Abstract
A 10-year-old domestic shorthair cat was spayed to remove bilateral ovarian masses and an enlarged uterus. Both ovaries were effaced by large, irregular, firm, glistening, white cystic masses filled with clear viscous fluid. The uterine lumen was filled with copious amounts of clear viscous fluid, and the uterus contained multiple, firm, glistening, white nodules. Histologic examination revealed an invasive spindle cell neoplasm with features of malignancy, and extensive hypocellular areas containing an alcian blue-positive myxoid matrix. Tumor cells expressed caldesmon, α-smooth muscle actin, and estrogen receptor but were negative for desmin. The animal was euthanized 1 month later because of suspected local tumor recurrence. At necropsy, the abdominal cavity contained 120 ml of mucoid ascites; multiple tumor nodules were present in the abdominal and thoracic cavities. The clinical behavior and gross, microscopic, and immunohistochemical findings established a diagnosis of myxoid leiomyosarcoma.
Uterine tumors are rare in cats, accounting for 0.29% of feline neoplasms in one report. 6 Previously reported feline uterine neoplasms include endometrial adenocarcinoma, mixed Mullerian tumor (adenosarcoma), leiomyoma, and leiomyosarcoma, of which leiomyomas occur most frequently. 4, 6 Myxoid leiomyosarcoma of the human uterus is a rare tumor; only 26 cases have been described in the literature, generally in women of perimenopausal age. 5, 8 Presenting complaints include an abdominopelvic mass, pain, and abnormal vaginal bleeding. Typical leiomyomas may occasionally coexist. The tumors are generally quite large at presentation (>8 cm). They are hypocellular spindle cell neoplasms with a myxoid matrix. The neoplastic cells often lack severe atypia, and mitotic indices are often low, in part owing to their low cellular density. The tumors frequently have an aggressive clinical course despite their generally bland microscopic appearance, with intra-abdominal recurrences being common. Distant metastases to the lung, liver, and brain have been reported. Myxoid leiomyosarcoma has previously been reported in the subcutis in a Sarus Crane 2 and in the gizzard of a chicken. 7
A 10-year-old sexually intact, female, domestic shorthair cat was presented for weight loss and anorexia. Physical examination revealed a small amount of blood-tinged, mucoid vaginal discharge and a markedly enlarged uterus, presumed to be cystic endometrial hyperplasia/pyometra complex. The cat was anemic, with a hematocrit of 25.3% and a mild neutrophilia (9398/µl, reference range 2500–8500/µl). The animal was prepped for ovariohysterectomy; exploration of the abdomen revealed that both ovaries were enlarged, measuring 4 × 4 × 4 cm and 6 × 5 × 5 cm, and effaced by proliferative mesenchymal tissue that imparted a shiny, white, and slightly gelatinous appearance (Fig. 1). Both ovarian masses were cystic on cut section and filled with viscous clear fluid. The uterus was markedly and diffusely enlarged with fluid-filled uterine horns, measuring 7 × 2 × 2 cm each, containing approximately 15 ml of a clear, thick viscous fluid. The endometrium was grossly hyperplastic, with a velvety proliferative appearance and several white intramural and exophytic polypoid masses measuring 3 to 10 mm as well as multiple endometrial cysts. The ovaries and uterus were removed and placed in 10% neutral buffered formalin for histologic examination. Examination of the abdomen was unremarkable, and the animal recovered uneventfully.
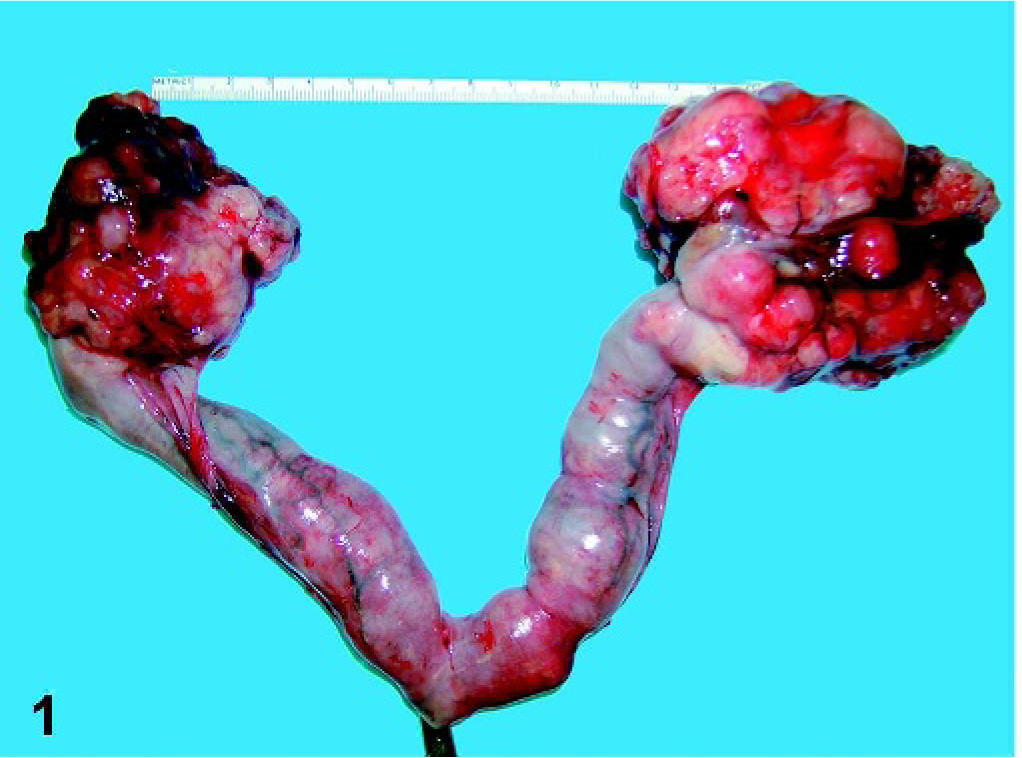
Ovaries and uterus; cat. Both ovaries are effaced by multinodular smooth glistening mesenchymal neoplasms. Uterus is diffusely enlarged and fluid-filled. Scale is in centimeters.
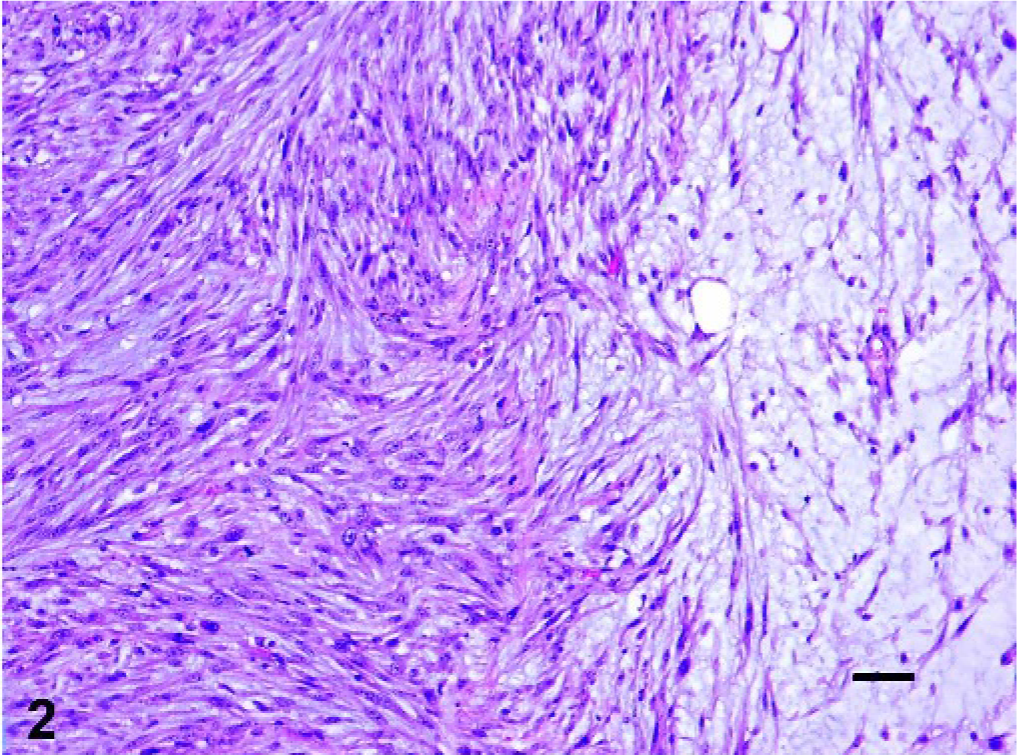
Uterus; cat. Fascicles of atypical smooth muscle cells merge with areas of low cellular density on a pale basophilic myxoid stroma. HE. Bar = 50 µm.
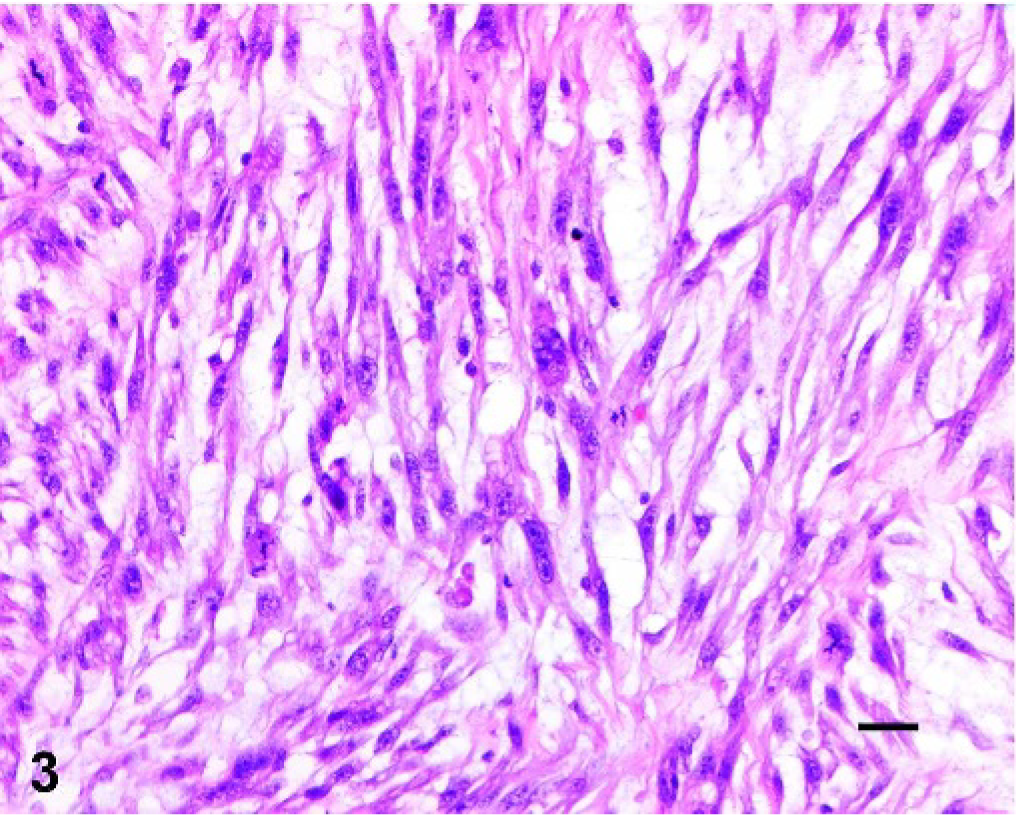
Uterus; cat. Anaplastic smooth muscle cells with multinucleate giant cells and mitotic figures. HE. Bar = 25 µm.
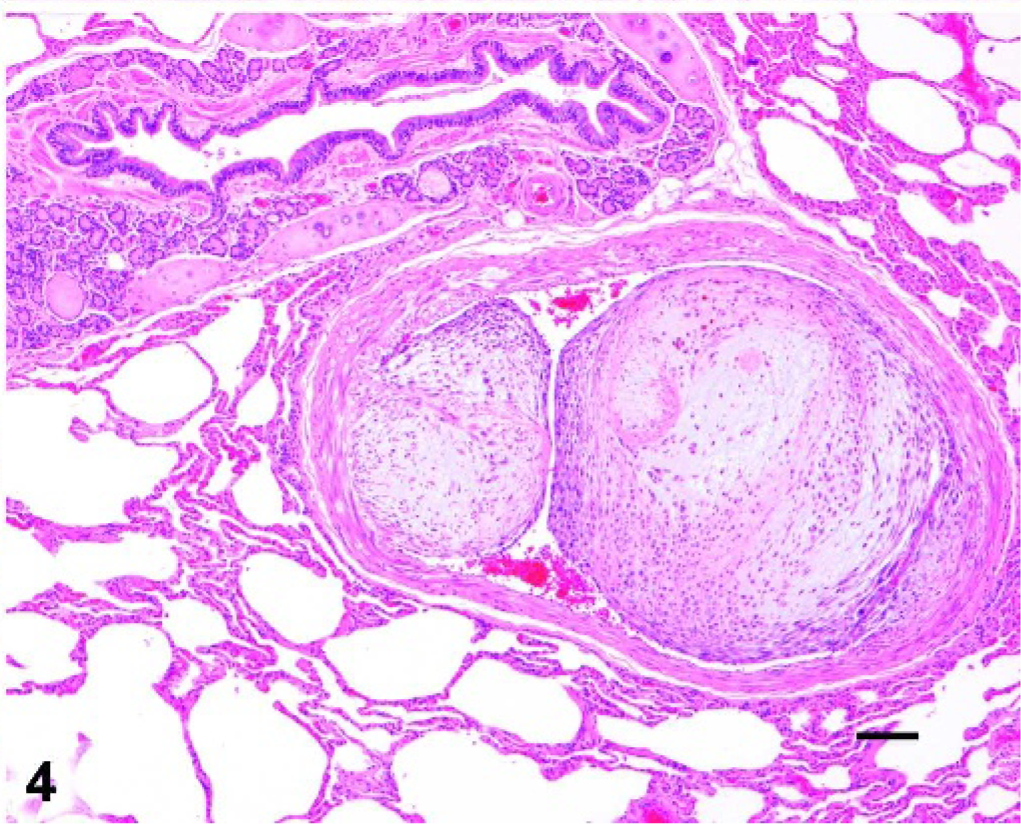
Lung; cat. A large neoplastic embolus is present within the vascular lumen and invading the wall of a pulmonary arteriole. Outer layers are densely cellular, while center is loose and myxoid. HE. Bar = 100 µm.
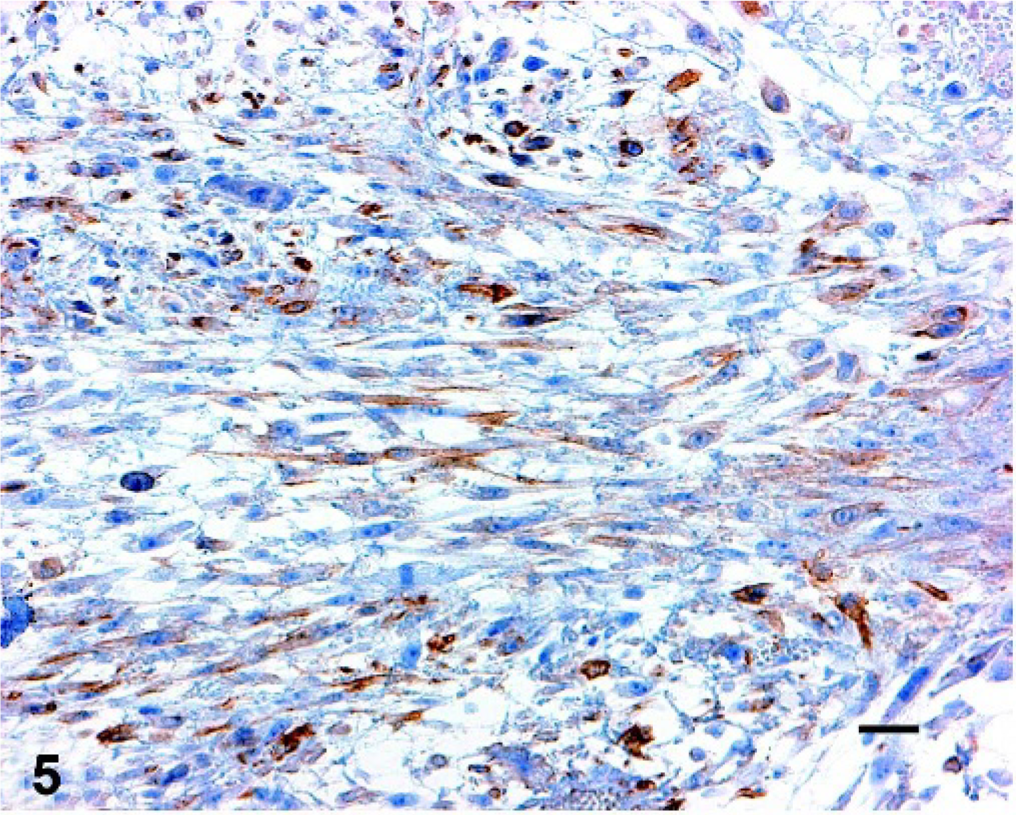
Uterus; cat. Immunohistochemistry shows positive reaction for α-smooth muscle actin. Bar = 25 µm.
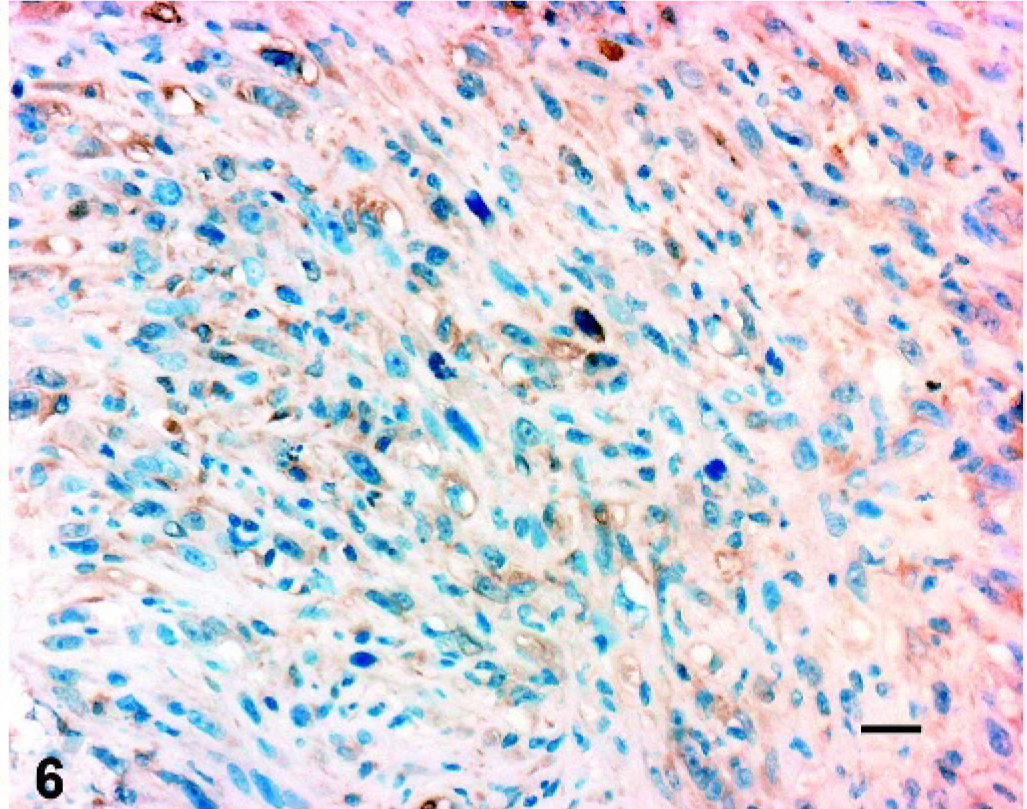
Uterus; cat. Immunohistochemistry shows positive reaction for caldesmon. Bar = 25 µm.
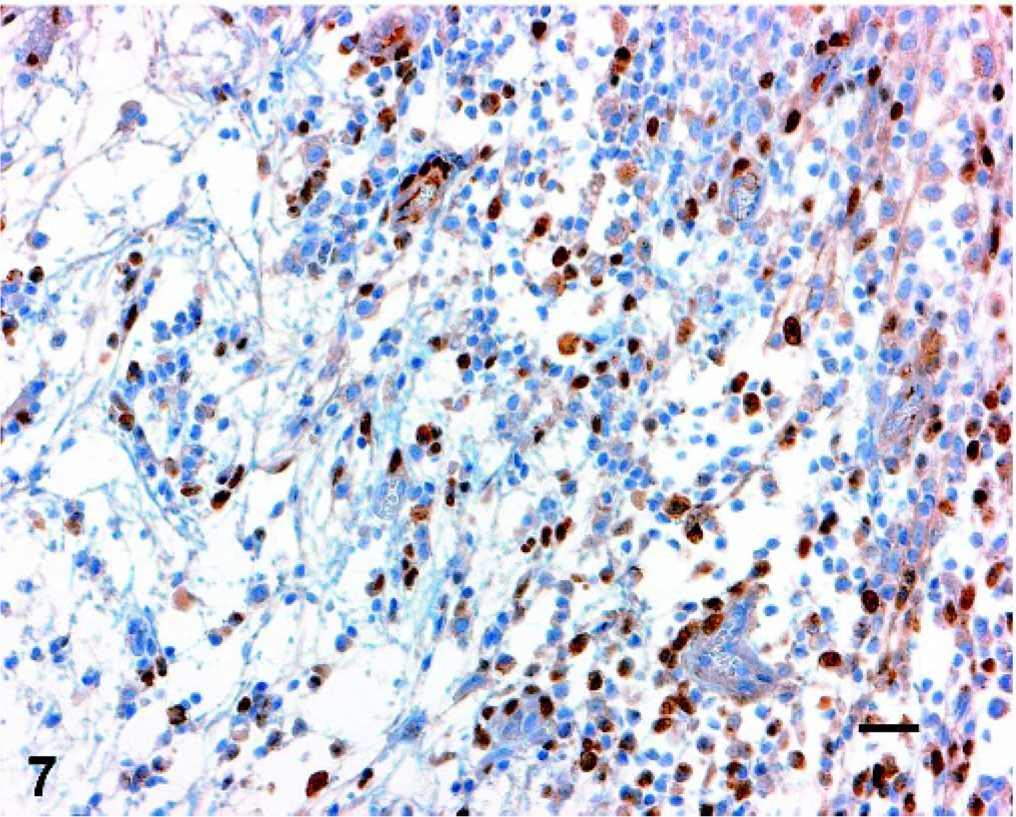
Uterus; cat. Immunohistochemistry shows positive nuclear reaction for estrogen receptor in some neoplastic cells. Bar = 25 µm.
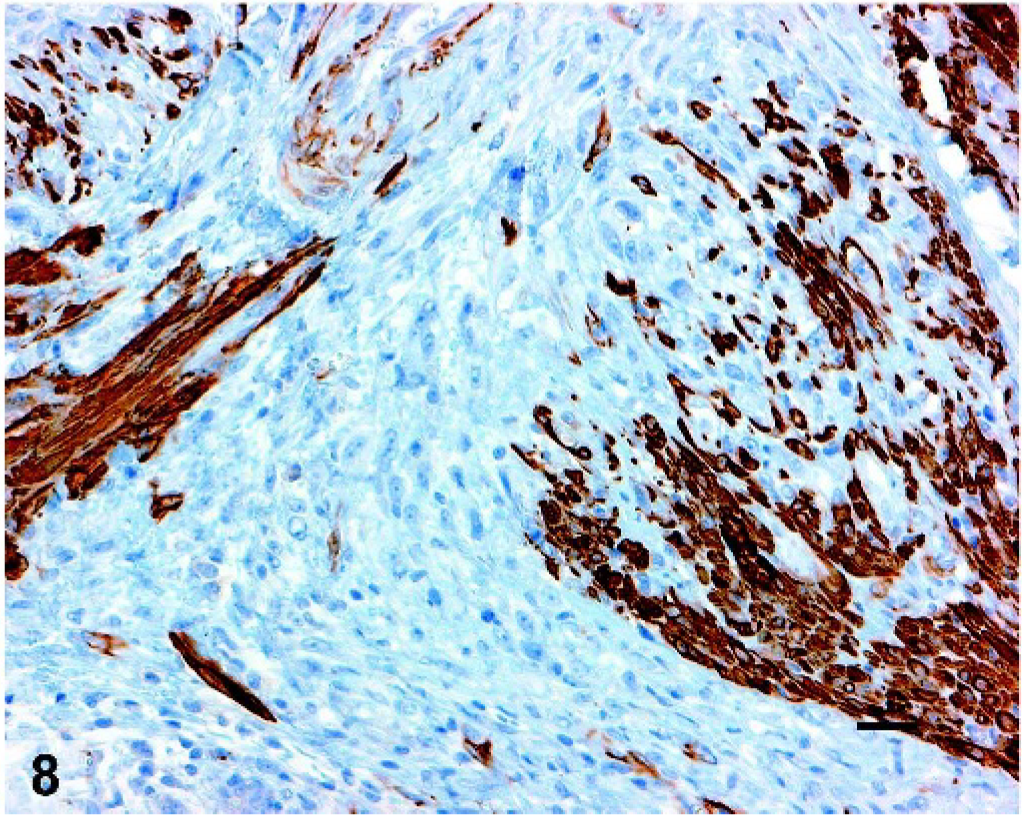
Uterus; cat. Neoplastic cells are negative by immunohistochemistry for desmin, although entrapped normal smooth muscle cells show positive reaction. Bar = 25 µm.
Following fixation, tissues were embedded in paraffin, sectioned at 4 µm, and stained with hematoxylin and eosin and alcian blue. For immunohistochemical studies, sections were deparaffinized and steamed in a citrate buffer for 5 minutes for antigen retrieval. Immunoperoxidase staining was performed with the automated Bio Tek-1000 immunostainer system (Bio Tek Solutions Inc., Santa Barbara, CA). All procedures were performed according to the manufacturer's protocols. The slides were then incubated with the following antibodies using the avidin-biotin peroxidase method: alpha-smooth muscle actin (clone 1A4, prediluted, Ventana Medical Systems Inc., Tucson, AZ), caldesmon (clone h-CD, 1 ° 250, Dako Corp., Carpinteria, CA), estrogen receptor (prediluted, clone 6F11, Ventana), progesterone receptor (clone 16, prediluted, Ventana), and desmin (clone D33, 1 ° 100, Dako). The staining of negative controls was the same except for substitution of the primary antibody with preimmunization mouse serum. The reaction product was visualized with 3,3′-diaminobenzidine chromagen and the sections were counterstained with 0.1% hematoxylin (Sigma, St. Louis, MO), dehydrated, and mounted.
Microscopic examination of the uterus demonstrated multiple variably-sized infiltrative masses originating in the myometrium with invasion of the endometrium to form sessile exophytic nodules. Masses were unencapsulated and poorly demarcated, with variable cellularity ranging from densely packed spindle cells arranged in interwoven short fascicles to loosely spaced spindle cells separated by copious amounts of pale basophilic, amorphous, alcian blue-positive matrix. Areas of low and high cellularity could frequently be observed to intermingle with a visible transition zone (Fig. 2). Cells had scant amounts of eosinophilic cytoplasm with occasional small clear cytoplasmic vacuoles and a moderately large central cigar-shaped nucleus containing finely stippled chromatin, with a single indistinct nucleolus. There was moderate to marked nuclear atypia present, with frequent karyomegaly and multinucleate giant cells (Fig. 3). The mitotic index ranged from 1 to 5 per high power field, with an average of 1–2 per 400× high power field (HPF). Greater mitotic activity was noted in the more cellular zones, and abnormal mitotic figures were readily identified.
There were multiple, large, irregular foci of acute coagulative necrosis and hemorrhage, and both ovaries were nearly completely effaced by neoplastic cells and coagulative necrosis. The endometrium showed diffuse, well-differentiated, simple hyperplasia with multiple cystic glands. There were several intravascular emboli in the uterus, which generally had a myxoid center with a more compact outer layer of smooth muscle cells. Similar emboli were present in the lung at necropsy (Fig. 4). The neoplastic cells demonstrated weak cytoplasmic staining for α-smooth muscle actin (Fig. 5) and caldesmon (Fig. 6), and weak nuclear expression of estrogen receptor (Fig. 7). The neoplastic cells were negative for desmin (normal myometrium demonstrated strong positive cytoplasmic staining; Fig. 8). Both neoplastic and normal uterine tissues lacked expression of progesterone receptor.
One month after ovariohysterectomy, abdominal palpation of the cat revealed a firm abdominal mass adherent to the body wall, assumed to be recurrence of the tumor. The animal was humanely killed with an overdose of sodium pentobarbital and immediately submitted for necropsy.
At necropsy, the abdomen was distended and contained 120 ml of a blood-tinged, clear viscous (mucoid) ascitic fluid with a total protein content of 4.0 g/dL. There were multiple, firm, shiny, white, glistening and gelatinous irregular sessile exophytic nodules, ranging from 0.7 to 3 cm in diameter scattered throughout the abdominal mesentery, with larger nodules characterized by a fluid-filled center containing clear viscous fluid. There was a large bilobed nodule, measuring 6 × 2 × 2 cm, which infiltrated both the abdominal wall and proximal jejunum, fixing the jejunum in place. A 2 × 1.5 × 1.5 cm nodule was present near the splenic artery invading the head of the spleen. A similarly sized nodule was present in the mesentery between the liver, gallbladder, and duodenum, causing partial compression of the common bile duct and resulting in a dilated gallbladder that was difficult to express. Tumor metastases were also visible on the abdominal surface of the spay incision, the abdominal surface of the diaphragm, and within the lungs. Additional gross findings included a 5 × 6 × 10 mm chronic cortical infarct in the cranial pole of the right kidney.
Tumor nodules observed at necropsy were histologically similar to uterine and ovarian masses, and neoplastic emboli were observed in multiple tissues, especially in the lung and spleen. Neoplastic emboli in the spleen were frequently observed in association with large, multifocal to coalescing areas of acute coagulative necrosis (infarcts), suggestive of ischemic events secondary to the emboli. In addition, there was a small focus of subacute coagulative necrosis rimmed by microglia and centered on an arteriole (infarct) in the white matter of the thalamus.
Additional histologic necropsy findings included nodular hyperplasia of the exocrine pancreas, amyloidosis of the endocrine pancreas, follicular atrophy and cysts in the thyroid, glomerulosclerosis and chronic interstitial nephritis in the kidneys, chronic lymphocytic enterocolitis, nonsuppurative cholangitis, and mild pulmonary changes consistent with feline asthma complex.
The feline neoplasm described herein is similar in all regards to the myxoid leiomyosarcoma of the human uterus. In particular, the invasive growth in the uterus and widespread metastases attest to the malignant behavior of the neoplasm, and the microscopic features and immunohistochemical evidence of smooth muscle differentiation confirm the diagnosis of myxoid leiomyosarcoma. The tumor is most consistent with a primary uterine neoplasm rather than a primary ovarian neoplasm based on the clinical presentation (uterine enlargement, vaginal discharge), tumor type (leiomyosarcoma is far more commonly a primary uterine than a primary ovarian neoplasm), and presence of bilateral ovarian tumors (characteristic of metastatic involvement even when large). Absence of desmin expression does not refute a diagnosis of a smooth muscle neoplasm because some leiomyosarcomas fail to express desmin 1, 3 ; other markers of smooth muscle differentiation (caldesmon, smooth muscle actin) were expressed by this tumor. The differential diagnosis includes myxoid leiomyoma, which is a benign, well-circumscribed smooth muscle neoplasm having myxoid features but lacking significant nuclear atypia and necrosis, and typically with a low mitotic index. Myxoid leiomyosarcoma should be considered in the differential diagnosis of uterine tumors producing mucoid ascites in animals.
Footnotes
Acknowledgements
The authors wish to thank Pat Wilcox for histology support, Barbara Domazet for immunohistochemistry support, and Dr. David Huso for assistance with photography.
