. 43,60
Two cases in our series were diagnosed as carcinosarcomas (cases 5 and 6). Mammary carcinosarcoma is a relatively rare entity in both dogs and cats. 14 It is composed of malignant epithelial cells and malignant mesenchymal cells intermingled with bundles of extracellular matrix. It always shows features of malignancies in both components, and in the dog, it most often presents as a carcinoma and osteosarcoma. 14,31,34,37 In our cases, the 2 cell components were evident, but the interstitial cells morphologically resembled the malignant myoepithelium of canine carcinoma and malignant myoepithelioma. 10,14,31,34,37 The definitive diagnosis was based on the fact that the interstitial cells exclusively expressed vimentin alone or vimentin and SMA. This is not definitive for the identification of myoepithelial cells, since vimentin and SMA also label mesenchymal cells (ie, myofibroblasts). 6,16,51,56,57 In the literature, there is a single case report describing a feline mammary sarcoma with muscle-like differentiation. 19 More recent studies in the human breast proved that SMA+ muscle-like differentiation can be induced in the stroma by downregulation of expression of micro-RNA 140, which is promoted by a high-fat diet as well as by high expression of ER. 55,62 It is curious that this case was the only one expressing high levels of ER (49%) in the luminal epithelial cells and that 11% of the cells expressed PR, which is uncommon for malignant feline mammary tumors. 3,52 No follow-up data were available for case 5, whereas case 6 developed neither relapses nor metastases, possibly indicating a tumor behavior that was not remarkably aggressive.
Combining our data with those from the literature, 3,13,14,31,39,43,45,59 biphasic and triphasic mammary tumors in dogs and cats are summarized in Supplemental Table S2. We believe it is important for standardization to clarify the cell components that are present in the different tumor subtypes. As recently proposed, 60 we categorize myoepithelial cells as round-to-oval suprabasal cells or as spindle-to-stellate interstitial cells. In addition, pleomorphic interstitial myoepithelial cells are characteristic of carcinoma and malignant myoepithelioma. Suprabasal and round-to-oval interstitial cells manifest a similar phenotype (vimentin+, p63+, CK14+, CK5/6+, calponin+, SMA+, pan-CK-low+, CK8/18–), whereas spindle-to-stellate and pleomorphic interstitial cells can lose expression of some myoepithelial cell markers. A combination of markers is therefore needed for their identification, and at least 2 lineage-specific markers need to be positive (eg, p63 and pan-CK, or CK5/6/14 or vimentin).
An association between mammary tumor histotype and clinical behavior has been identified in the dog and suggested in the cat. 35,37,43,45 An excellent prognosis has been described for carcinoma arising in benign mixed tumors, as well as a prolonged survival for complex and ductal carcinomas. 37,43,45 A much worse outcome has been associated with carcinoma and malignant myoepithelioma (pleomorphic interstitial myoepithelial cells) in the dog, although this tumor has a less aggressive behavior than other simple carcinomas. 39 Myoepithelial cells can also form monophasic tumors and have been described in dogs but not in cats. Benign myoepitheliomas are composed solely of spindle-to-stellate interstitial myoepithelial cells, whereas malignant myoepitheliomas contain exclusively pleomorphic interstitial myoepithelial cells. 13,14
In human breast cancer, myoepithelial cells can rarely (1%) be involved in certain breast cancer histotypes. 26 Myoepithelial cells in human breast cancer are not subdivided into subtypes by morphology, but a hierarchical model of myoepithelial cell differentiation proposes that precursor (undifferentiated), intermediate, or differentiated cells can be involved in neoplastic lesions producing variable morphologies and phenotypes. 36 Similarly to dogs and cats, biphasic epithelial-myoepithelial lesions of the human breast have a favorable prognosis. In contrast, malignant myoepithelioma (myoepithelial carcinoma), which is exclusively composed of undifferentiated myoepithelial cells, is more aggressive. 12,21,22,26,32 In humans, myoepithelial cells have been characterized for their tumor-suppressive functions, such as the release of several protease inhibitors and the expression of tumor-suppressive proteins. 9 However, a progressive switch into a less tumor-protective phenotype has been hypothesized when myoepithelial cells gradually lose their normal phenotype and organization. 8,9,17,18 This could explain the more aggressive behavior in all types of tumors containing malignant undifferentiated (pleomorphic) myoepithelial cells compared with those containing well-differentiated myoepithelial cells (ie, suprabasal, interstitial spindle-to-stellate, and interstitial round-to-oval myoepithelial cells). However, additional studies are needed to better define myoepithelial cell function and either their tumor-suppressing or tumor-promoting mechanisms in dogs and cats.
Conclusion
In this study, we demonstrated the existence of carcinoma and malignant myoepithelioma in the feline mammary gland, which potentially shows a less aggressive behavior than the more common feline simple carcinomas. We also summarized data from the literature to clarify the biphasic and triphasic nature of certain mammary tumor histotypes in the dog and in the cat. We showed that additional mammary tumor subtypes can be identified by combining morphology and IHC. We believe that even if it is complicated by many histotypes, a precise classification of feline mammary tumors is needed for the prediction of the clinical outcome and the therapeutic approach. We hope this will be useful for further analyses of the role of myoepithelial cells in human breast cancer and canine and feline mammary tumors, contributing to further understanding of the carcinogenetic process.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Sammarco_et_al - Biphasic Feline Mammary Carcinomas Including Carcinoma and Malignant Myoepithelioma
Supplemental Material, Combined_supplemental_materials-Sammarco_et_al for Biphasic Feline Mammary Carcinomas Including Carcinoma and Malignant Myoepithelioma by Alessandro Sammarco, Giovanni Finesso, Rossella Zanetti, Silvia Ferro, Roberta Rasotto, Diego Caliari, Michael H. Goldschmidt, Enrico Orvieto, Massimo Castagnaro, Laura Cavicchioli and Valentina Zappulli in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
