Abstract
Cyclooxygenase-2 (COX-2) is an inducible member of the family of cyclooxygenase enzymes that has been implicated in the genesis of numerous cancers. The role of COX-2 in canine mammary neoplasia remains to be more clearly elucidated. The goal of the study reported here was to determine whether a direct association between levels of COX-2 expression and tumor histologic subtype exists in canine mammary carcinoma. Immunohistochemical analysis was performed using a polyclonal antiprostaglandin G/H synthase 2 IgG COX-2 antibody. Sections from the kidneys of young dogs, which stain positive for COX-2 in the macula densa, served as positive controls. Slides were reviewed by a single pathologist, and were evaluated for COX-2 expression according to previously established scales. Positive-staining tumors were given a COX-2 staining distribution (on the basis of the percentage of positive staining cells in five 400x fields) and intensity score according to previously established scales. The product of the COX-2 staining distribution and intensity scores was calculated to create a COX-2 staining index. COX-2 expression was detected in 28 of 50 (56%) samples evaluated. Anaplastic carcinomas had a significantly higher COX-2 staining distribution, intensity, and index, compared with those for adenocarcinomas (P < 0.0001). The overall percentage of positive tumors (56%) was consistent with that of prior studies. To the authors' knowledge, these results indicate, for the first time, a direct association between COX-2 expression and tumor histologic subtype in canine mammary carcinomas. Future research directed at measuring tumor response in canine mammary carcinoma patients treated with a selective COX-2 inhibitor is indicated.
Mammary carcinoma is a common neoplasm in the female dog. There is strong association between reproductively intact status and development of mammary carcinoma, with a 0.05% incidence in females spayed before their first estrous cycle and an approximately 26% incidence in reproductively intact females. 20 Prognostic variables for this tumor type include size of the primary tumor, amount of local invasion, presence of metastatic disease, and histologic subtype of the tumor. 18, 27 Results of molecular studies have indicated that the presence of a high agryophilic nucleolar organizer region count and DNA aneuploidy also are negative prognostic factors in canine mammary carcinoma. 1, 6
Cyclooxygenase-2 (COX-2) is an inducible member of the family of cyclooxygenase enzymes that catalyze the conversion of arachadonic acid to precursor prostaglandins (PG) and thromboxanes (TX), 24 which are essential in the maintenance of cellular homeostasis. Physiologic responsibilities of PG and TX include preservation of renal blood flow and the gastrointestinal mucosal barrier. In the pathologic state, PG and TX promote inflammation. Inflammatory cells, such as macrophages and synoviocytes, have been documented to express COX-2, and subsequently, to increase concentrations of PG and TX as part of the inflammatory cascade. 7 Cellular conditions, such as hypoxia, cytokines (interleukin 6), oncogenes (ras and scr), and vascular endothelial growth factor (VEGF), lead to increased COX-2 expression. 24, 25 At the cellular level, higher activity of COX-2 helps promote tumor invasiveness that is mediated through increased activity of matrix metalloproteinases 2 and 9. 2 COX-2 promotes expression of the antiapoptotic protein bcl-2, thereby inhibiting cellular senescence. 5
The role of COX-2 in the progression of cancer has become an area of extensive research. COX-2 expression leads to cancer progression through the promotion of invasion, angiogenesis, and metastasis and the inhibition of apoptosis. Epidemiologic studies of human patients receiving long-term therapy with nonsteroidal anti-inflammatory drugs (NSAIDs) indicated a significantly decreased risk for development of colorectal carcinoma. 23 Those findings led to extensive investigation to elucidate the role of COX-2 in cancer. Human neoplasms that overexpress COX-2 include numerous carcinomas, 12 pediatric osteosarcomas and rhabdomyosarcomas, 3 and mucosal-associated lymphoid tissue lymphoma. 13 The results of those studies have led to numerous clinical trials and epidemiologic studies of the role of cyclooxygenase inhibitors, both COX-2 selective and non-selective COX inhibitors, as therapy and chemopreventatives. Results of recent research have documented decreased incidence of breast cancer in female patients with long-term use of NSAIDs. 8 However, to date, there has been only one study of the role of COX-2 in canine mammary carcinoma. 4
COX-2 has been documented to be overexpressed in canine squamous cell carcinoma, 17 transitional cell carcinoma, 9 renal carcinoma, 10 intestinal neoplasia, 14 mammary neoplasia, 4 and prostatic carcinoma. 26 The study by Dore et al. 4 indicated an association between degrees of COX-2 expression when comparing mammary adenomas with mammary carcinomas. However, to date and the author's knowledge, there have been no studies evaluating the association between levels of COX-2 expression and tumor histologic subtype of canine mammary carcinoma. Histologic subtype is a known prognostic variable in canine mammary carcinoma, with solid and anaplastic carcinomas carrying a worse prognosis, compared with that for tubular or papillary adenocarcinomas. 22 The aim of the study reported here was to determine whether there was a direct association between levels of COX-2 expression and histologic tumor subtype in a series of randomly selected cases of canine mammary carcinoma.
Materials and Methods
Cases of mammary carcinoma were examined by use of immunohistochemical analysis to determine levels of COX-2 expression. Cases were deemed eligible for selection if they had biopsy specimens submitted to the Laboratory of Pathology of the Veterinary Hospital of the University of Pennsylvania (VHUP). Medical records were reviewed until 50 eligible cases were collected. To have an accurate representation of the three tumor types studied (adenocarcinomas, solid carcinomas, and anaplastic carcinomas), cases were selected in the order in which they were archived. Biopsy specimens were collected from the pathology archives of the VHUP. Positive controls consisted of sections from the normal kidney of young dogs, in which the cells of the macula densa stained positive for COX-2. Negative controls, from the same source as the positive controls, were incubated in phosphate-buffered saline without primary antibody. All sides were reviewed by one of the authors (veterinary pathologist M. H. Goldschmidt) to confirm the initial diagnosis and verify the tumor histologic subtype.
To standardize the procedure and reduce technical error, immunohistochemical analysis was performed using the DAKO EnVision Plus technique and the DAKO Autostainer (DAKO Corporation, Carpenteria, CA). After processing in routine manner, paraffin-embedded tissues were cut at a thickness of 4 µm and mounted on ProbeOn Plus (DAKO Corporation) microscope slides. The tissues were heated to 60°C and were de-paraffinized using clearant and ethyl alcohol, followed by rinsing in deionized water and Tris-buffered saline. The primary antibody was COX-2 AB, a polyclonal antiprostaglandin endoperoxide synthase IgG (Product #PG 27 B, Oxford Biomedical Research, Ann Arbor, Michigan). The primary antibody was applied at room temperature for 30 minutes at a 1:250 dilution. The secondary antibody also was applied at room temperature for 30 minutes. This detection system uses the Envision+ (polyclonal) system (DAKO Corporation), which incorporates the secondary antibody with the enzyme detection in one step at a ready-to-use dilution. The usual avidin-biotin-complex method was replaced by use of a dextran polymer conjugated with horseradish peroxidase and goat antimouse secondary antibodies. Diaminobenzedine was used as the chromagen.
All positive-staining tumors were given a separate score for COX-2 staining distribution and intensity. Negative-staining tumors were given a 0 for both COX-2 staining distribution and intensity. The COX-2 staining distribution score was defined by the estimated percentage of positive-staining cells in five 400x fields. All tumors were given a COX-2 staining distribution from 0 to 4, with 0 = 0%, 1 = < 10%, 2 = 10–30%, 3 = 31–60%, and 4 = > 61% of cells staining positive for COX-2. The COX-2 staining intensity was defined as the strength of the signal in positive-staining tumors, with − = no signal, + = weak signal, ++ = moderate signal, and +++ = strong signal. The COX-2 staining index was obtained by multiplying the COX-2 staining distribution and intensity scores. This gave a range of COX-2 staining indices of 0–12. This grading and intensity scheme was adapted from a previously established scale for COX-2 immunoreactivity in canine mammary tumors. 4
To determine whether COX-2 staining distribution scores, intensity scores, and staining indices differed by tumor histologic subtype (anaplastic vs adenocarcinoma), a Student's t-test was performed. A value of P < 0.05 was considered statistically significant. All analyses were performed using SAS statistical software (version 8.2, SAS Institute, Cary, NC). Data are presented as frequencies and percentages for categorical data (e.g., tumor type) and as both mean and median with ranges for continuous data (e.g., COX-2 grade).
Results
Fifty cases were included in the analysis. Signalment data were available for 47 patients. Three of the biopsy specimens were referring veterinarian submissions and, consequently, lacked descriptive data. The characteristics of the sample population included 27 spayed females and 22 reproductively intact females. Mean age at the time of diagnosis was 10.2 (range: 2–18) years. Six of 47 patients had presented with evidence of metastatic disease. Regarding tumor characteristics, there were 37 adenocarcinomas, two solid carcinomas, and 11 anaplastic carcinomas.
COX-2 immunohistochemical analysis revealed positive staining in 28 of 50 (56%) tumors. Of the 50 tumors examined, 28 (56%) had some degree of positive COX-2 staining. The remaining 22 cases (44%) were negative for COX-2 expression. Seventeen of the 37 adenocarcinomas (46.0%), 0 of two solid carcinomas (0%), and 11 of 11 (100%) anaplastic carcinomas had some degree of positive COX-2 staining. The median COX-2 staining distribution score for the 17 adenocarcinomas was 1 (range: 1–3), and was 2 (range: 1–4) for the 11 anaplastic carcinomas. The staining index for the adenocarcinomas ranged from 0–4 (median 0; mean 1.0), and from 2–12 (median 6; mean 5.1) for the anaplastic carcinomas. A summary of the percentage of COX-2 positive tumors is presented in Fig. 1. Anaplastic tumors stained more densely for COX-2 distribution and intensity than did adenocarcinomas (P < 0.0001). An example of the staining characteristics of adenocarcinomas and anaplastic carcinomas, along with positive-control images is presented in Figs.2–4. Analysis of the staining index also revealed statistical significance, with anaplastic carcinomas having a higher overall staining index, compared with that of adenocarcinomas (P < 0.0001). The data for the two negative staining solid carcinomas was not included in the final analysis.
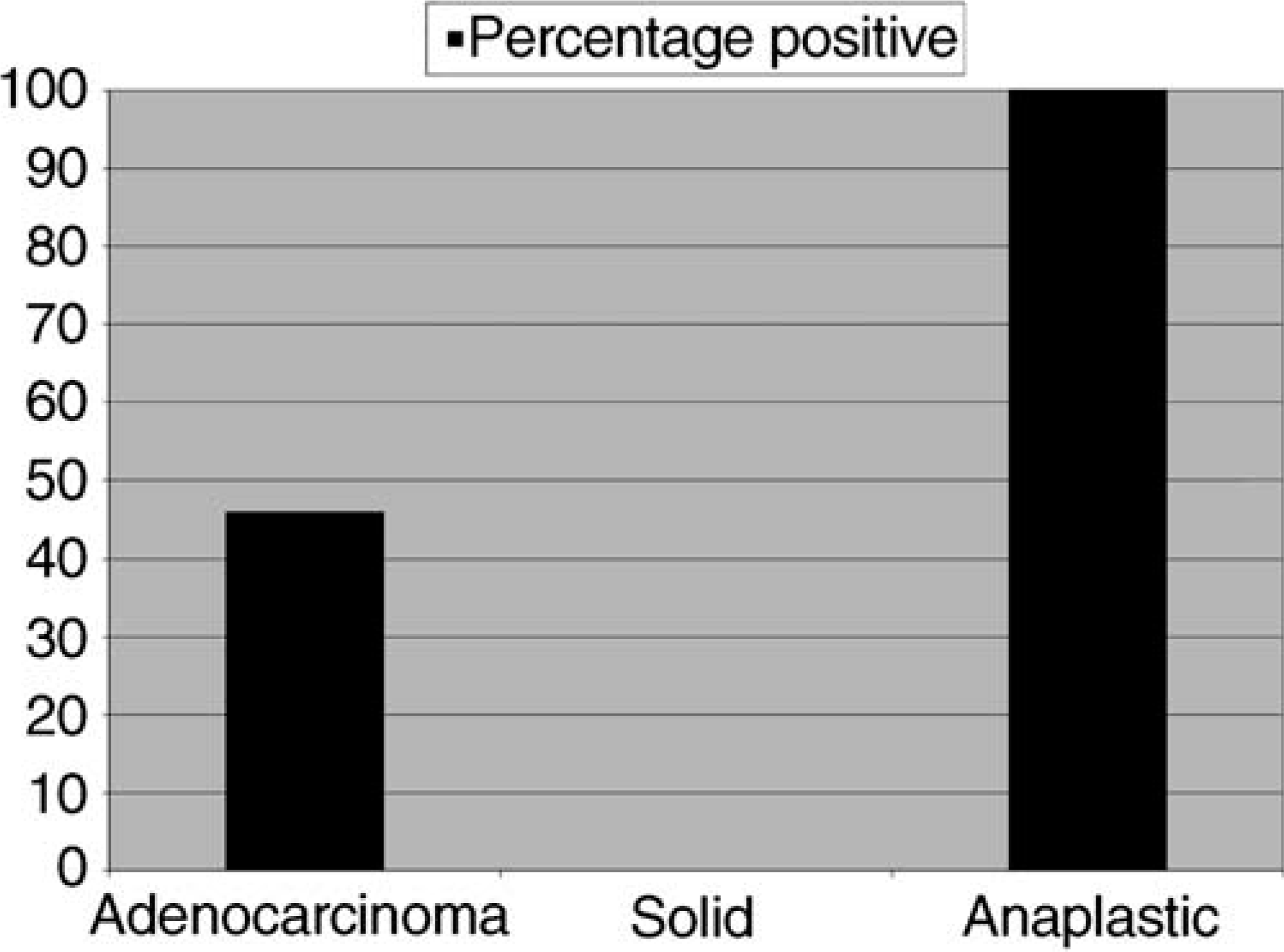
Percentage of positive tumors by histologic subtype.
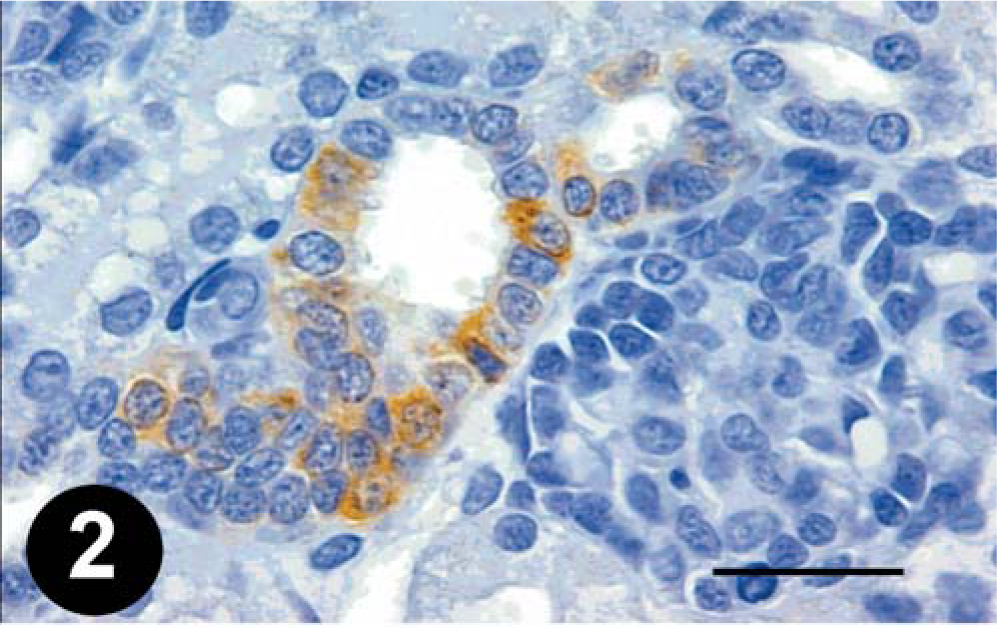
Kidney; young dog. COX-2 expression in the macula densa. Bar = 70 µm.
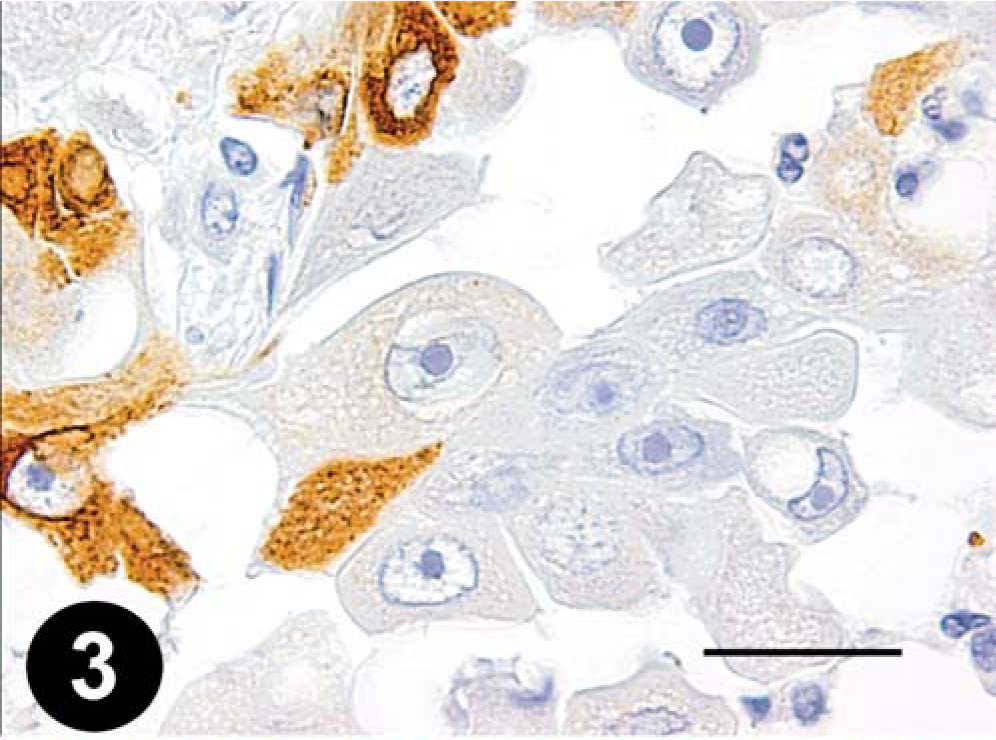
Mammary gland; dog. Minimal COX-2 staining in an adenocarcinoma. Bar = 35 µm.
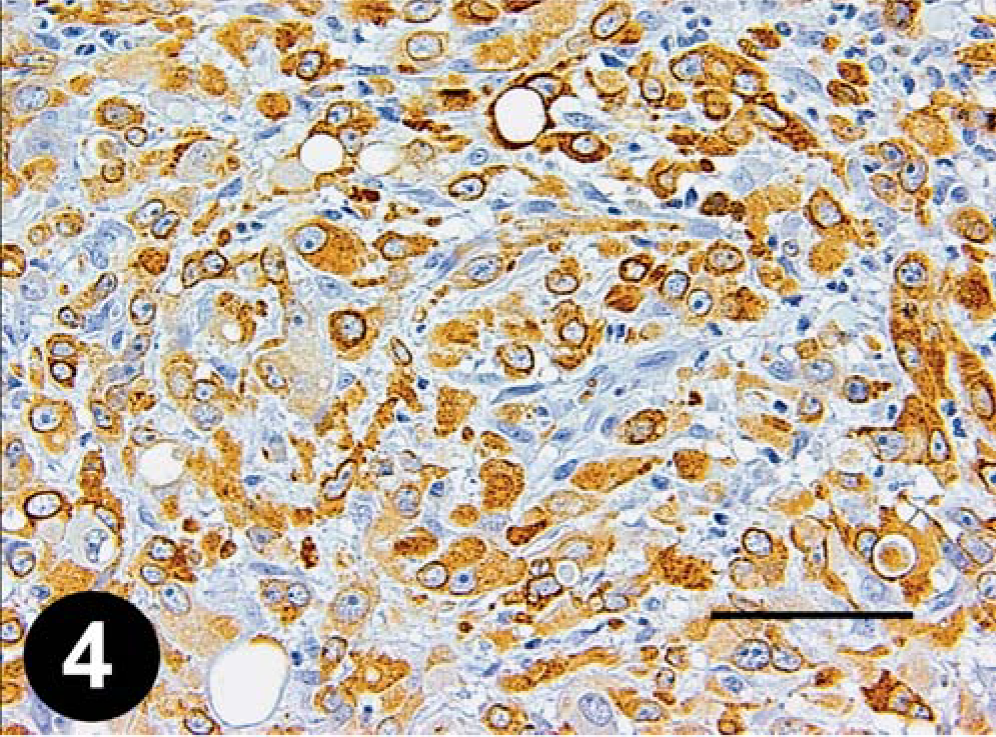
Mammary gland; dog. Strong COX-2 staining in an anaplastic carcinoma. Bar = 70 µm.
Discussion
The results of the study reported here document that COX-2 expression is related to tumor histologic subtype in canine mammary carcinoma. Our findings indicate that more poorly differentiated tumors express COX-2 with stronger intensity and frequency compared with those of more-differentiated tumors, such as adenocarcinomas. These results are similar to findings in human breast carcinoma, where Ristimaki et al. 19 found that higher levels of COX-2 expression were associated with more-undifferentiated tumor histologic subtype and poorer overall outcome. Earlier veterinary research indicated increased amounts of prostaglandin E2 in three of eight cases of canine mammary carcinoma, 15 and it is possible that the increased amounts of this prostaglandin in these tumors was due to increased COX-2 values. The findings of our study are similar to those of Dore et al., 4 with 56% of all canine mammary carcinomas having some degree of COX-2 expression in both studies. The two solid carcinomas of this study did not express COX-2; this may be a statistical aberrancy or there may be a difference in the biologic behavior of this histologic subtype. Further investigation of this difference is warranted.
COX-2 expression has been identified as a prognostic variable for both disease-free interval and survival in several human cancers. Specific tumor types in which COX-2 expression is an independent prognostic variable include gastric carcinoma, 21 pulmonary carcinoma, 11 and breast carcinoma. 19 The results of the aforementioned epidemiologic studies of colorectal tumors in long-term NSAID users has led to routine use of selective COX-2 inhibitors in high-risk patients. Such epidemiologic studies are not presently feasible in veterinary medicine. However, routine use of COX-2 inhibitors in patients at high risk for developing certain malignancies, such as breeding females, may potentially confer some protection against development of mammary tumors.
In this study, we chose to compare the most common histologic types of canine mammary carcinoma. Other types of mammary carcinoma, such as inflammatory mammary carcinoma, squamous cell carcinoma, and carcinosarcoma, were not examined. We eliminated inflammatory mammary carcinomas in our study population, as this is a unique histologic subtype of mammary carcinoma that carries a poor prognosis. 16 Due to the lack of follow-up information for the cases of this study, we were unable to associate COX-2 expression with disease-free interval or patient survival. Future work examining the prognostic value of COX-2 expression as an independent variable in determining disease-free interval and patient survival is indicated. The data presented here will help direct future clinical trials using COX-2 selective inhibitors. The role of COX-2 in the progression of canine mammary carcinoma needs to be more clearly elucidated. As research in human patients has documented that COX-2 expression is associated with patient outcome in breast carcinoma, 19 determining the role of COX-2 expression in disease-free interval and survival of canine patients with mammary carcinoma would be a logical future direction. In addition, evaluation of the therapeutic benefit of COX-2 inhibition in selected canine tumors is necessary prior to recommending them as treatment. Future evaluation of the use of COX-2 inhibitors as both therapeutic and chemopreventative agents is indicated.
Footnotes
Acknowledgements
We thank Dr. Anne Barger for technical assistance and the Commonwealth of Pennsylvania for contributions to this research.
This work was funded by the Departmental Research Resources Committee of the MJR-VHUP.
