Abstract
When compared with the canine species, feline mammary tumors (FMTs) are much less heterogeneous, with a predominance of simple malignant neoplasm. Benign FMTs are rare, and it is unclear if complex and mixed tumors exist in the feline. In this study, we selected for immunohistochemical analyses 12 FMTs that had unusual histologic features. A group of 8 (2 benign and 6 malignant) FMTs showed a biphasic epithelial/myoepithelial population and a very regular cord-like distribution in a “Chinese lettering” pattern, within ectatic ducts. A second group (2 benign and 2 malignant) had an intraductal epithelial papillary growth pattern with a basally located monolayer of myoepithelial cells and a supporting fibrovascular stroma. The myoepithelial component always produced a standard immunohistochemical signature. All malignancies were grade I, and the subjects were all alive at 1 year postdiagnosis. On the basis of their morphology, we propose that they be classified as feline ductal adenoma/carcinoma and feline intraductal papillary adenoma/carcinoma, respectively. They overlap with their canine counterparts and lack the typical myoepithelial differentiation patterns seen in canine complex neoplasms, and therefore, the term complex should be avoided in felines. This study will add new information on FMT classification and be useful for prognostic studies.
Feline mammary tumors (FMTs) are usually aggressive carcinomas (80%–90%) with a poor prognosis. 14 The most recent classification of FMTs published by the World Health Organization (WHO) 20 was based on morphologic criteria and distinguishes noninfiltrating in situ carcinomas by infiltrative simple carcinomas of different histologic subtypes, similar to the classification for human breast cancer (HBC). The most common type is an infiltrative aggressive carcinoma. 7,14 Among benign/dysplastic lesions, there is a high incidence in queens of a specific subtype classified as fibroadenoma/fibroadenomatous change, whereas other adenomas and duct papilloma are considered rare. 20 When compared with canine mammary tumors (CMTs), FMTs are much less heterogeneous, with a marked predominance of simple neoplasm composed of only 1 (luminal epithelial) cell type. 14,20 The existence of complex and mixed tumors in the feline species has not been precisely documented. Benign and malignant mixed tumors are reported very rarely in queens by some authors, 5,13,19 and more recently, feline mammary complex carcinomas have been described. 25,26 The role of myoepithelial cells in CMTs has been discussed, and their presence in complex and mixed tumors is associated with a less aggressive biological behavior. 20 The morphologic features of the myoepithelial component in complex and mixed CMTs are well characterized and can be easily recognized in standard hematoxylin and eosin (HE) sections. 11,20 In canine tumors, it is frequently a reactive/benign component even if CMT subtypes with malignant myoepithelium have been reported (carcinoma-and-malignant myoepithelioma; malignant myoepithelioma). 10,21,25 Several studies have investigated the immunohistochemical profile of myoepithelial cells, indicating that there is a progressive differentiation from basal/undifferentiated cells to well-differentiated myoepithelial cells that are able to enter osseous/condroid metaplastic transformation in canine mixed tumors. 1,9,17 Well-established immunohistochemical staining of differentiated canine mammary myoepithelial cells includes expression of cytokeratins (CK5_6 and CK14), calponin, p63, vimentin, and α-smooth muscle actin (α-SMA). 1,9,17,23,28 The presence of myoepithelial-like cells in FMTs has occasionally been described on the basis of immunohistochemical staining. 3,20 However, no morphologic evidence of the classical canine myoepithelial component has been reported in queens, indicating that there are species-specific differences. Nevertheless, up to now, the few studies describing feline mammary carcinomas with immunohistochemical evidence of myoepithelial differentiation found a better prognosis when compared with the more common simple tumors. 3,28 A new classification of CMTs has recently been proposed, including a detailed morphologic characterization for each tumor subtype. 11 Some new morphologic categories have been added such as ductal and intraductal papillary tumors and carcinoma and malignant myoepithelioma. Precise and standardized morphologic classification of neoplastic lesions is important to help with prognosis and comparison between studies. 6,18
The aim of this study was to better characterize feline mammary lesions with a double component composed of luminal epithelial cells and basal/myoepithelial cells. We also propose that they be classified as ductal and intraductal papillary tumors on the basis of similar neoplasms in CMTs and HBCs.
Materials and Methods
The mammary gland samples used in this study were retrieved from the archive of the Diagnostic Service of Veterinary Anatomical Pathology, Department of Comparative Biomedicine and Food Science, University of Padua, Italy. The neoplastic mammary samples included in this study were selected following morphologic diagnosis from a group of 74 FMTs surgically obtained by mastectomy from 73 queens. Samples were fixed in 10% neutral buffered formalin and embedded in paraffin. Sections (4 μm) were stained with HE for histopathologic evaluation based on the WHO classification. 20 Criteria for malignancies were cellular pleomorphism, loss of tissue architecture, multifocal areas of necrosis, mitoses, and infiltration of the peripheral subcutis. Specifically, in borderline lesions, a minimum of 3 mitoses per 10 high-power fields was considered a cutoff point for malignancy. Samples were reviewed by at least 2 certified veterinary pathologists. Malignant lesions were graded according to the scheme proposed by Castagnaro et al. 3
Immunohistochemical (IHC) analyses were conducted on the selected samples for cytokeratins (panCKs), cytokeratins 8 and 18 (CK8_18), cytokeratins 5 and 6 (CK5_6), cytokeratin 14 (CK14), p63, vimentin (vim), calponin (CALP), and α-SMA. For immunostaining, 4-μm wax-embedded sections were processed through an automatic immunostainer (BenchMark XT; Ventana Medical Systems, Tucson, AZ). The protocols included a high temperature (95°C), buffered antigen unmasking (cell conditioning [CC]), and/or an enzymatic retrieval (trypsin). All reagents, except primary antibodies, were supplied within the system. The incubation temperature for all primary antibodies was set within the system at 42°C, whereas the other specific features of the protocols (primary antibody, retrieval method and time, dilution and incubation time) were as follows: anti–human panCK (AE1/AE3; DakoCytomation, Carpinteria, CA), CC 30 minutes, 1:100, 16 minutes; anti–human CK8_18 (NCL-L-5D3; Novocastra, Buffalo Grove, IL), trypsin 8 minutes, 1:30, 24 minutes; anti–human CK5_6 (clone D5/16 B4; DakoCytomation), CC 30 minutes + trypsin 2 minutes, 1:100, 24 minutes; anti–human CK14 (NCL-LL002; Novocastra), CC 30 minutes, 1:20, 20 minutes; anti–human p63 (clone 4A4, sc-8431; Santa Cruz Biotechnology, Santa Cruz, CA), CC 30 minutes, 1:200, 20 minutes; anti–human vim (clone V9; DakoCytomation), CC 30 minutes, 1:150, 20 minutes; anti–human CALP (clone CALP; DakoCytomation), CC 30 minutes, 1:200, 12 minutes; and anti–human α-SMA (clone 1A4; DakoCytomation), no retrieval, 1:100, 16 minutes. The ultraView Universal DAB detection Kit (Ventana Medical Systems) was applied containing a secondary multimeric antibody and 3,3′-diaminobenzidine (DAB) as the chromogen. Positivity was demonstrated as brown cytoplasmic staining of neoplastic cells for all antibodies except p63, which showed nuclear staining. Normal feline mammary gland and skin included with some samples were used as positive controls. For negative controls, the antibody diluent (Ventana Medical Systems) was applied instead of the primary antibody. To stain the basal membranes and precisely localize the cell populations (suprabasal vs interstitial), standard histochemical periodic acid–Schiff (PAS) staining was superimposed on a selected subset (2 cases) of samples after processing for α-SMA IHC.
Clinical information was collected and 1-year follow-up data were registered when available.
Results
A total of 74 FMTs were analyzed, and 91.9% were malignant tumors. Of the 74 samples, a small subset (12 tumors) had some morphologic features that were not specifically described in any of the WHO tumor subtypes. These tumors were selected for further analyses. Based on the newly proposed CMT classification, ductal adenomas and intraductal papillary adenomas and carcinomas were diagnosed on morphologic evaluation and HE sections. 11 In addition, ductal carcinomas were confirmed after immunohistochemical analyses. Specifically, 2 ductal adenomas (Fig. 1), 2 intraductal papillary adenomas (Fig. 2), 6 ductal carcinomas (Figs. 3, 4), and 2 intraductal papillary carcinomas (Figs. 5, 6) were diagnosed. Ductal tumors were 11% and intraductal papillary tumors were 5% of the total examined. They were the only neoplastic lesions identified within the glandular tissue, except for 1 ductal carcinoma that was associated with a grade I tubulopapillary carcinoma. All malignant tumors were grade I. The average age of the cats with ductal or intraductal papillary neoplasms was 11 and 12 years, respectively.
Mammary gland; female cat. Ductal adenoma with neoplastic cells forming regular cords with the formation of slitlike lumina (Chinese lettering pattern). Hematoxylin and eosin (HE).
Ductal tumors showed densely cellular, well-defined, multinodular masses often growing within ectatic ducts. When malignant, the tumor was slightly infiltrative. The neoplastic component in benign ductal lesions typically formed regular cords with 2 distinct cellular components (luminal and basal) with the formation of slitlike lumina (Chinese lettering pattern) and with a minimal fibrous stroma (Fig. 1). The luminal cells formed a cuboidal monolayer with an underlying basal layer of oval to elongated cells that expanded into a more solid interstitial pattern in malignant lesions (Fig. 4). The luminal cells had a scant to moderate apical densely eosinophilic cytoplasm and a basal to central round nucleus with a single nucleolus. The basal layer was composed of cells with a minimal amount of clear cytoplasm, an oval open-faced nucleus, and indistinct cytoplasmic borders. Ductal carcinomas were more solid with less regular cords and increased numbers of basal cells that were embedded in a scant to moderate eosinophilic fibrillar (collagenous) matrix (Figs. 3, 4); both cell components had a higher mitotic index and more atypia than benign tumors, but mitoses were always less then 6 per 10 high-power fields, and marked anisocytosis and anisokaryosis were never evident. Squamous differentiation was not detected. One ductal carcinoma showed central areas of well-demarcated necrosis (comedonic appearance) with mineralization.
Intraductal papillary tumors showed a multinodular papillary growth pattern within ectactic ducts with the papillae supported by a fibrovascular stalk (Figs. 2, 5, and 6). The luminal cells were cuboidal to columnar with a moderate amount of apical cytoplasm and basal round to elongated nuclei with a single nucleolus. A basal monolayer of oval cells with open-faced nuclei (myoepithelial cells) was detectable with HE (Fig. 6). Within benign lesions, anisocytosis and anisokaryosis were minimal as well as mitoses; malignant tumors showed a more irregular papillary pattern, with increased pleomorphism and mitotic count. In the ductal and intraductal papillary neoplasms, the classical myoepithelial morphology 11 described for the canine species as interstitial/stromal (fusiform or stellate) 1 was never detected.
Using IHC, all ductal tumors showed variable expansion within the interstitium of the basal/myoepithelial component that was negative for CK8_18 (Fig. 7) and positive for all other markers (panCK, CK5_6, CK14, p63, vim, CALP, α-SMA) (Figs. 8–12). PanCK positivity was slightly lower than the luminal population. The luminal cells were exclusively panCK+ and CK8_18+ (Fig. 7). Similarly, intraductal papillary tumors had a basal/myoepithelial layer positive to all markers except CK8_18 but exclusively located underneath the luminal component without apparent expansion within the interstitium (Figs. 11, 12). This was confirmed when PAS staining was superimposed on the α-SMA IHC (Figs. 13, 14). In ductal tumors with PAS histochemistry, interstitial α-SMA+ myoepithelial cells were intermingled with PAS+ matrix residues (Fig. 13), whereas intraductal papillary lesions maintained a basal PAS+ linear staining (basal membrane) on top of which were the α-SMA+ (myoepithelial) cells (Fig. 14). Fibroblasts within the stroma were always vimentin positive and intermingled with scattered CALP+/α-SMA+ cells (myofibroblasts).
Mammary gland; female cat. Ductal adenoma: luminal and not basal cells show positivity for cytokeratins 8 and 18. 3,3′-Diaminobenzidine (DAB) immunohistochemistry with hematoxylin counterstain.
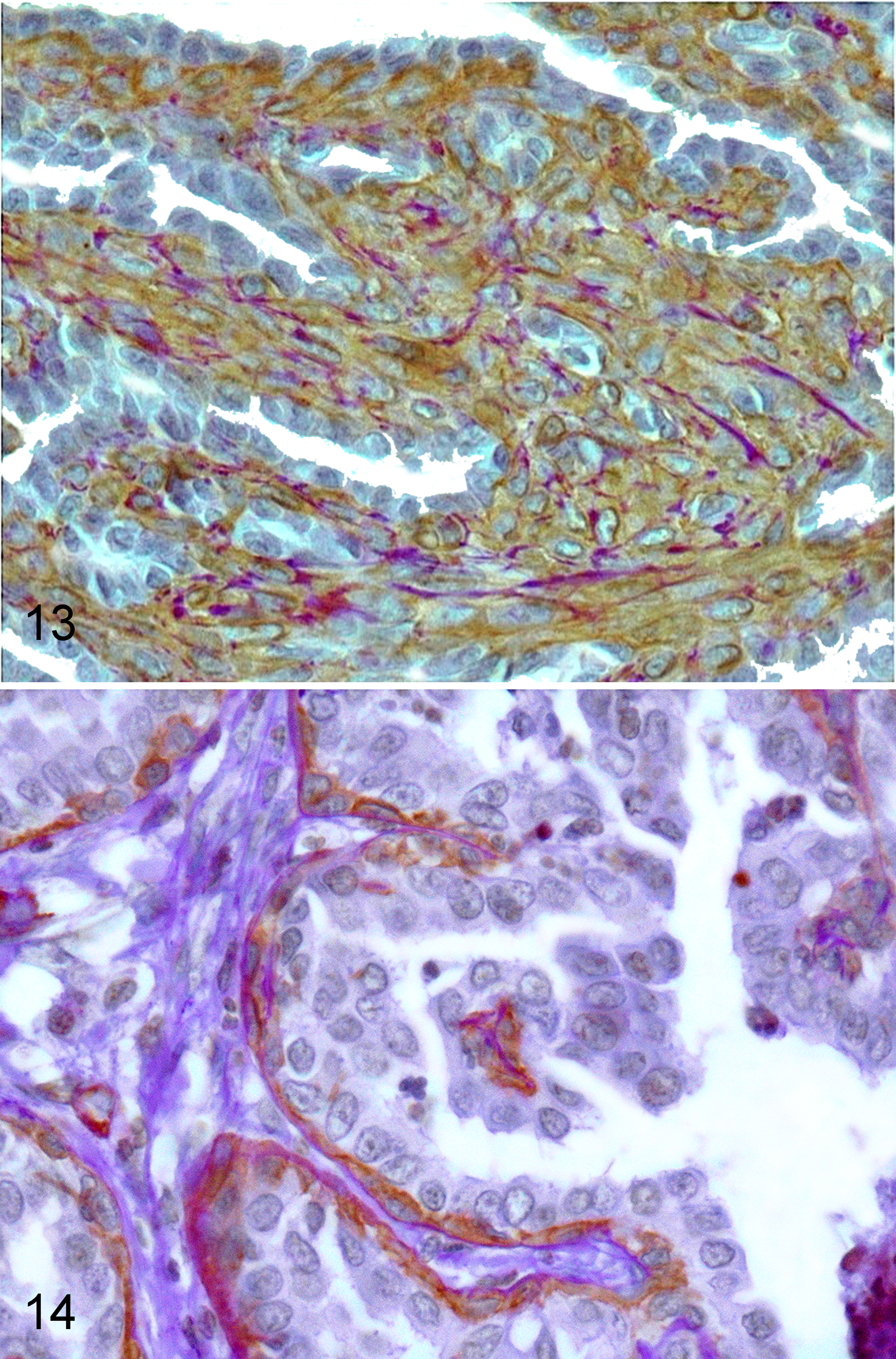
Mammary gland; female cat. Ductal adenoma: interstitial α-smooth muscle actin (α-SMA)+ basal (myoepithelial) cells are expanded within the interstitium and intermingled with periodic acid–Schiff (PAS)+ matrix residues. Routine PAS staining superimposed on 3,3′-diaminobenzidine (DAB) immunohistochemistry for α-SMA.
Insufficient follow-up data were collected for statistical analyses. However, all queens selected in this study were alive at 1 year postdiagnosis. Two queens with an intraductal papillary carcinoma developed new mammary lesions, but histology was not performed.
Discussion
We describe 12 neoplastic lesions in the mammary gland of cats that have morphologic features not characterized in detail in the most recent WHO classification. 20 Using both morphology and immunohistochemistry, 2 different histologic subtypes were identified. Specifically, a group of 8 tumors had features of the recently described “ductal adenoma/carcinoma” in CMTs, whereas 4 lesions had features of canine “intraductal papillary adenoma/carcinoma.” 11 In this series, 2 ductal and 2 intraductal papillary adenomas and 6 ductal and 2 intraductal papillary carcinomas were identified. All malignant neoplasms were grade I, and all subjects were alive 1 year postdiagnosis, without evidence of metastatic disease.
The subtypes classified as ductal tumors in this report were previously identified in the dog as “basaloid adenomas,” and a benign subtype with similar morphology was described in the cat as a “complex adenoma.” 20 In both species, this type of tumor has a dual cellular component of luminal epithelial cells and basal/myoepithelial cells organized in cords separated by slitlike lumina. The myoepithelial cells of the CMTs have been described as resting and proliferative suprabasal cells and spindle and stellate interstitial cells. 1 The basal/myoepithelial cells of the canine and feline ductal tumors maintain a “basal” (“basaloid”) differentiation, referable to the suprabasal proliferative myoepithelial cells of the dog. 1 Particularly, in the feline ductal tumors described here, these cells maintained the suprabasal proliferative morphology also when expanded into the interstitium. This is different from the well-described canine complex mammary tumors in which the myoepithelial cells expanding into the interstitium change their morphology into spindle or stellate. 1,9,11 The histologic features of the feline and canine mammary ductal tumors are very similar to human salivary, 4,8,26 breast, 2,24,26 and pulmonary 33 adenomyoepitheliomas/carcinomas and to feline/canine apocrine ductal carcinomas. 10,12 On the basis of these observations and in conjunction with the proposed classification of CMTs, we therefore suggest these neoplasms be classified as feline ductal adenoma/carcinoma. Similar lesions have occasionally been described in the literature and referred to as “feline complex carcinomas.” 3,27,28 Although only carcinomas were described, the findings were similar to ours since they were always low-grade malignant lesions with a better overall survival and disease-free survival. They represent 10% to 11% of all included FMTs. 3,27 In addition, Seixas and coauthors 27,28 found areas of squamous differentiation in some of their cases that we did not observe; nevertheless, when considering a small number of samples, some variability should be expected.
The protective role of the myoepithelial component of the common canine complex and mixed mammary tumors is well known and related to their less aggressive behavior. 19 This has been studied in the dog, in which the typical myoepithelium consists of spindle to stellate interstitial cells embedded in a myxoid matrix that are able to progressively lose some markers of expression and to differentiate into mesenchymal chondroid/osseous cells. 1,9 This type of differentiation was never detected in the feline lesions described above or in canine ductal adenoma/carcinoma or basaloid adenoma. 3,20,27,28 Some authors have suggested that also in those neoplasms in which the myoepithelial cells maintain a “basaloid” morphology, they confer a less aggressive behavior with a better prognosis. 27,28 The immunohistochemical signature displayed in this study by the myoepithelial cells (lowpanCK+/CK5_6+/CK14+/p63+/vim+/CALP+/α-SMA+/CK8_18–) matched the expression found in other studies in cats 17,28 and humans. 3,9,16,17,23,27,31 In the dog, this phenotype was described for the proliferative suprabasal myoepithelial cells. Conversely, this population is not detectable in the more common but aggressive simple FMTs that predominate in this species (80%–90%), indicating that there are some interesting differences from the dog. 19 Interestingly, in the ductal histologic subtypes discussed here and found in both the cat and dog, the myoepithelium was present and expanded but had a “basal” undifferentiated phenotype. This might be associated with a different site of origin (“ductal”) of the tumor and/or with specific differentiation pathways. We have also recently demonstrated a nuclear β-catenin staining within the basal/myoepithelial cells of the ductal feline tumors, but to our knowledge, no similar data are available for the dog. 34 Nuclear β-catenin expression has been described in human basal cell tumors of the salivary gland, and Wnt/β-catenin signaling has been related to both the maintenance of stem cells/undifferentiated pools and tumorigenesis. 15,30
In this study, a second histologic FMT subtype was included. This presented as a papillary intraductal growth similar to that previously described as the feline/canine “duct papilloma” and included in the classification exclusively in its benign form. 20 The newly proposed canine classification has renamed duct papilloma as intraductal papillary adenoma/carcinoma. 11 In the dog, this tumor may show myoepithelial proliferation (interstitial spindle to stellate), 1,11 but the papillary intraductal growth pattern distinguishes it from a complex carcinoma. 11 In our feline samples, an identical intraductal papillary proliferation was observed in which the myoepithelium was never expanded—as demonstrated by combined immunohistochemical and PAS staining—and neither did the “basaloid” phenotype or the classical interstitial canine morphology. It was more referable to the canine suprabasal resting cells. 1 Even though there were a small number of cases (5%) in our study, we identified both the benign and the malignant variant with a low grade of malignancy. On the basis of the specific morphology, we propose its incorporation into any new classification. As no data are available in the literature on this intraductal papillary FMT, further studies need to be undertaken on a larger number of cases.
In conclusion, in this study, we have described features of 2 uncommon types of FMTs and propose that they be classified as feline ductal adenoma/carcinoma and feline intraductal papillary adenoma/carcinoma. This study showed there are many similarities to their canine counterparts and stress that in the feline, myoepithelial differentiation found in the interstitium of canine mammary neoplasms is lacking. Therefore, the term complex, which typically refers to a biphasic tumor with both the epithelium and the interstitial myoepithelium commonly observed in the dogs, should not be applied. A recent study has recommended important guidelines for prognostic studies in veterinary oncology. 33 Specifically, there is a need for standardizing the diagnoses of mammary tumors. 6,18,20 –22,29,32 In this context, this study adds new information pertaining to FMT classification. This is useful for prognostic studies and for a better understanding of mammary tumor histogenesis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
