Abstract
Mammary tumors are the most common neoplasms in female dogs. Induction of cyclooxygenase-2 (COX-2) has been implicated in various cancers in humans. However, expression of COX-2 has not been investigated in canine mammary tumors. Normal mammary gland (n = 4), simple or complex adenomas (n = 63), and simple or complex adenocarcinomas (n = 84) were studied by immunohistochemistry. Results showed that COX-2 was not expressed in the normal gland but was detected in 24% of adenomas and in 56% of adenocarcinomas (P < 0.001). The incidence of COX-2 expression and the intensity of the COX-2 signal were higher in adenocarcinomas than in adenomas (P < 0.001). These results demonstrate for the first time that COX-2 is induced in a proportion of canine mammary tumors and that COX-2 expression is more frequent and more intense in malignant than in benign tumors, suggesting a potential role for COX-2 in canine mammary tumorigenesis.
Mammary tumors are the most common neoplasms in female dogs and the second most common tumors in dogs of both sexes. 15 Both benign and malignant tumors are frequently diagnosed, and malignant neoplasms represent approximately 50% of all mammary tumors. 15 Canine mammary tumors are age-dependent neoplasms, with a mean age of 6–7 years at onset. 1 All breeds of dogs can be affected, but certain breeds (sporting breeds for example) appear to show a higher prevalence. 15 Canine mammary tumors are hormone-dependent neoplasms, and early ovariectomy (i.e., before the first estrous cycle) has a major protective effect. 19 Indeed, progestin-induced biosynthesis of growth hormone has recently been proposed as one of the mechanisms that could promote mammary tumorigenesis in dogs. 26 Also, p53 mutations, a common acquired genetic abnormality found in human cancer, have recently been reported in mammary tumors of the dog. 16
The enzyme prostaglandin G/H synthase, also known as cyclooxygenase (COX), controls the first rate-limiting step in the synthesis of all prostaglandins from arachidonic acid. 21 COX is a homodimer composed of two subunits of about 70,000 d and one heme group. 25 The enzyme has two catalytic activities that mediate the conversion of arachidonic acid into a series of different prostanoids. Two isoforms of COX exist and are referred to as COX-1 and COX-2. 27 They share important similarities at the protein level but are derived from distinct genes and are encoded by messenger ribonucleic acid of different sizes. The two isoforms also differ markedly in their expression. COX-1 is present in a wide variety of tissues and is referred to as the constitutive form, whereas COX-2 is generally undetectable is most tissues but can be induced by a variety of agonists and is referred to as the inducible form. COX-1 and −2 are the targets of numerous nonsteroidal anti-inflammatory drugs (NSAIDs). 2
Mounting evidence suggests that COX-2 plays a role in various cancers in humans and in animals. 23 Increased expression of COX-2 was first demonstrated in human colorectal cancers but has now been reported in several different types of cancers in both humans and animals. 8 , 9 , 17 , 24 Recent studies looking at the expression of COX isoforms in human breast cancer reported high levels of COX-2 protein in some tumors. 4 , 18 , 22 Also, epidemiologic studies of the effects of NSAIDs on human breast cancer have found a statistically significant reduction of breast cancer risk with the use of NSAIDs. 5 , 20 Interestingly, overexpression of COX-2 in mice has recently been shown to be sufficient to induce mammary gland tumorigenesis. 12 In sharp contrast to this rapidly growing information on COX-2 in the oncogenesis of breast cancer, the expression and possible implication of COX-2 in mammary tumors in dogs remain uncharacterized.
One hundred forty-seven cases of canine mammary tumors submitted to the Département de pathologie et microbiologie of the Faculté de médecine vétérinaire (Université de Montréal, Québec, Canada) were included in the study. All cases were confirmed as mammary tumors by the examination of hematoxylin-eosin-saffran–stained (HES) sections by a veterinary pathologist (M. Doré). The histologic classification used was the World Health Organization International Histological Classification of Tumors of Domestic Animals. 13 Normal mammary tissues were obtained from four adult mongrels (aged between 3 and 4 years) euthanatized for reasons unrelated to health problems. All tissues studied were fixed in 10% neutral buffered formalin.
Immunohistochemical staining was performed using the Vectastain ABC kit (Vector Laboratories, Burlingame, CA), as described previously. 24 Briefly, formalin-fixed tissues were paraffin embedded, and 3-μm-thick sections were prepared and deparaffined through graded alcohol series. Endogenous peroxidase was quenched by incubating the slides in 0.3% hydrogen peroxide in methanol for 30 minutes. After rinsing in phosphate-buffered saline (PBS) for 15 minutes, sections were incubated with diluted normal goat serum for 20 minutes at room temperature. The anti–COX-2 antibody used was MF243, a polyclonal antibody that was raised in rabbits against ovine placental COX-2 and whose selectivity for COX-2 in dogs has previously been characterized. 24 MF243 diluted in PBS (1:7,500 dilution) was applied, and sections were incubated overnight at 4 C. Control sections were incubated with PBS or with nonimmune rabbit serum. After rinsing in PBS for 10 minutes, a biotinylated goat anti-rabbit antibody (1:222 dilution; Vector Laboratories) was applied, and sections were incubated for 45 minutes at room temperature. Sections were washed in PBS for 10 minutes and incubated with the avidin DH–biotinylated horseradish peroxidase H reagents for 45 minutes at room temperature. After PBS wash for 10 minutes, the reaction was revealed using diaminobenzidine tetrahydrochloride as the chromogen. Sections were counterstained with Gill's hematoxylin stain, mounted, and immunoreactivity was evaluated by two observers (M. Doré and J. Sirois). The percentage of COX-2–positive tumor cells was graded as 0 = <5%, 1 = 5–25%, 2 = 26–50%, 3 = 51–75%, and 4 = >75%, and the intensity of COX-2 immunoreactivity was graded as − = no, + = weak, ++ = moderate, and +++ = strong staining. Each COX-2 score represented the product of percentage of positive tumor cells and intensity, as described previously. 11 Chi-square and Pearson's tests were used to analyze frequencies of COX-2 expression between normal mammary tissues, mammary adenomas, and mammary adenocarcinomas, whereas Student's t-test was used to compare COX-2 scores and age of dogs between adenomas and adenocarcinomas. Statistical analyses were performed using the JMP Software (SAS Institute Inc., Cary, NC).
The age of dogs with mammary adenomas (including both simple and complex types) ranged from 2.8 to 13.6 years with a mean and standard deviation of 8.3 ± 2.4 years, whereas the age of dogs with adenocarcinomas (also including both simple and complex types) ranged from 4.7 to 15.2 with a mean and standard deviation of 9.6 ± 2.3 years (P < 0.01).
Normal mammary gland tissues (n = 4 dogs) were evaluated to determine whether COX-2 is expressed in this tissue. Results showed that normal canine mammary gland did not express COX-2 (Figs. 1A and 2). In contrast, 15 of 63 (24%) adenomas displayed some COX-2 expression (Fig. 1A; P < 0.001). Benign tumors included 23 simple and 40 complex adenomas, and COX-2 expression was found in 35% (8 of 23 cases) and 18% (7 of 40 cases) of simple and complex adenomas, respectively. In these tumors, COX-2 appeared as a diffuse pale cytoplasmic staining (Fig. 3). When the immunoreactive signal was evaluated in a semiquantitative manner, adenomas had a mean COX-2 score of 0.5 ± 0.3 (Fig. 1B).
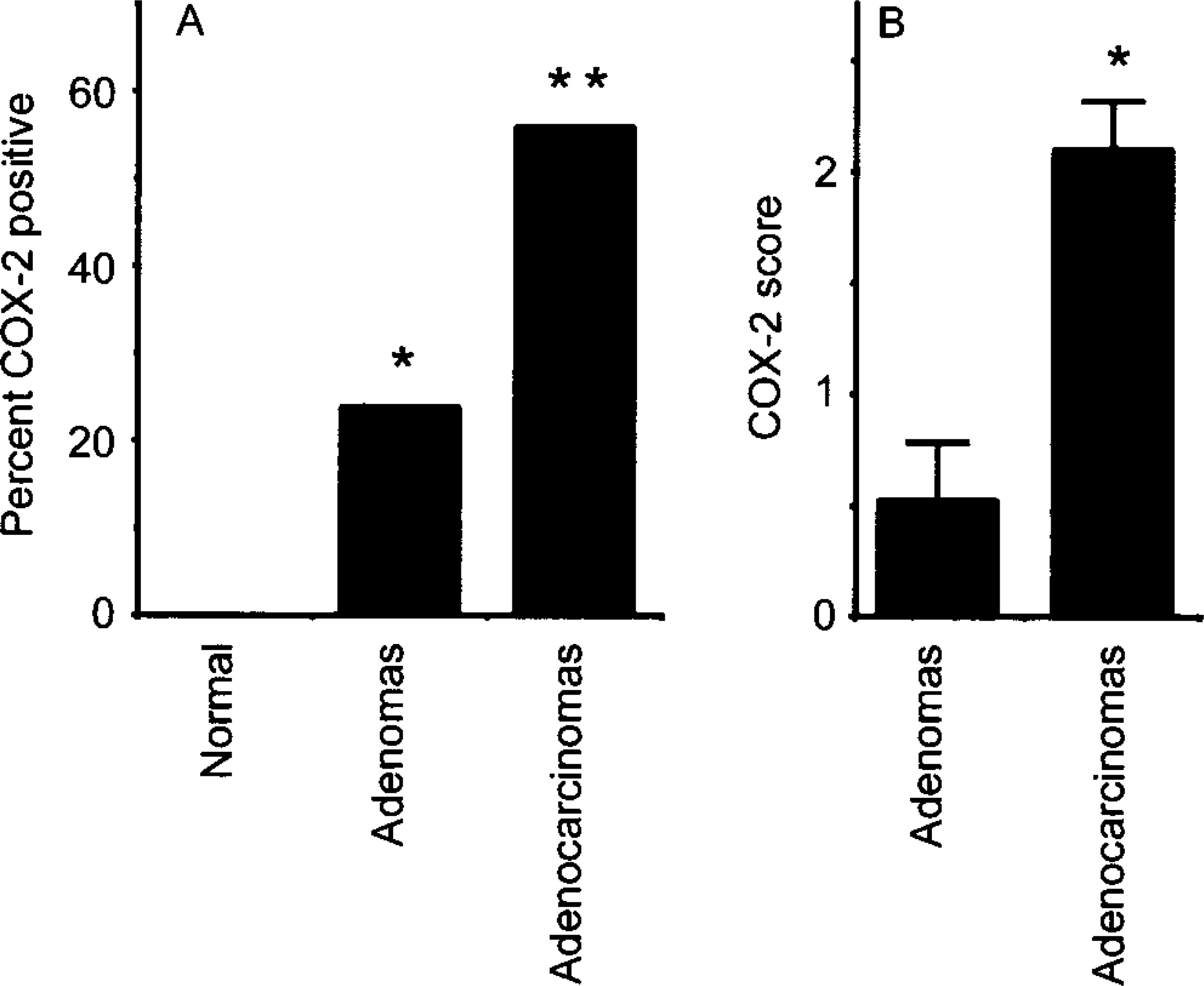
Expression of COX-2 in canine mammary tumors. Fig. 1A. Incidence of COX-2 expression in normal mammary tissues (n = 4), and in mammary adenomas (n = 63) and adenocarcinomas (n = 84). Bars marked with asterisk(s) are significantly different from normal tissues; the bar marked with two asterisks is significantly different from the one with one asterisk (P < 0.001). Fig. 1B. COX-2 scores (mean ± SEM; for more details see text) in adenomas and adenocarcinomas. The asterisk indicates a significant difference (P < 0.001).
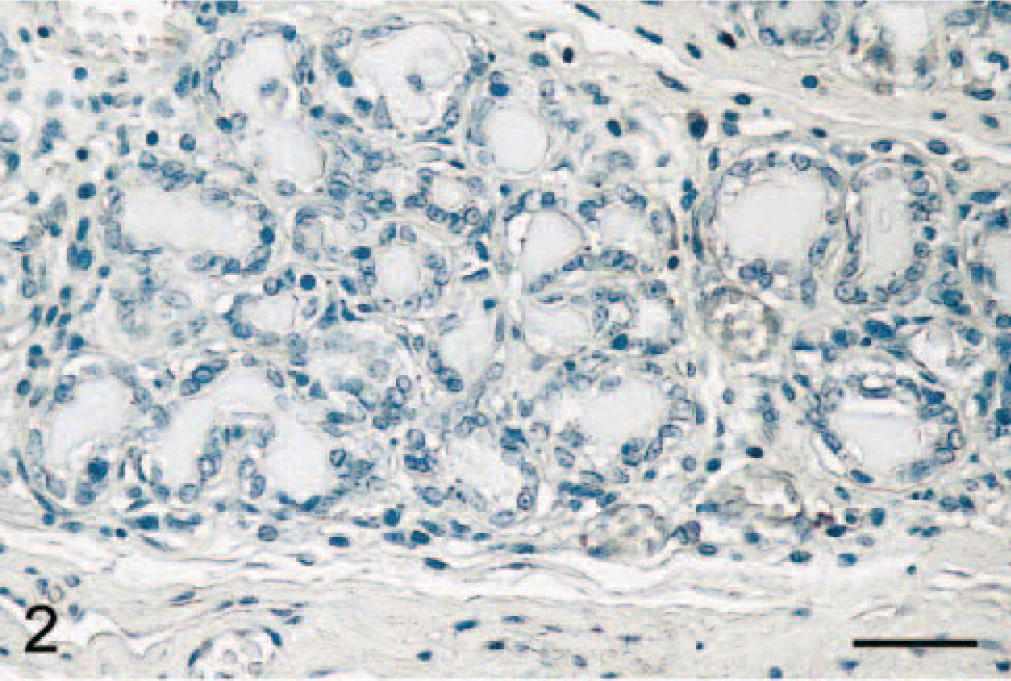
Mammary gland; dog. Epithelial cells from normal acini do not express COX-2. Avidin–biotin–peroxidase complex method, Gill's hematoxylin counterstain. Bar = 35 μm.
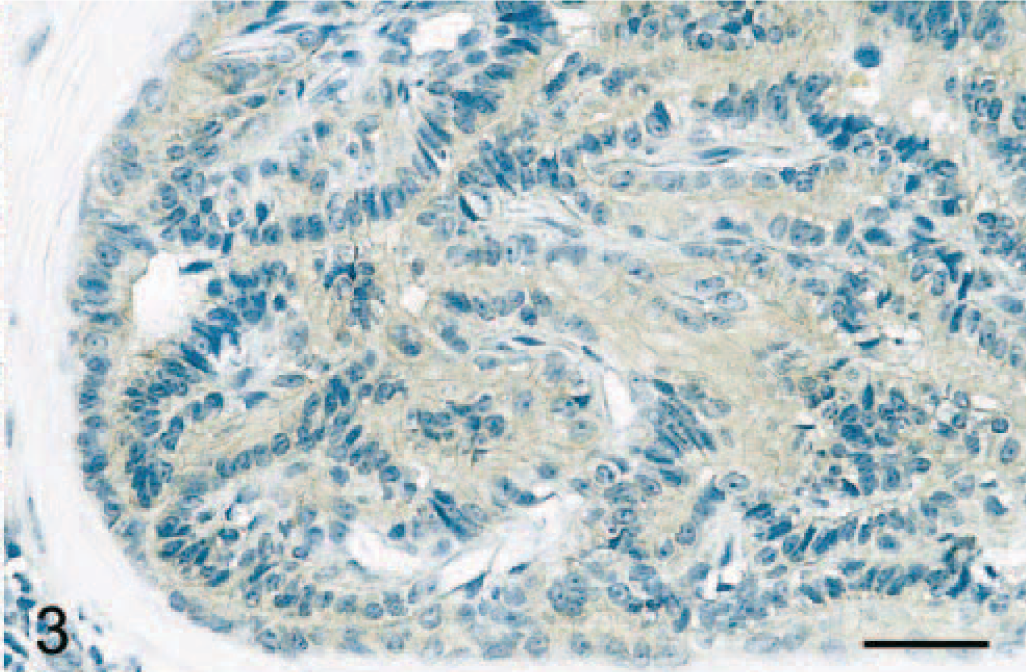
Mammary gland; dog. Adenoma. Neoplastic epithelial cells display a weak diffuse cytoplasmic COX-2 immunostaining. Avidin–biotin–peroxidase complex method, Gill's hematoxylin counterstain. Bar = 35 μm.
Immunostaining of malignant tumors revealed that 47 of 84 (56%) adenocarcinomas were COX-2 positive (Fig. 1A; P < 0.001). Malignant tumors included 45 simple and 39 complex adenocarcinomas, and COX-2 expression was observed in 47% (21 of 45 cases) and 67% (26 of 39 cases) of simple and complex adenocarcinomas, respectively (Tables 1 and 2). In both simple and complex adenocarcinomas, COX-2 expression was often localized to the cytoplasm of tumor cells (Figs. 4 and 5). In 14 tumors (of simple or complex type) where foci of necrosis were present, fusiform fibroblast-like cells bordering the necrotic area were often strongly positive for COX-2 (Fig. 6). Also, in 15 adenocarcinomas of the complex type, the cytoplasm of myoepithelial cells was COX-2 positive (Fig. 7). COX-2–expressing inflammatory cells were observed in the stroma of few adenocarcinomas. In most cases where nonneoplastic mammary lobules were present in the section, normal epithelial cells were COX-2 negative, except for patchy COX-2 cytoplasmic staining observed in 15 cases. Adenocarcinomas had a significantly higher mean COX-2 score (2.1 ± 0.2) compared with adenomas (Fig. 1B; P < 0.001). Fifteen of 84 (17.9%) adenocarcinomas displayed COX-2 scores of 5 and higher.
COX-2 expression in simple adenocarcinomas.
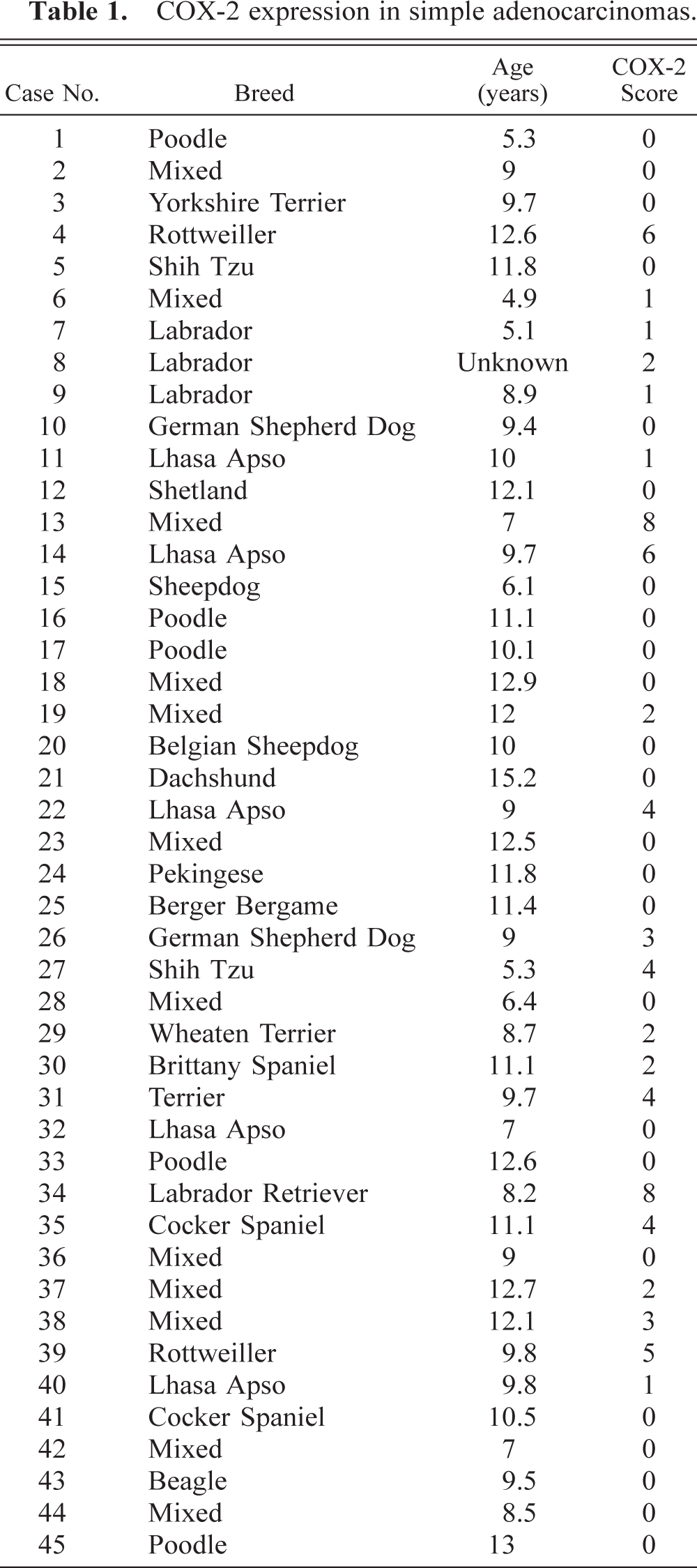
COX-2 expression in complex adenocarcinomas.
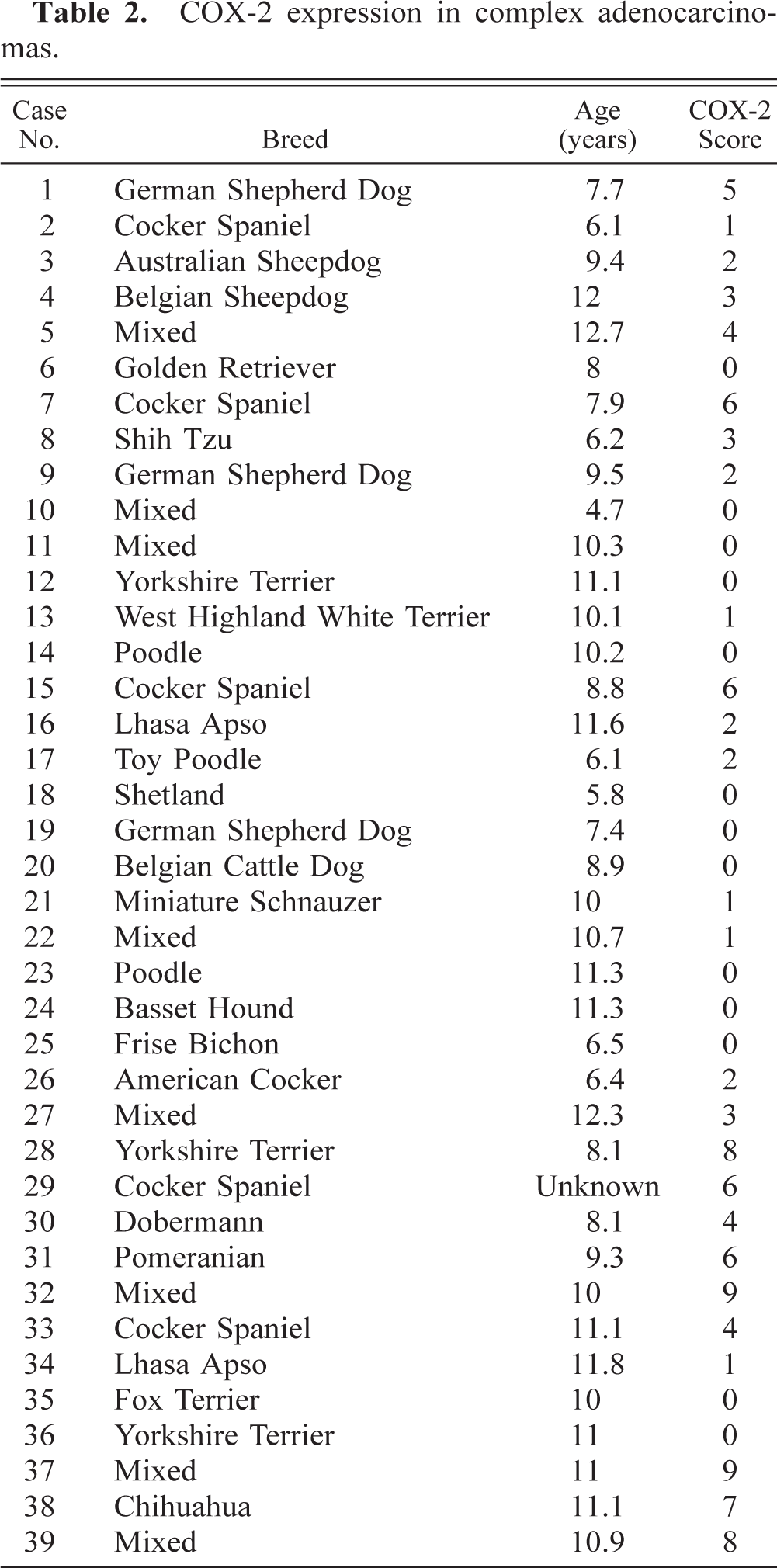
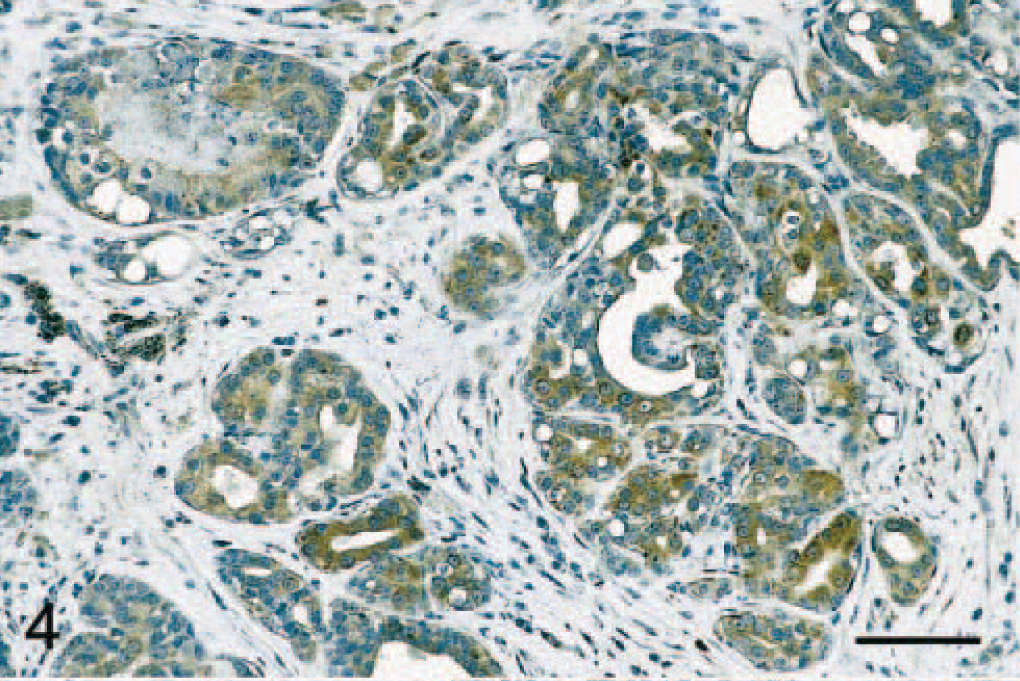
Mammary gland; dog. Adenocarcinoma. Moderate to intense COX-2 immunoreactivity is found in the cytoplasm of neoplastic eipthelial cells. Avidin–biotin–peroxidase complex method, Gill's hematoxylin counterstain. Bar = 70 μm.
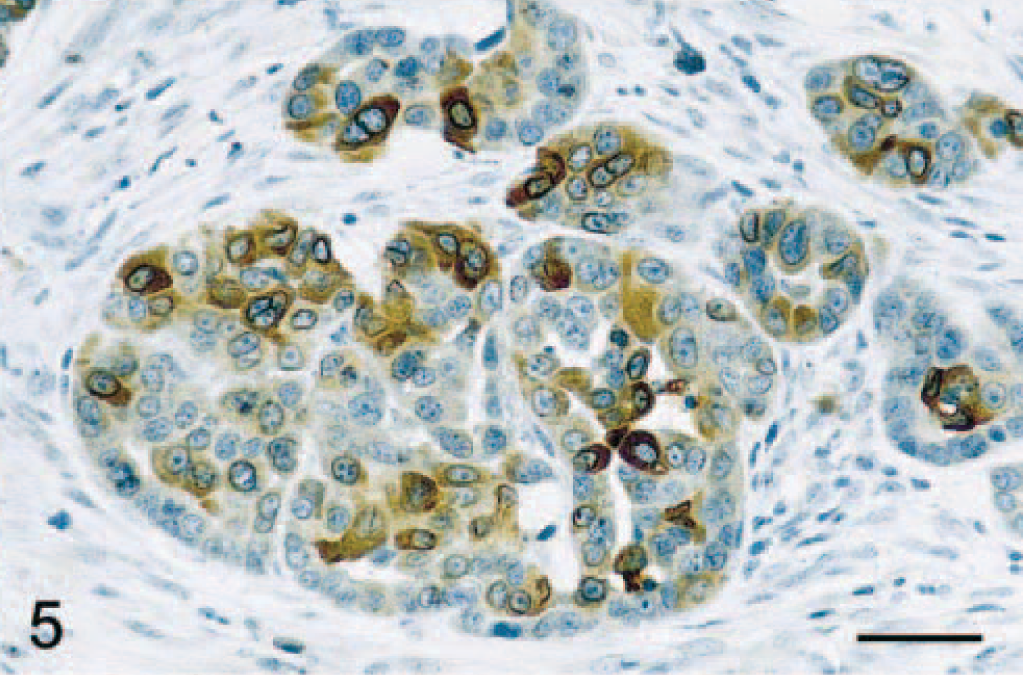
Mammary gland; dog. Adenocarcinoma. Intense COX-2 immunoreactivity is found in the cytoplasm of neoplastic epithelial cells. Avidin–biotin–peroxidase complex method, Gill's hematoxylin counterstain. Bar = 35 μm.
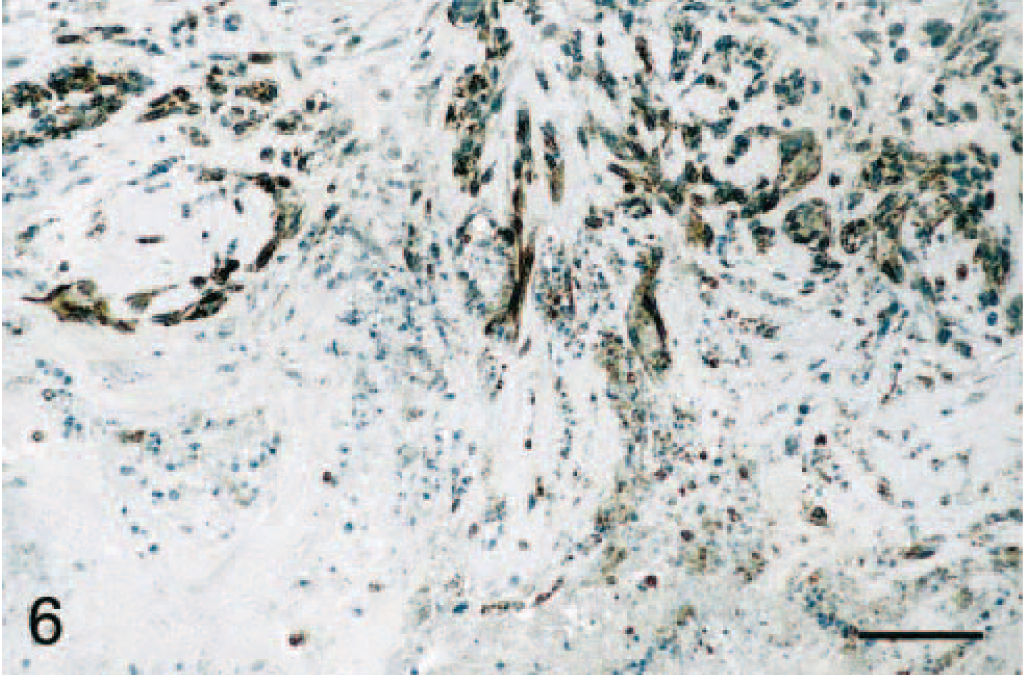
Mammary gland; dog. Adenocarcinoma. Fusiform cells bordering an area of necrosis are intensely COX-2 positive. Avidin–biotin–peroxidase complex method, Gill's hematoxylin counterstain. Bar = 70 μm.

Mammary gland; dog. Adenocarcinoma. COX-2 expression is present in myoepithelial cells of some tumors. Avidin–biotin–peroxidase complex method, Gill's hematoxylin counterstain. Bar = 35 μm.
This study is the first to document the expression of COX-2 in canine mammary tumors. Our results indicate that a small proportion (24%) of canine mammary adenomas weakly expressed COX-2, whereas more than half of the malignant tumors (56%) evaluated in the study displayed weak to strong COX-2 staining. In humans, there is some variation as to the reported incidence of COX-2 expression in breast cancer. Indeed, the first study of COX-2 expression in breast cancer reported only 2 positive cases out of 44 breast cancer specimens studied by immunoblot analysis. 7 However, Soslow et al. 22 later found that 7 of 17 (41.2%) invasive breast cancers expressed COX-2 protein. More recently, two studies detected COX-2 expression in 37.4% and 43% of invasive breast cancers, 4 , 18 whereas 62.5% of ductal carcinomas in situ were COX-2 positive by immunohistochemistry. 4 Our results reveal that COX-2 is also expressed in a significant proportion of mammary tumors in dogs, with moderate to high COX-2 expression being found in approximately 18% of adenocarcinomas. The induced expression of COX-2 in these tumors likely results in increased prostaglandin synthesis. Accordingly, a recent study reported elevated concentrations of prostaglandin E2 in three out of eight canine mammary carcinomas evaluated. 14
Morphologic classification of mammary tumors in dogs is complex with many proposed systems of classification, each of them including several different types of tumors. 3 , 13 , 15 We chose to look at COX-2 expression in two broad categories, benign versus malignant tumors, and to evaluate tumors of the simple and the complex types. We found that significantly more malignant tumors expressed COX-2 than did benign tumors and that the intensity of COX-2 expression was significantly higher in malignant tumors. No significant difference in COX-2 expression was present between tumors of the simple type versus the complex type. However, COX-2 staining was observed in the myoepithelial cells of some adenocarcinomas, suggesting that these cells also could contribute to prostaglandin synthesis in canine mammary tumors. Determination of COX-2 expression in other morphologic types of mammary tumors in dogs also could be informative.
The exact implication of COX-2 in canine mammary tumorigenesis is still unknown. Some epidemiologic evidence in humans indicates that the long-term use of NSAIDs can decrease the incidence of breast cancer, but no such information is available in dogs. Use of anti–COX-2 strategies for the treatment of breast cancer has also recently been proposed. 6 It is interesting to note that a phase I trial showed that administration of the NSAID piroxicam to dogs suffering from different naturally occurring tumors resulted in a partial remission in one of three animals displaying mammary adenocarcinoma. 10 One may speculate that this animal was harboring a mammary adenocarcinoma expressing COX-2, whereas the other two treated dogs might have had tumors with negative COX-2. Further studies will be needed to address this issue.
Our results demonstrate for the first time that a proportion of canine mammary tumors expresses significant amount of COX-2, suggesting the potential value of anti–COX-2 therapies for the treatment of this subset of canine mammary tumors.
Footnotes
Acknowledgements
We thank Dr. Stacia Kargman, Merck Frosst Centre for Therapeutic Research, Pointe-Claire-Dorval, Québec, Canada, for kindly providing antibody MF243, Ms. Danielle Rannou for technical assistance, and Dr. Alan K. Goff for assistance with the statistical analyses. This work was supported in part by grants from the American Kennel Club Canine Health Foundation (M. Doré and J. Sirois), the Natural Sciences and Engineering Council of Canada (M. Doré), and the Canadian Institutes for Health Research (CIHR) (J. Sirois). J. Sirois is the recipient of a CIHR Investigator Award.
