Abstract
The expression of tyrosine kinase receptors is attracting major interest in human and veterinary oncological pathology because of their role as targets for adjuvant therapies. Little is known about tyrosine kinase receptor (TKR) expression in canine liposarcoma (LP), a soft tissue sarcoma. The aim of this study was to evaluate the immunohistochemical expression of the TKRs fibroblast growth factor receptor 1 (FGFR1) and platelet-derived growth factor receptor–β (PDGFRβ); their ligands, fibroblast growth factor 2 (FGF2) and platelet-derived growth factor B (PDGFB); and c-kit in canine LP. Immunohistochemical labeling was categorized as high or low expression and compared with the mitotic count and MIB-1–based proliferation index. Fifty canine LPs were examined, classified, and graded. Fourteen cases were classified as well differentiated, 7 as myxoid, 25 as pleomorphic, and 4 as dedifferentiated. Seventeen cases were grade 1, 26 were grade 2, and 7 were grade 3. A high expression of FGF2, FGFR1, PDGFB, and PDGFRβ was identified in 62% (31/50), 68% (34/50), 81.6% (40/49), and 70.8% (34/48) of the cases, respectively. c-kit was expressed in 12.5% (6/48) of the cases. Mitotic count negatively correlated with FGF2 (R = –0.41; P < .01), being lower in cases with high FGF2 expression, and positively correlated with PDGFRβ (R = 0.33; P < .01), being higher in cases with high PDGFRβ expression. No other statistically significant correlations were identified. These results suggest that the PDGFRβ-mediated pathway may have a role in the progression of canine LP and may thus represent a promising target for adjuvant cancer therapies.
Canine liposarcoma (LP) is an uncommon soft tissue sarcoma that usually arises in the subcutis and of which several histological variants have been described: atypical lipoma, well-differentiated LP, myxoid LP, pleomorphic LP, and dedifferentiated LP. 4,21,24 LP recurrence has been reported in up to 28% of cases, but metastases are rare. 6,21 As for other soft tissue sarcomas (STSs), the main therapeutic option is surgical excision, 33 and the principal prognostic factors are completeness of surgical excision and histological grade. 13
The expression of growth factors and growth factor receptors has been evaluated with a particular focus on tyrosine kinase–type receptors in several canine STSs to assess these molecules or the corresponding receptors as potential targets for adjunctive tumor therapy to reduce the recurrence rate. 1,5,11,14,28
The growth factors with tyrosine kinase receptors most commonly investigated in both human and veterinary medicine include vascular endothelial growth factor (VEGF), platelet-derived growth factor B (PDGFB), and fibroblast growth factor 2 (FGF2). 1,5,9,11,14,28,36,42,47 In vitro studies have demonstrated that inhibition of the FGF2–fibroblast growth factor receptor 1 (FGFR1) pathway leads to a reduction of cell growth 53,54 and inhibition of angiogenesis. 32 Similarly, the inhibition of PDGF receptors reduces proliferation, angiogenesis, and expression of antiapoptotic molecules. 51 Human patients with STS have higher serum levels of FGF2 and VEGF compared with controls, 20,45,46 while VEGF serum levels decrease in dogs with STS after STS resection. 11
The expression of FGF2, PDGFB, and the corresponding receptors FGFR1 and PDGF receptor–β (PDGFRβ) has been described in several canine mesenchymal neoplasms, including vascular tumors, 3,48,55 Spirocerca lupi–associated sarcoma, 14 fibrosarcoma, 48 and perivascular wall tumors (PWTs). 5 Nevertheless, studies evaluating the expression of these molecules in canine LPs are lacking.
Another tyrosine kinase receptor whose expression has been investigated in both human and veterinary medicine is c-kit, which is the target for therapies in canine cancers such as mast cell tumors and is expressed in a small percentage of liposarcomas in humans. 22,27
The aim of this study was to evaluate the immunohistochemical expression of FGF2, FGFR1, PDGFB, PDGFRβ, and c-kit in canine LP and to compare expression levels with the mitotic count and proliferation index to identify a putative role of these pathways in LP growth.
Materials and Methods
Case Selection and Diagnosis
Canine liposarcoma cases were selected from the caseload of a previous retrospective study
4
and were classified according to a recent histological and immunohistochemical classification based on the following criteria: Atypical lipoma: a neoplasm composed of well-differentiated adipocytes and containing scattered atypical lipoblasts.
21
Well-differentiated LP: a neoplasm composed of round to polygonal cells arranged in solid sheets with little or no stromal collagen, in which most cells contain easily recognizable intracytoplasmic lipid droplets.
19,24
Cases with intermediate histological appearance with pleomorphic LP were classified as well differentiated when positive for MDM2 by immunohistochemistry.
4
Pleomorphic LP: a neoplasm characterized by marked cellular pleomorphism, variable numbers of multinucleated neoplastic cells, and scarce to absent intracellular lipids.
6,19,21,24
Cases with intermediate histological appearance with well-differentiated LP were classified as pleomorphic when negative for MDM2 by immunohistochemistry.
4
Myxoid LP: a neoplasm characterized by abundant background mucin resembling myxosarcoma, from which it is differentiated by the presence of cytoplasmic lipid-laden vacuoles and frequently containing a delicate anastomosing capillary vasculature.
19,21,24
Dedifferentiated LP: a neoplasm characterized by areas of well-differentiated LP juxtaposed to areas of nonlipogenic sarcoma usually resembling a fibrosarcoma or undifferentiated pleomorphic sarcoma.
4,16,18
Cases were graded according to the standard grading system applied to canine STS. 13 Mitotic count was evaluated in 10 high-power fields equating the standard area of 2.37 mm2. 38
Immunohistochemistry
Three-micrometer-thick sections were dewaxed and rehydrated. Endogenous peroxidase was blocked by immersion in H2O2 3% in methanol for 30 minutes. For antigen retrieval, sections were immersed in 200 ml citrate buffer (pH 6.0); heated in a microwave oven at 750 W for two 5-minute cycles for c-kit (Dako, Glostrup, Denmark), FGF2, FGFR1, PDGFB, and PDGFRβ (Santa Cruz Biotech, Santa Cruz, CA) and 4 cycles for Ki-67 (Dako); and cooled at room temperature for approximately 20 minutes. Source, type, and dilution of antibodies applied to canine LP are listed in Table 1.
List of Source, Type, and Dilution of Antibodies Applied to Canine Liposarcoma.
FGF2, fibroblast growth factor 2; FGFR1, fibroblast growth factor receptor 1; PDGFB, platelet-derived growth factor B; PDGFRβ, platelet-derived growth factor receptor–β.
The reaction was amplified by the avidin-biotin method (ABC kit elite; Vector, Burlingame, CA) and visualized with 3,3′-diaminobenzidine (0.04% for 4 minutes). Sections were counterstained with Papanicolaou hematoxylin, rinsed in tap water, dehydrated, and coverslipped. Sections of granulation tissue were used as positive controls for FGF2, FGFR1, PDGFB, and PDGFRβ, and sections of canine hyperplastic lymph node were used as positive controls for Ki-67. Negative controls comprised slides incubated with omission of the primary antibody. Expression of FGF2, FGFR1, PDGFB, and PDGFRβ was scored in a semiquantitative manner, examining the entire sample at intermediate magnification (200×), and assessing the percentage of positive neoplastic cells. FGF2, FGFR1, PDGFB, and PDGFRβ were considered highly expressed if labeling was present in more than 70% of neoplastic cells as previously reported. 5 Variation of the intensity of the labeling within a sample and labeling of stroma were recorded but not included in the scoring system. Expression of c-kit was classified as positive or negative as previously reported. 23,27 Ki-67 expression was evaluated as previously reported. 4
Statistical Analysis
Data were analyzed with Shapiro and Wilk’s W test for normality. Because data did not fit a normal distribution, the Spearman test was used to compare the grade, mitotic count, and Ki-67 labeling index of the group with high expression versus low expression of FGF2, FGFR1, PDGFB, and PDGFRβ and of the group of c-kit positive and negative. A conventional 5% level was used to define statistical significance.
Results
Fifty canine liposarcomas were collected. No atypical lipoma was diagnosed, 14 cases were classified as well differentiated LP, 7 as myxoid LP, 25 as pleomorphic LP, and 4 as dedifferentiated LP. All dedifferentiated liposarcomas were characterized by dedifferentiated areas resembling high-grade fibrosarcoma. Seventeen cases were grade 1, 26 were grade 2, and 7 were grade 3. Mitotic count ranged between 1 and 56 (median, 5), and Ki-67 labeling index ranged between 1.1% and 76.8% (median, 14.9%).
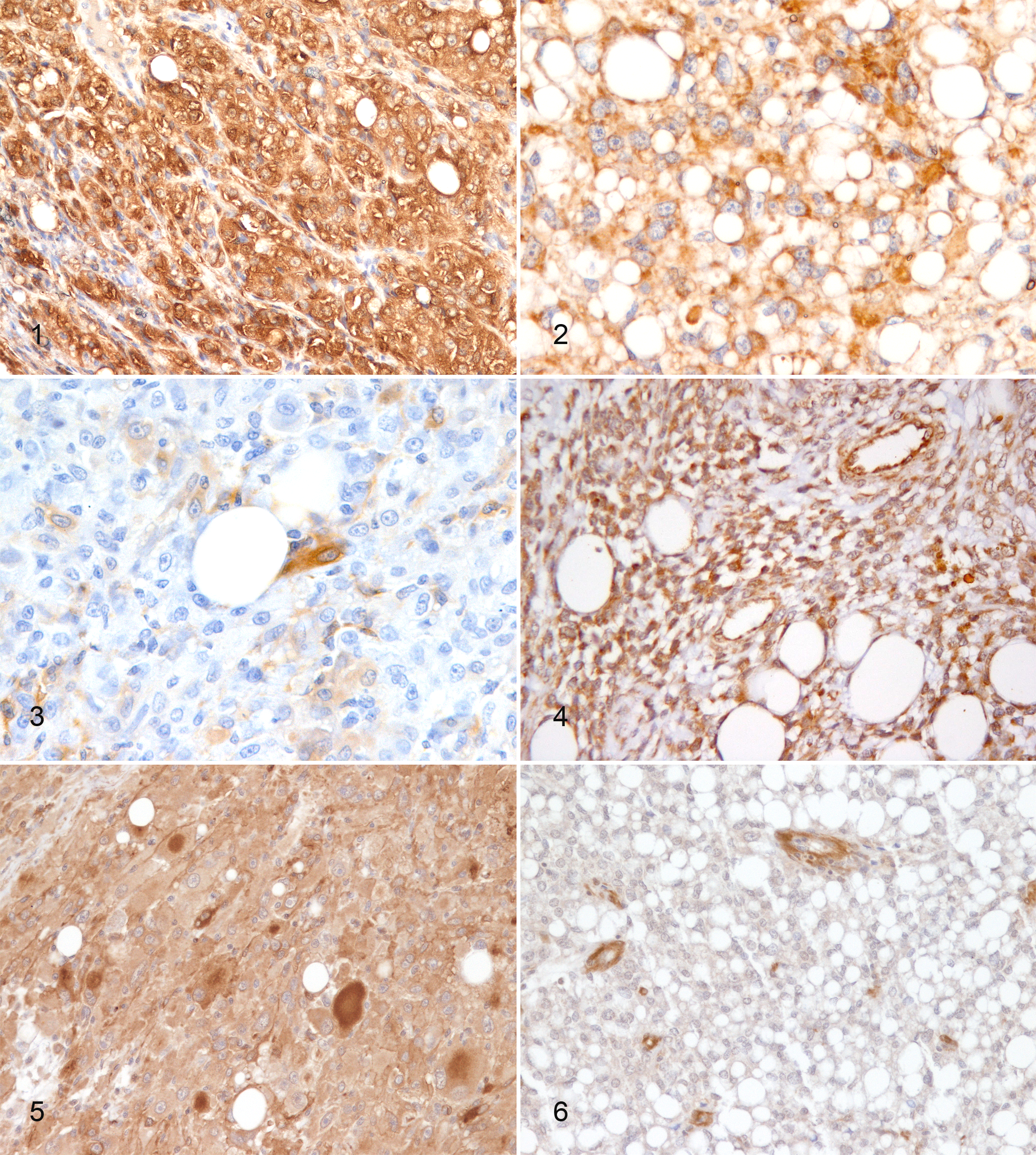
High expression of FGF2 and FGFR1 was identified in 31 cases (62%) (Fig. 1) and 34 cases (68%) (Fig. 2), respectively, and FGF2 low expression in 38% of cases (Fig. 3). PDGFB, PDGFRβ, and c-kit expression was not evaluated in 1, 2, and 2 cases, respectively, because of insufficient material in the original blocks. Therefore, PDGFB expression was assessed in 49 cases and PDGFRβ and c-kit expression in 48 cases. High PDGFB and PDGFRβ expression was identified in 40 cases (82%) (Fig. 4) and 34 cases (71%) (Fig. 5), respectively, and PDGFRb low expression in 38% of cases (Fig. 6). Six cases (13%) were c-kit positive and 42 (87%) were negative. The data regarding the expression of FGF2, FGFR1, PDGFB, PDGFRβ, and c-kit in canine liposarcomas are listed in Table 2.
Immunohistochemical Expression of FGF2, FGFR1, PDGFB, PDGFRβ, and c-kit in Canine Liposarcoma.

DDL, dedifferentiated liposarcoma; FGF2, fibroblast growth factor 2; FGFR1, fibroblast growth factor receptor 1; ML, myxoid liposarcoma; PDGFB, platelet-derived growth factor B; PDGFRβ, platelet-derived growth factor receptor–β; PL, pleomorphic liposarcoma; WDL, well-differentiated liposarcoma.
aTotal, n = 49 for PDGFB and 48 for PDGFRβ and c-kit; PL, n = 24 for PDGFRβ and c-kit; DDL, n = 3 for PDGFB, PDGFRβ, and c-kit.
For all markers evaluated, the labeling of neoplastic cells was cytoplasmic in all cases. Occasionally, labeling intensity varied among cells within the same tumor (Fig. 5). Stromal tissue was negative for all markers in all cases (Fig. 1).
Mitotic count negatively correlated with FGF2 (R = –0.41; P < .01), being lower in cases with high FGF2 expression, and positively correlated with PDGFRβ (R = 0.33; P < .01), being higher in cases with high PDGFRβ expression. No other statistically significant correlations were identified.
Discussion
This study detected expression of tyrosine kinase receptors FGFR1 and PDGFRβ and their ligands FGF2 and PDGFB in canine LPs. Most LPs examined were characterized by high FGF2 expression, chiefly in well-differentiated and pleomorphic LP histotypes. FGF2 expression has been reported in several canine neoplasms, including epithelial tumors such as prostatic carcinoma, 41 endocrine and thyroid carcinoma, 43 and mesenchymal neoplasms such as hemangiosarcoma 55 and fibrosarcoma. 14 FGF2 expression has also been investigated in human STS where FGF2 plasma levels were higher than in healthy controls. 20,45,46 All these data suggest that FGF2 may have a role in the occurrence and progression of several tumor types in both humans and dogs. Interest in FGF2 expression in cancer is linked to its putative target role for therapies using thalidomide, which has been demonstrated to inhibit FGF2-induced angiogenesis 10 and to be effective in the treatment of multiple myeloma 7,44,50 and has been tested in other solid tumors in humans. 31
Nevertheless, studies evaluating the prognostic impact of FGF2 have been contradictory: some studies report high FGF2 expression in neoplastic cells associated with a high proliferation rate and negative prognosis, 8,30,39 whereas others have identified a correlation between high FGF2 plasma levels and a low risk of recurrence. 20,45 These inconsistent findings have been explained hypothesizing that tumors using the FGF2 pathway might intrinsically have a less aggressive behavior 56 and that other more efficient pathways may induce cell proliferation in cases with a low FGF2 expression. 30 The same hypotheses may also explain the negative correlation observed in this work for canine LPs between high FGF2 expression and a lower mitotic count. This finding suggests that this pathway may be less efficient in inducing the proliferation of canine LP neoplastic cells, thus leading to a lower mitotic count in cases where FGF2 is highly expressed.
Most cases also highly expressed FGFR1, paralleling previous studies that identified expression of this receptor in canine mesenchymal tumors such as hemangiosarcoma 55 and PWTs, 5 as well as in canine liposarcoma and fibrosarcoma cell lines. 48 Nevertheless, FGFR1 was not correlated with mitotic count or the proliferation index in our study. This additional finding suggests that the FGF2-FGFR1 pathway is not relevant in the pathogenesis of canine LP. Likewise, c-kit does not seem to be primarily involved in stimulating cell proliferation since it was expressed in only a minority of canine liposarcomas, paralleling data reported in humans, 27 and a correlation with mitotic count and/or with Ki-67 labeling index was not evidenced.
By contrast, the PDGFB-PDGFRβ pathway seems to have a central role in canine LP progression. Both these molecules had greater expression than FGF2 and FGFR1, with high expression in more than 70% of the canine LPs tested. PDGF-PDGFRβ pathway expression has been demonstrated in several types of neoplasms, both in humans and dogs, 3 as it occurs in thyroid carcinoma, 34 glioma, 57 and renal cell carcinoma 49 in humans and in cutaneous and subcutaneous mast cell tumor 52 and lymphoma 2 in dogs. PDGFB and PDGFRβ expression has also been described in mesenchymal neoplasms in several species, including canine hemangiosarcoma 3 and PWTs, 5 feline injection site sarcoma, 29 and several STSs in humans, including dermatofibrosarcoma protuberans, 36 malignant peripheral nerve sheath tumor, 42 solitary fibrous tumor, 12 smooth muscle cell tumors, 40 and liposarcoma. 25,40 Interestingly, in most of the cases examined in this study, neoplastic cells expressed both PDGFB and PDGFRβ while stromal tissue was consistently negative, thus suggesting an autocrine rather than paracrine pathway for canine LP. Furthermore, our results suggest that the PDGF-PDGFRβ pathway may play a central role in the progression of canine LP, since the cases with high PDGFRβ expression had a significantly higher mitotic count. This finding is particularly interesting as PDGFRβ is a target for therapies in canine mast cell tumors, 22 osteosarcoma, 15,35 and epitheliotropic lymphoma. 26
Furthermore, the application of a multitargeted tyrosine kinase receptor inhibitor has been demonstrated to reduce STS growth in a murine model by reducing both the proliferation of neoplastic cells and angiogenesis. 56 Therefore, these therapies might provide a further option for the treatment of STSs after marginal/incomplete resection, a circumstance leading to a high risk of recurrence 13 that is usually managed with radiation therapy. 17,37 Further studies are needed to confirm the role of the PDGF-PDGFRβ pathway in the growth or progression of canine LP and to assess the efficacy of anti–tyrosine kinase therapy.
Surprisingly, the positive correlation between mitotic count and PDGFRβ and the negative correlation between mitotic count and FGF2 were not paralleled by similar correlations with the Ki-67 labeling index. These data should be confirmed by the evaluation of a larger caseload.
Summarizing, we have identified the expression of 2 tyrosine kinase receptor–mediated pathways in canine LP: FGF2-FGFR1 and PDGFB-PDGFRβ. PDGFB-PDGFRβ expression seems to be strictly involved in neoplastic cell proliferation and therefore represents the most promising target for specific therapies.
Footnotes
Acknowledgements
We thank A. Collins for editing the English text.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
