Abstract
The prognosis of canine soft-tissue sarcomas (STS) has traditionally been based on histologic grading. We have recently demonstrated the prognostic value of cellular proliferation markers in canine STS. Another method of predicting the behavior of neoplasms is intratumoral microvessel density (IMD), which is a measure of tumor angiogenesis. The prognostic significance of IMD has been documented in many human neoplasms and in a limited number of canine and feline neoplasms. To evaluate the prognostic value of IMD in canine STS, we studied 57 STS and compared IMD with histologic features, histologic grade, cellular proliferation, metastatic propensity, and survival. Using immunohistochemistry, the STS were labeled with anti—factor VIII-related antigen (FVIII-RA) and anti-CD31 antibodies to determine 3 IMD parameters: mean microvessel density, high microvessel density, and microvessel area. Using FVIII-RA and CD31, increasing IMD was statistically associated with increasing histologic grade, necrosis scores, and mitotic scores. Higher FVIII-RA IMD values were significantly associated with higher median argyrophilic nucleolar organizing region (AgNOR) values (as previously investigated) and increased metastatic propensity. Fibrosarcomas appear to be the least vascularized of STS. There is no correlation between IMD and survival. Our results indicate that IMD is of prognostic value for histologic grade, histologic features, cellular proliferation (based on AgNOR), and metastatic propensity of canine STS, specifically when using FVIII-RA as the endothelial marker. Assessing histologic grading, cellular proliferation, and IMD of canine STS at the time of diagnosis could therefore provide better prognostic information for the veterinary clinician.
Canine soft-tissue sarcomas (STS) are a heterogenous group of malignant neoplasms that are classified collectively, because they have similar histologic features and biologic behavior. A variety of neoplasms have been classified as STS, including fibrosarcoma, peripheral nerve sheath tumor, myxosarcoma, liposarcoma, leiomyosarcoma, rhabdomyosarcoma, malignant fibrous histiocytoma, and undifferentiated sarcoma. Histologically, these are all nonosseous, mesenchymal-derived or primitive ectodermic-derived neoplasms with a biologic behavior characterized by a tendency toward local invasion, a relatively high local recurrence rate, and a relatively low metastatic rate following conservative surgical excision. 3, 4, 10, 18, 26
The method most widely adopted in predicting the biologic behavior of canine STS is histologic grading, which is based on the number of mitoses, extent of cellular differentiation, and necrosis. 10 However, the use of histologic grading in the prognostication of many tumors in general is somewhat subjective and open to inconsistent interpretation and/or application between different pathologists. These particular problems have been acknowledged in the use of histologic grading in human STS specifically, 1 and similar problems were observed in the histologic grading of canine cutaneous mast cell tumors. 15 Additionally, histologic grading alone does not necessarily provide all the information that is important in predicting the biologic behavior of tumors, a problem that has also been specifically addressed in human STS. 1 The development of more objective morphometric methods of predicting the behavior of canine STS therefore might ameliorate some of these issues associated with histologic grading, as well as provide data that validates the continued use and significance of histologic grading in predicting the biologic behavior of canine STS. For example, we have recently investigated the prognostic value of cellular proliferation markers in canine STS. Specifically, we demonstrated positive associations between increasing Ki-67 and argyrophilic nucleolar organizing region (AgNOR) values with histologic grade and poorer survival. 5
Another method of predicting the behavior of neoplasms is intratumoral microvessel density (IMD), which is a measure of tumor angiogenesis, or the number of new intratumoral vessels in a neoplasm. For neoplasms to grow larger than 1–2 mm3, tumor angiogenesis is required for sufficient nutrient supply and waste removal for continued tumor growth. 6, 7, 28 By extension, tumor angiogenesis also plays an important role in tumor invasion and metastasis, by allowing neoplasms to induce the formation of their own vascular network that potentially facilitates breakdown of the extracellular matrix and allows direct hematogenous spread of neoplastic cells. Therefore, higher IMD values correlate with faster growing neoplasms, more malignant neoplasms, and poorer survival. 9, 13, 25, 28– 30
The prognostic significance of IMD has been documented in many human neoplasms, such as breast, prostate, ovarian, uterine, cervical, esophageal, and gastric cancers. 9 The prognostic value of IMD has been demonstrated in a limited number of canine and feline neoplasms, such as canine and feline mammary tumors, 8, 12, 19, 21 canine squamous cell carcinoma, 11 canine mast cell tumor, 16, 17 canine seminoma, 20 and canine osteosarcoma. 2 Determination of IMD is mainly based on identifying endothelial cells using molecular markers. 9, 29, 30 In canine and feline neoplasms, the main endothelial markers are factor VIII (von Willebrand's factor)–related antigen (FVIII-RA), 8, 12, 16, 17 CD31 (platelet endothelial cell adhesion molecule-1), 11, 19 and vascular endothelial growth factor. 12, 20, 21
The purposes of this study are to quantify IMD in canine STS using FVIII-RA and CD31, and to determine the value of IMD as a predictor of histologic features and grade, rate of cellular proliferation, metastatic propensity, and survival. Our hypotheses were: 1) IMD and histologic features/grade are positively associated; 2) IMD and cellular proliferation marker scores are positively associated; 3) IMD and metastatic propensity are positively associated; and 4) IMD and survival are negatively associated.
Materials and Methods
Samples
Sixty client-owned dogs with primary, naturally occurring STS were identified as potential cases for inclusion in the study. All samples had been submitted to the Department of Pathology of The Animal Medical Center between 1996 and 2002 for histologic examination. These cases had previously been studied for associations between histologic features, histologic grade, cellular proliferation, metastases, and survival data. 5 Histologic features (tumor type, degree of cellular differentiation, necrosis score, mitotic score, and surgical margins) were assessed by a single pathologist (T. J. S.). Grading was based on a modified human grading system previously described by Kuntz et al. (as presented in Table 1). 10 Cellular proliferation markers, comprising AgNOR, Ki-67, and proliferating cell nuclear antigen (PCNA) were assessed by a single pathologist (T. J. S.). Data regarding metastases and survival based on case records, client follow-up, and referral veterinarian follow-up was recorded.
The canine soft-tissue sarcoma (STS) grading scheme, as modified by Kuntz et al. 10 Grade I = cumulative score of 3 or 4. Grade II = cumulative score of 5 or 6. Grade III = cumulative score of 7, 8, or 9.

∗ Number of mitotic figures per 10 high power fields.
Of the 60 dogs that qualified for study inclusion, only 57 dogs were included in the study because archival tissue blocks for 3 dogs were unavailable for slide preparation.
Immunohistochemistry
Five-micron-thick formalin-fixed, paraffin-embedded sections were freshly cut and mounted on positively charged OptiPlus fluid barrier glass slides (Biogenex, San Ramon, CA). Adherent sections were dried for 30 minutes in a 65°C slide dryer, then manually deparaffinized with Histoclear (National Diagnostics, Atlanta, Georgia) and rehydrated in graded ethanol to water.
Following deparaffinization, adherent sections for CD31 immunohistochemistry were immersed in an epitope retrieval solution (Borg Decloaker, Biocare Medical, Walnut Creek, CA), heated to 120°C for 3 minutes in a pressure cooker (Decloaking Chamber, Biocare Medical, Walnut Creek, CA), cooled to room temperature, and rinsed with water for 5 minutes.
All subsequent immunolabeling was performed at room temperature using a computer-controlled automated system (Optimax Plus, Biogenex Laboratories, San Ramon, CA). Endogenous peroxidase activity was quenched for 10 minutes with 3% hydrogen peroxide. Sections for FVIII-RA immunolabeling were enzymatically retrieved using proteinase K (Zymed Laboratories, South San Francisco, CA) for 5 minutes.
Tissue sections were incubated with the primary antibody, at a concentration and duration that was determined previously by titration experiments. For FVIII-RA immunohistochemistry, polyclonal rabbit anti-human antibody (Dako Corporation, Carpinteria, CA) was used at a dilution of 1°2,000 and was incubated for 30 minutes. For CD31 immunohistochemistry, monoclonal mouse anti-human antibody clone JC70A (Dako Corporation, Carpinteria, CA) was used at a dilution of 1°25 and was incubated for 60 minutes.
Detection of primary antibody binding was performed using a modified biotin-streptavidin peroxidase technique. The secondary antibody complex, Biogenex biotinylated MultiLink (Biogenex, San Ramon, CA), was applied for 20 minutes, followed by 20-minute incubation with streptavidin-conjugated horseradish peroxidase (Biogenex, San Ramon, CA). Sections were incubated in 3,3′-diaminobenzidine (DAB) for 10 minutes and counterstained with Mayer's hematoxylin. Slides were washed with phosphate-based Optimax wash buffer (Biogenex, San Ramon, CA) between each reagent application. All slides were then manually dehydrated in increasing concentrations of ethanol, cleared in Histoclear (National Diagnostics, Atlanta, Georgia), and coverslipped with permanent mounting media (Cytoseal XYL, Stephens Scientific, Kalamazoo, MI).
Paraffin-embedded canine tissue arrays containing 32 different tissues (adipose tissue, adrenal gland, bone marrow, cerebrum, esophagus, gallbladder, granulation tissue, haired skin, heart, kidney, large intestine, liver, lung, lymph node, lymphoma, mammary gland, melanoma, nasal mucosa, sciatic nerve, oral mucosa, ovary, pancreas, salivary gland, small intestine, skeletal muscle, spleen, stomach, testes, thyroid gland, tongue, urinary bladder, and uterus) were used as positive control tissues. Endothelium, lining small and large arterial and venous vessels, were assessed for either cytoplasmic or cytoplasmic membrane staining. Nonendothelial tissues were assessed for background staining.
The same canine tissue arrays were used for negative controls. Anti-CD31 antibody was replaced with preimmune mouse serum (Dako Corporation, Carpinteria, CA) diluted to the same IgG concentration, whereas anti-FVIII-RA antibody was replaced with rabbit preimmune serum (Dako Corporation, Carpinteria, CA) diluted to the same protein concentration.
Quantification of intratumoral microvessel density
Immunolabeled sections were scanned at 100× magnification to identify vascular “hot spots,” which are regions of high vascular density within the tumor. 31 High-resolution photomicrographs of 3 nonoverlapping vascular “hot spots” from each section were taken at 200× magnification (2,590 × 1,920 pixel photomicrographs representing 0.37508 mm2) with a digital microscope camera system (Digital Sight DS-5M, Nikon, Melville, NY). Each photomicrograph was imported into and processed by digital imaging software (UTHSCSA ImageTool For Windows 3.00, San Antonio, TX).
Three IMD parameters were measured. Mean microvessel density (m-MVD) and highest microvessel density (h-MVD) were determined for each case, each expressed as the number of vessels per square millimeter. The number of microvessels were counted based on Weidner's method, which defines a countable microvessel as an immunopositive endothelial cell or endothelial-cell cluster that is separated from adjacent microvessels, regardless of the presence or absence of a lumen with or without red blood cells. 30 Microvessel area (MVA) for each case was then determined on the photomicrograph with the h-MVD, based on the total area of immunopositive labeled cells divided by the total area of the photomicrograph field (expressed as a percentage per 200× photomicrograph field). All IMD parameters were assessed by a single pathologist (R. H. Luong), who was blinded to the original data regarding histologic grade, cellular proliferation, metastases, and survival.
Statistical analyses
Computer software (Statview, SAS Institute Inc., Cary, NC) was used to perform all statistical analyses. Parametric one-way analysis of variance (ANOVA) was performed to compare IMD parameters with histologic grade, histologic features, cellular proliferation markers scores, and metastatic propensity. Additionally, the median values of IMD parameters were used as cut-offs to assign tumors into low and high value groups for each IMD parameter, which were subsequently compared with histologic grade, histologic features, cellular proliferation markers scores, and metastatic propensity by chi-square analysis. Simple logistical regression analysis was used to evaluate associations between IMD and histologic grade, IMD and cellular proliferation marker scores, and between IMD parameters.
Kaplan-Meier life table analysis was performed to calculate median survival times (MST), where survival time was defined as the number of days from surgical excision at the AMC to death. Dogs that died from non–STS-related disease or dogs that were still alive at the end of the study period were censored. Log rank testing and univariate Cox Proportional Hazards model were used to evaluate statistical significance between survival times and individual parameters associated with signalment, histologic grade, histologic features, cellular proliferation, IMD, metastatic data, and survival data. Stepwise multivariate Cox Proportional Hazards model was then used for all univariate variables with
Results
Of the 57 dogs included in this study, 37 (65%) were male and 20 (35%) were female. Eleven (19%) dogs were intact males, 26 (46%) were castrated males, 4 (7%) were intact females, and 16 (28%) were spayed females. The ages of the dogs ranged from 3.0 to 15.5 years, with a mean and median age of 9.8 and 9.7 years, respectively. A total of 23 dog breeds were represented, the most common breeds being mixed-breed (16), Golden Retriever (10), Rottweiler (4), Beagle (3), and German Shepherd (3).
Similar to our previous study,
5
the most common STS diagnosed was peripheral nerve sheath tumor (
The mean and median AgNOR scores (number of AgNOR dots per nucleus over 100 tumor nuclei) were 1.99 and 1.88 respectively, with a range of 1.10–3.35. The mean and median Ki-67 scores (number positive nuclei per 100 tumor nuclei) were 8.44 and 7.00 respectively, with a range of 0–37. The mean and median PCNA scores (number positive nuclei per 100 tumor nuclei) were 42 and 45 respectively, with a range of 11–68.
Data regarding development of metastatic disease owing to the STS was recorded in 50 (88%) of the 57 dogs. Seven (14%) of the 50 dogs developed metastases versus 43 dogs (86%) that did not develop metastatic disease.
Survival data was recorded in 54 (95%) of the 57 dogs. Twelve (22%) of the 54 dogs died of tumor-related causes, 7 (13%) of the 54 died of nontumor causes, and 35 (65%) of the 54 were alive at the end of the study period. Nontumor causes of death included splenic hemangiosarcoma with hemoabdomen, lymphoma, unspecified severe adult-onset seizures, central nervous system disease, an aortic body tumor with pleural effusion, and cardiac disease. The MST was not reached (>1,309 days), because 65% of the dogs were still alive at the end of the study period.
FVIII-RA IMD
The cytoplasmic membrane and cytoplasm of endothelial cells of control and case tissues displayed strong and specific brown immunopositivity when labeled with anti-FVIII-RA antibodies (Fig. 1). Background staining of nonendothelial cells was minimal, although plasma present within blood vessels displayed a general light brown homogenous immunopositivity as well. The m-MVD values ranged from 32 to 553 vessels/mm2, with a mean of 98, standard deviation (SD) of ± 105, and median 117. The h-MVD values ranged from 40 to 608 vessels/mm2 (mean 121 ± 115 SD, median 155). The MVA values ranged from 0.52 to 6.21% (mean 2.49 ± 1.45 SD, median 2.11).
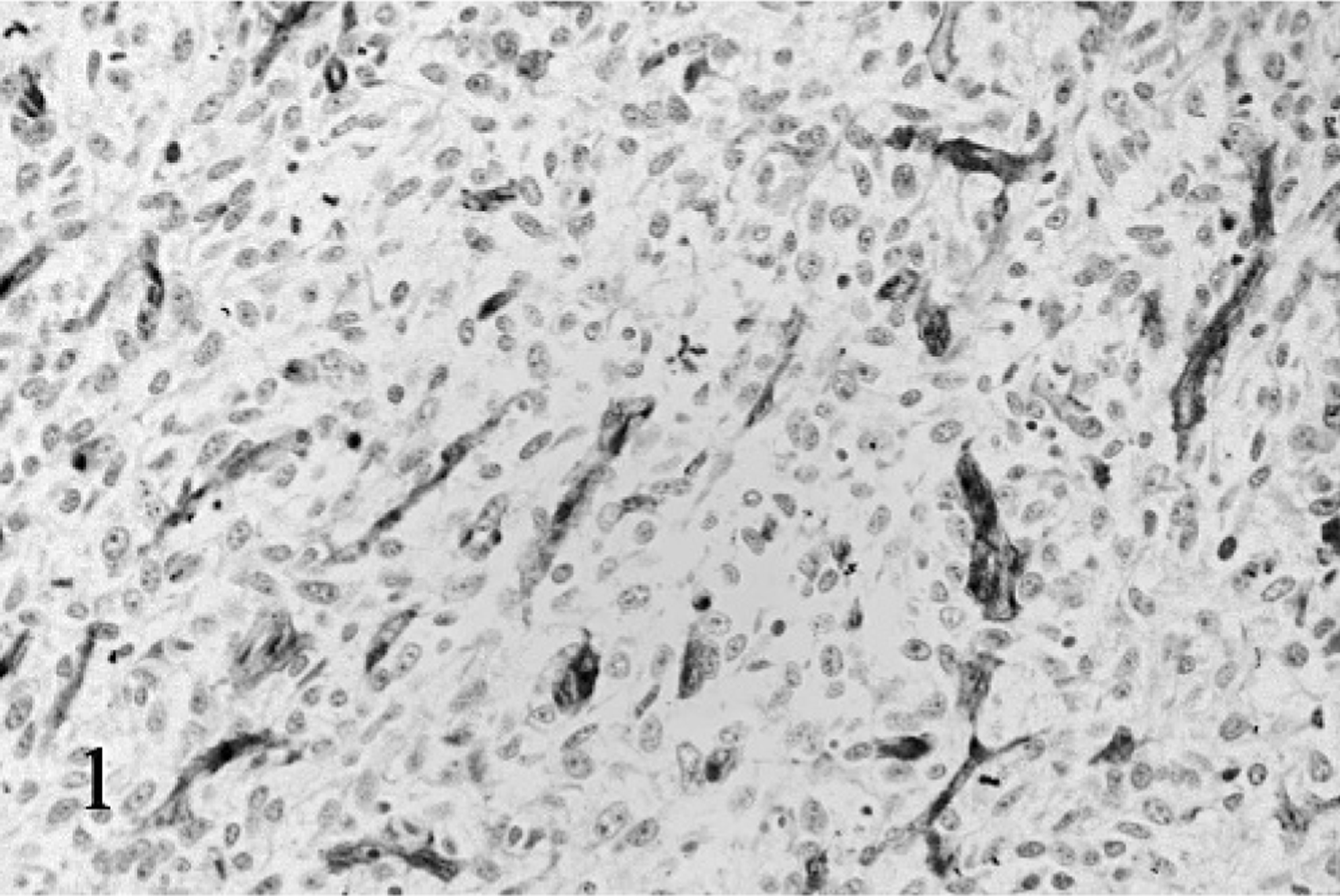
Soft-tissue sarcoma, dog, grade III, Case No. 1. An undifferentiated sarcoma immunolabeled with anti–factor VIII–related antigen antibody. Staining intensity is strong and specific. Streptavidin-biotin-peroxidase and hematoxylin.
FVIII-RA IMD versus histologic grade and features
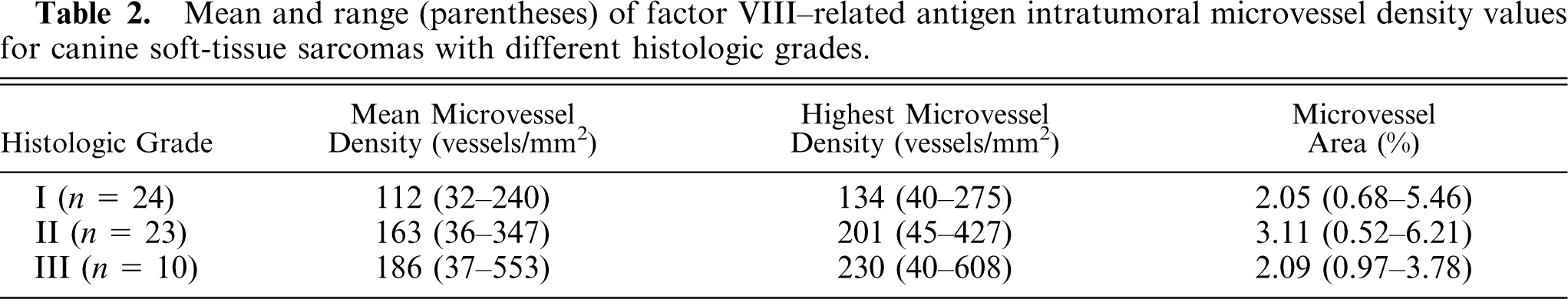
IMD results for tumors with different histologic grades are summarized in Table 2. Grade I tumors had significantly lower h-MVD values than grade II (
Mean and range (parentheses) of factor VIII–related antigen intratumoral microvessel density values for canine soft-tissue sarcomas with different histologic grades.
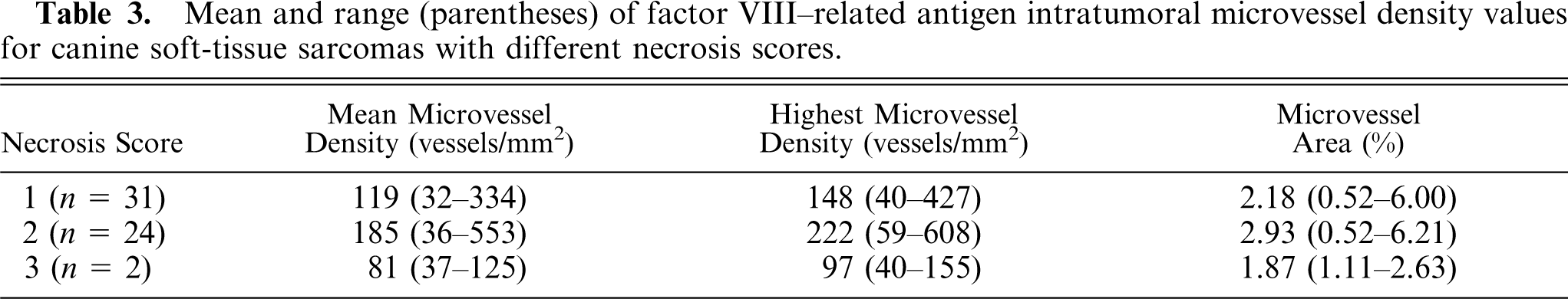
IMD results for tumors with different necrosis and mitotic scores are summarized in Tables 3 and 4, respectively. Tumors with necrosis scores of 2 had significantly higher m-MVD (
Mean and range (parentheses) of factor VIII–related antigen intratumoral microvessel density values for canine soft-tissue sarcomas with different necrosis scores.
Mean and range (parentheses) of factor VIII–related antigen intratumoral microvessel density values for canine soft-tissue sarcomas with different mitotic scores.

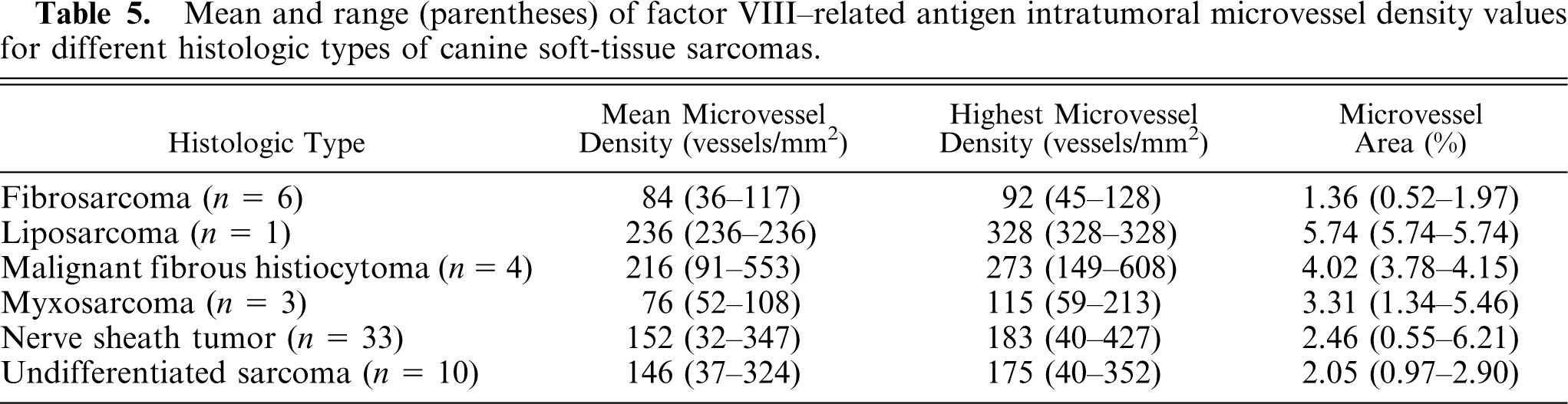
IMD values for different histologic types of STS are presented in Table 5. Significant differences in IMD values were found across tumor types. Fibrosarcoma had lower IMD values than malignant fibrous histiocytoma (m-MVD
Mean and range (parentheses) of factor VIII–related antigen intratumoral microvessel density values for different histologic types of canine soft-tissue sarcomas.
FVIII-RA IMD versus cellular proliferation marker scores
There was no significant association between IMD results and cellular proliferation marker scores. There was a positive association between increasing MVA and higher-than-median AgNOR values (
FVIII-RA IMD versus metastatic propensity
Table 6 summarizes the IMD values of tumors that developed metastatic disease versus tumors that did not develop metastatic disease. A statistically significant positive association existed between dogs that developed metastases compared to dogs without metastatic disease, based on m-MVD (
Mean and range (parentheses) of factor VIII–related antigen intratumoral microvessel density values for canine soft-tissue sarcomas with and without metastatic disease.
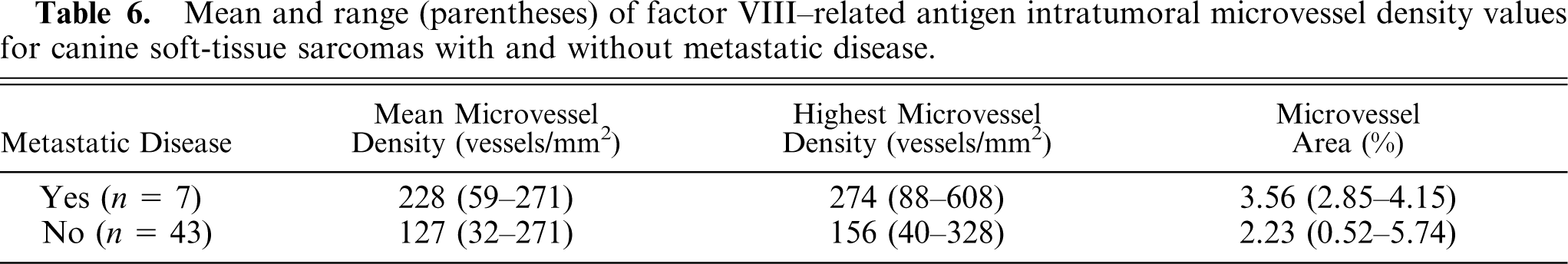
FVIII-RA IMD versus survival
There were no significant associations between IMD results and survival.
CD31 IMD
The cytoplasmic membrane of endothelial cells of control and case tissues displayed intermediate to strong and specific brown immunopositivity when labeled with anti-CD31 antibodies (Fig. 2). Background staining of nonendothelial cells was minimal. The m-MVD values ranged from 9 to 337 vessels/mm2 (mean 60 ± 73 SD, median 103). The h-MVD values ranged from 13 to 429 vessels/mm2 (mean 146 ± 90 SD, median 128). The MVA values ranged from 0.2 to 9.15% (mean 2.11 ± 1.69 SD, median 1.83).
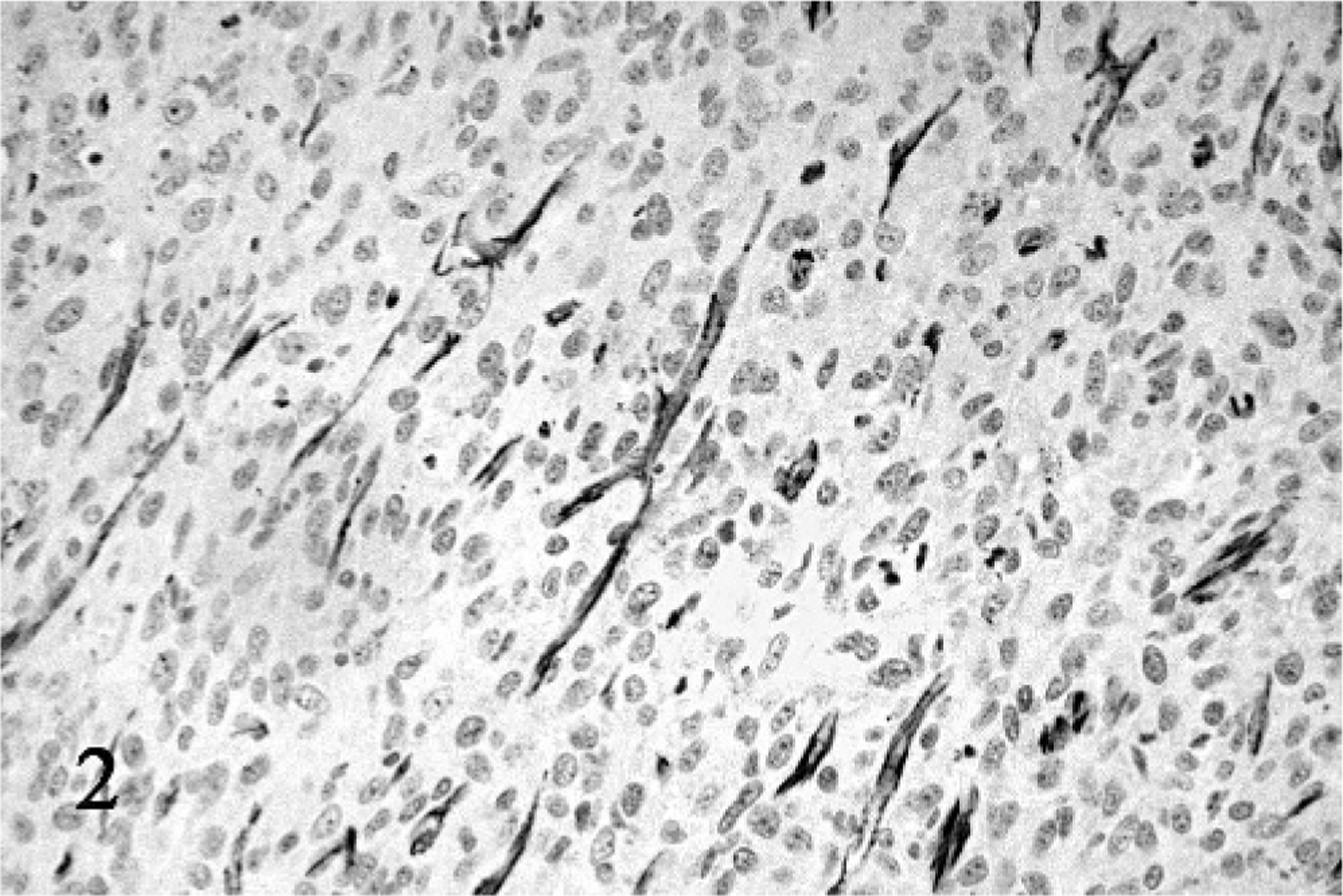
Soft-tissue sarcoma, dog, grade III, Case No. 1. An undifferentiated sarcoma immunolabeled with anti-CD31 antibody. Staining intensity is intermediate–strong and specific. Streptavidin-biotin-peroxidase and hematoxylin.
CD31 IMD versus histologic grade and features
IMD results for tumors with different histologic grades are summarized in Table 7. No significant differences between IMD parameters and histologic grade were found. Table 8 summarizes IMD results for tumors with different mitotic scores. Significantly higher h-MVD values were found in tumors with mitotic scores of 2 versus 1 (
Mean and range (parentheses) of CD31 intratumoral microvessel density values for canine soft-tissue sarcomas with different histologic grades.
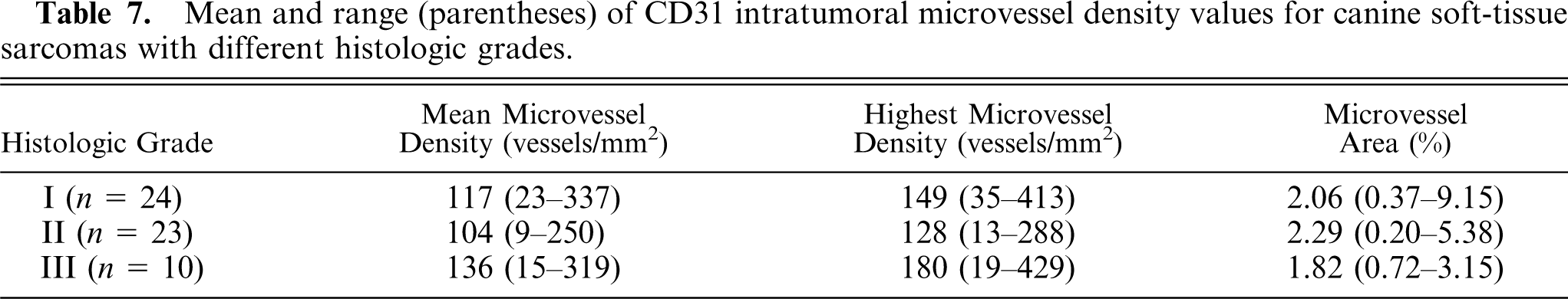
Mean and range (parentheses) of CD31 intratumoral microvessel density values for canine soft-tissue sarcomas with different mitotic scores.
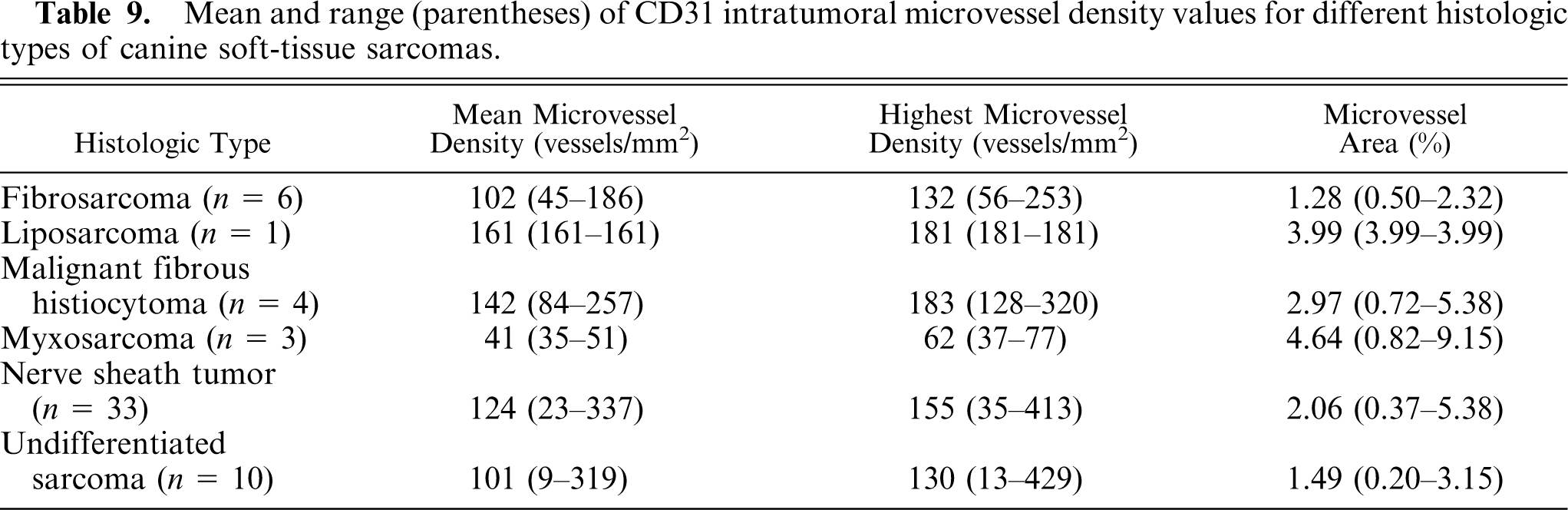
IMD values for different histologic types of STS are presented in Table 9. Significantly lower MVA values were found in fibrosarcoma compared to myxosarcoma (
Mean and range (parentheses) of CD31 intratumoral microvessel density values for different histologic types of canine soft-tissue sarcomas.
Tumors that were resected with >3-cm tumor-free margins had significantly lower h-MVD values compared to tumors with <3-cm tumor-free margins (
CD31 IMD versus cellular proliferation marker scores, metastatic propensity, and survival
There was no significant association between IMD results and cellular proliferation marker scores, development of metastases, or survival.
Comparison of FVIII-RA and CD31 IMD
Results from individual FVIII-RA and CD31 IMD parameters were compared with each other. A positive association was found between all individual FVIII-RA values, including m-MVD versus h-MVD (
Discussion
This study evaluated tumor angiogenesis in canine STS by determining IMD, based on immunolabeling of endothelium with anti-FVIII-RA and anti-CD31 antibodies. The aim of the study was to determine the significance of IMD as a predictor of histologic features and grade, rate of cellular proliferation, metastatic propensity, and survival.
We found that histologic grade of canine STS is positively associated with IMD. In particular, FVIII-RA IMD values predicted STS grade (with FVIII-RA h-MVD being the most consistent predictor) whereas none of the CD31 IMD values were predictive. Therefore, assessing FVIII-RA IMD appears to validate the use of the current histologic grading scheme for canine STS. IMD was also positively correlated with mitotic scores and necrosis scores, independent of grade. Specifically, FVIII-RA IMD values were positively correlated with increasing necrosis and mitotic scores, whereas CD31 IMD values were only of predictive value for increasing mitotic scores. This finding somewhat reinforces the results of previous canine STS studies, 5, 10 where it was found that these histologic features (particularly mitotic scores) should also be assessed independently of histologic grade to determine the clinical behavior of canine STS.
Schärz et al. reported variations in vascularity between different canine tumor types based on color Doppler and power Doppler ultrasonography. 23 Specifically, they reported significantly higher vascularization in canine oral squamous cell carcinoma compared to the other tumors in their study (including STS, bone tumors, melanomas, adenocarcinomas, acanthomatous epulis, and oral lymphosarcoma). Although most of their STS had low vessel density, they did not find statistical differences in vascularity between STS and other tumor types, and they did not report significant differences in vascularity between different types of STS. Our study (to the authors' knowledge) is the first to demonstrate statistical differences in tumor vascularization between different canine STS. Specifically, fibrosarcoma consistently had lower levels of vascularity compared to other types of STS, based on both FVIII-RA and CD31 immunolabeling. Additionally, it appears that based on FVIII-RA immunostaining that malignant fibrous histiocytomas and liposarcomas are more vascularized than other types of canine STS. However, the objective significance of our results is difficult to determine because sample numbers for some of the tumors are low (in particular, malignant fibrous histiocytoma and liposarcoma), which may influence the statistical significance of the results.
We found the degree to which canine STS can be adequately removed with tumor-free margins is also associated to IMD values. Immunohistochemistry using anti-CD31 antibodies reveals that tumors with higher IMD values were more likely to be removed without tumor-free surgical margins. This would support the hypothesis that tumors that are able to induce high rates of new blood vessel formation might facilitate tumor invasion into surrounding tissues. However, as we reported previously, 5 the number of cases with tumor present at surgical margins might be artificially increased in this study because not all surgeries performed were intended to be curative.
The results from this study indicate that canine STS with lower-than-median AgNOR values were more likely to have lower MVA than those with higher-than-median AgNOR values, based on FVIII-RA. This is somewhat suggestive that canine STS with higher proliferation rates induce higher rates of tumor angiogenesis, which is in accordance with the hypothesis that highly vascularized tumors are able to support higher tumor proliferation rates than those that are less highly vascularized growth. 6, 9, 13, 25, 28– 30
In our previous study, 5 we reported that increasing histologic grade was positively associated with metastatic propensity of canine STS. Based on FVIII-RA immunostaining, increasing IMD also appears to be predictive of the metastatic propensity of canine STS. This supports the consensus that more malignant tumors have higher tumor angiogenesis, which, in turn, facilitates hematogenous dissemination of neoplastic cells. 9, 13, 25, 28– 30 However, as we reported previously, 5 there was no standard method by which metastatic disease was recorded in these animals, so the metastatic propensity of the canine STS in this study might be falsely decreased. There was no significant association between IMD and survival data in our study. This is in agreement with limited IMD studies of human solid mesenchymal tumors. 9, 22
This study demonstrates that the use of anti-FVIII-RA antibodies in determining the significance of IMD in canine STS is far superior to the use of anti-CD31 antibodies, despite apparently high positive correlation between individual FVIII-RA and CD31 IMD results when compared with each other. We cannot readily account for these differences based on the data generated. One possible reason is that FVIII-RA is expressed on the surface of and within the cytoplasm of endothelium, as well as a soluble factor in plasma, 27 whereas CD31 is only expressed as a membrane-associated molecule. 14 This could potentially and artificially increase the ability of the pathologist and computer software in identifying plasma-filled vessels within tumors using FVIII-RA as an endothelial marker. However, we cannot substantiate this possibility from existing human or veterinary IMD studies. Additionally in human IMD reviews, it has been stated that polyclonal anti-FVIII-RA antibodies are more sensitive than monoclonal anti-FVIII-RA and monoclonal anti-CD31 antibodies. 9, 28 Whether this applies to the detection of FVIII-RA and CD31 in canine tissues is currently unknown.
There are other limitations to this study. First, this is a retrospective study that lacked standardized treatment and follow-up protocols. In many cases, the cause of death was not determined by necropsy, but rather by owner or clinical assessment. Second, although only primary canine STS were examined, some of these tumors represented local recurrences after previous treatment. The effect of the local recurrence of STS at primary sites on tumor angiogenesis is not known. Third, other endothelial markers could have been used that are more specific to measuring angiogenesis, such as endoglin (CD105). 7, 24 However, many of these alternative markers require the use of frozen sections, and therefore would have been unsuitable in our study because we only had access to paraffin-embedded tissues.
In conclusion, this study shows that there is prognostic significance in assessing tumor angiogenesis by IMD in canine STS. Specifically, the use of FVIII-RA as the endothelial marker for determining IMD is of predictive value for histologic grade, histologic features, cellular proliferation (as determined by AgNOR), and metastatic propensity of canine STS. Concurrently assessing histologic grading, AgNOR cellular proliferation, and FVIII-RA IMD of clinical cases of canine STS at the time of diagnosis could therefore provide better prognostic information for the veterinary clinician.
Footnotes
Acknowledgements
We thank Ms. N. Dube and Ms. C. Leung for technical assistance.
