Abstract
A 3-year-old neutered male Australian Shepherd mix dog presented with metastatic soft tissue sarcoma arising from a primary tumor in the left lumbar fascia. Two separate metastases to the lungs were characterized by neoplasia within bronchiolar walls, which caused obstruction of the bronchiolar lumina and atelectasis of adjacent alveoli, a characteristic feature of endobronchial metastasis. Neoplastic cells of the primary lumbar neoplasm, metastatic pulmonary lesions, and additional widespread metastatic masses identified postmortem were similarly immunoreactive for vimentin, but non-immunoreactive for cytokeratin, cluster of differentiation 18, synaptophysin, chromogranin, and desmin. The present report describes a naturally occurring case of endobronchial metastasis in a dog.
In human medicine, endobronchial metastasis (EBM) is defined as direct metastasis of an extrapulmonary neoplasm to the tracheal, bronchial, or bronchiolar wall, resulting in a neoplastic mass within the airway wall. The metastatic mass often elevates the respiratory epithelium, which may ulcerate, and may protrude into and/or compress the airway lumen, causing atelectasis of the corresponding lung fields and clinical signs related to airway obstruction. 9 In human beings, EBM has been reported in association with carcinomas of the breast, urinary bladder, prostate, and gastrointestinal tract (specifically colon) and is more rarely documented with sarcomas.1–4,6,7,13–15 Expectoration of airway casts containing neoplastic cells from EBM lesions may precede the diagnosis of the primary tumor or occur several years after treatment of a primary tumor.3,8,9,11,12 Findings of computed tomography imaging include atelectasis and obstruction or narrowing of airways adjacent to a pulmonary mass, but do not always demonstrate the intramural nature of the lesions; the latter is often identified by bronchoscopy or histology following surgical resection of the tumor.5,10 In human beings, EBM is associated with highly aggressive primary tumors of guarded to poor prognosis.
A 3-year-old male neutered Australian Shepherd mix dog was presented to the Oncology Service of Colorado State University’s Veterinary Teaching Hospital (Fort Collins, Colorado) for evaluation of a 10 cm × 10 cm × 17 cm mass in the fascia of the left lumbar region. A computed tomography (CT) scan of the abdomen demonstrated an expansile, well-demarcated, heterogeneously attenuating mass adjacent to the left lumbar vertebrae and compressing adjacent skeletal muscles. The left medial iliac lymph node and multiple mesenteric lymph nodes were enlarged and heterogeneously attenuating, with mixed contrast enhancement. An incisional biopsy of the lumbar mass yielded a diagnosis of grade III soft tissue sarcoma.
Stereotactic radiation therapy was elected due to the large size of the mass, its invasive nature, and its anatomic location. Preliminary therapy of 10 Gy × 3 fractions given over 5 days resulted in greater than 20% decrease in tumor volume. Following stereotactic radiation, the patient received doxorubicin chemotherapy (once every 3 weeks for a total of 5 treatments), which resulted in a progressive decrease in tumor size to approximately 5 cm in diameter. Five months after radiation therapy, the remaining mass was surgically removed en bloc.
Survey radiographs of the thorax taken 2 months after en bloc surgical excision of the left lumbar mass revealed 2 soft tissue nodules, 1 in the right middle and 1 in the right cranial lung lobe, which measured 1.7 cm and 0.7 cm in maximal dimension, respectively. Recheck radiographs 2 months later demonstrated dramatic progression of the lesion in the right middle lung field to approximately 10 cm in maximal diameter; the lesion in the right cranial lung lobe had not appreciably changed. By CT scan, the cranial lung lobe lesion appeared to abut a small bronchiole (Fig. 1). Atelectasis was noted in the pulmonary parenchyma adjacent to each lesion. A thoracotomy was performed, and the affected lobes were removed. The tracheobronchial lymph node, which was grossly normal at surgery, was also sampled for histology at the time of lobectomy. Grossly, the right middle lung lobe was expanded by a 9.4-cm-diameter, unencapsulated but discretely marginated mass. The mass was friable and mottled brown, tan, and red on cut section. Within the right cranial lung lobe, a 4.5-mm-diameter bronchiole was grossly occluded by a soft, tan mass. The parenchyma surrounding each of the metastatic masses was atelectatic but otherwise macroscopically unremarkable.
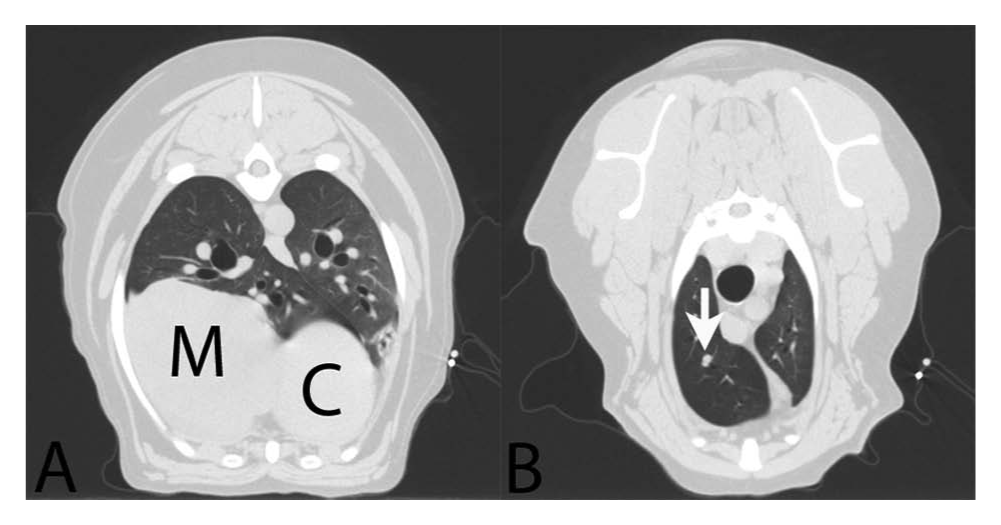
Dog; computer-assisted tomography.
The patient was prescribed metronomic cyclophosphamide (15 mg/m2 every 24 hr). One month after lung lobectomy, a new 7-cm soft tissue mass was detected in the thorax by survey radiography. No treatment was elected at that time. The patient presented with acute neurologic signs approximately 1 month later and died despite supportive care. Significant lesions at postmortem examination included discrete but unencapsulated soft tissue masses arising in the left lateral pericardium, both adrenal gland medullae, and the left temporal lobe of the brain. Each mass was mottled brown and red on cut section and contained variably sized foci of necrosis.
All tissue specimens for histology (including both biopsy and postmortem specimens) were fixed for >24 hr in 10% neutral buffered formalin, trimmed, processed routinely, sectioned at 4 µm, and stained with hematoxylin and eosin for histologic evaluation. Sections of the primary neoplasm, both pulmonary metastases, and metastatic lesions in the pericardium, adrenal glands, and brain were immunohistochemically stained using standard alkaline phosphatase methods for detection of cytokeratin a and vimentin. b Additional selected sections were stained for synaptophysin, c chromogranin, d cluster of differentiation (CD)18, e and desmin. f All immunohistochemistry was performed with an automated stainer. g Slides were deparaffinized, and antigen retrieval was performed with ER2 h epitope retrieval solution prior to incubation with the primary antibody (30 min, 37°C), secondary antibody (25 min, 37°C), and chromogen visualization. All immunohistochemistry preparations were counterstained with hematoxylin, dehydrated, and mounted in xylene-based mounting medium. Positive control tissues included the following canine tissues: pulmonary epithelium and cutaneous epidermis for cytokeratin, vascular endothelium and smooth muscle for vimentin, adrenal medulla for synaptophysin and chromogranin, lymph node for CD18, and skeletal muscle for desmin. For negative controls, nonspecific sera replaced specific antibodies in each immunohistochemical protocol.
The histologic features of the tissue from the incisional biopsy of the subcutaneous mass from the left craniodorsal hip were consistent with a grade III soft tissue sarcoma. The neoplasm was characterized by spindloid cells arranged in intersecting bundles and streams (Fig. 2). The cells had indistinct cell borders, moderate amounts of amphophilic staining cytoplasm, and ovoid nuclei with stippled chromatin and prominent nucleoli. Mitotic figures were common, and extensive areas of necrosis were present. Multiple veins within the tumor were characterized by segmental disruption or complete effacement of the endothelial cells by neoplastic cells, which occasionally bulged minimally into the vascular lumina. However, overt thromboembolism of neoplastic cells was not identified. Tissue excised from the left lumbar fascia after stereotactic radiation was almost completely necrotic with extensive histiocytic infiltration and fibroplasia. No viable neoplastic cells were identified.
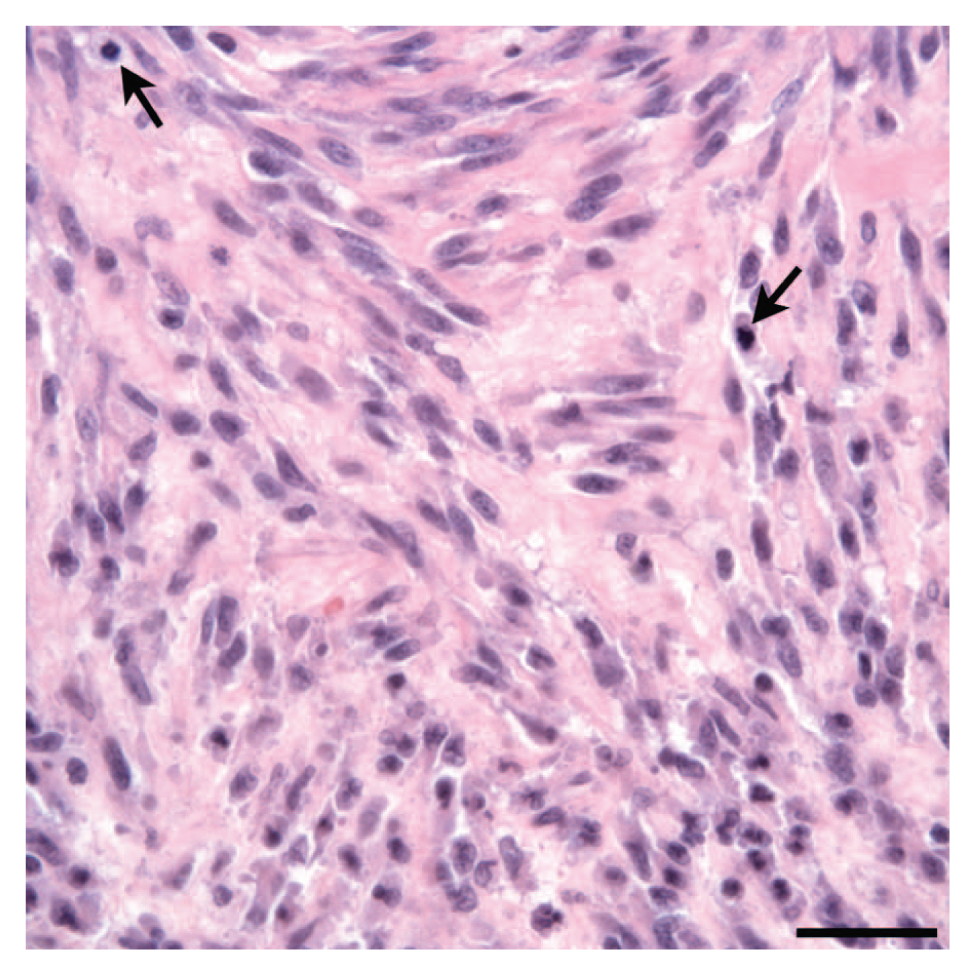
Dog; primary soft tissue sarcoma, left lumbar fascia. The neoplastic cells have indistinct cell borders, small amounts of eosinophilic cytoplasm, and spindloid nuclei with finely stippled chromatin and prominent nucleoli. Mitotic figures are common (arrows). The neoplastic cells are supported by moderate amounts of fibrovascular stroma. Hematoxylin and eosin. Bar = 50 µm.
Histologically, the large right middle lung lobe mass was composed of anaplastic cells arranged in broad sheets supported by scant, variably edematous fibrovascular matrix. The metastatic neoplastic cells effaced the muscular architecture of a small bronchiole (Fig. 3A). The bronchiole was lined by ciliated low cuboidal epithelium and squamous cells. Scant amounts of hemorrhage and proteinaceous fluid were present within the bronchiole lumen (Fig. 3B). The neoplastic cells were polygonal and spindloid with indistinct borders and small amounts of granular eosinophilic cytoplasm. Nuclei were round with stippled and clumped chromatin and a single prominent nucleolus (Fig. 3C). The mitotic rate was high with >50 mitoses per ten 400× fields. Neoplastic cells occasionally bulged into the lumina of incorporated blood vessels, and other vessels were occluded by fibrin thrombi. Scattered foci of necrosis and hemorrhage were present throughout the mass. The adjacent pulmonary parenchyma was atelectatic, and alveoli contained moderate numbers of foamy alveolar macrophages and hemosiderophages.
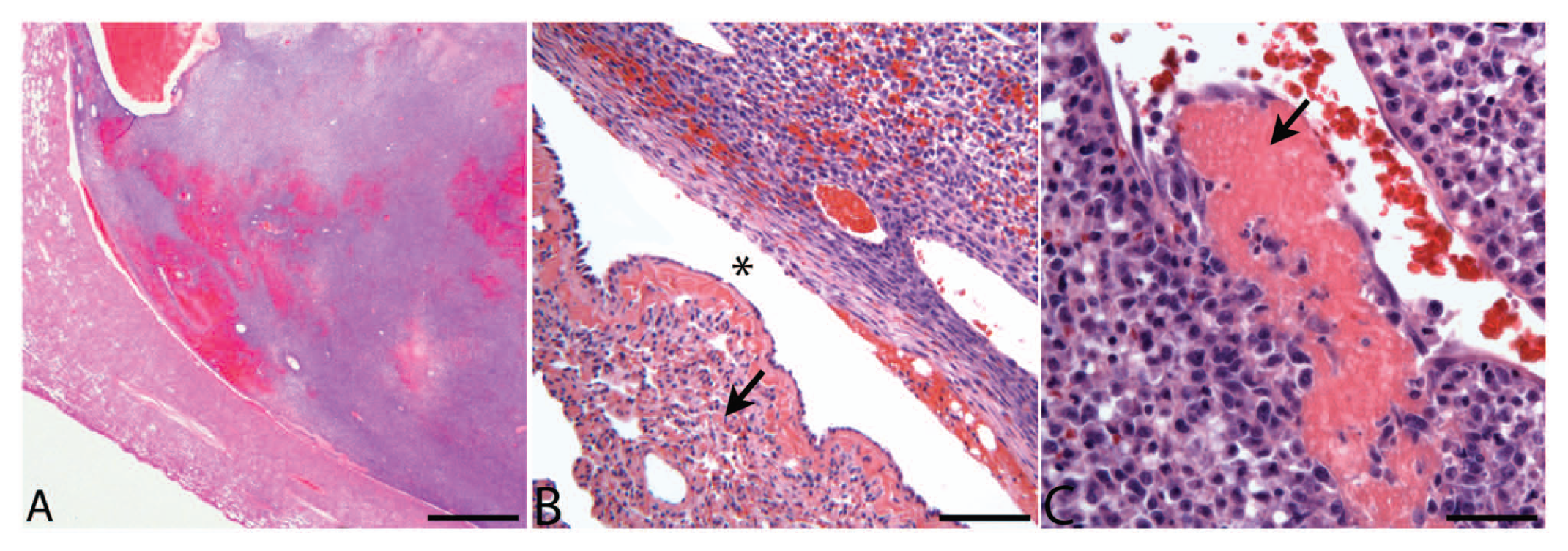
Dog; endobronchial metastasis.
Histology of the bronchiolar mass from the right cranial lung lobe revealed that the bronchiolar epithelium was elevated and partially effaced by a 7 mm × 3 mm mass within the bronchiolar wall. The mass was composed of neoplastic cells, granulation tissue and scattered lymphocytes, plasma cells, and hemosiderophages (Fig. 4A). The neoplasm was covered with mildly disorganized respiratory epithelium with extensive ulceration and multifocal squamous metaplasia (Fig. 4B). The lesion almost completely occluded the airway, and the adjacent alveoli were moderately atelectatic. The neoplastic cells were similar to those previously described (Fig. 4C). Over 100 step sections of this lesion were evaluated. Neoplastic cells were not identified in the bronchiolar muscle or adventitia or in the adjacent parenchyma outside of the airway. Since neoplastic cells subtended and elevated the bronchiolar epithelium, it was concluded that the neoplasm most likely metastasized to the bronchiolar submucosa at a site not represented in sectioning (perhaps due to tissue orientation in the paraffin block). Tracheobronchial lymph node sampled at the time of lobectomy was histologically unremarkable except for sinus drainage of hemorrhage and mild histiocytosis. Tumor cells in the primary left lumbar neoplasm, lung metastases, and metastases in the pericardium, adrenal glands, and left forebrain demonstrated weak to strong immunohistochemical staining for vimentin (Fig. 5A) and negative staining for cytokeratin (Fig. 5B), CD18, synaptophysin, chromogranin, and desmin (results not shown), consistent with soft tissue sarcoma.
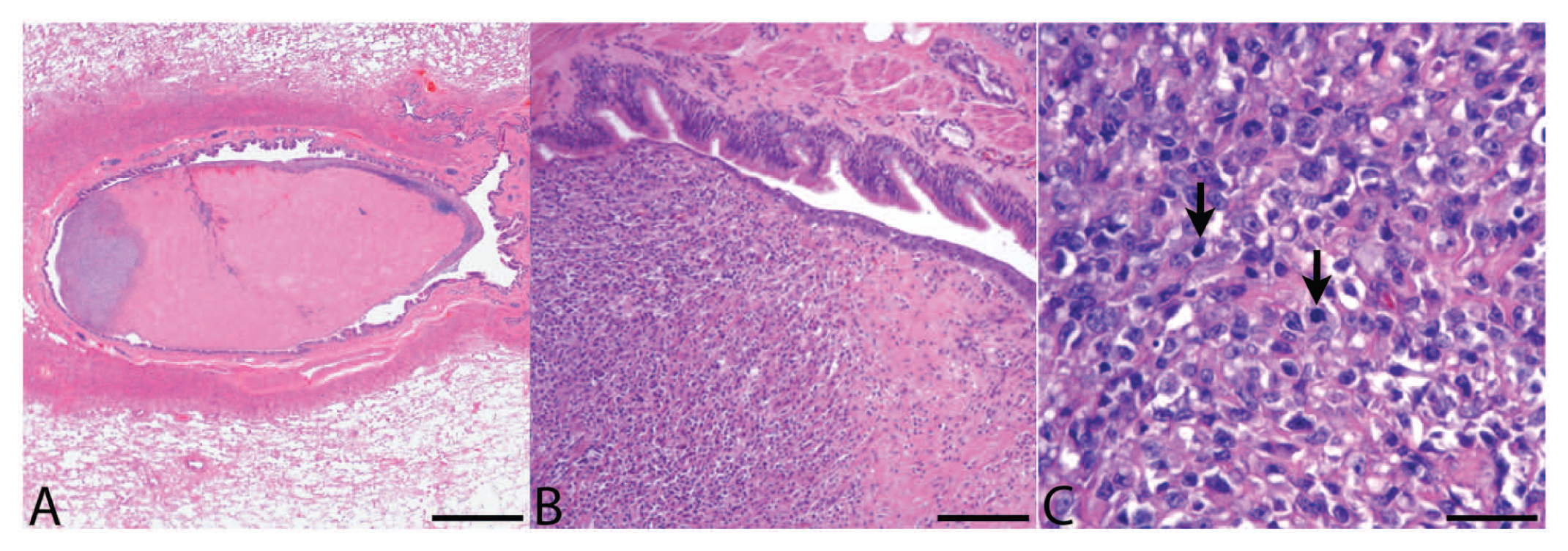
Dog; endobronchial metastasis.
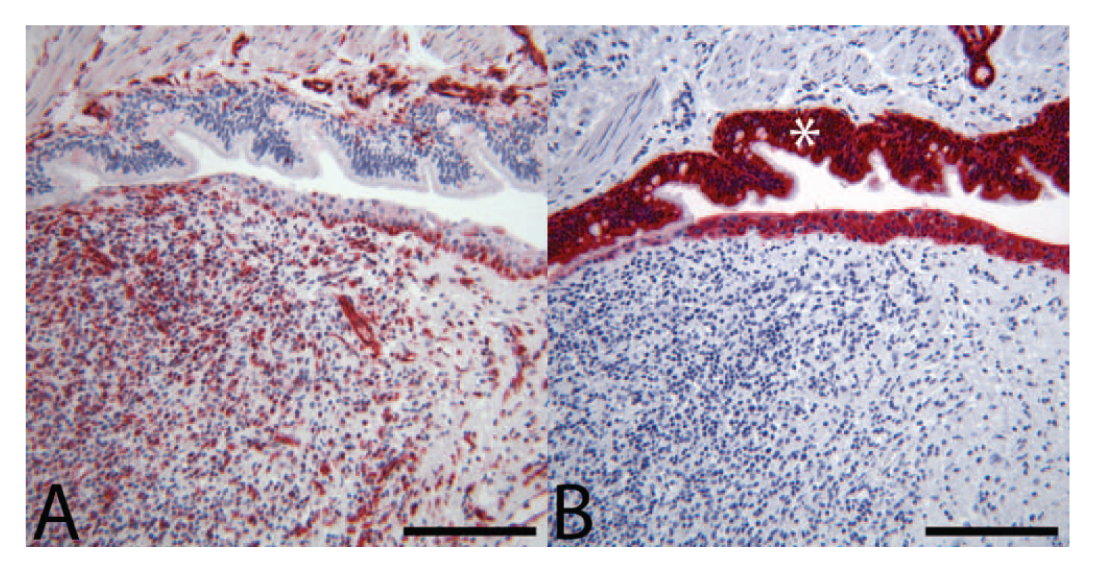
Dog; endobronchial metastasis.
The 2 metastatic pulmonary tumors demonstrate features of EBM. Both tumors were within the bronchiolar walls with variable local effacement of the bronchiolar architecture. The lesion in the right cranial and middle lung lobes expanded and compressed the bronchiolar lumina, resulting in partial occlusion of the airway and local pulmonary atelectasis. Both tumors were partially covered by attenuated to ulcerated respiratory epithelium. While the point of attachment of the bronchiole-associated mass in the right cranial lung lobe was not specifically identified, neoplastic cells were not evident in the bronchiolar muscularis or adventitia or surrounding pulmonary parenchyma. As the neoplasm clearly elevated the overlying epithelium, this strongly suggests that the tumor metastasized to the bronchiolar submucosa. Interestingly, despite gross and histologic evidence of atelectasis and compromise of bronchiolar lumina, the patient in the current report had no clinical signs associated with pulmonary disease.
Endobronchial metastasis is rare but well described in human literature and is associated with an overall poor prognosis. Evidence provided in the present report suggests that while rare, EBM may be associated with high-grade malignancies in domestic animal species as well. The patient in the present case died with disseminated metastatic disease despite aggressive radiation treatments, multiple surgeries, and chemotherapy.
Recognizing EBM in veterinary patients via CT scan or biopsy of lung lesions may help predict the clinical course and determine clinical therapy. In addition, recognizing EBM is important due to potential complicating clinical factors such as pulmonary atelectasis producing chronic cough, emphysema, and respiratory distress. Increased awareness of this entity in veterinary medicine will be necessary to determine the true incidence, clinical relevance, and appropriate treatment. The factors leading to specific tropism of tumor emboli to the bronchial or bronchiolar wall are unclear and merit further study. 6 In summary, the current report describes the case of a dog with metastatic soft tissue sarcoma and EBM in the lungs.
Footnotes
Acknowledgements
The authors thank Todd Bass for assistance with histology and immunohistochemistry and Charlie Kerlee and Jay Oaks for assistance with images.
a.
CK AE1/AE3, PA0909, Leica Microsystems Inc., Buffalo Grove, IL.
b.
VIM SRL33, PA033, Leica Microsystems Inc., Buffalo Grove, IL.
c.
27G12, PA0299, Leica Microsystems Inc., Buffalo Grove, IL.
d.
SP-1-Chromogranin A, Immunostar Inc., Hudson, WI.
e.
CA16.3C10-IgG, Dr. Peter Moore, School of Veterinary Medicine, University of California, Davis, Davis, CA.
f.
DE-R-11, PA0032, Leica Microsystems Inc., Buffalo Grove, IL.
g.
21.0051, Bond Max automated stainer, Leica Microsystems Inc., Buffalo Grove, IL.
h.
AR9640, Leica Microsystems Inc., Buffalo Grove, IL.
Declaration of conflicting interests
The author(s) declare that they do not have any conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
