Abstract
Objective
Given the implications of concurrent human papilloma viral infection (HPV) in the prognostic course and implications on therapeutic approached of patients with oral squamous cell carcinoma (OSCC), we seek to investigate the implications that P16 expression has on the clinical course and pathological appearance of patients with OSCC and concurrent infection.
Methods
Using S-P immunohistochemistry, we examined the expression of P16 and Ki67 in 460 patients with OSCC. We compared the expression of the protein between the tumor cells and normal epithelial mucosa within the same patient. The clinical and pathological characteristics (including gender, age, histological grade, lymph node metastasis, clinical stage, clinical recurrence, tumor diameter, Ki67 proliferation index) were analyzed by stratification statistically.
Results
In total 460 cases of OSCC were identified and expression of P16 was significantly higher in the OSCC group compared to the normal mucosal epithelial group (X2 = 60.545, p = .000). There also appear to be a gender predilection as the expression was higher in females compared to males (0.218 vs. 0.144, X2 = 3.921, p = .048). Younger age also appears to be a predictive factor as those under 35 years old had higher expression of the protein compared to those over 35 years old (0.294 vs. 0.157, X2 = 4.230, p = .040). P16 positivity showed a significant positive correlation with histologic grade (X2 = 4.114, p = .043). In addition, the positive rate of P16 was higher in patients with ki67 over 85% (0.455 vs. 0.160, X2 = 6.667, p = .023).
Conclusion
OSCC with HPV infection tends to occur more frequently in female patients and those under 35 years of age. HPV infection with expression of the P16 and ki67 protein may promote the proliferation and growth of OSCC at a higher frequency.
Introduction
Oral squamous cell carcinoma (OSCC) is one of the most common malignant tumors in oral and maxillofacial regions. There is a high incidence and the tumor has a tendency for early extensive lymph node metastasis. 1 Every year, there are about 300,000 new cases of oral cancers reported worldwide, and SCC accounts for about 80%–90% of those reported.2,3 Its carcinogenesis is dependent on a multifactorial process involving environment factors, viral infection and genetic alterations. 4 Clinically, OSCC predominantly affects men aged between 50 and 60 years, and is typically rare in patients under 40 years. However, in recent decades we have seen an increase in the incidence OSCC in younger patients. This can be partly explained by smoking and alcohol use, which have long been implicated as risk factors for OSCC, but 15%–20% of patients still develop OSCC without exposure to these risk factors. The role of viral infections in the carcinogenesis of OSCC is a possibility.5,6 Numerous studies have shown that various viruses are participating in the development and progression of OSCC, including human papillomavirus(HPV), 7 Epstein-Barrvirus (EBV), Herpes simplexvirus 1 (HSV-1), human cytomegalovirus(HCMV), human Herpesvirus-6 (HHV-6), hepatitis C virus(HCV), etc. Among these viruses, approximately 25% to 30% OSCCs have been associated with HPV. 8 HPV exhibits specific tropism for the squamous epithelium, which could result in dysregulation of cell proliferation, apoptosis and genetic stability. 9 Understanding the molecular mechanisms underlying HPV infection and its effects on tumorigenesis is crucial for developing effective prevention strategies and therapeutic interventions.
It is well known that p16 has good expression consistency with HPV infection. 8 In this study, by detecting the expression of P16 protein in OSCC samples, the role of HPV in the occurrence and development of OSCC was explored, and the correlation between HPV and clinicopathological features of OSCC was analyzed, which will hopefully provide evidence for the further targeted therapy for OSCC.
Methods
Case sources
All specimens were collected from 460 patients with OSCC who were surgically treated from the Sun Yat-Sen Memorial Hospital of Sun Yat-Sen University from October 2015 to November 2020. Patient’s age ranged from 19 to 94 years old (mean 57.18 years old). The patients were then stratified into two groups: < 35 years old and >=35 years old. Among those 460 patients, 405 cases of OSCC tissues and comparable normal adjacent mucosa, defined as more than 1 cm away from the cancer tissue, were selected for the study. All tumor tissue specimens were confirmed as SCC by pathologists according to diagnostic criteria of the World Health Organization. 1 Smokers, patients with lichen planus, patients with inconclusive biopsy results, and those with hemorrhagic and/or necrotic areas in their specimens were excluded. The adjacent tissues were normal mucosal or inflammatory lesions. In 367 patients this was their first OSCC, and in 93 this was a recurrence after local surgical resection. None of the 367 primary patients had received radiation or chemotherapy prior to surgery. The specific clinical and pathological characteristics of the selected cases are shown in Table 2. This study was approved by the Ethics Committee of the Sun Yat-Sen Memorial Hospital of the Sun Yat-Sen University (SYSKY-2022-018-01).
Main reagents and specimen treatment
The used immunohistochemical detection kit and mouse anti-human monoclonal antibodies P16 and Ki67 were purchased from Beijing Zhongshan Jinqiao Biotechnology Co. LTD. The working concentration of P16 was 1:1000 and that of Ki67 was 1:100. DAB chromogenic liquid was purchased from Fuzhou Maixin Biotechnology Company. All specimens were fixed with 10% percent neutral formalin and embedded in conventional paraffins. Gel coated white tablets with a thickness of 4 μm were cut for immunohistochemical detection.
Immunohistochemical detection and result determination of Ki67 and P16
Immunohistochemical staining was carried out according to the operation steps of SP kit, followed by DAB coloration. The brown termination reaction of positive cells was controlled by microscopic observation. Positive control (tonsil tissue) and blank control were set up for observation under light microscope, and photographs were taken for counting. In samples with P16 positivity, there was robust and uniform nuclear staining in at least 50% of normal mucosal or tumor cells. For samples with Ki67 positivity, they were categorized into two groups based on the number of positive cells: low expression (less than 85% of tumor cells) and high expression (at least 85% of tumor cells).
Statistical analysis
The 460 cases of OSCC were grouped according to gender, age group, tumor size, histological grade, number of lymph node metastases, clinical stage (2017 AJCC 8th Edition), and recurrence when enrolled. The SPSS 16.0 statistical software was used for analysis of the data. The Ki67 expression rate was expressed as the mean ± standard deviation (mean ± SD). The measurements were compared between normal mucosa and OSCC, and the data were tested by an independent sample T test consistent with a normal distribution and homogeneous variance. If the above conditions are not satisfied, Chi-squared test or exact probability method was used. The nonparametric rank summation test, Chi-squared test, or Fisher exact test were used to evaluate the correlation between clinicopathological parameters and P16 or Ki67 IHC scores in OSCC. In all cases, probability values less than 0.05 were taken to indicate a significant difference.
Results
Comparison of P16 protein expression in oral squamous cell carcinoma and normal tissues
Immunohistochemical results showed that P16 protein was mainly expressed in the nucleus (Figure 1). The expression rates of P16 protein in squamous cell carcinoma tissues were significantly higher than those expressed in the normal mucosal epithelial tissue (X2 = 60.545 analyzed via Fisher’s exact p = .000), and the difference was statistically significant, as shown in Table 1. Expression of P16 in oral squamous cell carcinoma and normal mucosal squamous epithelium: Morphological morphology of oral squamous cell carcinoma (A1-3) and expression of P16(B1-3); HE staining (C1) and P16 expression (C2) of normal squamous epithelium. ×200 magnification. The expression of P16 and Ki67 in normal mucosa and oral squamous cell carcinoma. *: X2 = 60.545, Fisher’s exact p = .000; #: t = 38.905, p = .000.

Comparison of proliferation index between oral squamous cell carcinoma and normal tissue
The expression of Ki67 was mainly found in the nucleus (Figure 2). The expression rates of Ki67 in squamous cell carcinoma tissues was significantly higher than those found in normal mucosal epithelium tissues (t = 38.905, p = .000), as shown in Table 1. Expression of Ki67 in oral squamous cell carcinoma and normal mucosal squamous epithelium: Morphological morphology (A1) and expression of Ki67 (B1) in oral squamous cell carcinoma. HE staining (A2) and Ki67 expression (B2) in normal squamous epithelium. ×200 magnification.
Relationship between P16 and Ki67 expression level in oral squamous cell carcinoma
Correlations between the expression level of P16 and the clinicopathological features in oral squamous cell carcinoma.
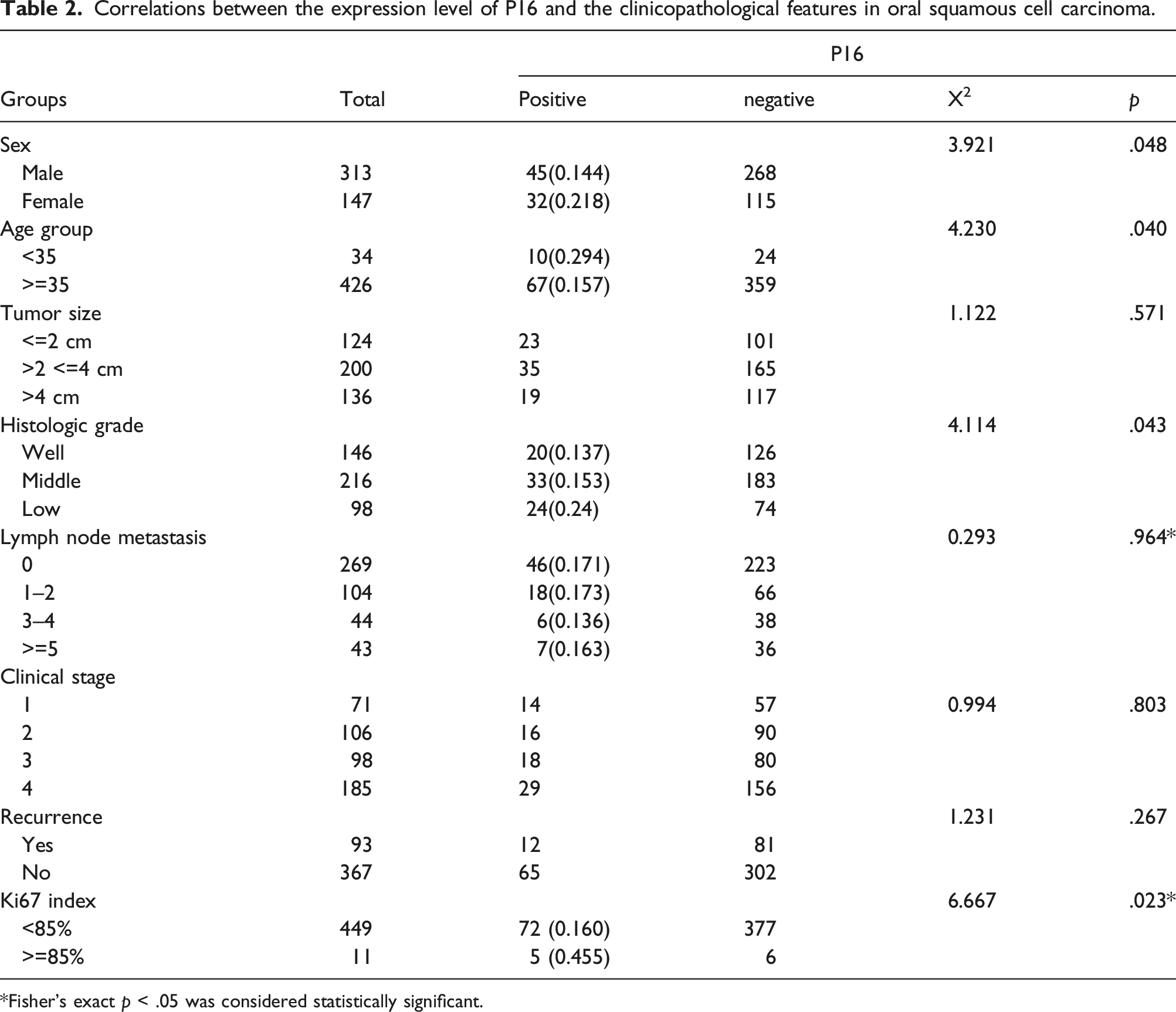
*Fisher’s exact p < .05 was considered statistically significant.
Relationship between P16 expression and clinicopathological features of oral squamous cell carcinoma
The 460 cases of OSCC underwent stratification analysis according to their gender, age, histological grade, lymph node metastasis, tumor recurrence and tumor diameter. P16 positivity was higher in female patients compared to male patients (0.218 vs. 0.144, X2 = 3.921, p = .048). Those under 35 years old also had a higher positivity rate compared to those over 35 years old (0.294 vs. 0.157, X2 = 4.230, p = .040). The rates of P16 expression was also significantly different among groups with high, medium and low histological grade (X2 = 4.114, p = .043, nonparametric rank sum test). There was an inverse relationship between P16 and histological grading. Those in the low histological grading had higher rates of P16 than in the high differentiation group (0.245 vs. 0.137, Z = −2.145, p = .032, nonparametric rank sum test). P16 positivity in high Ki67 expression group was higher than those in the low Ki67 expression group (0.455 vs. 0.160, X2 = 6.667, p = .023). Analysis demonstrated that the expression of P16 had no statistical difference in tumor diameter, lymph node metastasis, tumor recurrence and clinical stage (p > .05).
Logistic regression analysis of HPV infection risk factors in oral squamous cell carcinoma
Logistic regression analysis of the risk factors for human papillomavirus infection in oral squamous cell carcinoma.

Discussion
Previous report has confirmed HPV infection to be one of the major causes of head and neck cancer. 10 HPV infection has been implicated in the development and impacts the prognosis of some head and neck cancers, such as oropharyngeal squamous cell carcinoma, tonsillar carcinoma and other malignant tumors.11,12 Thus its role in the pathogenesis of OSCC is gradually becoming clear with the publication of more studies. Some researchers have utilized PCR, or other methods, to detect HPV infection rates in OSCC patients. 13 They found that the presence of HPV infection in patients was significantly associated with OSCC. Kreimer et al also demonstrated that the HPV infection rate was as high as 23.5% in a study involving 2642 OSCC. 10 Furthermore, Anaya-Saavedra et al revealed that the HPV infection rate in OSCC patients was significantly higher than that in the control group (43.5% vs. 13.7%) in a controlled study of 62 patients with OSCC. 14 Our study contributes additional evidence that confirms the relationship between OSCC and HPV infection. As our study demonstrates, HPV positivity was significantly higher in OSCC tissues than those found in the adjacent normal tissues. This further suggests that HPV infection plays a vital role in the carcinogenesis and development of oral squamous epithelium.
At present, there are only a few studies on the relationship between OSCC and HPV in China and other countries.15,16 HPV infection has only been confirmed in some patients with squamous cell carcinoma, and the clinicopathological characteristics have not been further analyzed. Whether HPV is an independent carcinogenic factor or synergistically promotes the carcinogenesis effects of other high-risk carcinogenic factors, as well as its specific carcinogenic mechanism, remains to be further studied. While some scholars believe that HPV-16 and HPV-18 infection plays a vital function in the development of OSCC, others have suggested that this consequence is affected by regional differences. Kansy’s meta-analysis of 527 case studies revealed that the overall HPV infection rate in OSCC cases was 30.1%, 17 with the most common subtypes being HPV-16 and HPV-18(25.4 and 18.1%, respectively). Coincidentally, Gupta et al conducted a statistical analysis of 50 studies from 1994 to 2014 and found that the incidence of HPV in OSCC ranged from 0% to 80%, with HPV-16 and HPV-18 being the most commonly detected types. 18 However, studies on HPV subtypes has demonstrated inconsistent conclusions. For example, TANDON et al found that positive expression of HPV18 × 107 was a risk factor for oral cancer, 19 while positive expression of P16 was a protective factor for oral cancer.20,21 In order to verify whether the effect of P16 on OSCC is promoting or inhibiting, more studies are needed to further clarify.
In view of the defect of sample size (small sample data) in previous studies, this study was conducted on 460 patient samples to detect the expression of HPV-16 virus in oral squamous cell carcinoma tissues by immunohistochemical method. Immunostaining against the P16 protein is a cost-effective method for the diagnosis of high-risk intra-tissue HPV infection. Diagnostic performance of p16 immunostaining is considered high enough to diagnose high-risk HPV infection in oropharyngeal squamous cell carcinoma, and according to the American College of Pathologists and the eighth edition of the TNM classification, this assay can be used as a surrogate marker for high-risk HPV infection.22,23 Stratified analysis was performed based on the clinicopathological characteristics of patients, including gender, age, histological grade, lymph node metastasis, clinical stage, clinical recurrence, tumor diameter, Ki67 index. We used logistic regression analysis and the entry method to gradually screen variables. Our results demonstrated that patients with OSCC and concurrent HPV infections tend to be females, and younger than 35 years old. In addition, these patients often have a high Ki67 proliferation index (defined as expression in > 85% of cells), suggesting that HPV infection appears to promote the proliferation and growth of oral squamous cell carcinoma tissues. Combined with previous studies, 24 high Ki67 index often represents active growth of tumor cells, suggesting that tumor cells have a high degree of malignancy.
Due to the limitation of experimental conditions, we have not yet been able to perform in-depth gene-level testing of head and neck tumor samples with high P16 expression, which reflects the limitation of our research work. More research work involving genetic testing will address these questions. All samples in this study were from collected from Southern China, and the sample size selected in this study has not been calculated and justified. There may be limitations in terms of sample size, ethnicity, and geography. Further multi-center, prospective studies may better address these questions.
Conclusions
This study demonstrates that HPV infection is associated with the histologic classification of OSCC. Poorly differentiated OSCC tissues were found to have higher HPV positive rate compared to well differentiated OSCC tissues. Further evidence revealed that HPV infection in oral squamous carcinoma tissues could promote the growth of the tumor, demonstrating its possible implication to the carcinogenic effects. The specific mechanism of HPV promoting tumor cell proliferation remains to be confirmed by more molecular biology experiments. Additionally, this study showed that there were differences in lymph node metastasis, recurrence and tumor size between HPV positive and negative squamous cell carcinoma groups, however there was no statistical significance (p < .05), possibly due to the smaller sample size impacting the ability to detect the difference. This area can be further researched in the future. The results provide new ideas for targeted therapy of OSCC by immunotherapy or virus-targeted therapy.
Footnotes
Acknowledgements
We thank Dr Zhi-quan Huang and his team for their technical support in oral and maxillofacial surgery.
Authors’contributions
Lin Wang designed the study and wrote the manuscript. Nian Jiang collected clinical information and performed the IHC assays. Chijen Lee Chen assisted the statistical analyses. All authors have read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Scientific and Technological Plan (No. 2011B031800116) and Medical Science and Technology Research Fund (No. A2011177) of Guangdong Province in China.
