Abstract
A 10-year-old female spayed domestic shorthaired cat presented with a subcutaneous tumor between the first and second phalanges of the left hind foot. Six months after excision, a similar tumor occurred on the medial aspect of the third phalanx of the same limb. Histologically, both tumors consisted of solid masses of spindle and round cells, many of which grew within endothelial-lined vessels. Tumor cells stained positively for smooth muscle actin and vimentin, but were negative for cytokeratin, S-100, desmin, synaptophysin, factor VIII-related antigen, and neuron-specific enolase. The diagnosis was dermal intravascular leiomyosarcoma.
Keywords
A 10-year-old domestic shorthaired cat was presented to the referring veterinarian because of a large swelling between the first and second phalanges of the left hind foot. The owner reported that the mass intermittently bled before presentation. The mass and associated phalanx were amputated at surgery and submitted to the New York State Diagnostic Laboratory for routine histopathologic examination. Six months later, a similar mass occurred on the medial aspect of the third phalanx of the same limb. The second mass and associated phalanx were resected and submitted for histopathologic evaluation. Specimens for histology were fixed in 10% neutral buffered formalin, processed routinely for paraffin embedding, and stained with hematoxylin and eosin. The phalanges were decalcified using citrate-buffered formic acid. Additional paraffin sections from both submissions were stained histochemically for smooth muscle actin, vimentin, desmin, S-100, neuron-specific enolase (NSE), factor VIII-related antigen, synaptophysin (Dako, Carpinteria, CA), and keratin AE1/3 (Boehringer Mannheim, Indianapolis, IN).
Both tumors were histologically similar and consisted of large highly cellular expansile dermal masses composed of round to spindle cell bundles, many of which were growing in vascular lumens (Fig. 1). Within the dermis, lobules of neoplastic cells replaced the adnexa, either leaving a Grenz zone consisting of dermal collagen, or directly abutted the basal epidermis. The margins of the tumor caused compression of the adjacent adnexa, but no invasion of underlying bone was observed. The neoplastic cells had moderate amounts of a granular or fibrillar lightly eosinophilic cytoplasm with indistinct cell margins. Nuclei were oval to fusiform with lacy or vesicular chromatin and 20 mitotic figures were found in 10 40× high-power fields. Intravascular growths were mostly attached to vessel walls and in some areas were composed of nests or anastomosing cords of cells surrounded by endothelium. Larger lobules were separated by a fine collagenous stroma that stained positively for collagen with Masson's trichrome. Histochemically, the neoplastic cells were strongly positive for smooth muscle actin (Fig. 2) and vimentin, but were negative for desmin, S-100, NSE, factor VIII-related antigen, synaptophysin, and keratin. The vascular channels containing tumor were lined by endothelial cells that stained positively for factor VIII-related antigen (Fig. 3). Endothelium surrounding nests composed of anastomosing cords of tumor was occasionally positive for factor VIII-related antigen. Both tumors appeared adequately excised.
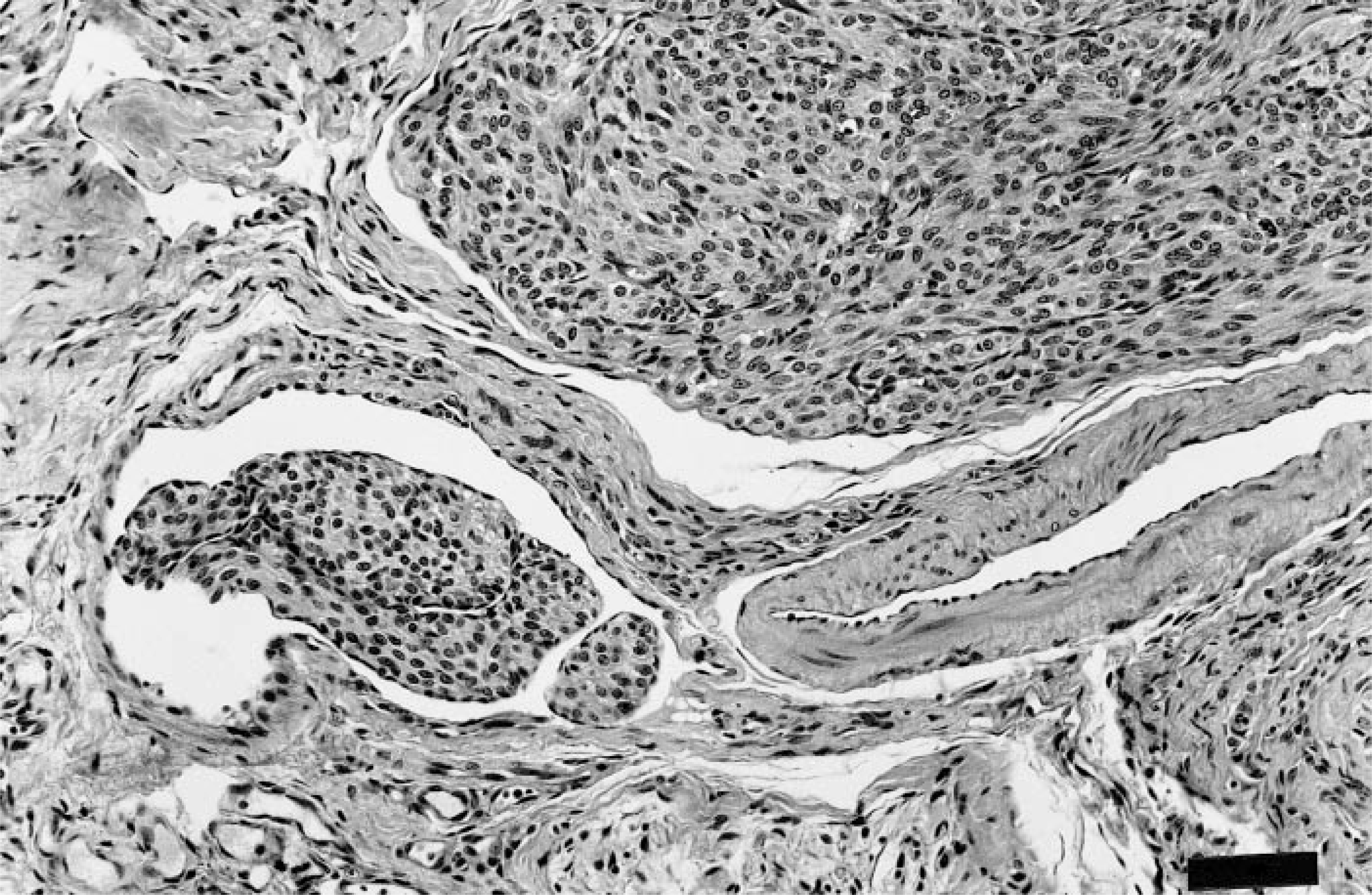
Intravascular leiomyosarcoma; cat. Bundles of neoplastic round to spindle cells growing adjacent to and within endothelial-lined channels. HE. Bar = 57 μm.
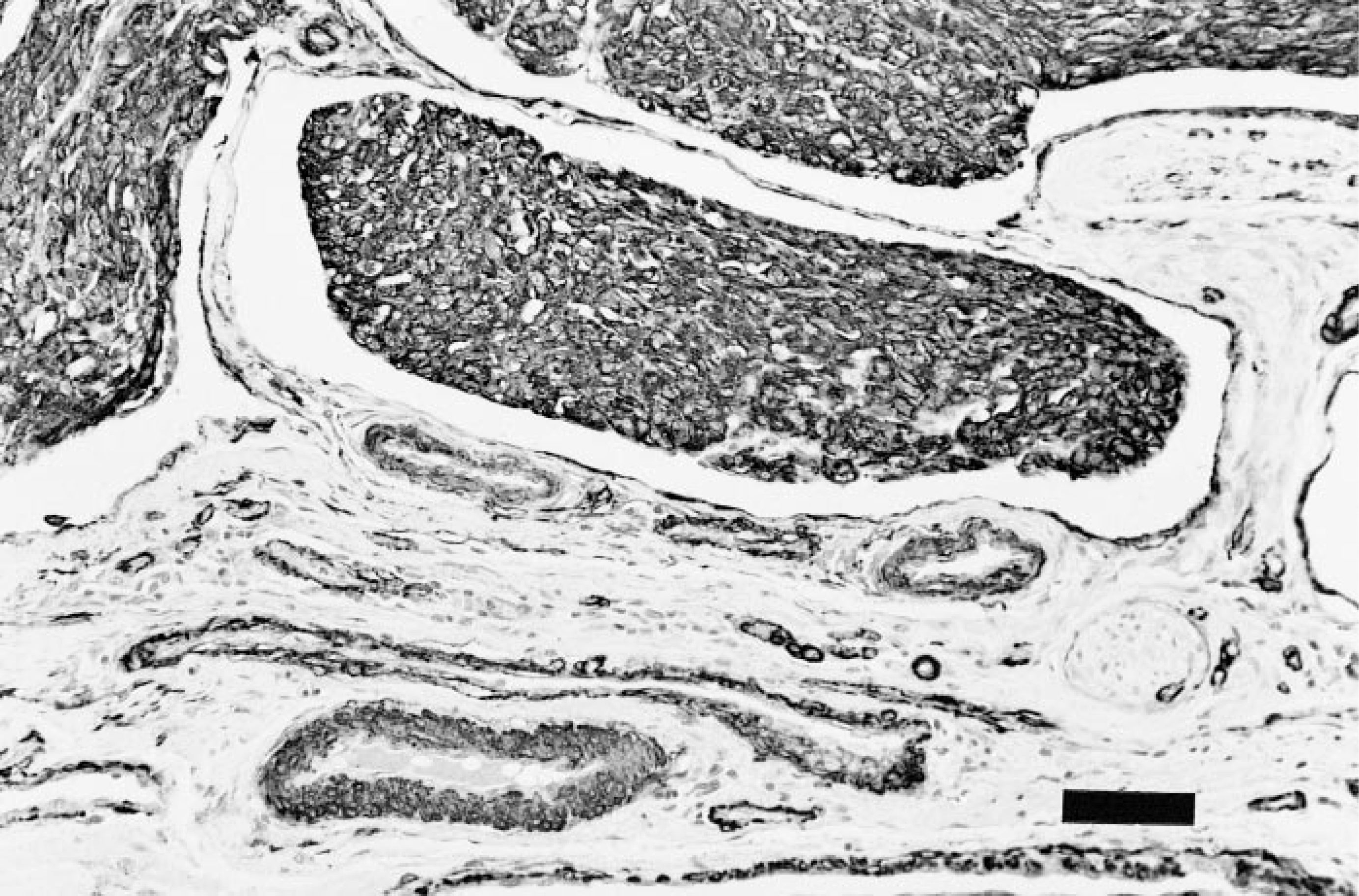
Intravascular leiomyosarcoma; cat. Immunoperoxidase staining for smooth muscle actin. Neoplastic cells react strongly, as does smooth muscle of adjacent blood vessels. Meyer's hematoxylin counterstain. Bar = 57 μm.
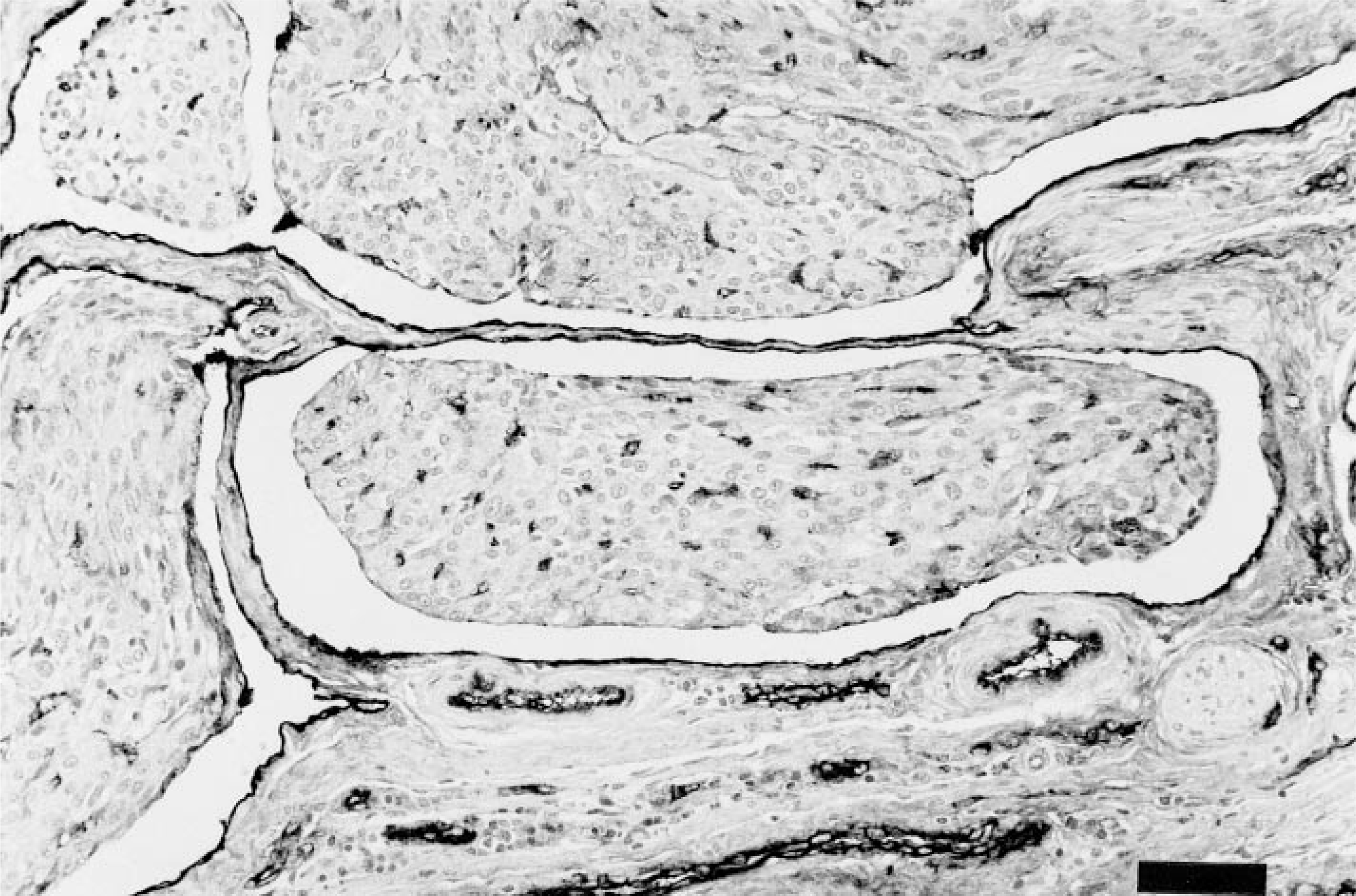
Intravascular leiomyosarcoma; cat. Immunoperoxidase staining for factor VIII-related antigen. Endothelium lining the vascular channels stains positively, as does endothelium within the tunica of adjacent capillaries. Meyer's hematoxylin counterstain. Bar = 57 μm.
Intravascular smooth muscle tumors are a rare finding in animals, with only a single case reported from the bladder of a cat.9 This previous case report describes an intravenous leiomyoma in a 5-year-old male Siamese cat with an intramural bladder mass. Neoplastic cells were evident growing within ectatic veins and endothelial-lined channels that resembled lymphatics. The intraluminal growths were either attached to vessel walls or as solid cords that floated within vascular lumens. Extravascular growth was not observed in any of the sections examined. Mitoses were uncommon (a total of one per 10 high-power fields), and metastases were not observed in a sublumbar lymph node that was examined. The cat was clinically normal 25 months after excision of the mass, confirming the apparent benign nature of the tumor.
The histochemical findings and morphology of the present neoplasm raise the possibility of glomus tumor in the differential diagnosis. Glomus tumor is a rare neoplasm of humans that is derived from the modified smooth muscle cells of the glomus body.3 The normal glomus body is a specialized arteriovenous anastomosis that serves in thermoregulation. Most glomus tumors involve the subungual region of the fingers, although they have been recorded from sites where the normal glomus body is sparse or even absent (chest wall, eyelid, stomach, and bone). Histologically, the cells comprising glomus tumors are quite distinctive. They are rounded or polygonal, with centrally placed, sharply defined ovoid nuclei, and amphophilic to lightly eosinophilic cytoplasm. The presence of intermixed small nerve fibers is also characteristic. Subsets of glomus tumor are recognized, including glomangiomyoma, in which there is a gradual transition from the classical glomus cell to fusiform cells resembling smooth muscle. Immunohistochemical studies of both human and canine glomus tumor reveal consistent positive staining for smooth muscle actin and vimentin, with rare human cases also staining positive for desmin.2,11 Although the immunohistochemical staining characteristics of the present neoplasm were similar to human and canine glomus tumor, intravascular growth of glomus tumor is rare1 and the absence of admixed neural elements is more consistent with an intravenous smooth muscle neoplasm than with glomus tumor. The lack of desmin immunoreactivity in the present case is not inconsistent with a smooth muscle origin, because vascular smooth muscle may not express desmin.7 Reports of feline cutaneous smooth muscle neoplasia are confined to multiple piloleiomyomas.5 These tumors showed no vascular invasion or intravascular growth and were presumed to have arisen from the arrector pili muscles. The origin of the present tumor is uncertain, given the presence of both intra- and extravascular growth. The intravascular growth observed may reflect vascular invasion of a cutaneous tumor, or de novo origin from vascular smooth muscle, with subsequent invasion of the dermis. In humans, intravenous leiomyomatosis is a rare condition in which nodules of smooth muscle grow within the veins of the myometrium. The condition may result from either vascular invasion of a leiomyoma, or directly by transformation of the smooth muscle of the vein itself.8 In either case, these lesions are not considered to have metastatic potential, but occasional patients have developed extensions of the process into the pelvic veins, vena cava, right atrium, and even the lung. Determining suitable criteria of malignancy in smooth muscle tumors is problematic. In both human and veterinary pathology, mitotic activity is generally considered one of the most accurate and reproducible features in predicting metastatic potential.4,10 However, comparisons between human smooth muscle tumors of the uterus and soft tissues revealed that uterine lesions with a total of five to nine mitoses per 10 high-power fields are considered borderline, whereas a similar number of mitoses in a soft tissue tumor would signify the capability for metastasis. This highlights the difficulty in comparing data between smooth muscle tumors of differing anatomical locations. In considering the present tumor, we favor a diagnosis of leiomyosarcoma over leiomyoma. Although the tumor was well differentiated with no evidence of cytologic atypia, the mitotic index was comparable with that observed in a study of 80 human dermal leiomyosarcomas, in which two mitoses per 10 high-power fields warranted a malignant diagnosis.4 A separate study also revealed that at least one quarter of human leiomyosarcomas of peripheral soft tissue arose from or involved a vein.6 The latter finding confirms the potential difficulty in establishing the origin of some human peripheral soft tissue leiomyosarcomas, a situation similar to the present case. Ultimately, only clinical follow-up of a significant number of similar tumors will allow for the formulation of accurate histologic criteria for predicting malignancy. Unfortunately, data for feline cutaneous smooth muscle neoplasia is lacking, given the rarity of documented cases.
The occurrence of the same tumor type within an adjacent toe 6 months after excision of the first raises the possibility of intravascular extension as the mechanism of spread. This would seem more likely than metastasis, because most human cutaneous leiomyosarcomas metastasize to local lymph nodes or lung. When last examined (6 months after excision of the second mass), the cat remained healthy with no evidence of recurrence or metastasis.
