Abstract
Cyclooxygenase-2 (COX-2) can be overexpressed at inflammatory sites, leading to the generation of proinflammatory prostanoids. Selective inhibitors of COX-2 have potential use in treating inflammatory conditions including ophthalmic diseases in veterinary medicine. Keratitis is considered the most common inflammatory eye disease in dogs. In this study we evaluated the expression of COX-2 in normal dog eyes and in dog eyes with keratitis by immunohistochemistry using isoform-specific antibodies. In the normal eye (n = 4), no COX-2 immunoreactivity was observed in the cornea. In keratitis, COX-2 (n = 12) expression was observed in all corneal layers (epithelium, stromal cells, and endothelium). COX-2 immunoreactivity was also noted in the stromal and epithelial cells of the iris and the stromal cells of the trabecular meshwork. These data indicate that COX-2 may play a pathophysiologic role in keratitis and suggest potential therapeutic implications of prostaglandin modulation in inflammatory eye diseases.
The initiation, progression, and sequelae of keratitis are mediated by inflammatory cytokines released by either the damaged tissue or infiltrating inflammatory cells, such as neutrophils and macrophages. 5 Inflammatory mediators involved in ocular disease have recently become of interest as potential therapeutic targets for the inhibition of inflammation in the eye, including nitric oxide synthase, tumor necrosis factor (TNF), interleukin-1 (IL-1), and cyclooxygenase-2 (COX-2)–generated prostanoids. 7,12 Prostanoids are the products of arachadonic acid (AA) metabolism by the COX enzymes. 5,6 Two isoforms of COX have been identified, COX-1 and COX-2. COX-1 is constitutively expressed in many tissues, whereas COX-2 is induced by inflammatory mediators such as endotoxin, hypoxia, 5 basic fibroblast growth factor, 10 and interleukin-1β 10 to form prostaglandins (PGs). In the eye, PGs are believed to act in a paracrine fashion to regulate blood flow, production of aqueous humor, and angiogenesis 14 and to modify the blood aqueous barrier. 9 COX-2 is evident immunohistochemically in the ciliary process epithelium in normal humans 19 and in the iris and ciliary process in normal rabbits. 9
COX-2 expression increases in ocular diseases such as glaucoma, immune-mediated uveitis, 14 and herpetic keratitis. 8 Chronic inflammation promotes corneal fibrosis, pigmentation, and neovascularization resulting in corneal scarring, 25 and disruption of the blood ocular barrier may lead to chronic immune-mediated uveitis. 25 Therefore, inhibition of the progression of inflammation is important to prevent chronic ocular damage. To better understand the distribution of COX-2 in keratitis, we examined the expression of COX-2 in both normal dog eyes and dog eyes with keratitis.
Materials and Methods
Tissue samples
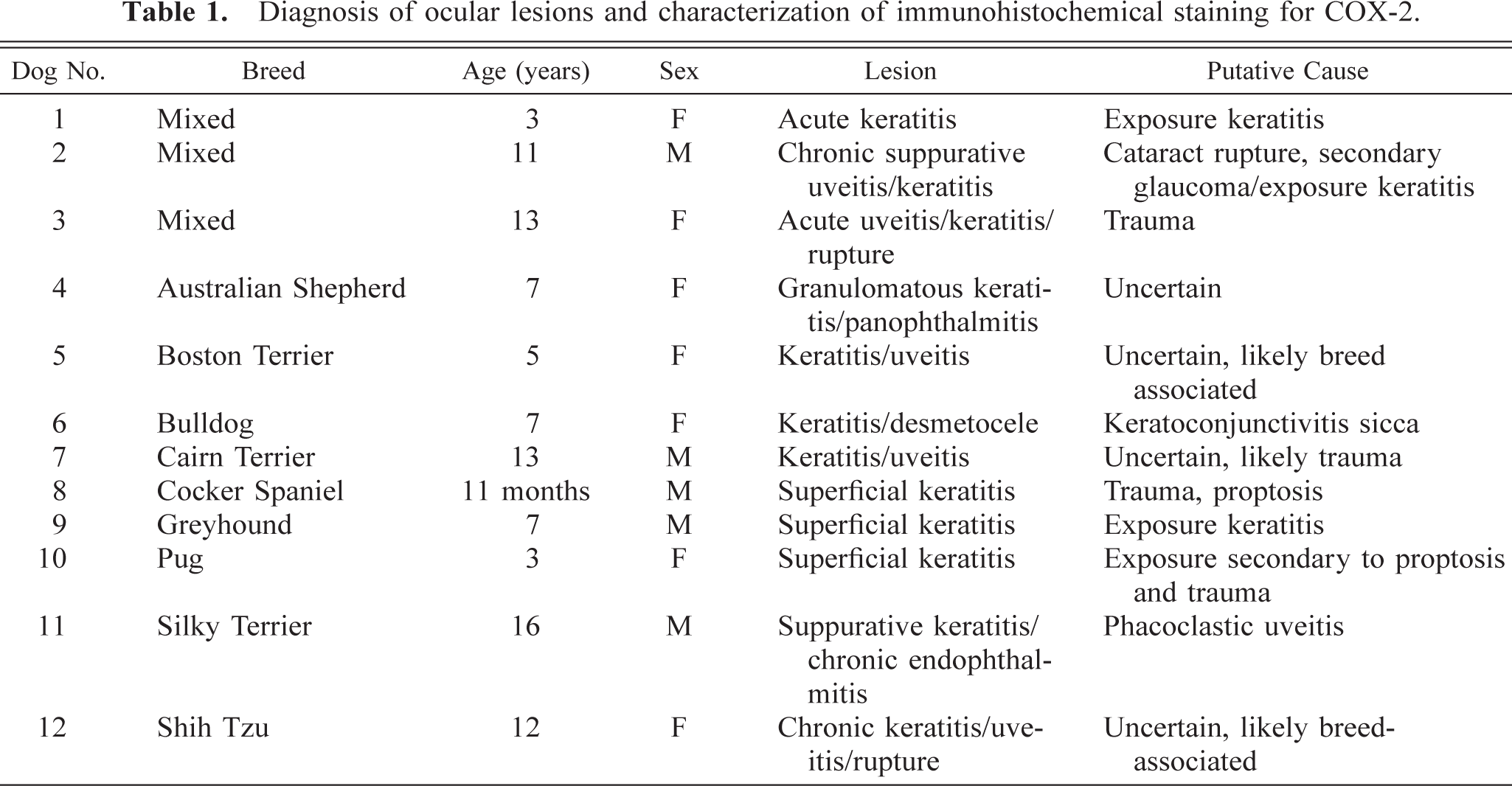
Four normal and 12 keratitic dog eyes were chosen randomly from necropsy and biopsy specimens from the veterinary teaching hospital at Ohio State University School of Veterinary Medicine and from normal research Beagles at Pharmacia, Inc. (Skokie, IL) during the years 1996 to 2000. Tissues had been fixed in 10% neutral buffered formalin (NBF) for 16 to 24 hours and paraffin embedded, except in the cases of 2 of 4 control dog eyes and 1 of 12 keratitis eyes, which may have been fixed in 10% NBF for up to 5 days. The dogs with keratitis ranged in age from 3 to 16 years, with seven females and five males, involving both purebred and mixed-breed dogs (Table 1). Control eyes were from a Brussels Griffon (age unknown), a mixed breed dog (age unknown), and two normal research Beagles (∼6 months of age) (fixed in Davidson's fixative).
Diagnosis of ocular lesions and characterization of immunohistochemical staining for COX-2.
Immunohistochemistry
Five-micrometer tissue sections were deparaffinized in xylene and rehydrated through decreasing gradients of ethanol. Intrinsic peroxidase activity was inhibited by 20 minutes incubation in 3% H2O2 in methanol followed by washes in deionized H2O (dH2O). Slides were placed in 95°C target retrieval solution (DAKO, Carpinteria, CA) for 20 minutes and allowed to cool to room temperature and washed in dH2O and then in Tris-buffered saline with Tween (TBST; DAKO). Tissues for COX-2 staining were blocked for endogenous avidin–biotin activity (avidin/biotin blocking kit, Vector Laboratories, Inc., Burlingame, CA). Tissues for COX-2 immunohistochemistry (1 : 400, polyclonal rabbit anti-human, Oxford Biomedical Research Inc., Oxford, MI) were incubated overnight at room temperature. Negative-control slides were treated with rabbit preimmune serum (Oxford Biomedical Research) rather than primary antibodies. Slides were then washed in TBST and incubated for 30 minutes in biotinylated anti-mouse or anti-rabbit immunoglobulin (IgG) (1 : 1,000, Jackson Immuno Research Laboratories, Inc., West Grove, PA). Tissues for COX-2 staining were incubated in swine anti-rabbit IgGs (1 : 200, DAKO) for 30 minutes, washed in TBST, and exposed to streptavidin–horseradish peroxidase (1 : 1,000, DAKO) for 30 minutes, followed by TBST wash and 5 minutes exposure to diaminobenzidene. Tissues were counterstained with methyl green or alcian blue. Positive control tissue for COX-2 was neonatal pig kidney.
Results
Histology
Histologic lesions and putative causes are presented in Table 1. Eleven of the 12 animals had suppurative and ulcerative keratitis, and six of these had acute to chronic anterior uveitis. The causes of keratitis included trauma, keratoconjunctivitis sicca, proptosis, phacoclastic uveitis, and bacterial infections. A primary cause for keratitis was not evident in three cases. It is likely that the eye lesions in these dogs represented trauma or breed-associated keratitis secondary to exophthalmia.
COX-2 immunohistochemistry
No COX-2 expression was evident immunohistochemically in the cornea or anterior chamber in the normal dog eyes examined (Fig. 1). Conversely, COX-2 protein was expressed in the cornea of all 12 dog eyes with keratitis (Figs. 2, 3). COX-2 staining was moderate to marked in the cornea and involved one or all corneal layers: the epithelium, corneal endothelium, and stromal keratocytes (Table 2). Occasionally, this staining was present in the stromal cells of the filtration angle and in the iris epithelium or stroma (Table 3). Intensity and distribution of COX-2 immunohistochemical staining correlated positively to severity of ocular damage.
Immunohistochemical localization of COX-2 in ocular tissues of dogs with corneal lesions.

Summary of COX-2 immunohistochemistry (IHC) in dog eyes with keratitis.
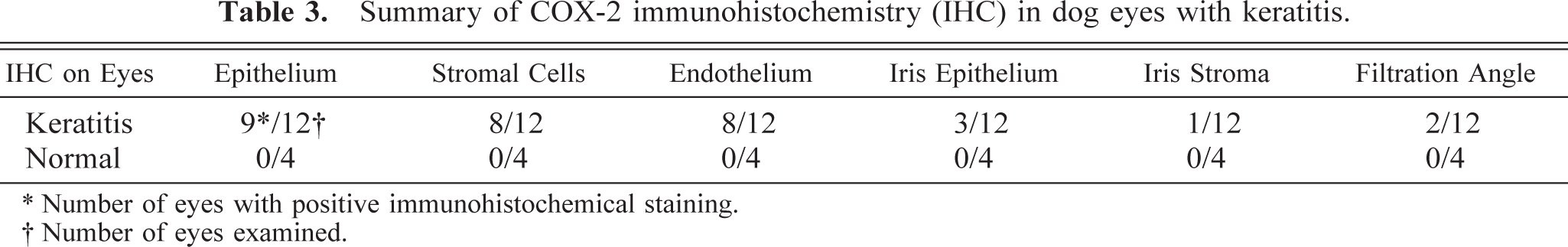
∗ Number of eyes with positive immunohistochemical staining.
† Number of eyes examined.

Normal cornea; Brussels Griffon. Note that no COX-2 staining is evident in any layer of cornea. Streptavidin–biotin complex method. Bar = 40 µm.
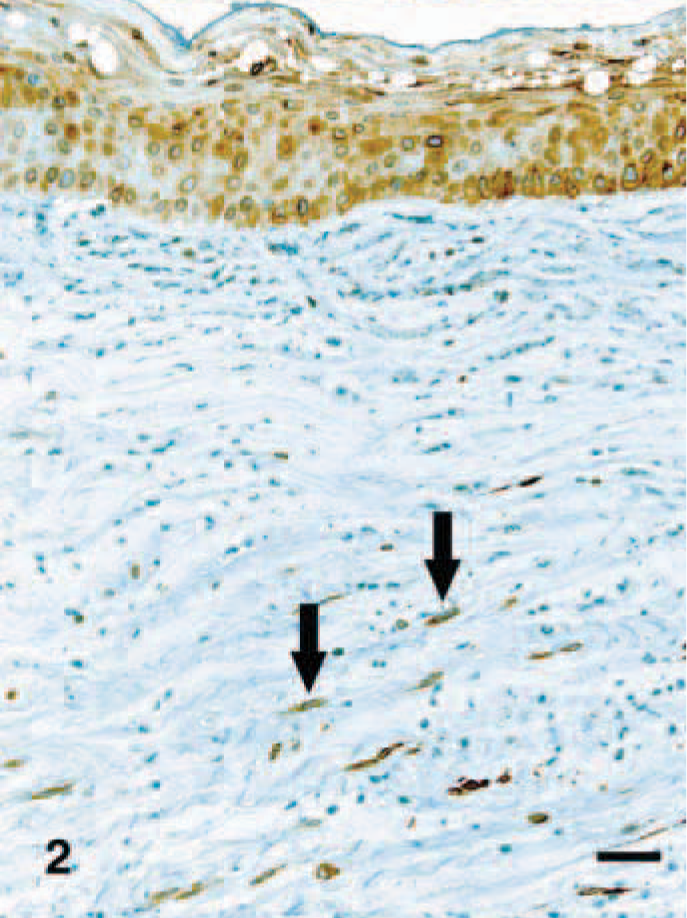
Cornea; Pug with keratitis. Corneal epithelial cells and stromal keratocytes (arrows) have marked cytoplasmic staining for COX-2. Streptavidin–biotin complex method. Bar = 40 µm.

Cornea; Pug with keratitis. Corneal endothelial cells (arrows) have marked cytoplasmic staining for COX-2. Streptavidin–biotin complex method. Bar = 40 µm.
Discussion
Our study demonstrated that COX-2 expression increased in the canine cornea, and to a lesser extent in the stroma of the iris and filtration angle, in clinical cases of keratitis. In the cornea, COX-2 was expressed in the epithelium, endothelium, and stromal cells of the inflamed cornea. Conversely, no expression of COX-2 was evident in the cornea or anterior segment of the normal dog eye. This finding is consistent with previous studies in dogs (J. Marshall, personal communication). Other studies that have examined COX-2 expression in normal eyes by immunohistochemistry have reported only minimal staining in the iris stroma (rabbit) 9 or ciliary process epithelium (human and rabbit), but COX-2 expression in normal cornea of dogs has not been reported. 9,19 Differences in staining distribution (iris and ciliary process) may be reflective of species variation, tissue fixation procedure, or the antibodies used to localize COX-2. Collectively, these data indicate that COX-2 expression is markedly increased in all layers of cornea during an inflammatory insult.
The role of COX in the normal eye has not been entirely elucidated. Differences in the capacity of different portions of the eye to synthesize AA metabolites have been demonstrated. 17 For example, it has been reported that the cornea and the lens have a lower capacity than the anterior uvea to synthesize AA metabolites. 17 Prostaglandin E2 (PGE2) synthesis has been demonstrated in the lens, cornea, ciliary epithelium, and iris. 30,31 PG receptors have been identified in the corneal epithelium and endothelium, the trabecular meshwork, the lens epithelium, and the epithelium and endothelium of the anterior segment. 4,27,30,31 In addition, there are variations in the distribution of the various receptor subtypes in the eye. 30,31 Lack of significant constitutive COX-2 expression in the normal eyes suggests that COX-1 is responsible for normal PG-dependent processes in the eye, 24 similar to its roles in other locations such as the gastrointestinal tract and the kidney. Studies using topical nonsteroidal anti-inflammatory drugs (NSAIDs) in the eye, such as diclofenac and Voltaren, which nonselectively inhibit both COX isoforms, have been associated with the development of corneal ulcers in patients after cataract surgery or photorefractive keratectomy. 24 These ocular lesions are proposed to be mediated by vascular mechanisms similar to that of NSAID-associated gastrointestinal ulceration. 24 PGs also play an important role in the normal flow of aqueous humor from the eye, and PG analogs have been used extensively for the treatment of open-angle glaucoma in humans. 22 In particular, PGE2 is thought to be important for maintaining the outflow of aqueous humor. 19 Additionally, PGE2 stimulates corneal epithelial proliferation and maintains corneal epithelial cell morphology in vitro. 24
Inflammatory mediators such as IL-1 and TNF-α induce COX-2 but not COX-1 expression. Metabolites of AA produced in damaged tissues by COX-2, such as PGE2 and prostacyclin (PGI2), may be important in the initiation and progression of ocular inflammation. 1,3 COX-2 metabolites have vasoactive properties and have been shown to be important in angiogenesis. 16 Because COX-2 has been implicated in angiogenesis, 18,20,28 it is likely that it plays an important role in corneal neovascularization after injury. In studies examining corneal wound healing in vitro, it was found that PGE2 was increased in corneal endothelial cells in response to injury and returned to normal levels after recovery. 16 Interestingly, PGE2 has also been demonstrated to inhibit endothelial cell migration in response to experimental corneal wounds 15 and may also inhibit endothelial cell proliferation. In rabbit eyes, increases in ocular PGs cause vasodilation and disrupt the blood-aqueous barrier, which promotes inflammation. 3 PGI2, in addition to having vasodilatory and platelet-aggregating effects, also enhances the chemotactic effects of other cytokines, thereby promoting the inflammatory process. 25 COX-2 inhibitors in rabbits have been reported to decrease neutrophilic infiltrates into the iris and ciliary body after intraocular endotoxin exposure, indicating a potential role of COX-2 in ocular inflammation. 21 In addition, pain associated with inflammation is due, in part, to the effects of PGE2, which promotes pain hypersensitivity. 25
In addition to having vascular effects, COX-2 metabolites likely play a role in both tissue remodeling and tissue damage after corneal trauma. The role of COX-2 metabolites in corneal re-epithelialization after wounding is less clear. In an in vitro corneal wound–healing model, indomethacin and flurbiprofen did not alter corneal re-epithelialization, whereas lipoxygenase inhibitors markedly delayed re-epithelialization. 13 Studies have demonstrated that after corneal trauma, the inflammatory mediator platelet-activating factor (PAF) accumulates in the cornea, as do the prostanoids PGE2, PGD2, and PGF2alpha. 23 The increase in prostanoids has been hypothesized to be, in part, the result of the activation of COX-2 by PAF. 23 PAF is important in the activation of proteolytic cascades, which may lead to corneal defects and ulcers. PAF increases uroplasminogen activator (uPA), matrix metalloproteinase-9 (MMP-9), and MMP-1, 23 and PAF antagonists have demonstrated protective effects against acute and chronic anterior segment inflammation in animal models of ocular injury. 2 These changes may be mediated by COX-2 because inhibition of COX-2 will decrease PAF-induced MMP-1, MMP-9, and uPA. 23 Therefore, the use of COX-2 inhibitors may be necessary to reduce ocular inflammation and damage secondary to excessive or abnormal remodeling.
Whereas the role of many inflammatory cytokines such as IL-1, TNF-alpha, IL-6, IL-8, granulocyte-macrophage colony stimulating factor, and leukotriene B4 in mediating ocular inflammation has been extensively examined in the literature, 11,26,29 studies on the role of eicosanoids have been less prominent. Based on the findings in this study and in others, ocular PGs in corneal and uveal inflammation may be an important therapeutic target, and further investigations should be undertaken.
Footnotes
Acknowledgements
We thank Dusko Trajkovic, James Yan, and Janet Harmon for technical assistance, Sheila Saldeen for excellent proof reading and formatting assistance, and Alan Opsahl and Josh Bunger for their photographic expertise.
