Abstract
Spindle cell lipoma (SCL) is a benign neoplasm of the adipose tissue that may resemble an undifferentiated soft tissue sarcoma (STS). This report describes the histopathological features of 6 SCLs in dogs. All SCLs were located in the subcutis and were composed of bland, occasionally vacuolated spindle cells intermixed with ropey collagen and myxoid matrix. Sudan IV stain performed in 1 case demonstrated the lipid content of vacuoles. Mature adipocytes represented less than 10% of the neoplasm in 3 cases and were absent in the remaining 3. Average mitotic count in 10 high-power fields was 0.17. Neoplastic cells were immunohistochemically positive for vimentin and negative for S100 protein, smooth muscle actin, factor VIII-ra, and MDM2. Awareness of SCL and its specific histopathological features is essential to diagnose this specific tumor. Further studies are needed to document the biological behavior of these tumors in dogs.
Spindle cell lipoma (SCL) is a well-characterized benign neoplasm of adipose tissue described in man. SCL usually arises as a solitary subcutaneous lesion typically located on the back, posterior neck, and shoulder in men 45 to 60 years of age. 3 Histologically, SCL is characterized by a variable admixture of adipocytes and bland bipolar spindle cells with inconspicuous nucleoli and rare to absent mitotic figures. 3 Neoplastic cells are admixed with a characteristic so-called ropey collagen and embedded in myxoid matrix. 3,7 Some tumors are predominantly or exclusively composed of spindle cells, lacking any identifiable lipomatous component (the so-called fat-free/fat-poor spindle cell lipoma) and thereby representing a diagnostic challenge. 2,7
SCLs are rarely recognized in dogs and are described as lesions composed of a mixture of adipocytes and spindle cells with a low mitotic rate. 5 Thus, the histopathological features allowing the differential diagnosis of SCL from other canine soft tissue sarcoma (STS) are poorly categorized. The aim of this report is to describe the microscopic features of 6 cases of canine SCLs.
Cases were retrieved from the archives of 2 academic institutions including signalment and macroscopic description of the lesions. Hematoxylin and eosin–stained sections were reviewed. The mitotic count was evaluated in 10 high-power fields (HPF) equating the standard area of 2.37 mm2. 6 Histochemical stains (Masson’s trichrome, periodic acid–Schiff [PAS], Alcian blue at pH 2.5) were performed in all cases. Wet tissue was available in 1 case, and a portion of the neoplasm was snap-frozen in isopentane cooled at the temperature of liquid nitrogen and cryosectioned to perform Sudan IV stain for lipids. For all cases, immunohistochemistry was performed applying primary antibodies recognizing vimentin (mouse monoclonal, clone V9, dilution 1:600; Dako, Glostrup, Denmark), smooth muscle actin (mouse monoclonal, clone 1A4, dilution 1:450; Dako), MDM2 (mouse monoclonal, clone 2A10, dilution 1:100; Abcam, Cambridge, UK), S100 (rabbit polyclonal, dilution 1:2400; Dako), factor VIII-ra (rabbit polyclonal, dilution 1:2000; Dako), and Ki-67 (mouse monoclonal, clone MIB1, dilution 1:600; Dako). Heat-induced antigen retrieval using pH 6.0 citrate buffer was performed for all the antibodies with the exception of S100, which was incubated with pH 7.6, 0.05% trypsin solution for 15 minutes at 37°C. Ki-67 labeling index was determined by counting a minimum of 1000 cells for each case in the areas with the highest proliferation rate.
Dogs included 2 mongrels, 2 Labrador Retrievers, 1 German Shepherd, and 1 Schnauzer. Two dogs were intact females, 1 was an intact male, 1 was a castrated male, and in 1 case the sex was not recorded. Age was 13 years in 2 dogs, 12 years in 3 dogs, and 9 years in 1 dog (mean, 11.8 years; median, 12 years). All tumors were solitary subcutaneous lesions located in the mammary region (2 cases), thorax (1 case), hip (1 case), or proximal hind limb (1 case). In 1 case, the specific location of origin was not recorded. Tumor size ranged from 2 to 11 cm in diameter (mean, 6.6 cm; median, 6.25 cm).
Tumors were subcutaneous, well demarcated and pseudocapsulated, expansile, and moderately cellular. Microscopic appearance was characterized by bland spindle cells with scarce eosinophilic cytoplasm and central to paracentral oval nucleus (Fig. 1). Cells were intermixed with a moderate to elevated amount of alcianophilic, PAS-negative, myxoid matrix (Fig. 2) and abundant collagen characterized by variably sized and haphazardly arranged fibers (so-called ropey collagen) (Fig. 1). In the areas with higher cellularity, cells were spindle-shaped and plump, with an eccentric oval nucleus and an inconspicuous nucleolus, and occasionally contained a single intracytoplasmic vacuole, recapitulating “developing” adipocytes (Fig. 3). Sudan IV staining demonstrated cytoplasmic positivity in 1 case, confirming the lipid content of the vacuoles (Fig. 3). Three cases contained small numbers of mature adipocytes (less than 10%), single or in small aggregates (fat-poor cases) (Fig. 4), whereas mature adipocytes were not identified in the remaining 3. Multifocal areas of myxoid change were present in all cases together with irregular branching spaces simulating vascular spaces and resembling the pseudoangiomatous pattern described in spindle cell lipoma in man. 3 Mitotic count in 10 HPF was 0 in 5 cases and 1 in 1 case (mean, 0.17). A small number of lymphocytes and plasma cells were observed at the periphery of the lesion in 5 cases. MIB-1 labeling index ranged between 0.9 and 5.6 (mean, 2.77). All cases expressed vimentin and were negative for S100, smooth muscle actin, factor VIII-ra, and MDM2. The cells lining the branching spaces within the neoplasm were factor VIII-ra negative, confirming the nonvascular nature of these spaces.
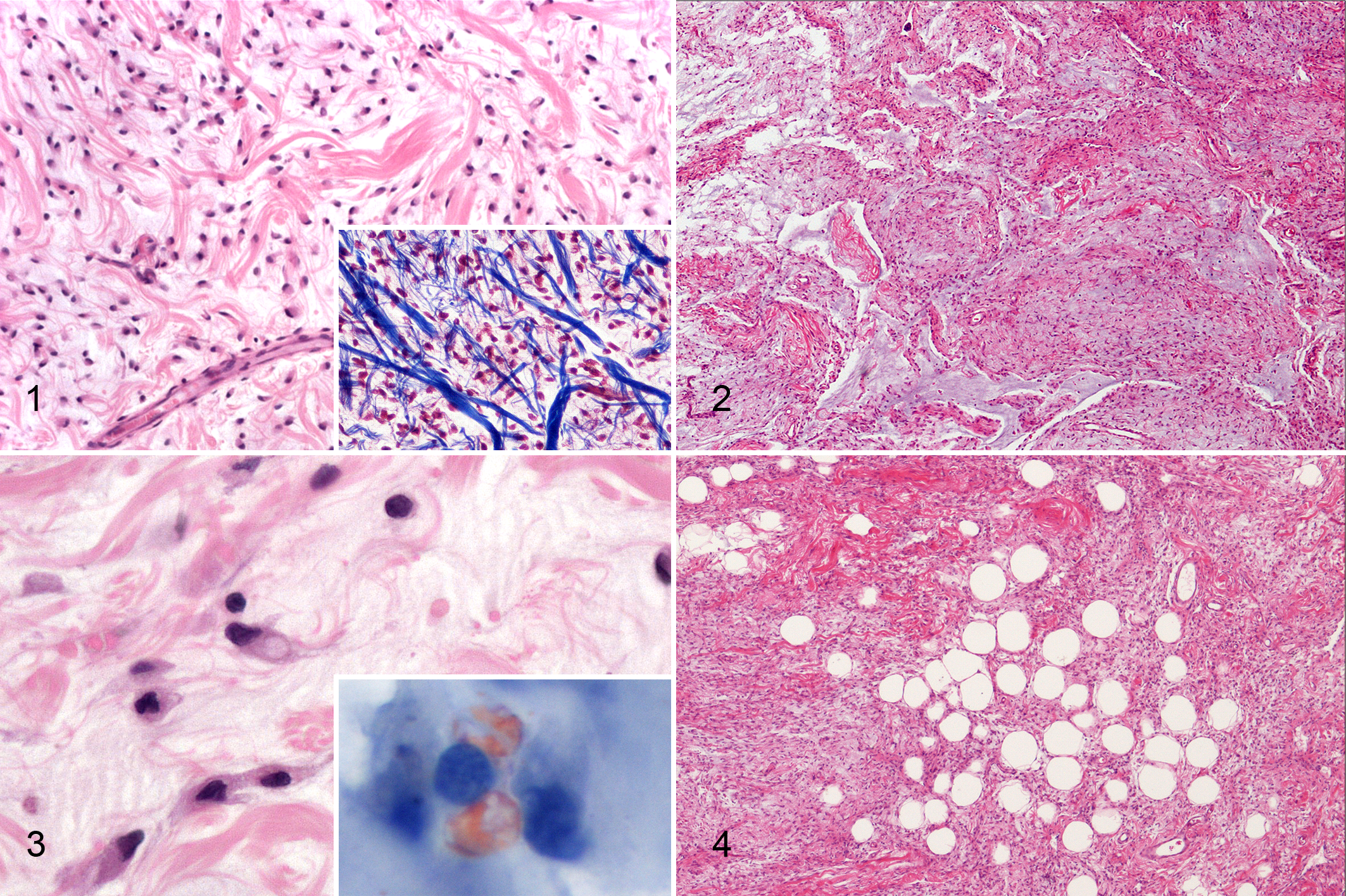
The main differential diagnoses included fibrolipoma, atypical lipoma, myxoid and dedifferentiated liposarcoma, fibrosarcoma, peripheral nerve sheath tumor, canine soft tissue sarcoma with myoid differentiation (smooth muscle tumor, myofibrobastic tumor), and nodular fasciitis. Fibrolipoma is characterized by the presence of dissecting fibrous tissue in an otherwise typical lipoma, 5 while in our SCLs, the amount of well-differentiated adipocytes was scarce and the collagen fibers had the characteristic ropey appearance, rather than the classical appearance of mature collagen. Atypical lipomas are composed by well-differentiated adipocytes intermixed with rare atypical lipoblasts, 5 and this diagnosis was excluded based on the inconsistent presence of only rare and scatted mature adipocytes. Myxoid and dedifferentiated liposarcoma are characterized by lipoblasts featuring single or multiple sharply defined cytoplasmic vacuoles deforming a hyperchromatic nucleus and a high proliferation rate. 1 These morphological features were absent in our SCLs, which were characterized by low to absent mitoses and low MIB-1 labeling index, and adipocytic differentiation was evident only in the areas containing mature and well-differentiated adipocytes. All these features in conjunction ruled out the diagnosis of myxoid or dedifferentiated liposarcoma. 1 Furthermore, all SCL cases were immunohistochemically negative for MDM2, which is expressed in dedifferentiated canine liposarcoma. 1 Well-differentiated liposarcoma was excluded based on the absence of intracellular vacuoles in most of the cells and the abundant collagen. 3,5
Lack of S100, factor VIII-ra, and smooth muscle actin expression did not support perineural, endothelial, or smooth muscle/myofibroblastic differentiation of the neoplasms.
In human medicine, SCL is part of a morphological spectrum including pleomorphic lipoma, a neoplasm characterized by mature adipocytes intermixed with atypical multinucleated cells, that nevertheless was not identified in our caseload. 3
A tumor with fibrous differentiation could not be excluded based on immunohistochemical reactivity alone. Nevertheless, compared with a fibroma, the cellularity of the SCL cases was higher and the collagen fibers were less dense, more variable in size, and more haphazardly arranged. The bland appearance of the neoplastic cells, the occasional intracytoplasmic vacuoles, and the low cell proliferation were inconsistent with a diagnosis of fibrosarcoma, despite the “vimentin-only” phenotype. 4
Nodular fasciitis is considered a reactive, nonneoplastic lesion in dogs composed of fibroblasts and myofibroblasts that are therefore assumed to express smooth muscle actin, which was absent in all our cases. Furthermore, nodular fasciitis is characterized by typically jagged or spiked margins and a “cell culture” appearance of the fibroblasts, 4 and both these features were absent in our cases, which showed an expansive growth of bipolar cells with oval nuclear morphology.
The histopathological features described in this caseload (bland spindle cells, immature adipocytes, rare mature adipocytes, ropey collagen, pseudo-angiomatous pattern, and myxoid matrix) paralleled descriptions of SCL in man. 2,3,7 Identification of these morphological hallmarks is required for SCL diagnosis. Awareness of these features and of this specific tumor is essential to avoid a misdiagnosis of STS.
Footnotes
Acknowledgements
We thank Ms A. Collins for editing the English text.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
