Abstract
Small intestinal adenocarcinomas are uncommon neoplasms that are rarely reported in nonhuman primates. These neoplasms are also rare in humans, although they are thought to share a similar pathogenesis with the more common colorectal carcinoma. Herein the authors report the clinical, histologic, immunohistochemical, and molecular characteristics of small intestinal adenocarcinoma in 10 common marmosets (Callithrix jacchus). Retrospective analysis of necropsy records revealed small intestinal carcinoma to be the most common neoplastic cause of morbidity and mortality in aged common marmosets. The average age of affected animals was 6.6 years old, and there was no sex predilection. Nine of 10 (90%) tumors arose within the proximal small intestine near the interface with the duodenum. All cases were characterized by disorganization, loss of polarity, and proliferation of neoplastic epithelial cells along the crypt to midvillous interface. Two of 10 (20%) were defined as carcinoma in situ. Eight of 10 (80%) had some degree of invasion, with lymphatic invasion and lymph node metastasis present in 6 of 10 (60%) animals. Immunohistochemically, 10 of 10 (100%) expressed cytokeratin; 7 of 9 (77%) expressed E-cadherin; and 8 of 9 (88%) expressed β-catenin. The expression of E-cadherin and β-catenin was decreased in the cell membrane and increased in the cytoplasm. No Helicobacter-like bacteria were observed via silver stain, and callitrichine herpesvirus 3 was detected by polymerase chain reaction with equal frequency from neoplastic and nonneoplastic intestinal sections. The tumors described in this population illustrate comparable features to human cases of small intestine carcinoma and may serve as a potential animal model for small intestinal carcinomas.
Gastrointestinal carcinomas are relatively common in humans; however, the bulk of these neoplasms arise in the colon and rectum with a small percentage in the small intestine. 4,9 Those that arise in the small intestine are more prevalent in the duodenum compared to the mid- and distal jejunum and are directly linked to advanced age; however, no other causative factors have been found in human medicine. 7 In nonhuman primates, a similar scenario exists in which gastrointestinal carcinomas are most commonly described in the large intestine whereas those arising in the small intestine are described only rarely. 1,2,18 The prototypical gastrointestinal carcinomas in nonhuman primates are ileocecal adenocarcinomas in the rhesus macaque (Macaca mulatta) and colorectal adenocarcinoma in aged cotton-top tamarins (Saguinus oedipus). The proposed pathogenesis of colorectal adenocarcinoma in cotton-top tamarins involves an association between chronic lymphoplasmacytic colitis and the development of crypt dysplasia that progresses to carcinoma. 10 Other New World primates, including the common marmoset (Callithrix jacchus), have rarely been reported to develop small intestinal carcinomas; however, too few cases have been analyzed to determine any specific trends. 1,2
In humans, a variety of infectious, genetic, and environmental factors can contribute to gastrointestinal tumorigenesis. Bacteria, such as Helicobacter sp, are implicated in the development of gastric and colonic carcinomas in humans and immunodeficient mice. 7,12 Helicobacter species have been recovered from the feces and gastric mucosa of common marmosets; however, no known disease association exists. 5,16,20 In the cotton-top tamarin, novel intestinal Helicobacter species have been isolated from animals with chronic colitis. 16 However, the relationship with bacterial infection, colitis, and eventual tumor formation is not known, and whether a similar scenario exists in other nonhuman primate species is purely speculative. Epstein-Barr virus, a human gamma herpesvirus most commonly associated with lymphoproliferative diseases (including lymphosarcoma), is also associated with gastric adenocarcinoma formation. 17 An Epstein-Barr-related herpesvirus (callitrichine herpesvirus 3 [CHV-3]) has been isolated from common marmosets with lymphosarcoma; however, an association with other malignancies in the common marmoset has not been determined. 3
There are broad similarities in the histologic appearance of large intestinal carcinoma from small intestinal carcinoma, and a similar molecular pathogenesis is proposed for the two sites. Commonly implicated in the pathogenesis of colorectal carcinoma is derangement and dysregulation of the Wnt pathway. 7,21 Wnts are signaling proteins that prevent β-catenin degradation in the cytoplasm, thereby allowing β-catenin to translocate to the nucleus and accumulate in a complex with various transcription factors that in turn regulate target gene transcription. 13,14 If Wnt signaling is absent, excess β-catenin is degraded within the cytoplasm so that excess transcription factor activation does not occur. 13,14 A second function of β-catenin exists outside the Wnt pathway and involves binding to the cytoplasmic domain of type I cadherins. This complex facilitates linkage to the actin cytoskeleton and allows for normal cellular structural organization. 13,14 Alterations in the conventional Wnt pathway will lead to accumulation of β-catenin and modified progression through the cell cycle. Perturbed interaction between β-catenin and type I cadherins can destabilize cell–cell interactions and promote loss of cell cohesion, an important aspect of metastatic spread. In human colorectal carcinogenesis, the primary dysregulation of the Wnt signaling pathway occurs through mutations in the adenomatous polyposis coli (APC) gene, a potent tumor suppressor. 14 Normally, the APC protein complexes with β-catenin and causes phosphorylation of β-catenin, rendering it marked for degradation. 14 Therefore, β-catenin can exert control on both nuclear transcription and cell adhesion, and it is central to the pathogenesis of gastrointestinal tumors.
Herein we characterize the spontaneous development of small intestinal carcinomas in 10 common marmosets. We use silver stains to assess the presence of Helicobacter-like bacteria; immunohistochemistry for CD3, CD20, cytokeratin, E-cadherin, and β-catenin; and polymerase chain reaction (PCR) for CHV-3 to better elucidate the pathogenesis of these tumors in the common marmoset.
Materials and Methods
Necropsy records at the New England Primate Research Center were searched for the 1991–2009 period for intestinal malignancies in common marmosets. Excluding neonates and research necropsies, 384 common marmosets were necropsied during this time. All cases recorded were localized to the small intestine. All animals were necropsied within 24 hours of death, and representative sections of all major organs were collected, fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 5 μm, and stained using hematoxylin and eosin (HE). Additional sections of affected intestine were stained via the Warthin-Starry method.
To characterize the neoplasms with immunohistochemical methods, we used standard immunoperoxidase staining for CD3, CD20, E-cadherin, cytokeratin AE1/AE3, and β-catenin. Formalin-fixed, paraffin-embedded sections were deparaffinized, rehydrated, and subsequently blocked with hydrogen peroxide. Pretreatment for all antibodies involved microwaving for 20 minutes in 0.01M citrate buffer, followed by 20 minutes of cooling. All steps were followed by a tris-buffered saline wash. Before application of primary antibodies, all slides were treated with Dako (Carpinteria, CA) protein block for 10 minutes. Sections were incubated with anti-human CD3 (Dako; polyclonal, 1:600, 30 minutes at room temperature), anti-human CD20 (Dako; monoclonal, 1:175, overnight in refrigerator), anti-human E-cadherin (Labvision, Fremont, CA; monoclonal, 1:100, overnight in refrigerator), anti-human cytokeratin AE1/AE3 (Dako; monoclonal, 1:140, overnight in refrigerator), and anti-human β-catenin (Santa Cruz Biotechnology, Santa Cruz, CA; monoclonal, 1:100, overnight in refrigerator). Slides were then incubated with secondary antibody biotinylated goat anti-rabbit (Vector Laboratories, Burlingame, CA; 1:200, 30 minutes at room temperature) for CD3 and E-cadherin and biotinylated horse anti-mouse (Vector Laboratories; 1:200, 30 minutes at room temperature) for CD20, cytokeratin AE1/AE3, and β-catenin. This was followed by a 30-minute incubation at room temperature with Vectastain ABC Elite (Vector Laboratories; CD3, E-cadherin, cytokeratin AE1/AE3, and β-catenin) or Vectastain ABC Standard (Vector Laboratories; CD20). All slides were developed with DAB chromagen (Dako) and counterstained with Mayer’s hematoxylin. In all cases, step sections were incubated with isotype-specific irrelevant antibodies for negative controls. Positive controls consisted of sections of lymph node (CD3 and CD20) and small intestine (E-cadherin, cytokeratin AE1/AE3, and β-catenin) from age-matched common marmosets.
Isolation of DNA from affected frozen small intestine was performed on case Nos. 5 and 8 and was carried out using the DNeasy Tissue Kit (Qiagen, Valencia, CA). To amplify DNA, degenerate herpesvirus primers were used to screen the sections of frozen small intestine. The degenerate primers corresponded to DFASA (5′-GTGTTCGACTTYGCNAGYYTNTAYCC-3′) and GDTD1B (5′-CGGCATGCGACAAACACGGAGTCNGTRRCNCCRTA-3′). 15 Amplifications were carried out in 25 μl of 1× GoTag Green Master Mix (Promega, San Diego, CA) with 20 pmoles of each primer and 50 ng of template DNA. The cycling condition consisted of 2 minutes denaturation at 94°C, followed by 35 cycles of 1-minute denaturation at 94°C, 1 minute of annealing at 60°C, and 1 minute of extension at 72°C, followed by a final 10-minute extension step at 72°C. An aliquot (2 to 5%) of these amplification products (DFASA–GDTD1B) was then used as a template in a subsequent nested PCR with the primer pool VYGA (5′- ACGTGCAACGCGGTGTAYGGNYTNACNGG-3′) and GDTD1B. The products of the secondary nested PCR amplification were electrophoresed in a 2% agarose gel and visualized by irradiation with ultraviolet light in the presence of ethidium bromide. The nested PCR product (236 base pairs) was cloned into pCR 2.1-TOPO vector (Invitrogen) and sequenced (Retrogen, Inc, San Diego, CA).
To compare the relative levels of viral replication in neoplastic and nonneoplastic gastrointestinal tract, a 10-fold serial dilution was made with the DNA samples, and a 306–base pair fragment was amplified with CHV-3-specific primers (F: 5′-TTGACTTCGCCAGCCTCTAT-3′ and R: 5′-TTGCAGGTGCACTTGATAGC-3′) derived from DNA sequences from the GenBank (accession No. AF319782.2). The cycling condition consisted of a 3-minute denaturation at 94°C; followed by 5 cycles of 30 seconds at 94°C, 30 seconds at 60°C, and 30 seconds at 72°C; followed with 35 cycles of 30 seconds at 94°C, 30 seconds at 55°C, and 30 seconds at 72°C; followed by a final 10-minute extension step at 72°C.
Results
Clinical and Gross Findings
Table 1 presents age and sex data, clinical findings, and gross findings for the 10 animals. Nine of 10 animals were euthanized because of intractable diarrhea (which was variably bloody) and weight loss, with the average age at euthanasia being 7.6 years (range, 3.2 to 13.4). Animals with small intestinal adenocarcinoma corresponded to 3.8% of the total nonexperimental and nonneonatal common marmosets necropsied during this period. Six animals were female and 4 were male. Clinical chemistry analysis revealed hypoalbuminemia in 4 of 10 animals. Five of 10 animals had a mildly decreased hematocrit. Fecal cultures were performed on 7 animals, with Escherichia coli (5 of 7), Klebsiella sp (3 of 7), Enterococcus sp (2 of 7), and Proteus mirabilis (1 of 7) isolated. One animal was fecal positive for Giardia by antigen capture (Triage, Biosite, San Diego, CA).
Clinical and Gross Findings for the 10 Marmosets
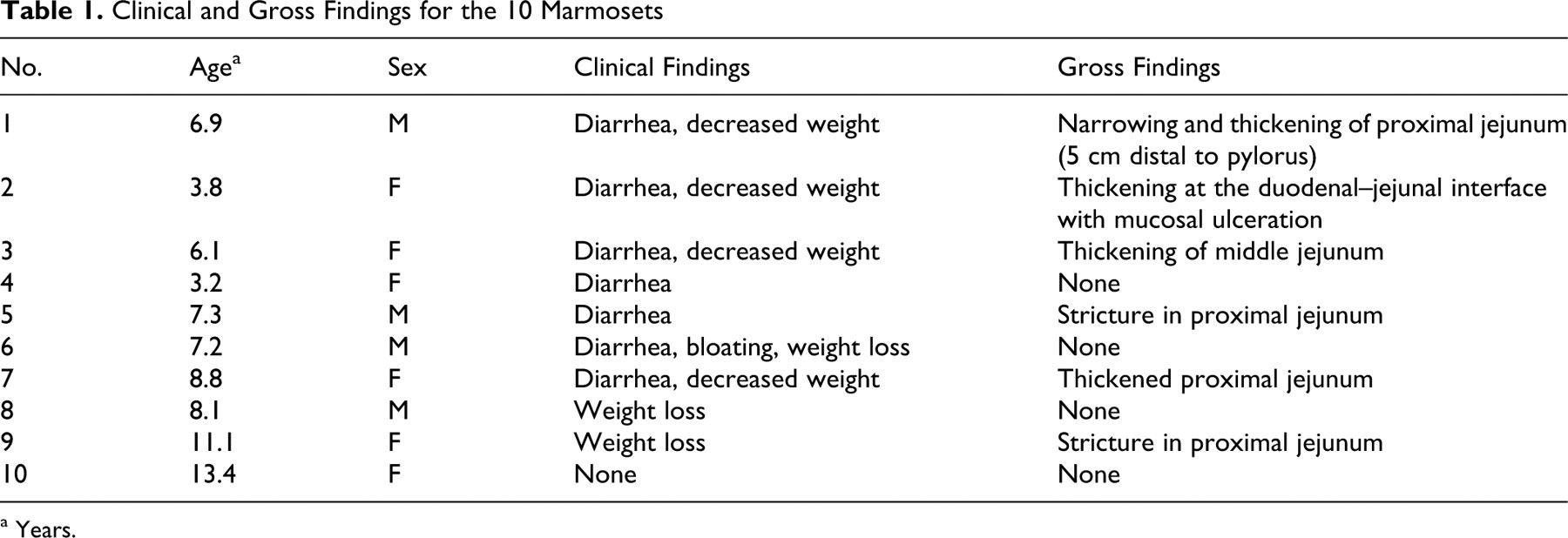
a Years.
Grossly, 5 of 10 animals were noted to have thickening of the distal duodenum and proximal jejunum, with stricture observed in 2 of 10 animals. One of 10 was noted to have thickening in the middle jejunum. Gross lesions in the small intestine were not identified in 4 of 10 animals.
Histologic Findings
In 9 of 10 cases, there was a locally extensive region of hypercellularity along the crypt to midvillus zone of the duodenum and proximal jejunum. In one of the cases (case No. 3), there was a similar region in the middle jejunum. Two of the cases (case Nos. 4 and 8) were classified as carcinoma in situ in which there was no obvious breach of the basement membrane zone but clear disorganization in the overlying crypt region (Fig. 1). Irrespective of invasiveness, all cases were characterized by loss of cell polarity and effacement of the crypts and villi by a homogeneous population of neoplastic epithelial cells that multifocally breached the basement membrane zone and infiltrated into the tunica submucosa and smooth muscle layers (Fig. 2). Neoplastic cells had moderate amounts of deeply basophilic cytoplasm and large prominent nuclei with 1 to 2 eccentrically located nucleoli. Mitotic figures were variable but averaged 0 to 1 per high power field. Roughly 50% of the neoplastic cells in all cases had large solitary intracytoplasmic clear vacuoles that marginated the nucleus (signet ring cells) (Fig. 3). In 1 case (case No. 10), the neoplastic cells were admixed with abundant mucin (Fig. 4). The neoplastic cells were embedded in a relatively scant stroma, except for 1 case (case No. 5), in which they were embedded in an abundant scirrhous response, leading to marked narrowing of the intestinal lumina (Fig. 5). In 3 affected animals (case Nos. 1, 3, and 10), the neoplasm was extensively ulcerated and covered by a thick plaque of degenerate neutrophils, sloughed epithelial cells, and hemorrhage.
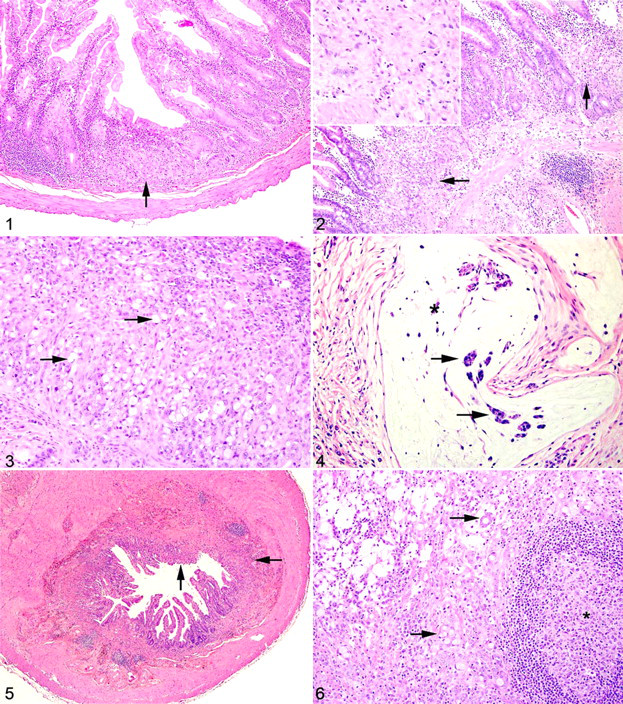
Proximal jejunum; common marmoset, case No. 4. Focally, the crypts are disorganized with loss of cell polarity (arrow). The lamina propria is filled with moderate numbers of lymphocytes and plasma cells. HE.
In 6 of 10 cases (case Nos. 1, 3, 5, 6, 9, 10), there were nests and acini of neoplastic cells within the submucosal lymphatics, and they were multifocally expanding the subcapsular sinuses of the draining mesenteric lymph nodes (Fig. 6). Case Nos. 4, 6, and 9 (3 of 10) were accompanied by a moderate enteritis characterized by large numbers of small lymphocytes and fewer plasma cells that expanded the lamina propria in both the affected and unaffected portions of the small intestine. Additional comorbid histologic findings included cholecystitis (1 of 10), mesangioproliferative glomerulonephritis (2 of 10), atherosclerosis (1 of 10), pancreaticodochitis (1 of 10), and typhlitis with attaching and effacing E coli (1 of 10).
Histochemical and Immunohistochemical Findings
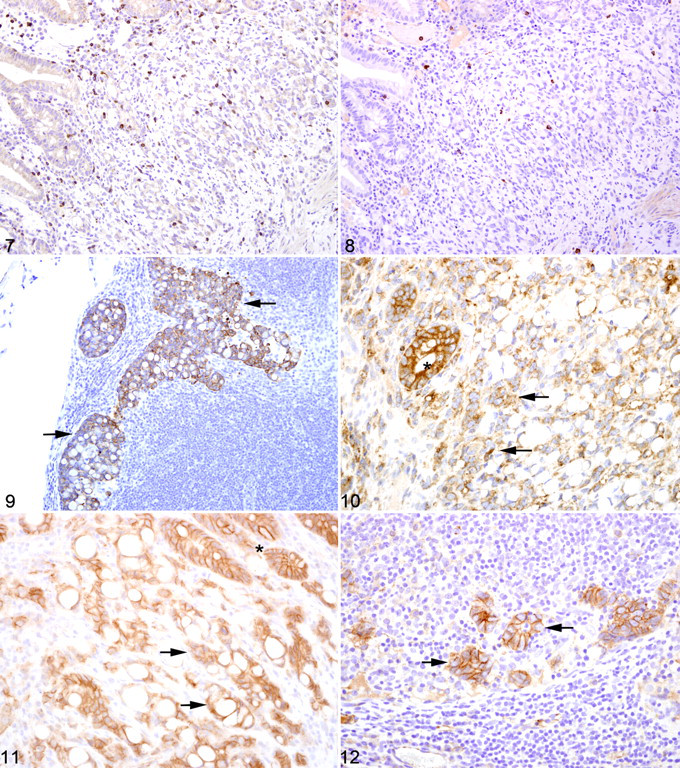
Warthin-Starry staining of all cases revealed no spiral bacteria noted within the affected and unaffected sections of small intestine. Paraffin-embedded neoplastic small intestine was not available from case No. 2 and therefore not included in the immunohistochemical studies. Nearly three-quarters (75%) of the inflammatory cells in the affected small intestine had strong membranous CD3 immunoreactivity (Fig. 7). Approximately 15% of the inflammatory cells in the affected small intestine had strong membranous CD20 immunoreactivity (Fig. 8 ), whereas roughly 10% of the inflammatory cells did not stain for either CD3 or CD20. There was no difference in the percentage of CD3- and CD20-positive cells in the affected and unaffected sections of small intestine in the 9 cases. Cytokeratin immunoreactivity was strong and diffusely cytoplasmic in the neoplastic cells and highlighted the invasiveness and lymph node metastasis of the neoplastic epithelial cells (Fig. 9). E-cadherin immunoreactivity was detected in 7 of 9 cases examined. There was no E-cadherin immunoreactivity in neoplastic and nonneoplastic tissue from case Nos. 3 and 4. E-cadherin immunoreactivity was predominately and strongly associated with the cell membrane and, to a lesser degree, the cytoplasm in the normal sections of small intestine. In the affected sections as well as the affected lymph nodes, neoplastic cells had scant to absent membrane staining and increased intracytoplasmic, often punctuate to granular, staining (Fig. 10). Aggregates of neoplastic cells (ie, pseudoacini) often retained strong membrane staining and only scattered intracytoplasmic immunoreactivity. β-Catenin immunoreactivity was detected in 8 of 9 cases examined. No immunoreactivity to β-catenin was recorded in case No. 4. Normal small intestine had uniform strong cell membrane staining with faint cytoplasmic staining. In the neoplastic cells, there was decreased cell membrane staining with increased cytoplasmic immunoreactivity and minimal nuclear reactivity (Fig. 11 ). Neoplastic cells that formed pseudoacini often retained strong cytoplasmic membrane staining (Fig. 12 ).
Sequencing Analysis
DNA isolated from paraffin-embedded and fresh-frozen neoplastic small intestine and unaffected large intestine from case Nos. 5 and 8 were screened for the presence of CHV-3 DNA using nested PCR. Positive results were obtained from the neoplastic small intestine and unaffected large intestine in both cases. Sequence analysis of amplicons generated with the nested broad-based herpesvirus primers indicated alignment with CHV-3 and revealed 94% identity with CHV-3 sequences previously reported. The PCR product was distinct from Epstein-Barr virus and other Old World primate lymphocryptoviruses. After serial dilution of DNA isolated from the two regions of interest, there was no difference in the ability to detect CHV-3 between neoplastic small intestine and large intestine from both animals tested, indicating that viral copy number was equivalent in neoplastic and nonneoplastic tissue.
Discussion
In primate species, carcinomas confined to the small intestine are exceedingly rare and have been reported in 2 rhesus macaques (M mulatta) and 4 common marmosets. 1,2,6,11 In those previously reported, the location of tumor development varied and included the proximal small intestine (n = 2), the mid-small intestine (n = 1), and the ileum (n = 1). 1,2 Herein we report the clinical, histologic, immunohistochemical, and molecular characteristics of 10 cases of small intestinal adenocarcinoma in common marmosets (C jacchus). Interestingly, 9 of the 10 animals reported herein developed small intestine adenocarcinoma in the proximal jejunum, immediately distal to the duodenum, certainly suggesting an increased propensity for tumors to develop at this site in the common marmoset. There were no consistent blood work findings in animals with tumors, and fecal cultures, when performed, revealed a mixed population of common intestinal bacteria. Six of the cases had gross lesions of thickening and structure at the site of the neoplasm. Four were considered incidental findings on histologic examination; however, of these 4, only 2 were categorized as carcinoma in situ, whereas the other 2 had spread extensively into the muscle layers. Many of the cases had only moderate invasion into the underlying submucosa and muscle layers, and carcinomatosis was not present in any of the studied animals; however, most cases had marked lymphatic invasion and metastasis to mesenteric lymph nodes. This indicates that although these tumors are small and confined to a specific region, metastatic spread is a common and likely an early event. Signet ring cell differentiation was typical in all cases; however, only 1 case had large areas of mucin deposition. Signet ring cells are more common in cases of gastric and colonic carcinoma; however, in one large human study, 37% of the total cases of small intestinal adenocarcinoma were classified as signet ring carcinoma. 4 Currently in human medicine, there is no prognostic significance to the presence of signet ring cells in cases of small intestinal carcinoma, and where a similar scenario exists in the common marmoset is not known. 4 Mucin deposition has been described in cases of small intestinal carcinoma, although it is more commonly observed in large intestinal carcinomas. 1
Although the small intestine has a similar epithelial turnover and proliferative rate as the large intestine, epithelial tumors of the former are rare compared to their colonic counterpart. The reason for this is not known; however, various hypotheses have been put forth: First, digesta in the small intestine has a faster transit time when compared to that of the large intestine, thereby decreasing exposure to preformed and metabolized toxins; second, digesta is more liquid than that in the large intestine, leading to less mucosal irritation; third, the small intestine has a lower total bacterial count than does the colon; fourth, there are higher levels of immunoglobulin A in the small intestine, in addition to more abundant mucosal-associated lymphoid tissue, which provides a cytoprotective effect through immune surveillance; and, finally, stem cells that line the crypts are farther away from the lumen (ie, versus their counterparts in the colon) and therefore exposed to fewer luminal carcinogens. 7,8 For humans, many of these proposed hypotheses are thought to play a role, but the true pathogenesis of spontaneous small intestinal adenocarcinoma remains poorly defined, although chronic inflammation, bacterial and viral infection, and genetic mutations all likely play a role in promoting tumor development.
Chronic inflammation progressing to intestinal carcinoma is well described for humans and cotton-top tamarins (S oedipus). In the cotton-top tamarin, chronic lymphoplasmacytic colitis is a common cause of wasting in middle to advanced age. A large proportion of these animals with chronic colitis progress through dysplasia and eventual carcinoma. 10 This pathogenesis mimics human ulcerative colitis–carcinoma progression. In 3 of 10 cases presented herein, the tumor was accompanied by a moderate lymphoplasmacytic enteritis. The majority of lymphocytes were T cells with only a small percentage of B cells. There was no difference in the percentage of inflammatory cells in the neoplastic sections of small intestine and the nonneoplastic regions. It is unlikely that the chronic inflammation induced the tumor formation in these 3 animals; it is likely, however, that the inflammation contributed to an altered local environment that potentiated tumor growth.
In humans, Epstein-Barr virus has been associated with the genesis of gastric carcinoma; however, there has been no association made between Epstein-Barr virus infection and small intestine adenocarcinoma. 17,19 We analyzed tissue from 3 cases reported herein for the presence of CHV-3, a lymphocryptovirus related to Epstein-Barr virus that has been recovered from common marmosets with lymphosarcoma. 3 All 3 cases had CHV-3 DNA in both the neoplastic small intestine and the normal colon. Even after titrated PCR, the levels of virus remained the same in both locations, indicating that although these animals had been exposed to and infected with CHV-3, it likely plays no role in the pathogenesis of small intestinal carcinomas in this species. Helicobacter spp have been associated with chronic enteritis, intestinal metaplasia, and gastric carcinoma in a variety of species, including humans and immunodeficient mice. 7,12 Helicobacter spp have been recovered from the feces and gastric mucosa of marmosets, although no disease association has been made. 5,20 No spiral bacteria were detected in the cases presented herein, and they are unlikely to play a role in the pathogenesis.
The molecular basis of small intestinal adenocarcinomas in humans is not as well characterized as the more prevalent colorectal adenocarcinoma; however, many similarities exist between carcinomas at the two sites. In the colorectal region, progression of adenomas to carcinomas involves the deregulation of both oncogenes and tumor suppressor genes. These include APC genes, P53, K-ras, and a variety of DNA mismatch repair genes (including hMSH2 and PMS2). 8,9,14 Also of importance in the pathogenesis of colorectal carcinomas is dysregulation of Wnt signaling. The most commonly observed aberrant Wnt signaling is via altered β-catenin function. β-Catenin serves a dual-functioning protein that acts as a transcription factor activator as well as a link between membranous proteins (eg, cadherins) and the cytoskeleton. 13 Altered levels of β-catenin can lead to a shift in balance either toward increased nuclear gene expression or altered cell adhesion. In the current study, several cases had no immunoreactivity to E-cadherin and β-catenin, which was interpreted as being secondary to altered fixation times rather than true loss of expression. In those cases that did display E-cadherin and β-catenin expression, there was decreased cell membrane staining and increased cytoplasmic staining in the tumor cells, compared to the normal small intestinal epithelium. This cell membrane staining was preserved in cohesive populations of cells. Although this does not prove that derangement in either protein is what triggers the initiation of small intestinal adenocarcinoma development in common marmosets, the decreased expression suggests that loss of cell–cell adhesion is an early change in the pathogenesis of these tumors.
In conclusion, we report 10 cases of small intestinal adenocarcinoma in common marmosets (C jacchus). These tumors were almost entirely located within the proximal small intestine, immediately distal to the duodenum. Signet ring cell differentiation, lymphatic invasion, and metastatic spread to regional lymph nodes were present in the majority of cases. There was no association between expression of callitrichine herpesvirus 3 and tumor development. Similarly, there was no association between the presence of inflammation or Helicobacter in regard to tumor development. Immunohistochemistry for E-cadherin and β-catenin indicates decreased cell membrane immunoreactivity for both of these proteins with a concomitant increase in cytoplasmic staining, suggesting an important role for these proteins in tumorigenesis.
Footnotes
Acknowledgements
We wish to thank Kristen Toohey for assistance with photography and Liz Curran for tissue procurement.
Declaration of Conflict of Interest
The authors declared that they had no conflicts of interests with respect to their authorship or the publication of this article.
Financial Disclosure/Funding
This research was funded, in part, by National Institutes of Health grants RR00168 and RR07000.
