Abstract
An aged mixed-breed goat doe was presented with a 9-mo history of serosanguineous vaginal discharge. Vaginal speculum examination revealed serosanguineous discharge but otherwise no abnormalities. Transrectal ultrasonography showed normal ovaries and multifocal cystic lesions within the uterus. Ovariohysterectomy was recommended because of a strong suspicion of neoplasia. Multiple, non-resectable masses were noted in and around the uterus intraoperatively, and euthanasia was elected. Autopsy revealed multiple masses within the uterus, cervix, and lung parenchyma. Histologically, the masses within the uterus represented a likely collision tumor of primary adenocarcinoma and leiomyosarcoma. Our report highlights the importance of obtaining biopsy samples of all masses because the lesions described showed significantly different biological behavior. This information is vital to guide treatment and prognosis.
The prevalence of neoplasia in a retrospective study of 100 goats was 8.7%, and included lymphoma, squamous cell carcinoma, thymoma, and mammary adenocarcinoma as the most common tumors. 6 Six tumors of the reproductive tract were reported, with 3 does diagnosed with adenocarcinoma of the cervix or uterus and 3 diagnosed with leiomyoma of the uterus or vagina. One doe with adenocarcinoma had distant metastases. 6
A collision tumor is the presence of 2 types of tumors with areas of mingling within 1 organ. This phenomenon is rare in animals, but has been reported in the rabbit and hamster.2,3 We describe herein a case of uterine adenocarcinoma and leiomyosarcoma in a goat.
An ~8-y-old female mixed-breed doe was presented to the Cornell University Hospital for Animals (Ithaca, New York) with a history of serosanguineous vaginal discharge for at least 9 mo. She had been obtained by a rescue organization and had exhibited the discharge since rescue. She was housed on pasture with multiple other animals, none of whom showed signs of illness.
On presentation, she exhibited mild tachycardia and pale mucous membranes. Serosanguineous vaginal discharge and perivulvar staining were noted. Her physical examination was otherwise unremarkable. She was also noted to have mild stranguria and an abnormal posture during urination.
No masses were visualized on vaginal speculum examination but copious serosanguineous discharge was present. Vaginal cytology was obtained and revealed cuboidal epithelial cells (most consistent with endometrial cells) admixed with low numbers of non-degenerate neutrophils. No neoplastic cells were noted.
Transrectal ultrasound was performed using a 5-MHz prostate probe and revealed multifocal, anechoic, cystic structures diffusely in the uterus. Both ovaries appeared normal. Given the signalment and chronicity of the vulvar discharge, neoplasia was strongly suspected and ovariohysterectomy was recommended.
The doe was anesthetized, and the surgical site was prepared aseptically. A standard ventral midline incision was made. The left ovary was identified and exteriorized, and the ovarian pedicle was ligated. The left uterine horn was dilated. Palpation of the right ovary and right horn of the uterus revealed multiple masses extending to and along the dorsal body wall. Given the inability to completely resect these masses, euthanasia was elected.
Autopsy revealed that the cervix was partially effaced by a large (1.5 × 1.5 × 2 cm), infiltrative, multinodular mass that extended into the uterine body (Fig. 1). The mass was gray to white, firm, and smooth on the surface and when sectioned. The cranial portion of the left uterine horn was dilated and filled with clear, thin fluid (unilateral hydrometra). A second, ill-defined mass occluded the left uterine horn distal to the segmental dilation. This mass measured ~0.8 × 0.5 × 1 cm. The ovaries and uterine tubes were normal. The right and left ureters were compressed by the uterine tumors and were dilated (bilateral hydroureters). In the lung, dozens of randomly distributed, irregularly shaped, well-demarcated, raised masses with an occasional central depression (umbilication), 0.5–2 cm diameter, were noted on the pleural surfaces and throughout the parenchyma. Multiple deposits of fibrin were present on the pleural surface (Fig. 2), as were multiple fibrous adhesions to the thoracic wall.
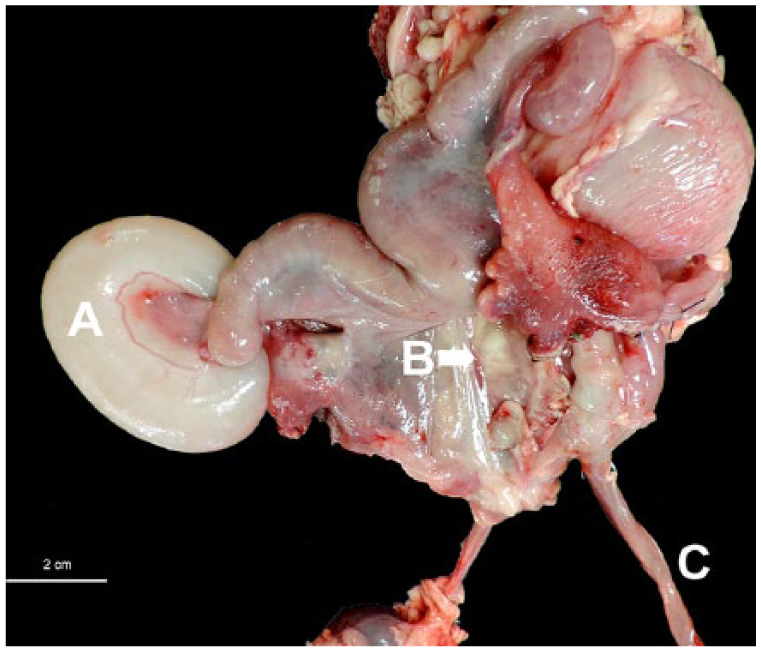
Goat uterus with a dilated proximal portion of the left uterine horn (A, hydrometra, confirmed by histopathology); an ill-defined mass occluded the uterine horn distal to this dilation. A multinodular mass in the area of the cervix (B) and hydroureter (C). Bar = 2 cm.
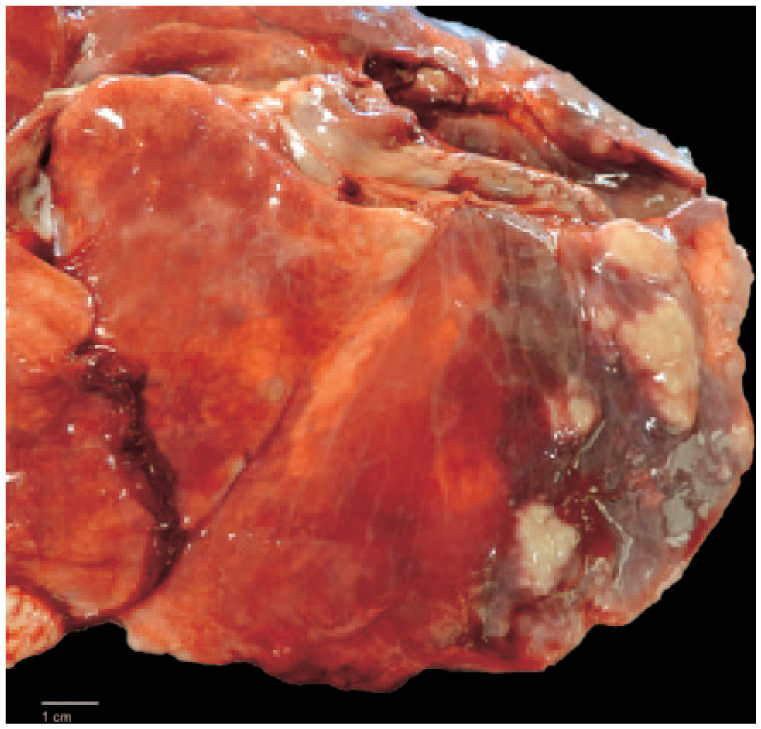
Multiple adenocarcinoma metastases within the lung parenchyma. Bar = 1 cm.
Histopathology revealed a multilobulated, well-demarcated, unencapsulated, locally invasive, uterine neoplasm composed of 2 different intermingled spindyloid and epithelial cell populations (Fig. 3). Lobules of spindle cells were arranged in broad interlacing bundles supported by moderate amounts of collagenous stroma. These cells had indistinct cell borders, moderate amounts of vacuolated or fibrillar eosinophilic cytoplasm, blunt-ended oval nuclei with finely granulated chromatin, and single eosinophilic nucleoli. One mitotic figure per 10 high power fields (HPF; 400×) was noted. Anisocytosis and anisokaryosis were mild. Rare karyomegalic cells and multinucleate cells admixed with areas of necrosis and hemorrhage were present within these areas. These areas of the tumor were most consistent with uterine leiomyosarcoma (Fig. 3C).
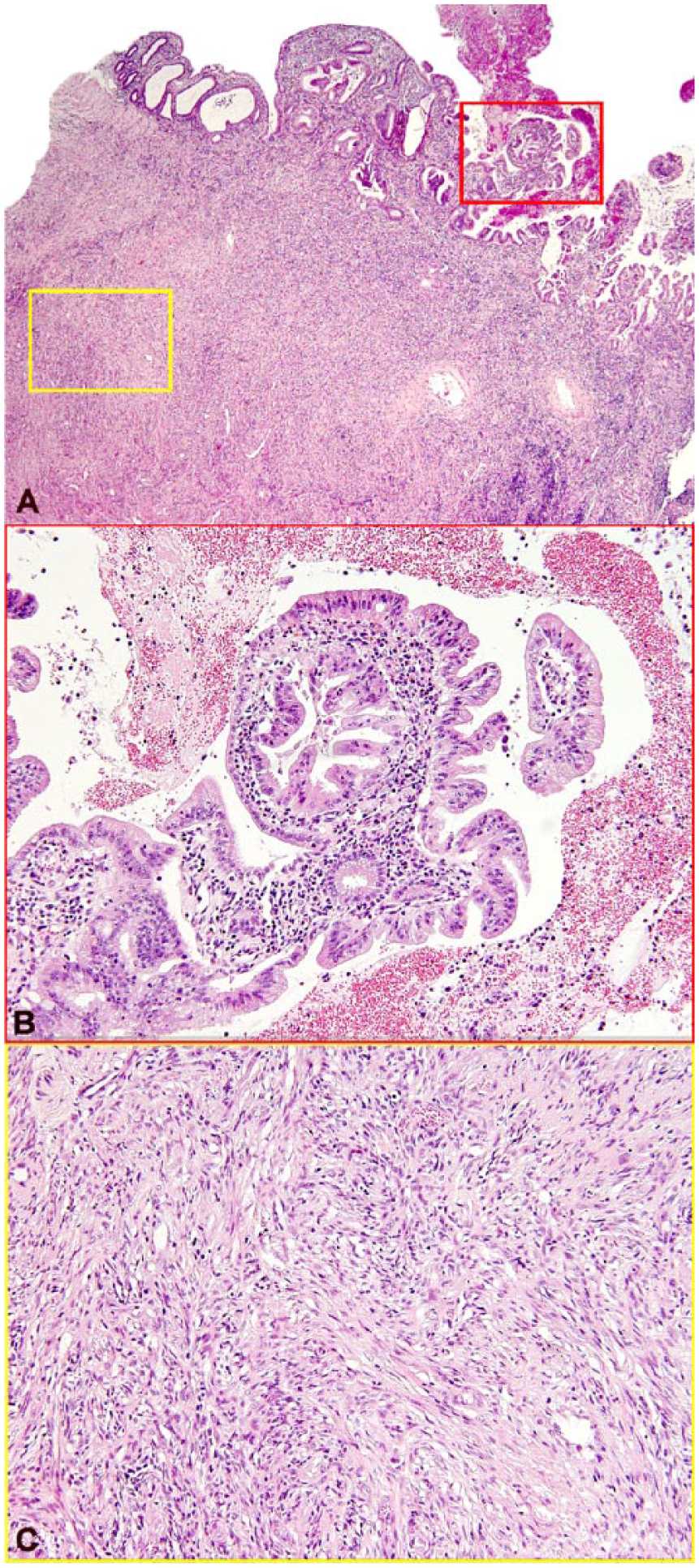
A. An adenocarcinoma, arising from the endometrium and composed of tubules and lobules of epithelial cells, collides with a leiomyosarcoma, composed of spindle cells arranged haphazardly or in streams arising from the myometrium. H&E. B. Red box inset from panel A. Papillary projection of uterine adenocarcinoma into the uterine lumen. C. Yellow box inset from panel A. Uterine leiomyosarcoma spindyloid cells forming interlacing bundles.
Epithelial portions of the mass consisted of papillary projections that protruded into the uterine lumen as well as lobules of densely packed tubular and glandular structures supported by dense collagenous stroma that infiltrated the subjacent myometrium. Papillary projections and tubular structures were covered or lined by a single layer of cuboidal-to-columnar neoplastic epithelial cells supported by variable amounts of loose vascular collagenous stroma. Neoplastic cells had a moderate amount of eosinophilic cytoplasm, indistinct cell borders, and polygonal nuclei with one inconspicuous nucleolus. Anisocytosis and anisokaryosis were severe, with 3 mitotic figures per 10 HPF. Large amounts of sloughed cells, inflammatory debris, and necrosis were present among these infiltrates, and tumor emboli were noted in uterine lymphatic vessels. These infiltrates were most consistent with uterine adenocarcinoma (Fig. 3B). Approximately 70–80% of the mass was comprised of spindle cell infiltrates consistent with leiomyosarcoma, with the remaining mass consisting of adenocarcinoma. The leiomyosarcoma emerged from the myometrium and reached the endometrium, where the adenocarcinoma emerged and infiltrated the underlying leiomyosarcoma.
Extensive intermingling of the 2 neoplastic cell populations was highlighted by immunohistochemistry (Fig. 4A). Neoplastic mesenchymal cells exhibited strong intracytoplasmic immunoreactivity to α–smooth muscle actin (Fig. 4B) and did not stain with cytokeratin AE1/AE3. Neoplastic epithelial cells stained with cytokeratin AE1/AE3 with strong intracytoplasmic immunoreactivity, whereas spindyloid populations did not (Fig. 4C). Multifocally, the endometrial glands formed cysts of various sizes occasionally filled with neutrophils, cellular debris, and foamy macrophages, which was interpreted as cystic endometrial hyperplasia with developing pyometra.
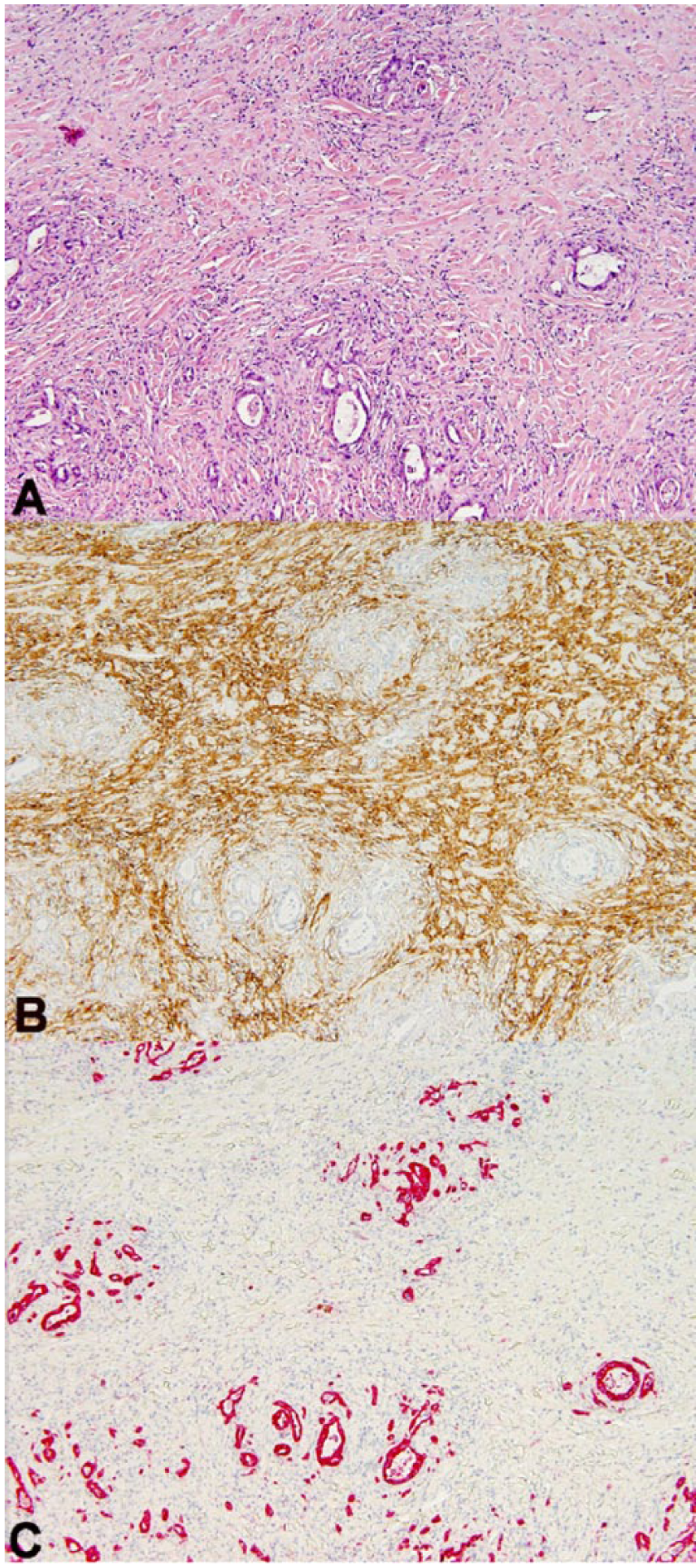
(A) An area of neoplasia with extensive areas of intermingling. H&E. (B) The leiomyosarcoma is positive for α–smooth muscle actin (α-SMA), but the adenocarcinoma is negative. Immunohistochemistry (IHC), α-SMA. (C) The cytokeratin-positive adenocarcinoma forms isolated islands; spindle cells are negative. IHC, cytokeratin AE1/AE3.
The nodules within the lungs were well-demarcated, unencapsulated, infiltrative, moderately cellular epithelial neoplasms that compressed and replaced the lung parenchyma, similar in appearance to the epithelial tumor populations within the uterine mass. These lesions were consistent with metastasis from the uterine adenocarcinoma (Fig. 5).
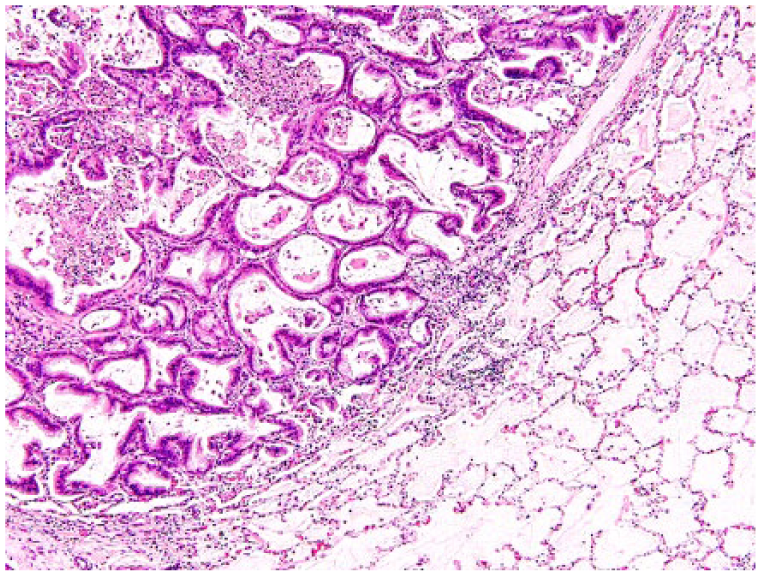
Metastatic uterine adenocarcinoma in the lung: well-defined tubular structures compress the lung parenchyma and are surrounded by moderate numbers of neutrophils admixed with fibrin. H&E.
Serosanguineous vaginal discharge is often noted in animals with uterine neoplasia.1,2,10–12 Other clinical signs of uterine neoplasia include weight loss, lethargy, depression, and abdominal pain.10,12 In this case, given the chronicity of vaginal discharge and the biological behavior of these neoplasms, the leiomyosarcoma most likely developed first followed by the adenocarcinoma. A scirrhous response has been excluded and a diagnosis of collision tumor favored because areas with no epithelial component (leiomyosarcoma) are continuous with areas infiltrated by epithelial cells (adenocarcinoma). The mesenchymal component of this neoplasm was diagnosed as leiomyosarcoma (instead of leiomyoma) given the high degree of infiltration, the presence of 1 mitotic figure per 10 HPF, karyomegalic cells, and areas of necrosis and hemorrhage within the neoplasm.
Carcinosarcoma was considered the primary differential diagnosis in this case. In humans, carcinosarcoma is considered a uterine carcinoma in which areas of malignant sarcomatous differentiation are present. The epithelial component of the mass is thought to drive this process; both mesenchymal and epithelial populations exhibit features consistent with a “high-grade” malignancy with sharp demarcations between these populations. True collision tumors are thought to arise from 2 different cell populations in close proximity that subsequently intermingle. A collision tumor in this case was thought more likely because the masses clearly represent 2 distinct populations of relatively well-differentiated neoplastic cells that intermingle in areas. However, when both cell populations are relatively well-differentiated, as in this case, definitive differentiation between a carcinosarcoma and a collision tumor can be difficult. 7
Abnormal posture during urination was also noted in this patient. Pollakiuria and abdominal straining in goats with pathologic enlargement of the uterus has been reported, and is postulated to occur secondary to mechanical irritation of the urinary bladder.1,8,12 The present case was also diagnosed with bilateral hydroureters secondary to compression of the ureters by uterine mass lesions, which may have contributed to this patient’s abdominal straining.
Collision tumor within the uterus has been reported in humans, with abdominal pain and abnormal genital bleeding being the most common presenting complaints.4,5,9 Treatment typically consists of hysterectomy with or without adjunct hormonal therapy, chemotherapy, and radiation therapy.4,5,9 A collision tumor of a granular cell tumor and a uterine adenocarcinoma has been reported in a hamster. 2 The hamster only required ovariohysterectomy for treatment. 2 Unfortunately, the disease described in our case was advanced and euthanasia was elected.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
