Abstract
A 10-year-old, female, pot-bellied pig (Sus scrofa) experienced a 3-month history of reduced appetite, dysphagia, and weight loss. Clinical examination revealed a mass in the left part of the oral cavity extending from the hard to the soft palate. At necropsy, a firm, white, poorly demarcated ulcerated mass at the left hard and soft palate with metastases to the left retropharyngeal lymph node and the lung was observed. Additional findings included a uterine adenocarcinoma, a hepatocellular adenoma, and nodular hyperplasias in spleen and adrenal glands. Histologically, the poorly demarcated, infiltrative growing oral mass consisted of islands, cords, and single epithelial cells with moderate squamous differentiation. Cells were strongly positive for cytokeratin by immunohistochemistry. Similar cells were found in the left retropharyngeal lymph node and the lung. The present findings represent the first report of a metastasizing oral squamous cell carcinoma in a pig.
Keywords
Neoplasms in pigs have been reported only infrequently and include uterine leiomyoma and fibroma, malignant lymphoma, melanoma, hemangiosarcoma, hepatocholangioadenoma, hepatocellular carcinoma, and osteoma in decreasing frequency. 1, 4, 10, 11 Squamous cell carcinoma represents a common neoplasm in some domestic species including dogs and cats. In addition, it has been described in cows and horses but not in pigs. 6, 7, 12 This tumor entity can be observed in various locations including eyelid, conjunctiva, pinna, oral cavity, vulva, stomach, and skin. 12 However, there are specific preferential locations for each species. 12 Although the cause triggering development of squamous cell carcinomas remains undetermined in most cases, chronic physical irritation (i.e., chronic epidermal injury), UV light, and virus infection have been shown to play a causative role in most cases. 12 In addition, carcinogens contained in tobacco, coal tar, and soot as well as arsenic and smegma have been shown experimentally or by epidemiologic data analysis to play an important role as promotor. 12 Uterine adenocarcinoma, a common finding in rabbit and cattle, has been described only rarely in other domestic animals including the sow. 8 Metastases can be found frequently in regional lymph nodes and lung. 8
A 10-year-old pot-bellied pig (Sus scrofa) showed a 3-month history of weight loss and dysphagia. Clinical examination revealed an ulcerated, infiltrative growing mass in the hard and soft palate of the left oral cavity. Owing to poor prognosis, the animal was killed and submitted for necropsy. Tissue samples from various organs including the mass of the oral cavity and the left retropharyngeal lymph node were collected at necropsy, fixed in 10% buffered formalin, and embedded in paraffin wax. 9 Tissue sections were stained with hematoxylin and eosin (HE) and used for immunohistochemistry by applying the ABC-method using primary antibodies directed against cytokeratin (DakoCytomation, Glostrup, Denmark), and vimentin (DakoCytomation, Glostrup, Denmark). 9 In brief, sections were dewaxed and antigens were demasked by Pronase E and Tween treatment (cytokeratin; 20 minutes), Triton treatment (vimentin; 0,25%, 30 minutes) as described. 9
Gross pathology revealed a multifocal, firm, white, ulcerated, poorly demarcated mass measuring 15.0 × 7.2 × 2.4 cm in size, in the left side of the oral cavity in the hard and soft palate (Fig. 1). The neoplasm reached from the ethmoid bone and the left internal pterygoid muscle to the rostral palate with subpalatinal extension to the canines. Furthermore, a diffuse infiltration into the left cheek was noticed. All premolars and molars of the left side and the third molar of the right side were missing. An abscess at the ethmoid bone, adjacent to the neoplasm was detected. In addition, a tumor mass, measuring 8.0 × 4.0 × 4.0 cm, was found in the region of the left lateral retropharyngeal lymph node. The lung was slightly increased in size, firm, and reddish with prominent interlobular septae.
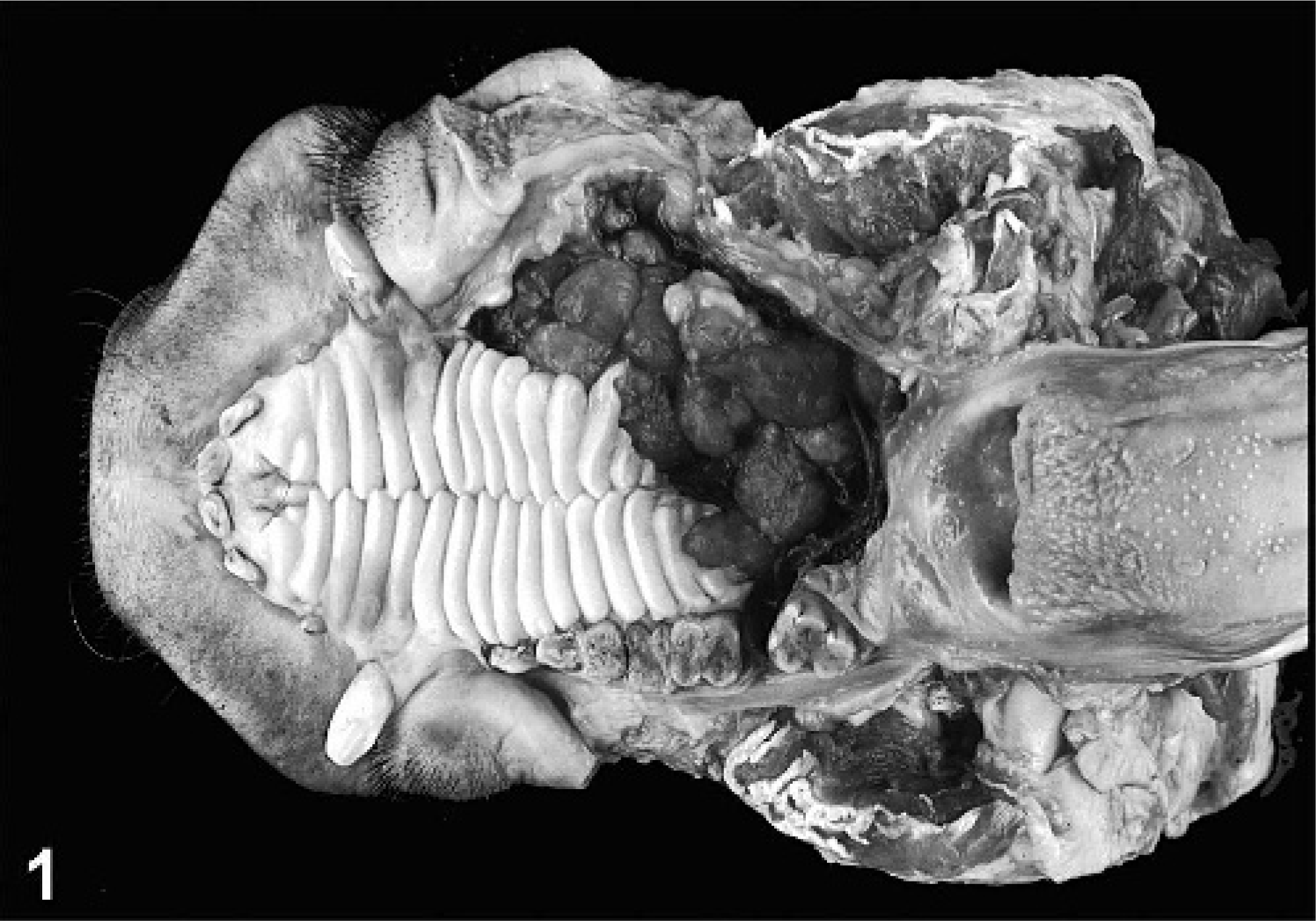
Head, porcine oral squamous cell carcinoma. Poorly demarcated, ulcerated infiltrative growing tumor in the hard and soft palate of left oral cavity.
An exophytic uterine mass, 8.0 × 1.5 × 1.5 cm in diameter, with subtotal obstruction of the lumen was found at the uterine bifurcation. The largest part of the tumor was localized in the left uterine horn. Both uterine horns showed, in addition, a mild mucometra. The liver exhibited a focal, nodular neoplasm measuring 0.4 × 0.9 × 0.5 cm in size. Multifocal nodular hyperplasias were detected in spleen and adrenal glands, measuring 1.0 cm and 0.2 to 0.3 cm in diameter, respectively. In addition, moderate bilateral nephrolithiasis, moderate bilateral endocardiosis, and one gastric trichobezoar, 8.0 × 4.0 × 3.0 cm in size, were observed.
Histologic examination of the oral cavity revealed a densely cellular, poorly demarcated, unencapsulated mass consisting of a moderate fibro-vascular stroma and numerous neoplastic epithelial cells arranged in sparsely-packed islands, cords, or as single cells, infiltrating into the submucosa. The tumor cells, round to oval, sometimes polygonal, between 10 × 20 µm and 15 × 30 µm in size, with distinct cell borders, exhibited a moderate amount of a pale eosinophilic cytoplasm and possessed a centrally located round to oval nucleus (Fig. 2). Tumor cells were further characterized by a finely stippled chromatin pattern, two to five nucleoli and one to two mitoses per high power field (HPF). Prominent squamous cell differentiation was found in a few locations. A few bizarre mitotic figures were also found. Additional findings included loss of preexisting bone tissue as a result of tumor-cell infiltration, crater-like ulcerations with secondary superficial bacterial infection, multifocal areas of necrosis, and a moderate, diffuse, lympho-histiocytic, neutrophilic, focal granulomatous inflammation with few multinucleated giant cells. In the left retropharyngeal lymph node and most frequently in the interlobular pulmonary septae, tumor-cell infiltration, similar to the one described in the oral cavity, was associated with lymphocytes and plasma cells (Fig. 3).
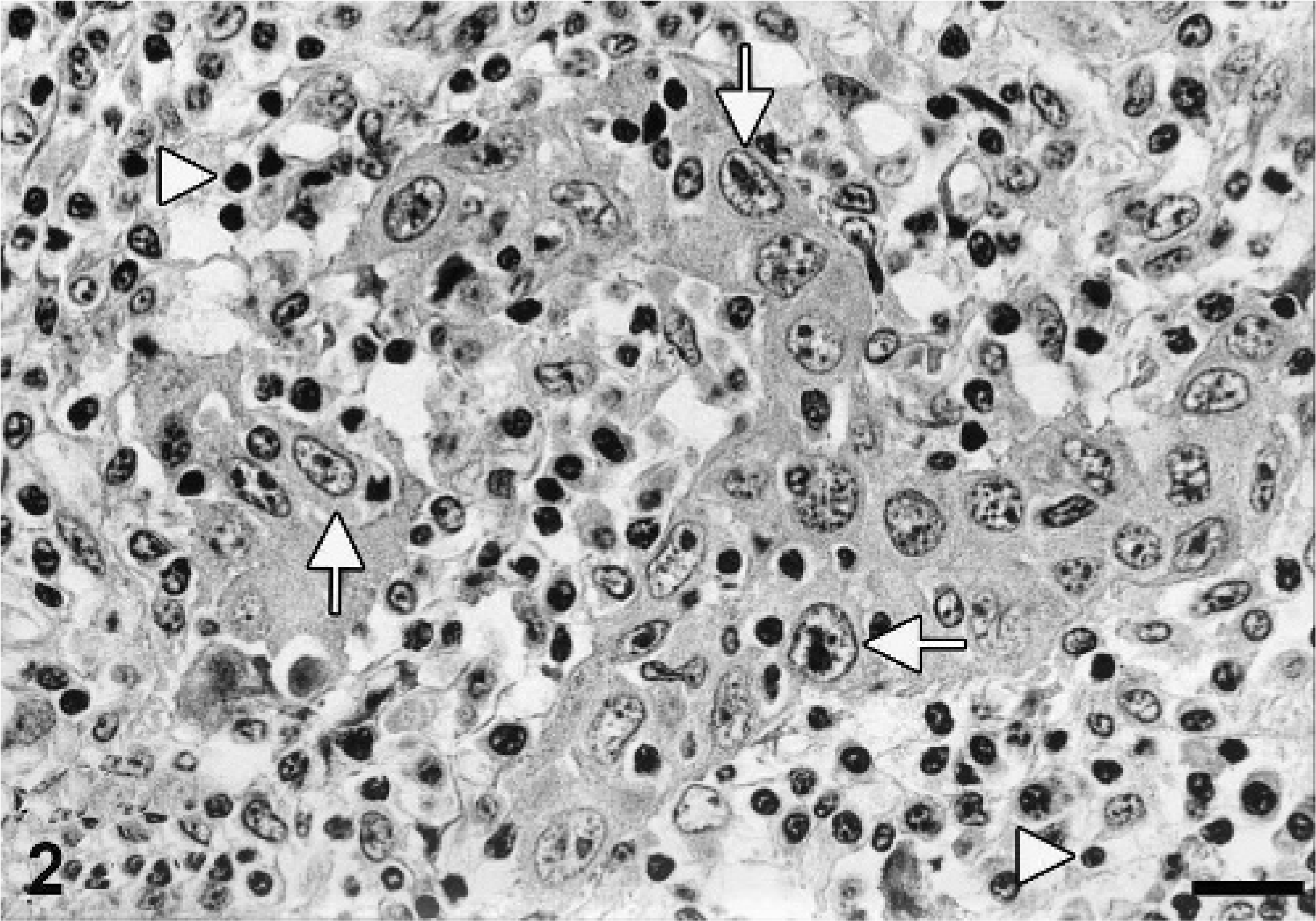
Oral cavity, porcine oral squamous cell carcinoma. Tumor cells (arrows) with distinct cell borders; finely stippled chromatin pattern embedded in a severe lymphocytic, plasmacytic, and histiocytic inflammation (arrowheads). HE. Bar = 18.5 µm.
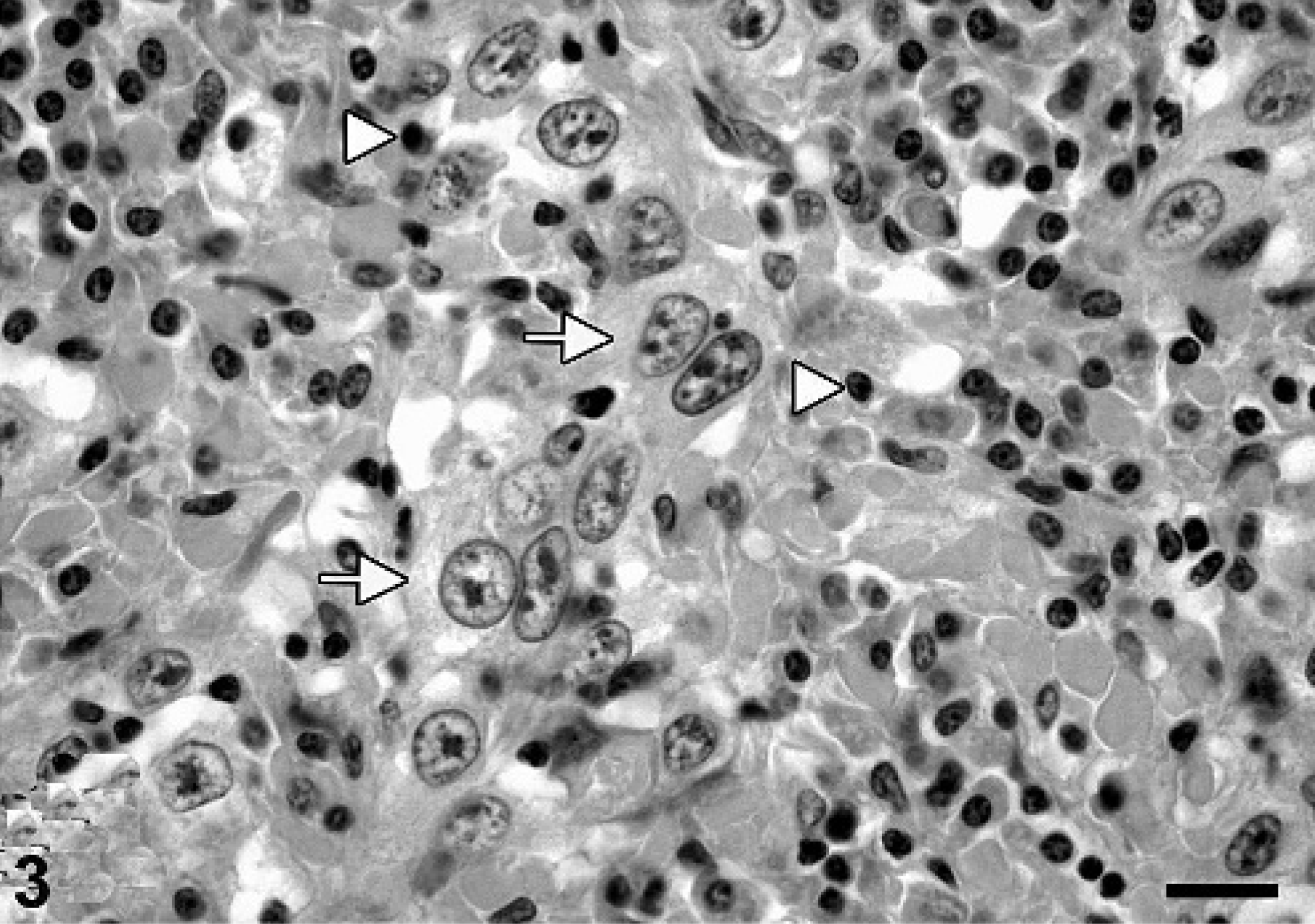
Lung, porcine metastasizing oral squamous cell carcinoma. Metastatic tumor cells (arrows) and associated inflammatory infiltration (arrowheads) consisting of lymphocytes and plasma cells in the interlobular septae. HE. Bar = 29 µm.
The poorly demarcated uterine mass was localized predominantly in the endometrium. This tumor consisted of densely packed cells displaying a nodular, papillary, tubular, and adenoid growth pattern with focal squamous cell metaplasia (Figs. 4, 5). The columnar tumor cells, 10 × 15 µm in size, consisted of poorly defined cell borders and a round to oval, basally localized nucleus with finely stippled chromatin and an abundant, eosinophilic, granular cytoplasm. Nuclei contained one to two basophilic nucleoli and the mitotic activity was approximately one mitosis per HPF. In addition, scant amounts of a fibro-vascular stroma were noticed. The well-demarcated hepatic mass consisted of hepatocyte-like cells and lacked the lobular organ-specific micro-architecture including central veins and bile ducts. Cells showed a nodular, expansile growth and were encapsulated by a thin rim consisting of fibroblasts and collagen. The hepatocyte-like tumor cells were round to polygonal, measuring 15 to 20 µm in diameter; consisted of a moderate amount of an eosinophilic, cloudy cytoplasm; and displayed well-defined cell borders. Nuclei were round, centrally located, and showed a finely stippled chromatin. Each cell contained one prominent nucleolus, and the mitotic activity was less than one mitotic figure per HPF. Single cell necrosis was infrequently observed. Additional findings included mild, multifocal, lympho-histiocytic inflammation, and compression atrophy of the adjacent hepatic tissue.
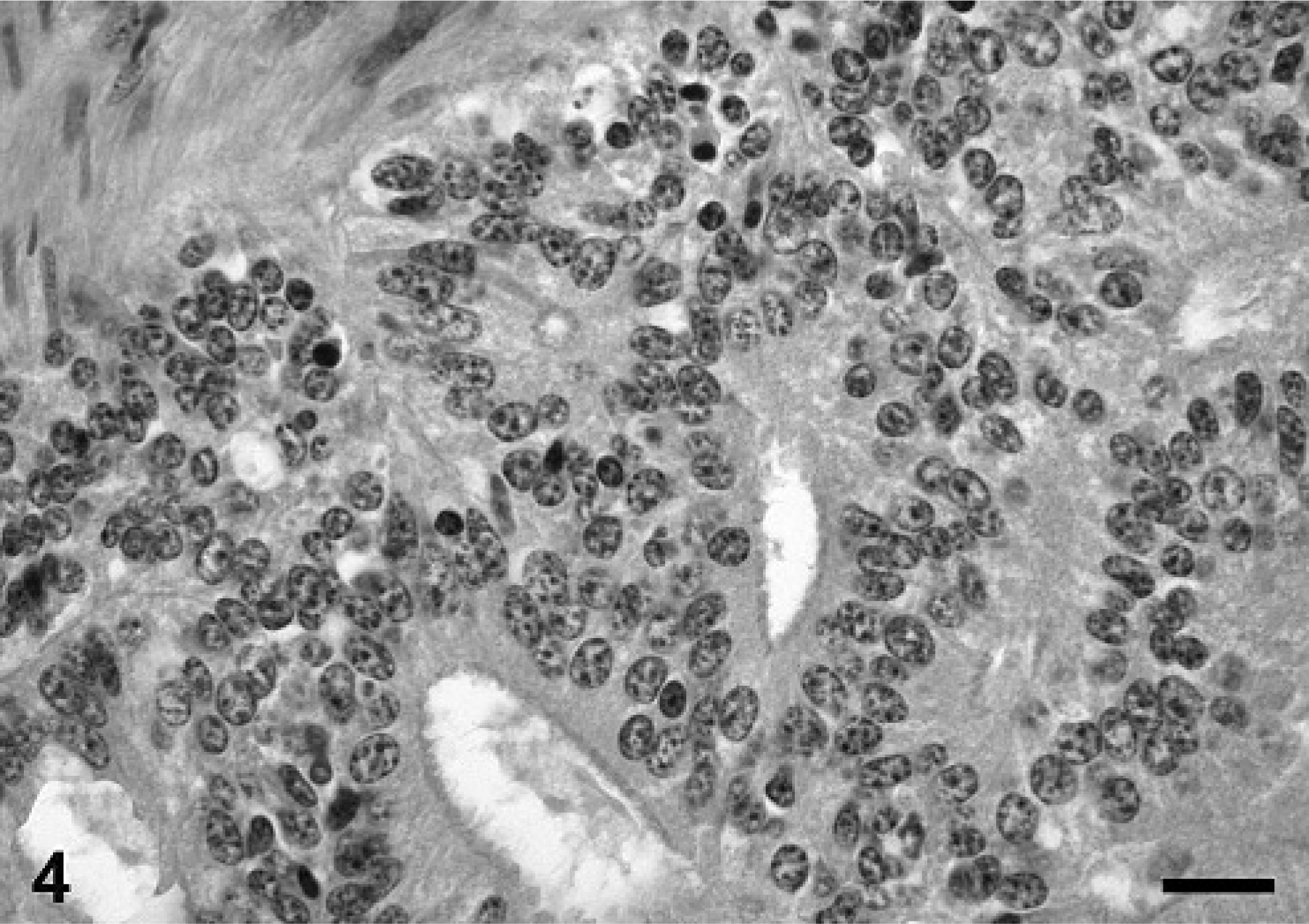
Uterus, porcine uterine adenocarcinoma. Tumor cells displaying nodular, papillary, tubular, and adenoid growth pattern. HE. Bar = 58 µm.
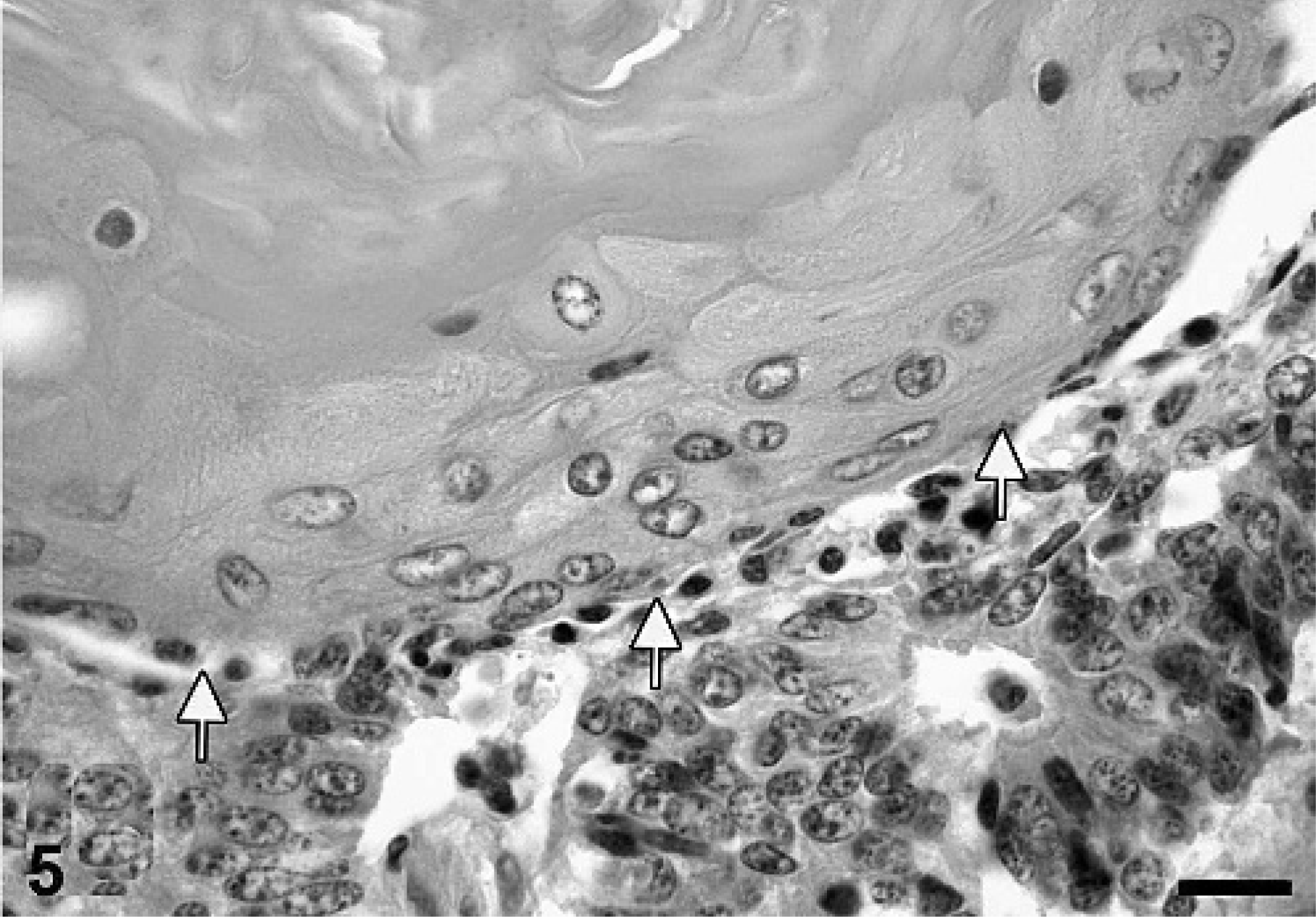
Uterus, porcine uterine adenocarcinoma. Focal squamous metaplasia (arrows). HE. Bar = 29 µm.
Immunohistochemistry revealed a positive cytokeratin reaction of tumor cells in the oral cavity, lymph node, and lung (Fig. 6). The number of positive cells varied in the oral cavity and the metastases from 30 to 50%. A similar positive immunoreactivity was observed in the uterine adenocarcinoma. All tumor cells were negative for vimentin. Squamous cell carcinoma is a relatively common neoplasm of most domestic species including cat, dog, horse, and cattle 12 ; however, it has not been described in the pig so far. Squamous cell carcinomas of the oral cavity are the most or second most common malignant tumors in cats and dogs, respectively. 7 Sites of tumor origin include tongue, gingiva, and tonsil. 7 In general, aged animals have a higher risk to develop squamous cell carcinomas than do younger ones. The reported mean age for feline and canine oral squamous cell carcinomas ranges from 11.6 to 12.5 and 8 to 10 years, respectively. 7
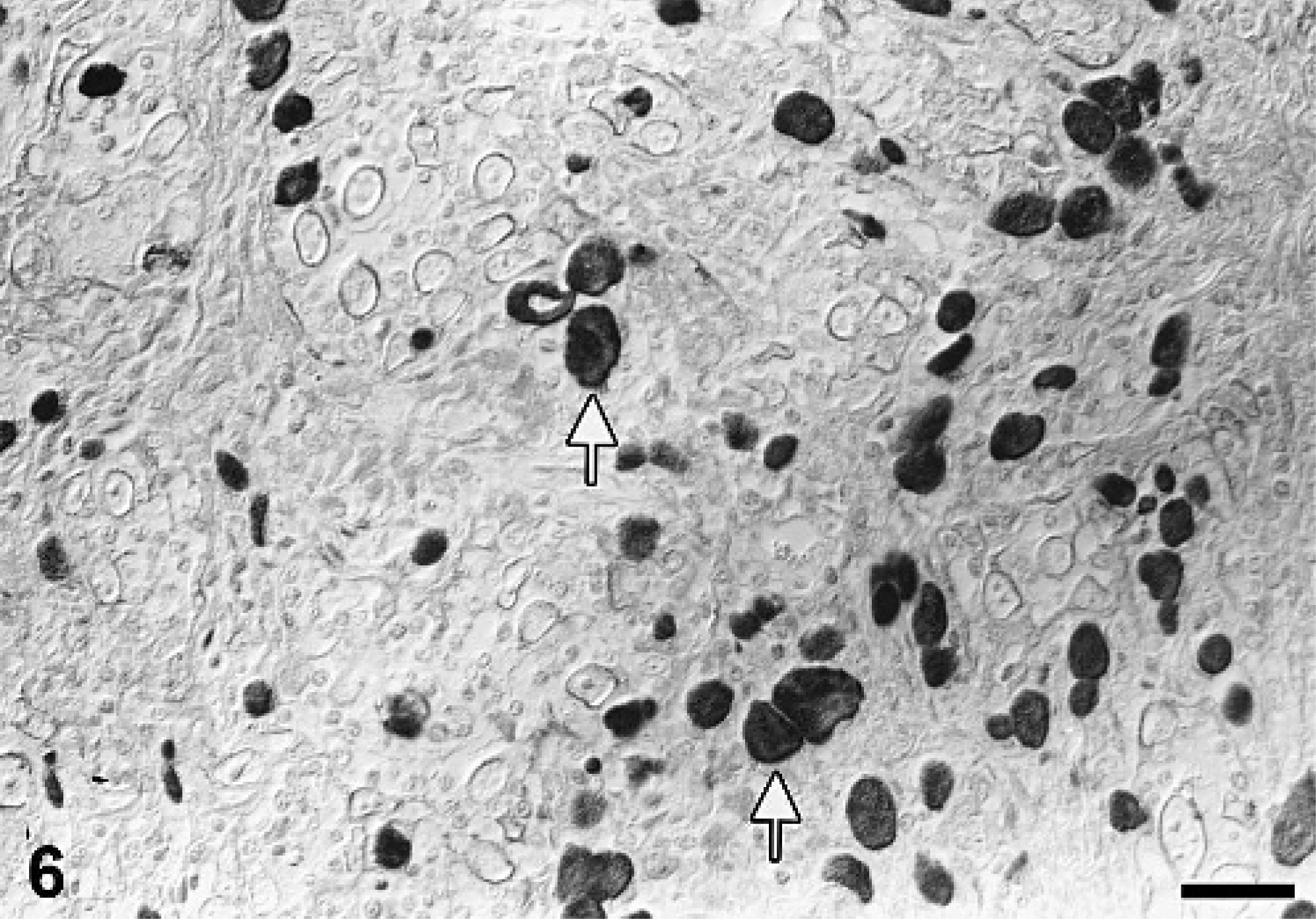
Oral cavity, porcine oral squamous cell carcinoma. Cytokeratin positive tumor cells (arrow). Cytokeratin-specific antibody, ABC method, slightly counterstained with hematoxylin. Bar = 29 µm.
Squamous cell carcinomas grow locally invasive and cause severe destruction of the adjacent tissue including periodontal structures, and secondary tooth loss similar to the present case. 7 Owing to the advanced lesions and severe secondary changes, the exact site of origin of the presented tumor remains undetermined. The primary site could have been the gingiva or palate with local invasion, which spread into the ipsilateral retropharyngeal lymph node; however, a tonsillar origin cannot be excluded completely.
Squamous cell carcinoma may also have an extension to the nasal cavity, and ulcerations are frequently complicated by secondary bacterial infections similar to the present case. 7 Lymphatic metastasis of feline and canine oral squamous cell carcinomas may occur by massaging tumor cells into vessel by the tongue. 7 Only 5–10% of canine gingival squamous cell carcinomas metastasize into regional lymph nodes and only 3% to distant sites. 7 In contrast, tonsillar squamous cell carcinoma of the dog metastases frequently and very early in the course of the disease into the mandibular lymph node. Ninety-eight percent spread to the regional lymph nodes, and 63% show distant metastases in lung, liver, and spleen. 7 In this 10-year-old, female, pot-bellied pig, spatial distribution of the primary tumor and metastases, ulcerations, and extension into ethmoidal structures are reminiscent of metastasizing oral squamous cell carcinoma of tonsillar origin in cats and dogs. The cause of the present case remains undetermined.
Though genital tract tumors including uterine leiomyomas and fibromas are frequently noted in aging pigs, endometrial adenocarcinomas have been described only rarely. 1, 5, 11 The latter is a rare tumor in many domestic animals except for cattle and rabbits. 8 The incidence of endometrial adenocarcinomas is more frequent in older animals compared with younger. 8 Squamous cell metaplasia as observed in the uterine adenocarcinoma of the present pot-bellied pig has also been described in humans. 2 Unfortunately, immunohistochemical results did not allow to distinguish between a carcinoma of endometrial or oral cavity origin, therefore a metastasis of the squamous cell carcinoma within the uterine adenocarcinoma cannot be ruled out completely.
Hepatocellular adenomas are described in dogs, cattle, sheep, cats, and pigs. 3 The occurrence of benign hepatocellular neoplasms is less frequently reported than the malignant counterpart. 3 Some authors reported an increased incidence of hepatocellular adenoma in younger pigs in contrast to other species. 3 In the present pot-bellied pig, the adenoma was differentiated from nodular hyperplasia and carcinoma by lack of infiltrative growth and anisokaryosis as well as the presence of a capsule. In conclusion, this report describes for the first time a metastasizing oral squamous cell carcinoma in a 10-year-old pot-bellied pig.
Footnotes
Acknowledgements
We would like to thank Mrs. P. Grünig for excellent technical assistance and Mrs. B. Behrens for photographic support.
