Abstract
Mammary cancer is the most common cancer in female dogs. Induction of cyclooxygenase-2 (COX-2), a key enzyme in prostaglandins (PGs) biosynthesis, has been demonstrated in various cancers in humans and dogs, including mammary cancer. The objective of this study was to investigate the expression and regulation of COX-2 in canine mammary epithelial cells. Cell lines derived from normal and neoplastic canine mammary glands were cultured in the absence or presence of phorbol 12-myristate 13-acetate (PMA), and immunoblots, immunocytochemistry, radioimmunoassays, and a cell proliferation assay were used to study COX-2 expression and PGs production. Results showed that the neoplastic cell line CMT12 constitutively overexpressed COX-2 protein whereas other mammary cell lines expressed low to undetectable basal levels of COX-2 protein. Basal PGE2 production was significantly higher (P < .05) in CMT12 compared to other cell lines. Levels of COX-2 protein in CMT12 decreased in a time-dependent manner with serum starvation, and PMA stimulation induced a strong time-dependent increase in COX-2 protein. Treatment of CMT12 cells with NS-398 (a specific COX-2 inhibitor) significantly blocked PGE2 synthesis and reduced cell proliferation (P < .05). These results indicate that some neoplastic canine mammary cell lines constitutively overexpress COX-2, and that COX-2 inhibition decreases PGE2 production and cell proliferation, supporting a role for COX-2 and PGs in canine mammary oncogenesis.
Mammary tumors are the most common neoplasms in female dogs, with a reported incidence varying between 198 and 622.6 cases/100,000 dogs per year. 4, 6, 18, 36 Both benign and malignant tumors are frequently diagnosed, and malignant neoplasms represent approximately 50% of all mammary tumors. 17, 20, 36 Malignant tumors can recur following surgical excision or can metastasize to distant organs such as lymph nodes, lung, and liver. 40 Canine mammary tumors are age-dependent neoplasms, with a mean age at onset of 6–7 years. 9 All breeds of dogs can be affected, but certain breeds (English Setter, Chihuahua, miniature Poodle, and Afghan Hound for example) appear to show a higher prevalence. 18, 36 They are hormone-dependent neoplasms, and early ovariectomy (i.e., before the first estrous cycle) has a protective effect. 18, 46 Immunohistochemical studies have demonstrated the presence of estrogen and progesterone receptors on canine mammary tumors, but their prognostic significance is yet to be established. 33, 39 Also, progestin-dependent synthesis of mammary growth hormone has been proposed as one of the mechanisms potentially involved in mammary tumorigenesis in dogs. 60
Prostaglandins are lipid mediators that are involved in many physiologic and pathologic processes including tumorigenesis. The first rate-limiting step in the synthesis of all prostaglandins is controlled by the enzyme prostaglandin G/H synthase, also known as cyclooxygenase (COX). 15, 51 COX mediates the conversion of arachidonic acid into PGH2, a common precursor for the synthesis of all prostanoids. Two isoforms of COX (COX-1 and COX-2) have been identified. 52 COX-1 is present in a wide variety of tissues and is often referred to as the constitutive form. In contrast, COX-2 is generally undetectable in most tissues but can be induced by different agonists, and is referred to as the inducible form. Overexpression of COX-2 has been reported in several types of cancer in humans, and selective targeting of COX-2 appears as a promising new therapeutic avenue for the prevention and treatment of cancer. 2, 55, 59
Epidemiologic studies have reported that the regular use of nonsteroidal anti-inflammatory drugs (NSAIDs) may protect against breast cancer. 21, 47, 48 The role of COX-2 in mammary tumorigenesis was recently highlighted by a study in transgenic mice demonstrating that COX-2 overexpression alone is sufficient to cause breast tumor formation. 32 Several reports have also demonstrated that COX-2 is overexpressed in breast cancer. 19, 34, 42, 44, 53 In dogs, COX-2 overexpression has so far been reported in renal, urinary, prostatic, intestinal, bone, and mammary cancers as well as in squamous cell carcinomas. 12, 22, 26, 27, 35, 38, 43, 58 In a recently published study of 84 cases of canine mammary carcinomas, we found that more than half of the carcinomas displayed weak to strong COX-2 staining. 12 Moreover, Heller et al. 22 showed an association between COX-2 expression and mammary tumor histologic subtype in dogs with anaplastic carcinomas expressing significantly higher levels of COX-2 compared with adenocarcinomas. These studies suggest that COX-2 expression could play a role in canine mammary oncogenesis. The objective of the present study was to characterize an in vitro model of canine neoplastic mammary cell lines to investigate the expression, regulation, and possible role of COX-2 overexpression in canine mammary cancer.
Materials and Methods
Diaminobenzidine tetrahydrochloride (DAB) and dimethyl sulfoxide (DMSO) were obtained from Sigma Aldrich Canada (Oakville, Ontario, Canada); Hybond polyvinylidene difluoride membranes 0.45 μm (PVDF) were purchased from ICN Pharmaceuticals (Montreal, Quebec, Canada); Kodak Bio-Max X-ray film was obtained from Eastman Kodak Company (Rochester, NY, USA); ECL plus kit and enhanced chemiluminescence detection, Western blotting detection system were obtained from Amersham Biosciences (Baie D'Urfe, Canada); Vectastain ABC kit was purchased from Vector Laboratories (Burlington, Ontario, Canada); anti-rabbit Ig, horseradish peroxidase linked, whole antibody from donkey was obtained from Amersham Biosciences; the cell proliferation reagent WST-1 was purchased from Roche Diagnostics; NS-398 was purchased from Cayman Chemical CO (Ann Arbor, MI, USA); Dulbecco's Modified Eagle's Medium (DMEM), Leibovitz (L-15), Hepes tampon, phosphate buffered saline (PBS), and Hanks' balanced salt solution (HBSS) were purchased from Gibco BRL Life Technologies, Inc. (Grand Island, NY, USA); EGF was obtained from Peprotec Canada, Inc. (Ottawa, Ontario, Canada), Bio-Rad protein assay and electrophoretic reagents were purchased from Bio-Rad Laboratories (Richmond, CA, USA). Quik Hyb solution was from Stratagene (Cedar Creek, TX, USA) and Prime-a-Gene Labeling system was purchased from Promega Corporation (Madison, WI, USA).
Cell culture
The cell lines CF35, CF33, and CF41 were obtained from ATCC (Manassas, VA, USA). They were cultured in DMEM supplemented with 10% fetal calf serum and penicillin G (100 U/ml)-streptomycin (100 μg/ml). The cell lines CMT12, CMT9, and CMT28 were established from dogs with spontaneous mammary carcinomas. 61 They were cultured in L-15 medium supplemented with 10% fetal calf serum and penicillin G (100 U/ml)-streptomycin (100 μg/ml). Cells were incubated at 37°C in 5% CO2, 95% air, and passaged by exposure to trypsin/EDTA (0.05/0.02% w/v) in Dulbecco's phosphate buffered saline (PBS).
Solubilized cell extracts and immunoblot analysis
Solubilized cell extracts were prepared as previously described. 50 Briefly, cells were homogenized on ice in TED homogenization buffer (50 mM Tris, 10 mM EDTA, 1 mM DEDTC, pH 8.0) supplemented with 2 mM octyl glucoside, and centrifuged at 30,000 × g for 1 hour at 4°C. The crude pellets containing membranes, nuclei, and mitochondria were sonicated (5 seconds/cycle; 4 cycles) in TED sonication buffer (20 mM Tris, 50 mM EDTA, 0.1 mM DEDTC, pH 8.0) containing 32 mM octyl glucoside. The sonicates were centrifuged at 13,000 × g for 25 minutes at 4°C. The supernatants (solubilized cell extracts) were stored at -70°C until immunoblot analysis. The protein concentration in each extract was determined by the method of Bradford (Bio-Rad Protein Assay). Proteins were resolved by one-dimensional sodium dodecyl sulfatepolyacrylamide gel electrophoresis (SDS-PAGE), and electrophoretically transferred to Hybond polyvinylidene difluoride (PVDF) membranes. Blocking of membranes was done using 5% nonfat dry milk in 0.1% TTBS (0.1% Tween-20, 10 mM Tris-buffered saline, pH 7.5) for 1 hour at room temperature, then washed twice for 2 minutes at room temperature with 0.1% TTBS. After blockage, membranes were incubated with a selective anti-COX-2 antibody (MF243 at 1°7,500 dilution) diluted in 0.05% TTBS (0.05% Tween-20, 10 mM Tris-buffered saline, pH 7.5) containing 2% nonfat dry milk for 2 hours at room temperature. Membranes were incubated with a horseradish-peroxidase-labeled donkey anti-rabbit secondary antibody (1°15,000 dilution) for 1 hour at room temperature. The membranes were washed and the bound secondary antibody was detected using the enhanced chemiluminescence (ECL) detection kit. The signal was visualized on Kodak Bio-Max X-ray film.
Prostaglandin E2 radioimmunoassay
Concentrations of PGE2 were measured directly in culture media, as previously described. 31 The antiserum was purchased from Assay Designs Inc. (Ann Arbor, MI, USA); its cross-reactivities against PGE1, PGF1α, PGF2α, and 6-keto PGF1α were 70%, 1.4%, 0.7%, and 0.6%, respectively. The sensitivity of the assay was 40 pg/ml, and the intra- and interassay coefficients of variation were 6.3% and 8.6%, respectively.
Immunocytochemistry
Cells were cultured in 8-chamber glass slides (Lab-Tek II) until they reached 70% confluency. Unstimulated cells were cultured in complete medium. For PMA stimulation, cells were cultured in medium without serum for 40 hours before addition of PMA (5 ng/ml) for 24 hours. After treatment, cells were fixed with 95% ethanol, 5% acetic acid, and immunocytochemical staining for COX-2 (antibody MF243, Merck Frosst Centre for Therapeutic Research, Pointe-Claire-Dorval, Quebec, Canada) or COX-1 (antibody 8223) was performed using the Vectastain avidin°biotin complex (ABC kit), as previously described. 5, 58 The specificity of these polyclonal antibodies for canine COX-1 and -2 has been documented. 58 Control staining was done with rabbit serum or PBS.
Cell proliferation assay
Cell proliferation was measured using an assay based on the cleavage of the tetrazolium salt WST-1 to a soluble formazan salt by metabolically active cells, as directed by the manufacturer (Roche Diagnostics, Laval, Canada). For this assay, cells were seeded in 96-well plates at a density of 30,000 cells/well. After 24 hours of culture in complete medium, NS-398 dissolved in DMSO was added to the cells. Control wells received DMSO at the highest concentration used with the inhibitor. After 48 hours of culture with the inhibitor, WST-1 reagent was added to each well and the plate was incubated for 4 hours at 37°C. Spectrophotometric absorbance of the samples was measured with a microtiter plate reader at a wavelength between 550 and 600 nm.
Statistical analyses
Experiments were performed in duplicate or triplicate. One-way ANOVA was used to compare PGE2 production between different cell lines, and to test the effect of serum starvation, agonist, and inhibitor treatment on PGE2 production and cell proliferation. When ANOVAs indicated significant differences (P < .05), the Dunnett's test was used for multiple comparisons. Data expressed as percentages were arcsine transferred before being analyzed. Statistical analyses were performed using the computer program JMP (SAS Institute, Inc., Cary, NC, USA).
Results
COX-2 expression and PGE2 synthesis in canine mammary cell lines
Six different canine mammary cell lines were initially characterized. Five of them (CF33, CF41, CMT9, CMT12, and CMT28) are derived from canine mammary tumors, and 1 cell line (CF35) is from a normal canine mammary gland. Basal expression of COX-2 in these cell lines cultured in complete medium was determined by immunoblot analysis using a selective anti-COX-2 antibody, and results showed that basal levels of COX-2 expression varied between the different cell lines (Fig. 1A). The cell line CMT12 displayed a strong COX-2 immunoreactive signal whereas all other neoplastic cell lines showed faint to undetectable COX-2 expression (Fig. 1A). A weak COX-2 signal was detected in CF35, the cell line derived from normal mammary tissue (Fig. 1A). The production of PGE2 by these cell lines was determined, and results paralleled those of COX-2 protein expression with PGE2 synthesis by CMT12 being the highest of all cell lines (Fig. 1B). To characterize the pattern of COX-2 expression in situ, immunocytochemistry was performed on CF35 and CMT12 cells cultured in complete media. In agreement with the immunoblot results, COX-2 protein was weakly present in the cytoplasm of CF35 cells (Fig. 2A) while CMT12 cells displayed strong cytoplasmic COX-2 immunoreactivity (Fig. 2C). No COX-1 was present in CMT12 (Fig. 2D) or CF35 cells (data not shown).
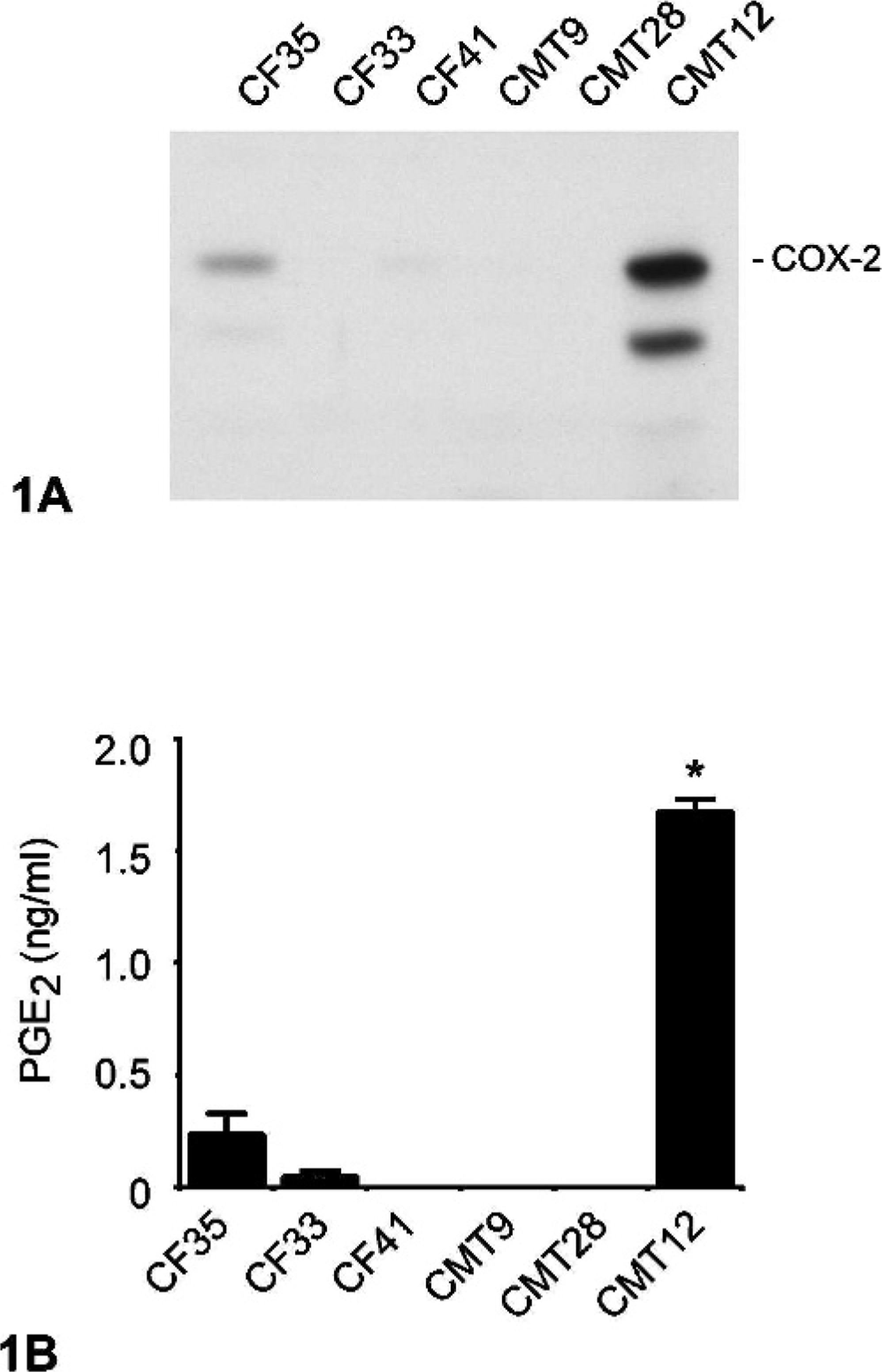
COX-2 expression and PGE2 synthesis in normal and neoplastic canine mammary epithelial cells. Normal (CF35) and neoplastic (CF33, CF41, CMT9, CMT28, and CMT12) canine mammary epithelial cells were cultured in complete medium, as described in Materials and Methods.
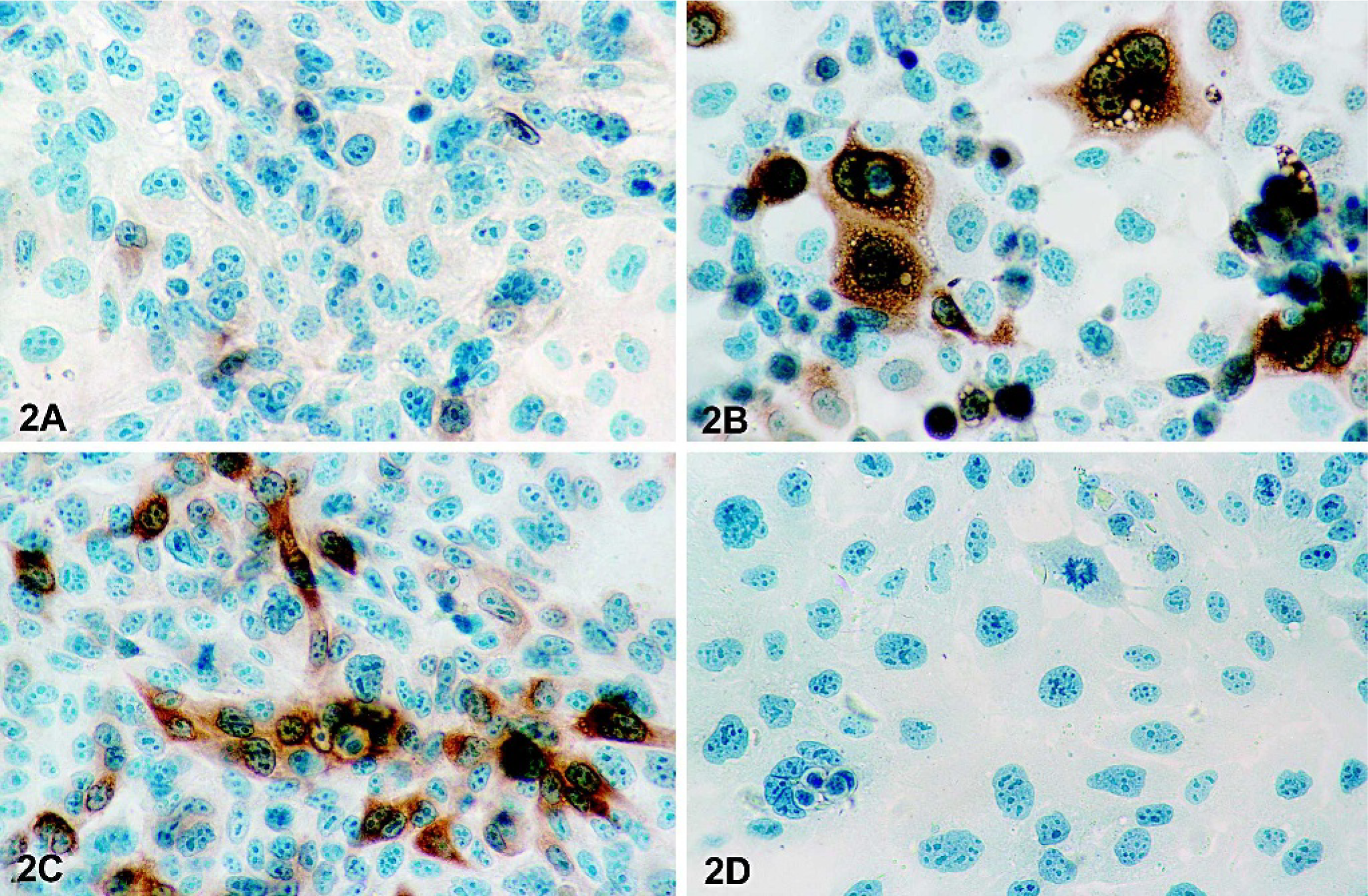
Immunocytochemical detection of COX-2 in normal and neoplastic canine mammary epithelial cells. Immunocytochemistry was performed on normal (CF35) or neoplastic (CMT12) canine mammary epithelial cells cultured for 24 hours in the absence (control) or presence of 5 ng/ml PMA, as described in Materials and Methods.
Cell lines CF35 and CMT12 were selected to determine the effect of serum starvation on COX-2 protein levels. Results showed undetectable levels of COX-2 in CF35 cells before and after serum starvation (Fig. 3A) while a time-dependent reduction of COX-2 expression was observed in CMT12 cells after 24, 48, and 72 hours of serum starvation (Fig. 3B). Following 24 hours of serum starvation, COX-2 protein was markedly reduced in CMT12 cells and a faint signal was present after 48 hours (Fig. 3B). Synthesis of PGE2 by both cell lines dropped to very low levels following 24 hours of serum starvation (Fig. 3C, D).
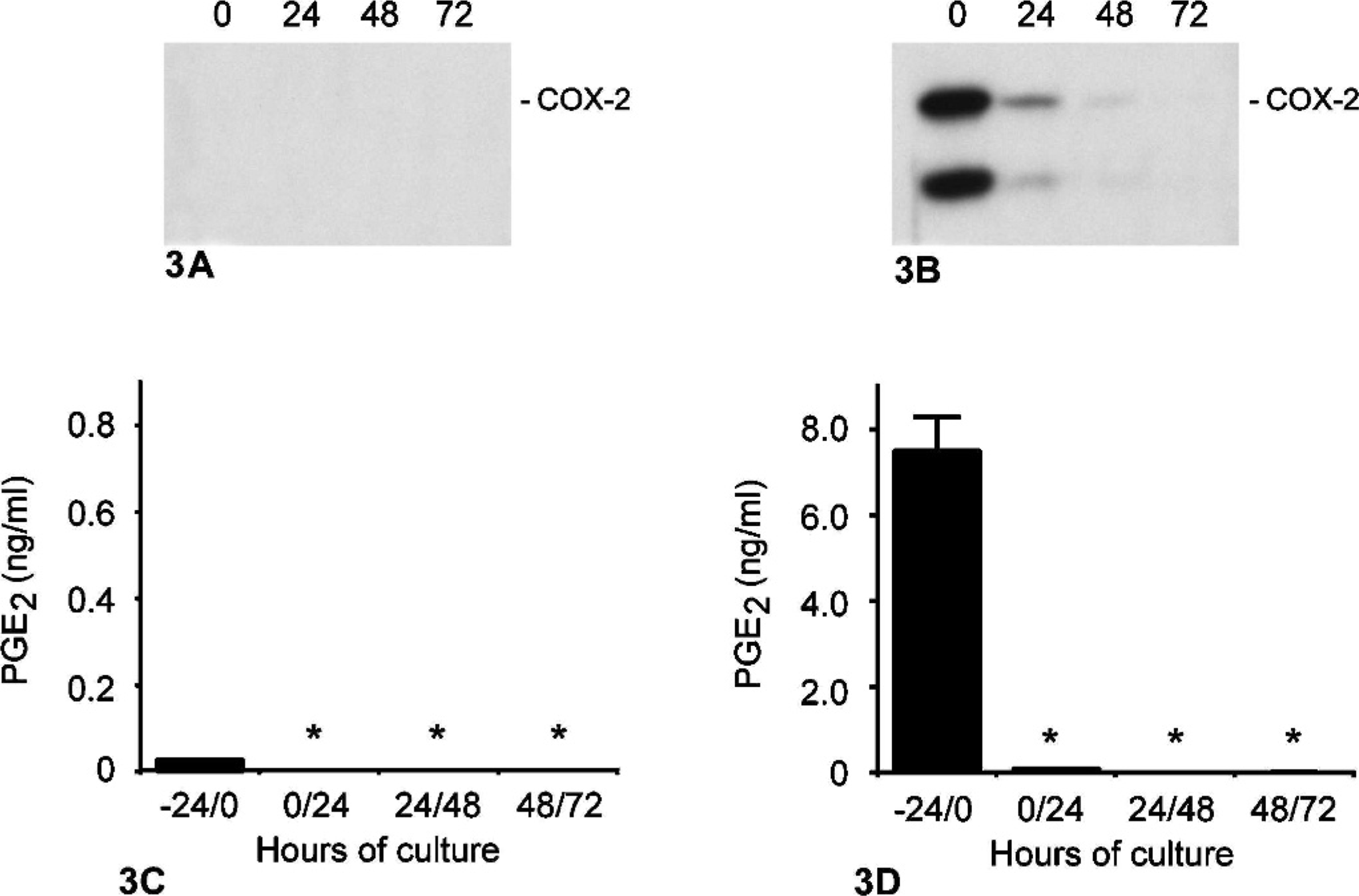
Effect of serum deprivation on COX-2 expression and PGE2 synthesis in normal and neoplastic canine mammary epithelial cells. Canine mammary epithelial cells were cultured in complete medium (0 hours) or in serum-deprived medium for 24, 48, or 72 hours, as described in Materials and Methods. Cell extracts were prepared from cultures of normal (A; CF35) and neoplastic (B; CMT12), and proteins (100 μg/lane) were analyzed by one-dimensional SDS-PAGE and immunoblotting techniques using a COX-2 selective antibody. Markers on the right indicate migration of intact COX-2 protein (Mr = 72,000). Concentrations of PGE2 in media of normal (C; CF35) and neoplastic (D; CMT12) were determined by radioimmunoassays. Bars marked with an asterisk are significantly different from -24/0 hours time point (P < .05).
PMA-dependent regulation of COX-2 expression and PGE2 synthesis in canine mammary cell lines
To investigate the regulation of COX-2 in canine mammary cells, CF35 and CMT12 cells were serum-starved for 40 hours to decrease endogenous expression of COX-2 protein, and cells were cultured in the absence or presence of the phorbol ester PMA for 1, 3, 6, 12, or 24 hours. In control cultures of CF35 and CMT12, levels of COX-2 remained low and stable (Fig. 4A, B [controls]). Stimulation with PMA (5 ng/ml) resulted in a time-dependent induction of COX-2 in both cell lines (Fig. 4A, B [PMA]). Induction of COX-2 by PMA was first observed at 6 hours in CF35 cells, and became maximal at 12 and 24 hours. Immunocytochemical localization of COX-2 revealed an intense cytoplasmic staining in CF35 cells following 24 hours of PMA stimulation (Fig. 2B). In CMT12 cells, COX-2 levels had slightly increased after 3 hours, and a marked induction was present between 6 and 24 hours of PMA stimulation, being maximal at 24 hours (Fig. 4B [PMA]). The induction of COX-2 by PMA appeared stronger in CMT12 compared to CF35 cells, as previously observed for basal COX-2 expression. Production of PGE2 was measured in agonist-stimulated cultures. PGE2 production remained low and unchanged between 1 and 24 hours in control cultures of CF35 and CMT12 cells (Fig. 5A, B [controls]), but treatment with PMA caused a significant increase in PGE2 synthesis in both cell lines (Fig. 5A, B [PMA]). The effect of PMA on PGE2 production by CMT12 was clearly more pronounced than on CF35, with levels at 24 hours being 288-fold higher in CMT12 than in CF35 cells (14.4 ± 0.6 versus 0.05 ± 0.0 ng/ml, respectively) (Fig. 5A, B [PMA]).
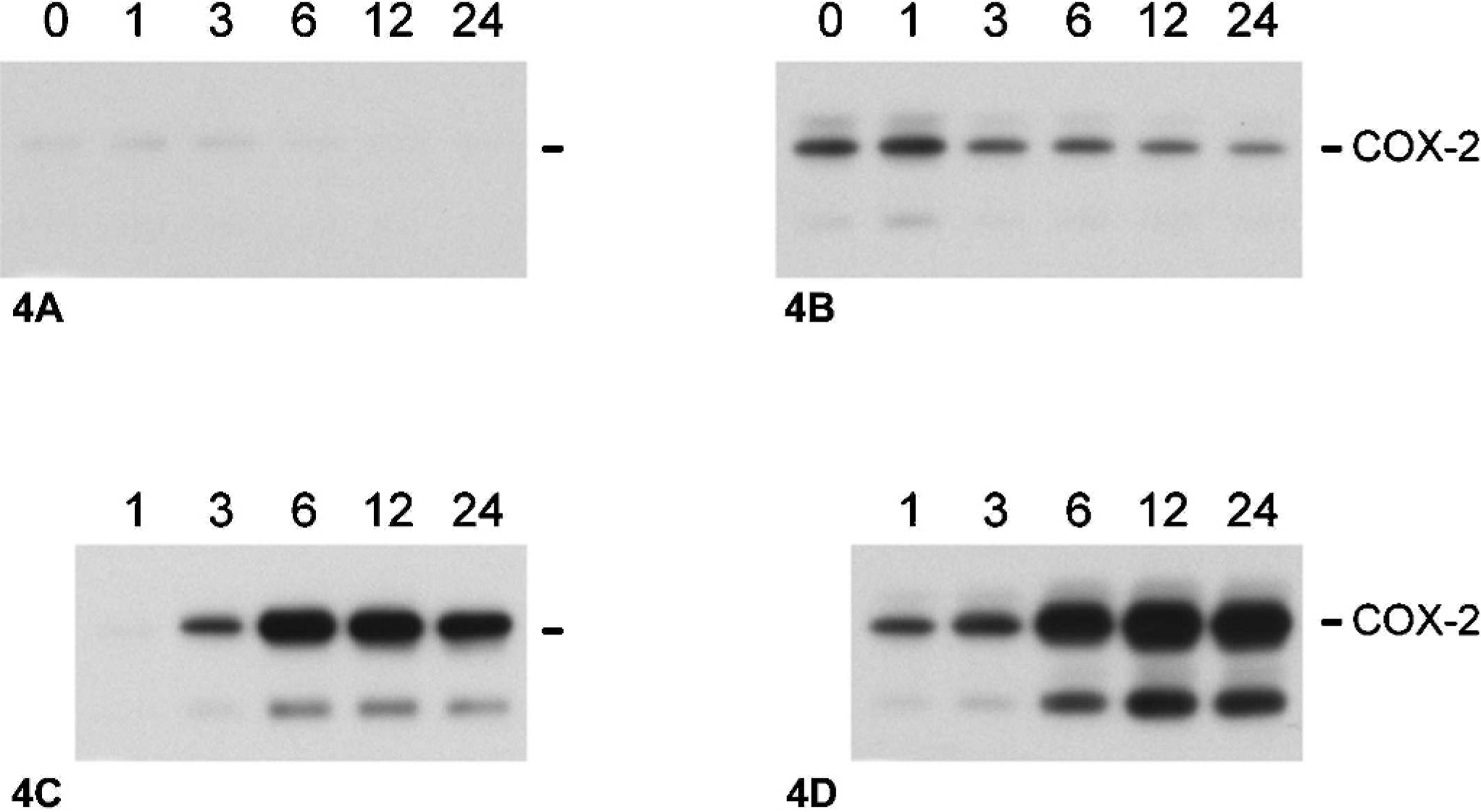
Effect of phorbol 12-myristate 13-acetate (PMA) on COX-2 protein in normal and neoplastic canine mammary epithelial cells. Normal and neoplastic canine mammary epithelial cells were serum starved for 40 hours, then cultured in the absence (A, normal; B, neoplastic) or presence (C, normal; D, neoplastic) of 5 ng/ml PMA for 1, 3, 6, 12, or 24 hours, as described in Materials and Methods. Cell extracts were prepared from cultures, and proteins (100 μg/lane) were analyzed by one-dimensional SDS-PAGE, and immunoblotting techniques using a COX-2 selective antibody. Markers on the right indicate migration of intact COX-2 protein (Mr = 72,000).
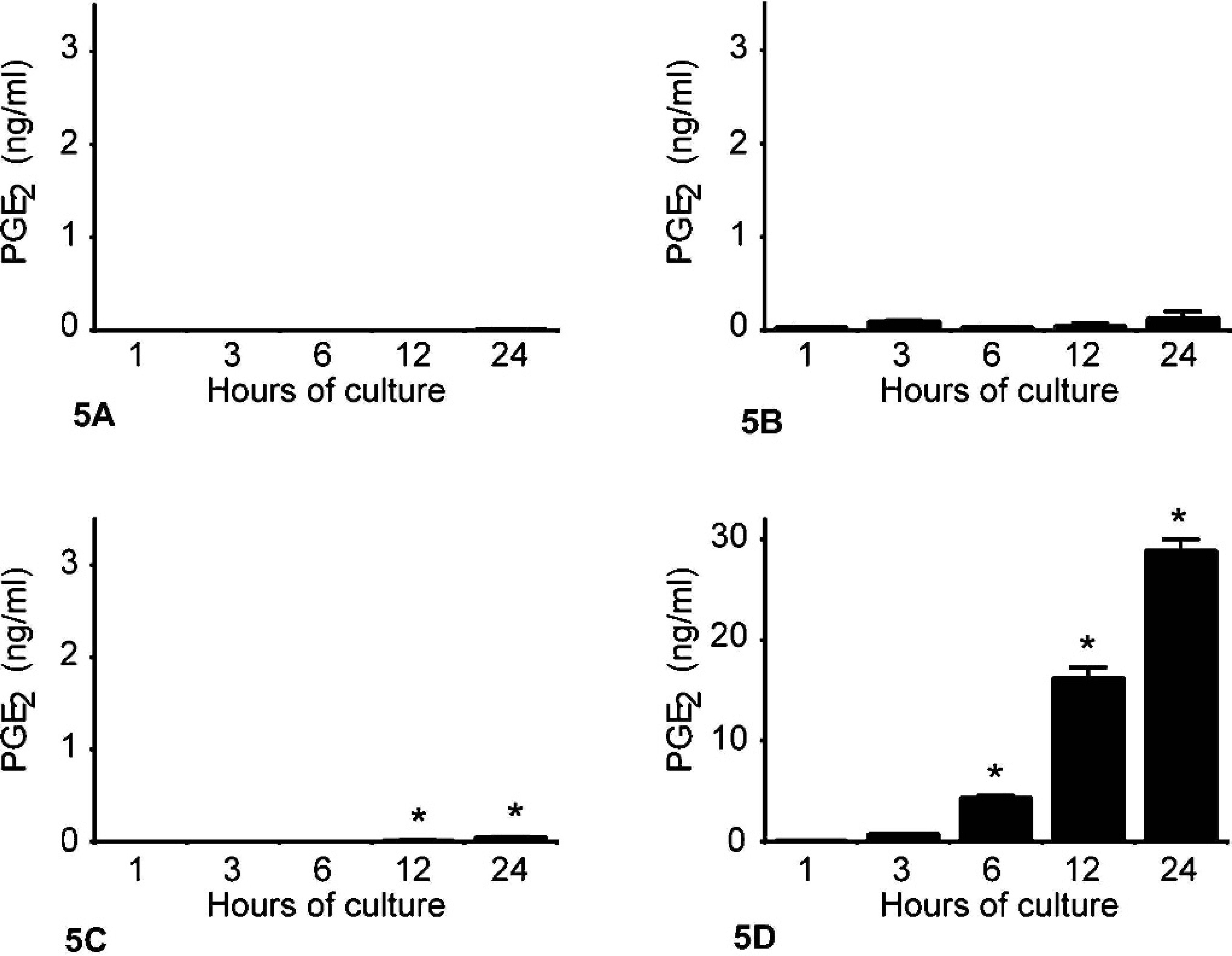
Effect of PMA on PGE2 synthesis by normal and neoplastic canine mammary epithelial cells. Normal and neoplastic canine mammary epithelial cells were serum starved for 40 hours, then cultured in the absence (A, normal; B, neoplastic) or presence (C, normal; D, neoplastic) of 5 ng/ml PMA for 1, 3, 6, 12, or 24 hours, as described in Materials and Methods. Concentrations of PGE2 in media were determined by radioimmunoassays. Bars marked with an asterisk are significantly different from 1 hour time point (P < .05).
Effect of COX-2 inhibition on PGE2 synthesis and cell proliferation
NS-398 is a selective COX-2 inhibitor, 16 and its effect on PGE2 synthesis in CMT12 cells was evaluated. NS-398 caused a dose-dependent and dramatic inhibition of PGE2 synthesis in CMT12 after 48 hours of treatment (Fig. 6). A significant inhibition was observed at a concentration as low as 1 μM of NS-398 (Fig. 6). The effect of this COX-2 inhibitor on cell proliferation was also tested. Results showed that NS-398 caused a dose-dependent reduction of cell proliferation, with a significant inhibitory effect at 1 μM of NS-398 (Fig. 7).
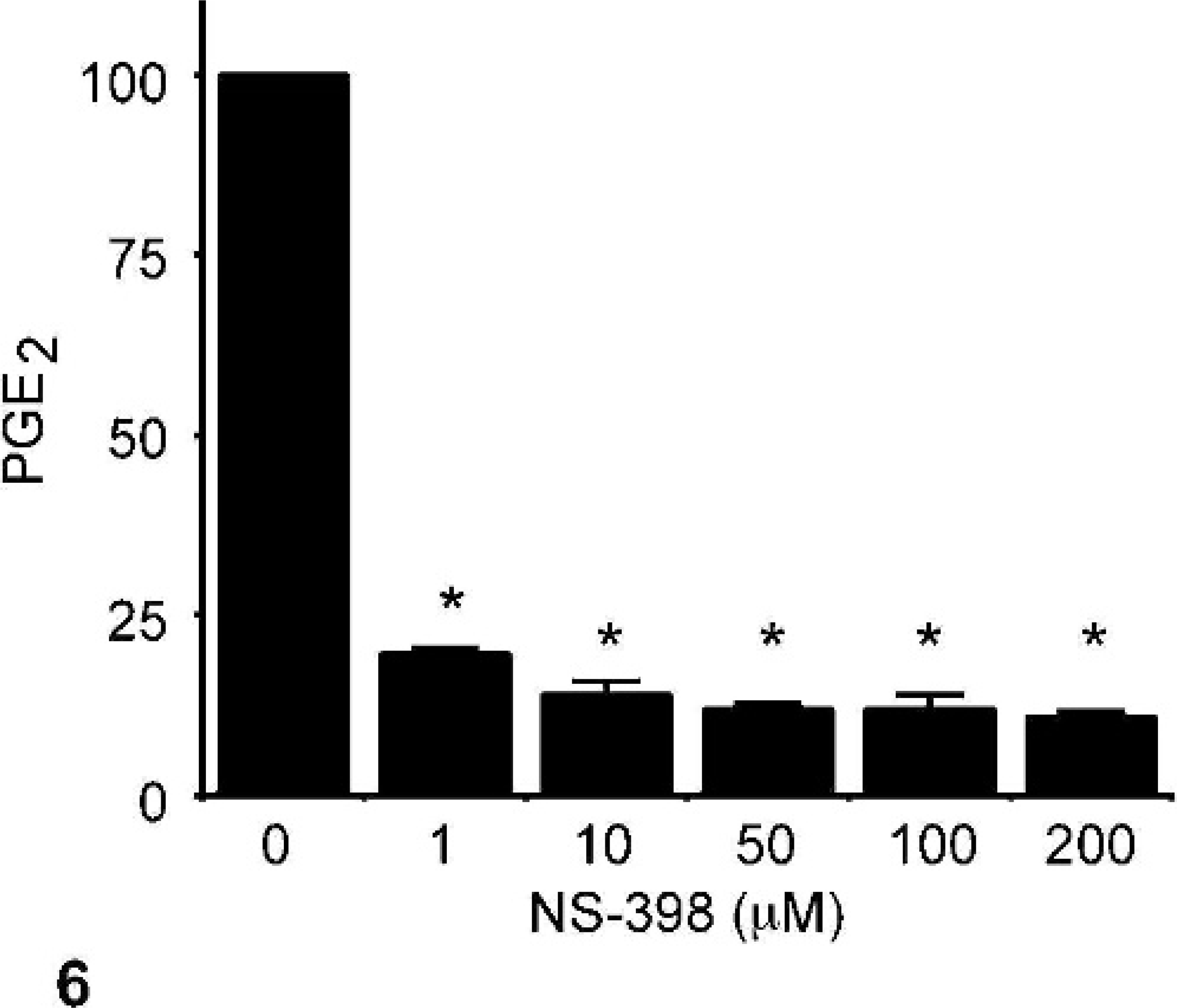
Effect of COX inhibition on PGE2 synthesis by neoplastic canine mammary epithelial cells. Neoplastic canine mammary cells (CMT12) were cultured in 24-well plates in the absence (0) or presence of 1, 10, 50, 100, or 200 μg/ml NS-398 for 48 hours, as described in Materials and Methods. Concentrations of PGE2 in culture medium were determined by radioimmunoassay. Bars marked with an asterisk are significantly different from control without inhibitor (P < .05).
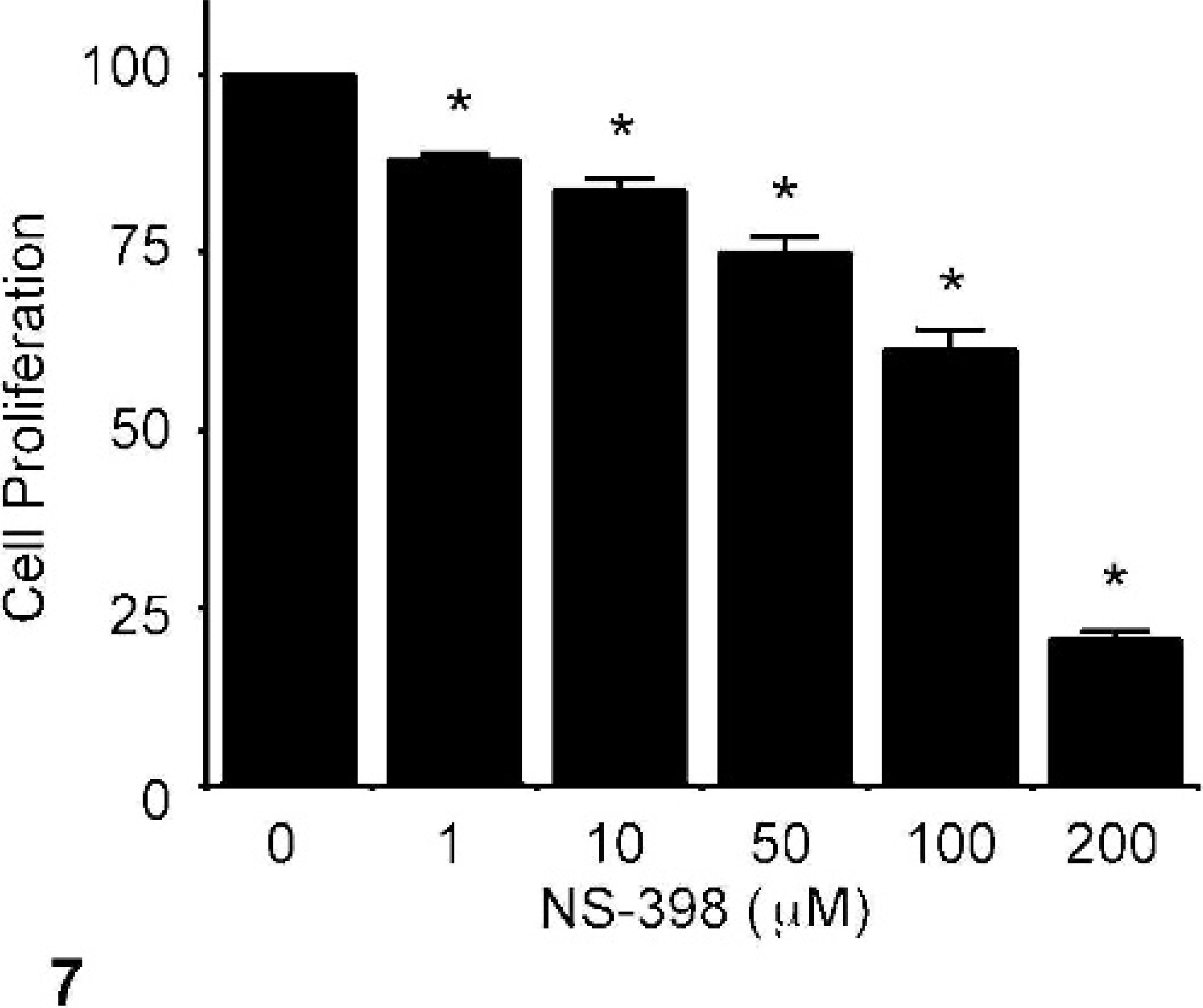
Effect of COX inhibition on cell proliferation of neoplastic canine mammary cells. Neoplastic canine mammary cells (CMT12) were cultured in the absence (0) or presence of 1, 10, 50, 100, or 200 μg/ml NS-398 for 48 hours, as described in Materials and Methods. Results for cell proliferation are presented as percentages compared to control cultures without inhibitor. Bars marked with an asterisk are significantly different from control without inhibitor (P < .05).
Discussion
The present study characterizes for the first time the expression and regulation of COX-2 in canine mammary cell lines. Our results indicate that, of the 5 canine neoplastic cell lines that were analyzed, 1 (CMT12) strongly overexpressed COX-2. The prevalence of COX-2 expression among the cell lines tested (1 out of 5 cell lines; 20%) is lower than recently reported in vivo findings on COX-2 expression in canine mammary tumors. 12, 22 Indeed, in a study of 84 canine malignant mammary tumors, we found that approximately half of the tumors were COX-2 positive. 12 However, COX-2 expression was not uniform in the tumors, with an average of 25–50% of neoplastic cells being COX-2 positive. Similarly, Heller et al. 22 found that 46% of the 37 adenocarcinomas they evaluated were COX-2 positive. The absence of COX-2 proteins in several cell lines likely reflects the negative COX-2 status of the tumor cells from which they were initially derived. In humans, the reported incidence of COX-2 expression in breast cancer tissues varies greatly, ranging from 4.5 to 100% depending on the study. 19, 25, 34, 42, 44, 53 A proposed explanation for this important variation is that COX-2 positive breast tumor could represent a subset of breast tumors that are also HER-2-positive. 56 Two studies have so far looked at HER-2 expression in canine mammary tumors, and reported detection of HER-2 in 73.9 and 19.1% of malignant tumors. 1, 45 The HER-2 status of the COX-2-positive malignant tumors in dogs will need to be investigated.
Of the 5 neoplastic cell lines, CMT12 showed the highest constitutive COX-2 expression and synthesized elevated amount of PGE2. PGE2 is the predominant prostaglandin metabolite produced by COX-2, and elevated levels of PGE2 have been found in some canine mammary tumors. 37 PGs have widespread effects and one of the possible consequences of increased PGE2 synthesis by tumor cells is to stimulate cell proliferation. PGE2 has been shown to induce mitogenesis in mammary epithelial cells in the presence of EGF. 3 Evidence suggests that prostaglandins overproduction could also stimulate proliferation through estrogen production. 11 In particular, PGE2 has been demonstrated to increase the aromatase gene CYP19 that is responsible for estrogen synthesis, and a positive correlation has been reported between COX expression and aromatase in human breast cancer specimens. 7, 8 Additionally, COX-2 expression has been associated with inhibition of apoptosis in different cell types, 24, 49 a mechanism that could also contribute to the reduced cell proliferation observed with the COX-2 inhibitors. PGE2 can also affect the immune system by depressing cell proliferation of T and B cells and cytokine synthesis, and by altering antigen processing by dendritic cells, 54, 62 and appears to be implicated in the angiogenic process. A significant correlation has been reported between COX-2 and the vascular growth factor (VEGF) mRNA copy numbers in invasive breast cancer specimen. 28 The effects of COX-2-derived PGs on cell proliferation, apoptosis, and vascular growth likely contribute to the tumorigenic process.
Inhibition of COX-2 with NS-398 resulted in a drastic reduction of PGE2 synthesis by CMT12 cells, even at the lowest concentration of inhibitor tested (1 μM). In comparison, its effect on cell proliferation was not as pronounced, and only the highest concentration of inhibitor (200 μM) was able to decrease cell proliferation as much as PGE2 synthesis. Similarly, Kundu et al. 30 reported that the growth inhibition of a murine mammary tumor cell line was observed only at concentrations of NS-398 greater than those required to ablate PGE2 synthesis. This suggests that the inhibitory effect of these COX inhibitors on cell proliferation may involve mechanisms that are dependent and independent of PGE2 synthesis.
The mechanisms underlying the constitutive overexpression of COX-2 observed in CMT12 cells are unknown. COX-2 expression is regulated at the transcriptional and post-transcriptional levels, and can also be regulated by the rate of protein synthesis and degradation. 23 The human COX-2 promoter contains multiple transcription factor binding sites, including a cAMP response element, and potential binding sites for Myb, nuclear factor interleukin-6, nuclear factor 6B, and Ets factors. 23 The relative importance of these different factors is likely to vary in different tissues. A recent study revealed that PEA3 and cAMP response elements are required for HER-2/neu-mediated induction of transcription in human mammary epithelial cells. 56 Canine COX-2 has been characterized and sequenced. 5 The canine COX-2 coding region encodes a 604–amino acid protein, which is identical in length to that of human and all other known mammalian COX-2 proteins except ovine COX-2 that has only 603 residues. 63 Comparisons between canine and human COX-2 revealed an 89% identity at the amino acid level, with all putative structural and functional domains implicated in COX-2 function conserved in the canine protein. 6 The canine COX-2 promoter remains to be characterized. Analyses and comparison of the canine COX-2 promoter between the different cell lines could help to understand the basis for the constitutive expression of COX-2 in some mammary cell lines.
In contrast to colon cancer where epidemiologic evidence has established a correlation between the long-term use of NSAIDs and a reduction in colon cancer incidence, such a link is not as clear for breast cancer. Indeed, some studies found no correlation between the use of NSAIDs and breast cancer incidence. 13, 41, 57 However, several studies have found an association between the use of NSAIDs and a reduction in the risk of breast cancer. 10, 14, 21, 47, 48 In dogs, only 1 clinical study has so far investigated the use of NSAIDs and various cancers in dogs. 29 In this study, a partial remission was reported in 1 of 3 dogs displaying mammary carcinomas that were treated with piroxicam, a COX inhibitor. 29
In summary, we have shown that, as observed for canine mammary adenocarcinomas in vivo, there is variation in the level of COX-2 expression between different cell lines of canine mammary epithelial cells. Malignant mammary cells overexpressing COX-2 also produced elevated amount of PGE2 that could contribute to the tumorigenic process, and the prostaglandin synthesis could be blocked by a selective COX-2 inhibitor. Further understanding of the role played by prostaglandins in canine mammary tumors may offer new chemopreventive or chemotherapeutic avenues.
Footnotes
Acknowledgements
We thank Dr. Stacia Kargman, Merck Frosst Centre for Therapeutic Research, Pointe-Claire-Dorval, Québec, for kindly providing antibody MF243, and Ms. Danielle Rannou for technical assistance. This work was supported in part by grants from the American Kennel Club/Canine Health Foundation (MD and JS), the Natural Sciences and Engineering Council of Canada (MD), and the Canadian Institutes for Health Research (CIHR) (JS). JS is the recipient of a CIHR Investigator Award.
