Abstract
Vascular endothelial growth factor (VEGF) is a dimeric protein that stimulates angiogenesis in vitro and in vivo by inducing endothelial cell proliferation and migration. In this immunohistochemical study, VEGF-immunolabeled cells were counted in a series of 10 benign and 40 malignant canine mammary tumors. The morphologic pattern of VEGF positivity (intensity of immunolabeling and VEGF granule size and distribution) was also evaluated. A low number of cells weakly positive for VEGF with few and small granules polarized to the luminal pole was detected in benign neoplasms. In contrast, in malignancies a high number of VEGF-positive cells had strong immunolabeling, often with large granules found diffusely in the cytoplasm. This level of immunolabeling was more pronounced in the less differentiated, more malignant phenotypes (grade 3). Macrophages, which can synthesize VEGF, were strongly positive. Stromal and myoepithelial cells were negative. VEGF data were correlated statistically with intratumoral microvessel density (number of newly formed microvessels) and both measures were greater in less differentiated malignant neoplasms, demonstrating that angiogenesis and malignancy increase together. VEGF appears to be a powerful angiogenic factor in canine mammary tumors.
Angiogenesis, the formation of new vessels from the preexisting vascular bed, is a physiologic process that is tightly regulated by stimulatory and inhibitory factors that function in a delicate balance.8,10 In tumors, the balance falls in favor of stimulatory factors, and the angiogenesis increases. Among these factors, vascular endothelial growth factor (VEGF) is the most thoroughly investigated in human oncology.4,13,21,25,29 It is a dimeric protein that stimulates migration and proliferation of vascular endothelial cells, key steps in angiogenesis.5,7,15 It is produced by neoplastic cells, macrophages, plasma cells, and sometimes also by lymphocytes and exerts paracrine effects by binding specific tyrosinase receptors localized on vascular endothelial cells, but it may also exert autocrine effects by stimulating neoplastic cell growth.17
In many human malignant tumors, overexpression of VEGF has been correlated with metastasis,27 and experimental studies using animal models have demonstrated that the inhibition of VEGF production by administration of anti-VEGF antibodies was associated with a reduction of the neoplastic growth.12 Therefore, VEGF is considered an important angiogenic factor in human oncology.
Because this angiogenic factor is still poorly investigated in veterinary oncology,18 the aim of the present study was to evaluate VEGF expression in a series of canine mammary tumors and to correlate this expression with the differentiation grade and microvessel density of neoplasms.
Materials and Methods
Samples
Fifty samples of canine mammary tumors (10 benign and 40 malignant) were examined. Tumors were classified using The World Health Organization criteria9 (Table 1) and were graded as moderately to poorly differentiated (grades 1–3) by two independent observers. The parameters used were 1) extent of tubule formation (for tubular and papillary adenocarcinomas), 2) hyperchromatism and number of mitoses, and 3) irregularity of size, shape, and staining of nuclei.19
WHO classification of canine mammary tumors used in this study.
Among the malignant tumors, two of 18 tubular adenocarcinomas, three of six solid carcinomas, two of three squamous cell carcinomas, and one of three malignant mixed tumors, all grade 3, had already metastasized to the inguinal lymph nodes at the time of diagnosis. Two of the three early metastatic solid carcinomas metastasized to the lung after two years. Three of 10 tubular adenocarcinomas (grade 2) and two of six solid carcinomas (grade 3) that had not metastasized at the time of diagnosis spread to lungs, liver, and bones after 2 years.
Immunohistochemistry
All samples were fixed in 10% neutral buffered formalin and embedded in paraffin. Sections 5 μm thick were deparaffinized in xylene, dehydrated in graded alcohols, and washed in 0.01 M phosphate-buffered saline (PBS), pH 7.2–7.4. Endogenous peroxidase was blocked with 0.3% hydrogen peroxide in absolute methanol for 30 minutes. Before the immunohistochemical procedure, samples in ethylenediamine tetraacetic acid were heated three times for 5 minutes each at 750 W in a microwave oven. The primary antibody was a monoclonal mouse anti-human VEGF Ab-3 (clone JH121, NeoMarkers).
The antibody was diluted 1:200 in Tris-HCl buffer containing 0.5% bovine serum albumin and 0.015 M sodium azide (Dako). The sections were incubated overnight at 4 C. The immunolabeling procedure included negative control sections incubated in PBS without primary antibody. Biotinylated anti-mouse, anti-rabbit, and anti-goat immunoglobulins (LSAB Kit; Dako) diluted in PBS were used as secondary antibodies; they were applied for 30 minutes. After washing in PBS, the sections were incubated in streptavidin conjugated to horseradish peroxidase in Tris-HCl buffer containing 0.015% sodium azide (LSAB Kit; Dako) for 30 minutes. To demonstrate the immunolabeling, 3,3′-diaminobenzidine tetrahydrochloride was used as a chromogen, and hematoxylin was used as a counterstain.
Scoring of immunoreactivity
A count of VEGF-positive cells was performed by two observers at 400× magnification (40× objective + 10× ocular). At least 20 fields per tumor were examined, and a minimum of 1,000 neoplastic cells were counted. The results were expressed as a percentage. VEGF intensity was scored as follows: + = strong; ± = weak. VEGF granule size was scored as SG (small granules) when the immunoreactivity was expressed by small cytoplasmic brown granules and LG (large granules) when the immunoreactivity was expressed by large cytoplasmic brown granules. VEGF distribution was also scored as P (polarized) when the VEGF granules were restricted to the luminal pole of neoplastic cells and C (cytoplasmic) when the VEGF granules were located diffusely in the cytoplasm. Microvessel density was assessed by image analysis system as previously described.23 Twenty fields per tumor were randomly chosen, and every immunolabeled intratumoral vessel was counted on a 400× field using an automated (Image Pro Plus 1) image analysis system (Sistema MONO; Immagini e Computer, Milan, Italy).
Statistical analysis
VEGF-positive cell number and microvessel density, expressed as means and SDs, were both compared with differentiation grade of mammary neoplasms by analysis of variance (ANOVA). VEGF-positive cell number and microvessel density were compared with each other among the benign mammary neoplasms and different malignant grades using the linear regression.
Results
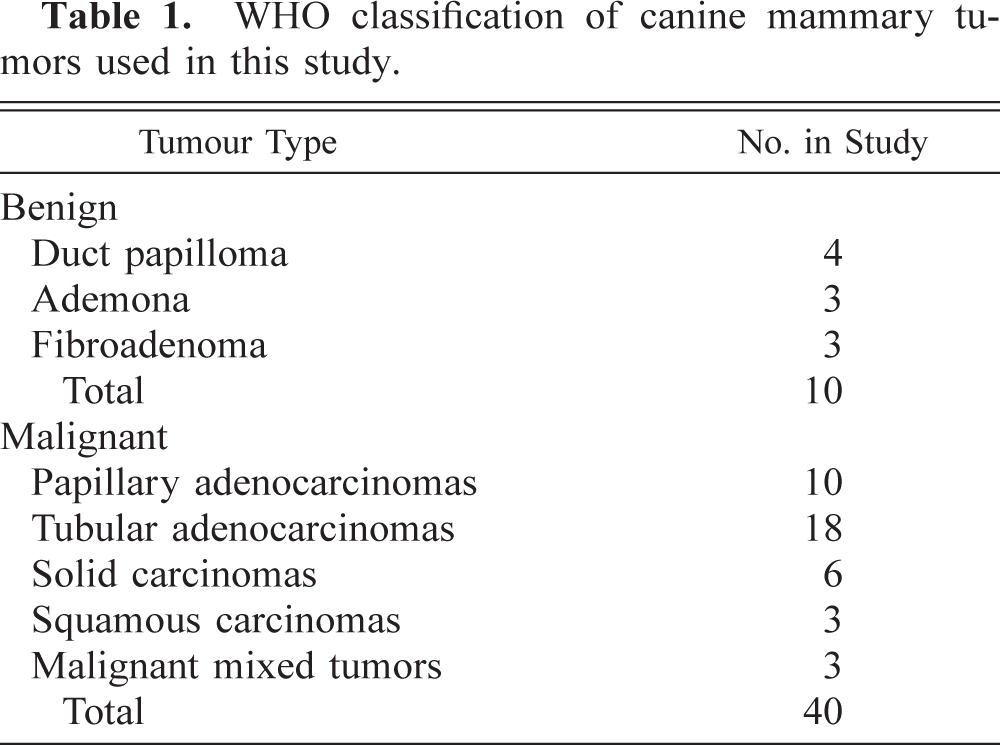
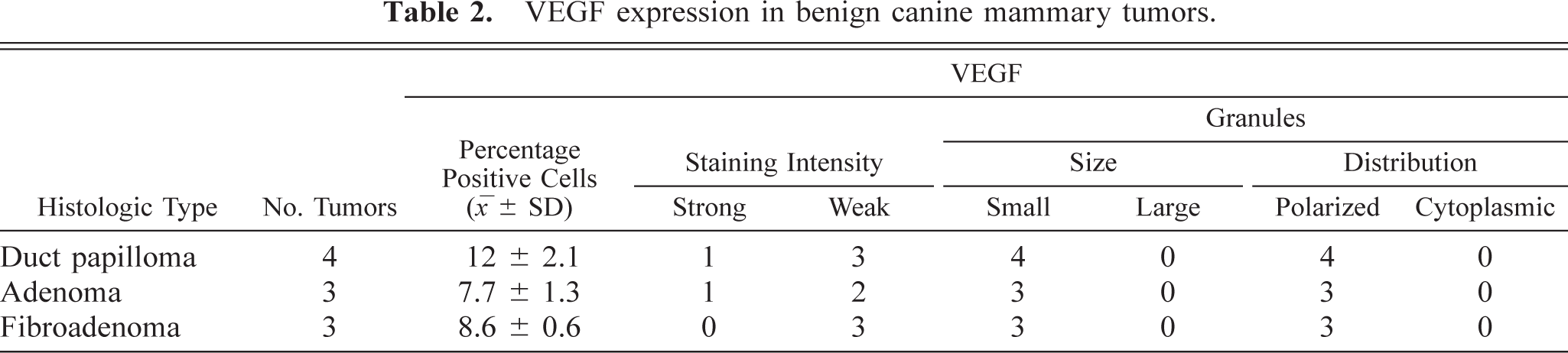
In benign tumors (Table 2), neoplastic cells expressed weak, fine granular immunolabeling that was polarized to the luminal pole (Fig. 1). The percentage of positive cells ranged from 8.4% to 15.2% (9.7% ± 2.5%). In six of 14 grade 1 tumors (Table 3), neoplastic cells expressed strong VEGF immunolabeling (+); in the remaining eight samples, the VEGF intensity was weak (±). In most of grade 1 tumors, VEGF granules were small and polarized to the luminal cell pole (SG/P). Percentage of positive cells ranged from 11.3% to 28.1% (15.2% + 5.5%). Ten of 12 grade 2 tumors (Table 3) expressed strong VEGF immunolabeling (+). VEGF granules were small and polarized in six samples (SG/P) and large and diffuse in the cytoplasm in the others (LG/C) (Fig. 2). Percentage of positive cells ranged from 14.7% to 32.7% (23.2% ± 5.1%). Eleven of 14 grade 3 tumors (Table 3) expressed strong VEGF immunolabeling (+). VEGF granules were large and diffuse in the cell cytoplasms (LG/C) (Fig. 3). The percentage of positive cells ranged from 24.3% to 37.4% (33% ± 4.4%).
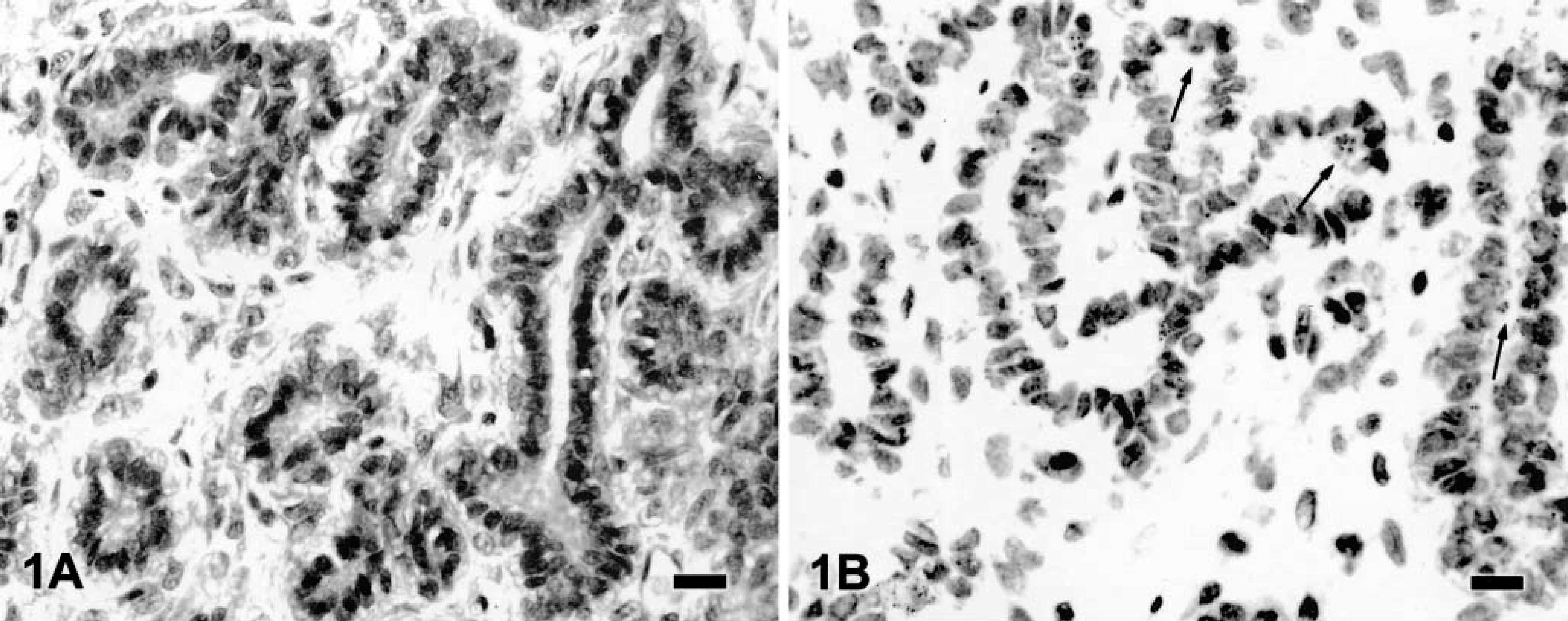
Canine mammary fibroadenoma; tumor No. 9. Weak VEGF immunolabeling is seen in a few neoplastic cells showing small abluminal granules (SG/P) (arrows).
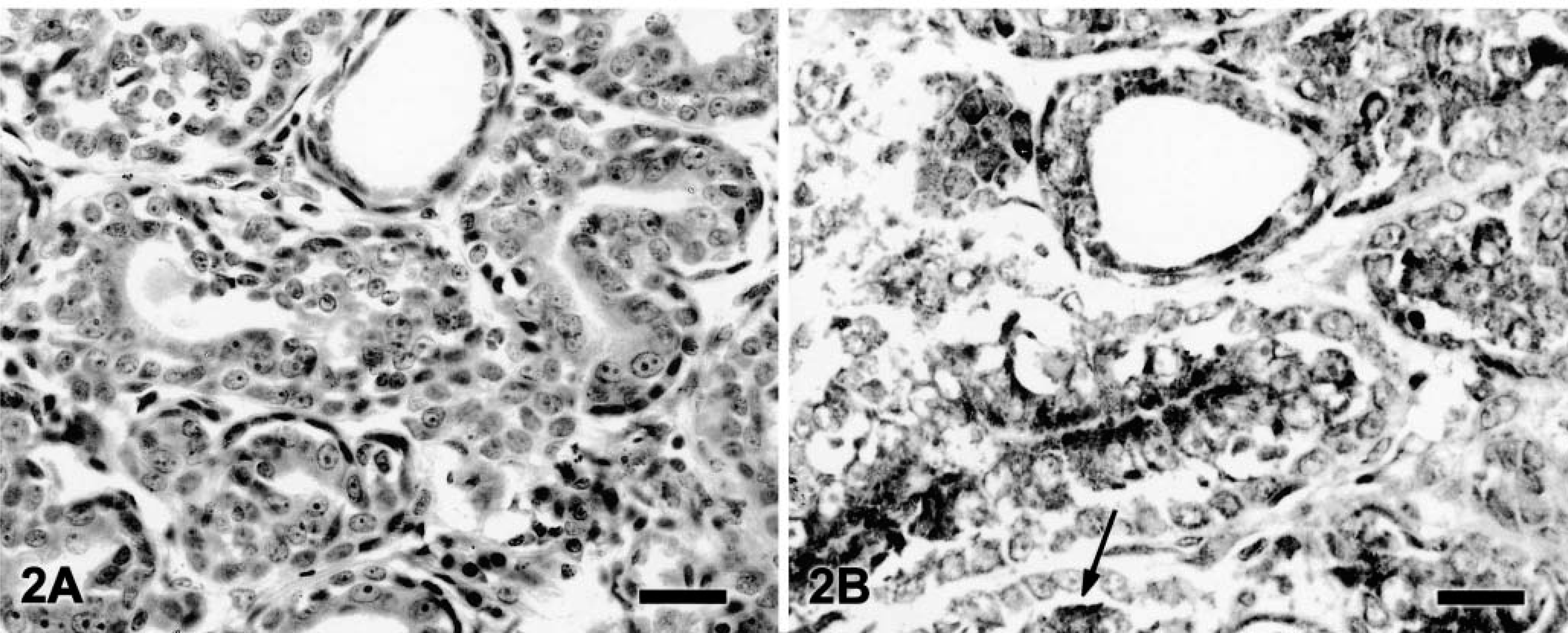
Canine grade 2 tubular adenocarcinoma; tumor No. 24. Neoplastic cells show strong VEGF immunolabeling expressed by small and polarized granules. Some neoplastic cells showing strong diffuse immunolabeling are also present (arrow).
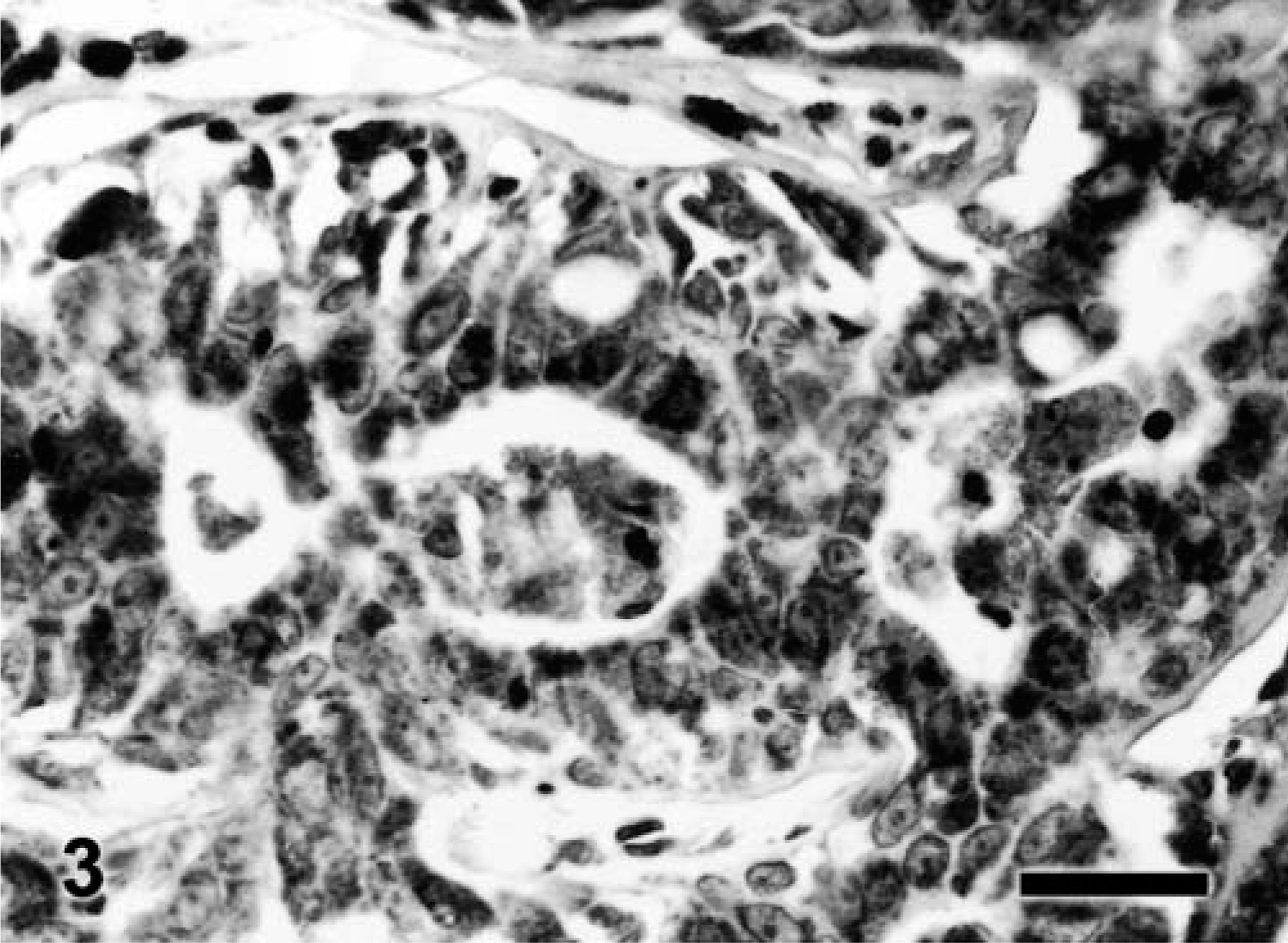
Canine grade 3 tubular adenocarcinoma; tumor No. 28. Many neoplastic cells show strong diffuse VEGF immunolabeling. Streptavidin–biotin–peroxidase complex method. Bar = 10 μm.
VEGF expression in benign canine mammary tumors.
VEGF expression in malignant canine mammary tumors.
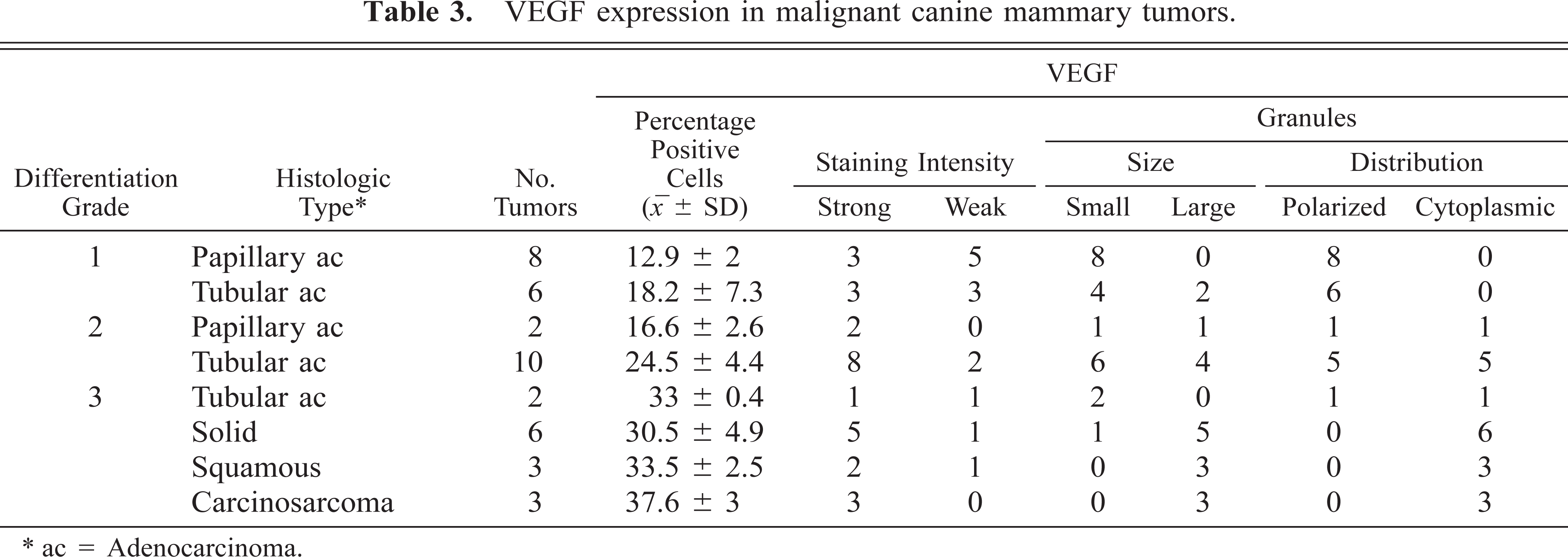
ac = Adenocarcinoma.
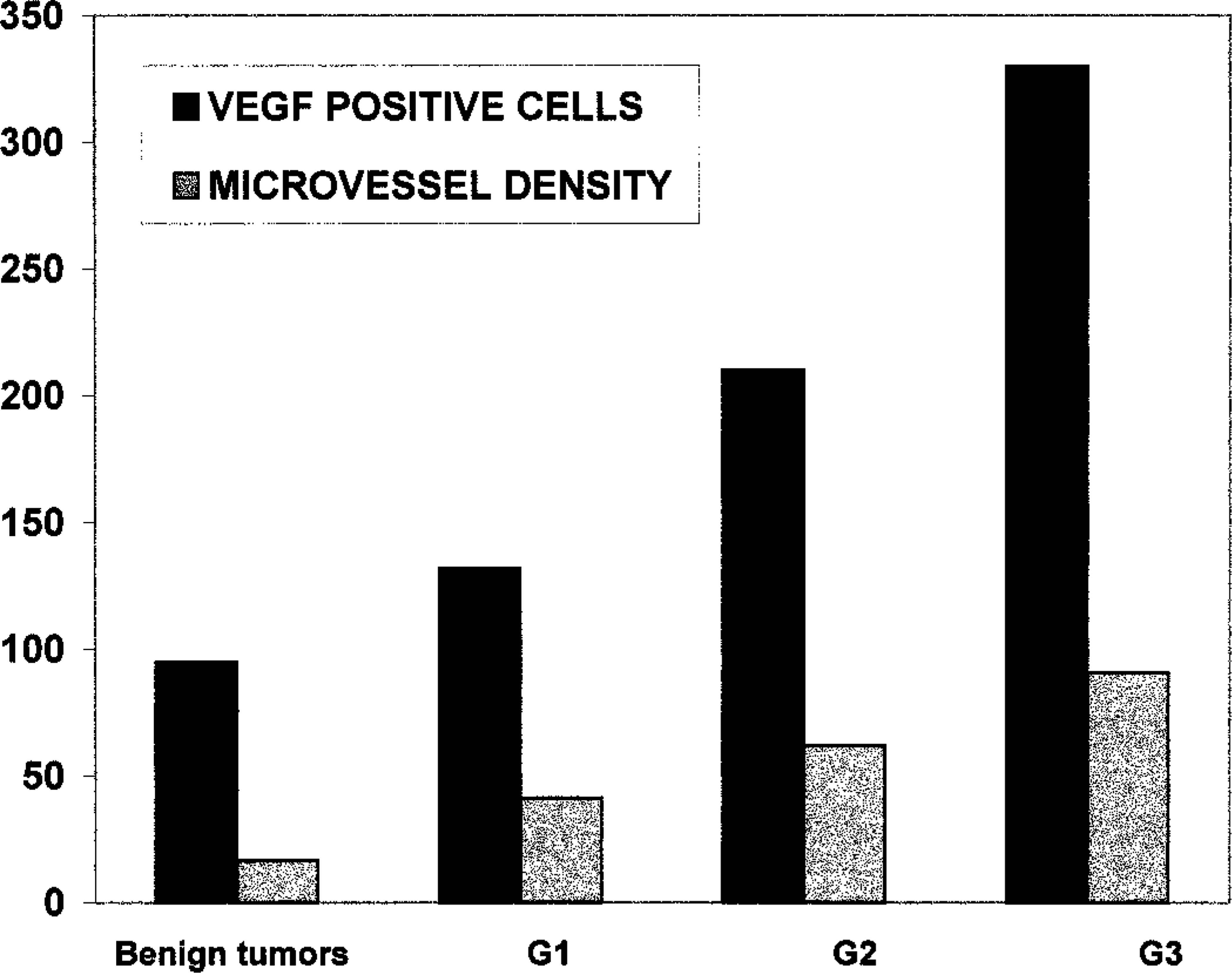
In many malignant neoplasms, strong and diffuse immunolabeling was expressed by neoplastic cells located near necrotic centers. Macrophages, which were present at the periphery and often infiltrated the neoplastic proliferations, expressed strong and diffuse immunolabeling and were used as positive controls. Stromal and myoepithelial cells were negative. The number of VEGF-positive cells was higher in malignant than in benign tumors (
Correlation of number of VEGE positive cells and microvessel density with differentiation grade of tumor.

Microvessel density was greater in malignant than in benign tumors (
VEGF-positive cells and microvessel density were correlated with each other for both benign and malignant tumors (
Correlation of VEGF-positive cells with microvessel density for benign and malignant (grades G1–G3) canine mammary tumors.
Discussion
In tumors, increased VEGF synthesis is considered one of the first expressions of the angiogenetic switch, which contributes to acquisition of malignant characteristics such as rapid growth and metastasis.3,10 In this study, an increase in VEGF production, as indicated by a high number of positive cells associated with a loss of VEGF granule polarization, was observed in malignant tumors, especially those that were less differentiated. Loss of polarization of VEGF granules, which was more evident in grade 2 and grade 3 tumors than in benign or grade 1 neoplasms, could be a consequence of the enhanced VEGF synthesis and VEGF accumulation in neoplastic cells. The increase in VEGF synthesis could be linked to the activation of oncogenes encoding VEGF6 or to the loss of suppressor genes encoding VEGF inhibitors.22
Upregulation of VEGF could be also linked to the hypoxic condition present in the necrotic compartments. VEGF expression is highest in tumors with significant necrosis.24 Hypoxia can increase transcription of the VEGF-encoding gene and can stabilize its mRNA product,11 which could explain the strong immunolabeling observed in neoplastic cells located near the necrotic regions of malignant tumors. The hypoxic response also could act synergistically with other tumor-related factors, including cytokines such as tumor necrosis factor α produced by macrophages and other inflammatory cells, to upregulate VEGF in neoplasms. In this regard, macrophages, which can also synthesize VEGF, could play an important role in VEGF-mediated angiogenesis.14,16
In this study, the upregulation of VEGF also was correlated with microvessel density, in agreement with results of some human studies.13,26,27
In a previous evaluation of mammary neoplastic vasculature,23 the values for microvessel areas and perimeters were lower in less differentiated malignant tumors than in more differentiated malignant and benign tumors. VEGF stimulates endothelial cell proliferation and migration, but many other angiogenic factors are necessary for mature, functioning new vessels. Therefore, in tumors, VEGF-induced endothelial cell proliferation may not be adequately balanced by other angiogenic factor synthesis. As a consequence, in malignant tumors, microvessels become aberrant, neoplastic cells outgrow the vascular oxygen supply, and the result is ischemic necrosis.1
Endothelial cell proliferation induced by VEGF promotes metastasis by 1) inducing formation of new vessels, 2) producing collagenase and other degradative enzymes that facilitate neoplastic cell escape from the mass into the vasculature,20 and 3) stimulating neoplastic cell growth.28 Therefore, the more VEGF, the more endothelial cells, and the greater the risk for tumor growth and dissemination. This proposed sequence of events provides a theoretical explanation for the success of VEGF inhibition in experimental tumor therapy.2,12 In this regard, canine mammary neoplasms may be useful models for therapeutic trials.
Footnotes
Acknowledgements
This work was supported by grants from the Ministero della Ricerca Scientifica e Tecnologica 1997. We thank Mr. R. Ilsami for technical assistance.
